Abstract
Background:
Defects in modulating wild-type (wt) p53 and survivin are associated with a resistant disease in acute lymphoblastic leukemia (ALL). Yet, no wt-p53 and survivin modulating drugs have been approved for clinical application in ALL. Here, we investigated if in vitro eicosapentaenoic acid (EPA) concentrations equal to human plasma levels are able to target wt-p53 and survivin.
Methods:
Wt-p53 Molt-4 cells (ALL cell line) were treated with 50, 100, 150, and 200 µM of EPA after which cell number, viability, proliferation rate, survivin expression, wt-p53 accumulation, caspase-3 activation, and apoptosis were evaluated.
Results:
After 48- and 72-h treatments with EPA at concentrations ranging from 50 to 200 µM, cell proliferation rates were measured to be 71.5–32.6% and 68.2–13.7% and metabolic activities were measured to be 77–44% and 71–26%, respectively. Treatment with 50–200 µM of EPA for 48 h resulted in 14.1–74.6% and 69.5–45.5% decreases in survivin mRNA and protein levels, respectively. EPA induced 1.3–6 and 1.9–20-fold increases in caspase-3 activation and wt-p53 accumulation, respectively. Increase in wt-p53/survivin and caspase-3/survivin ratios from 1 in untreated cells to 20.3 and 5.8 was measured for 150 µM of EPA. Low necrotic rates ranging from 0.3% to 2.8% and an increase in the number of total apoptotic cells (early + late) ranging from 9.8% to 81% were also observed with increasing EPA concentrations.
Conclusion:
EPA induces strongly wt-p53 with a remarkable decrease in survivin expression, representing an attractive compound to modulate wt-p53 and survivin in ALL cells.
Keywords
Introduction
Acute lymphoblastic leukemia (ALL) is the commonest childhood cancer and an important cause of morbidity from hematological malignancies in Iran and the world. 1,2 Unfortunately, current therapies for treatment of ALL do not eliminate malignant cells, leading to disease relapse. 3 It has been shown that overexpression of survivin gene that belongs to the inhibitor of apoptosis (IAP) genes family correlates with relapse and refractory ALL disease. 4 Survivin blocks induction of apoptosis by a variety of apoptotic triggers. 5 Survivin has been reported to bind directly to and inhibit caspase-3 and -7, which acts as terminal effectors in apoptotic protease cascades. 5,6 This molecule is undetectable in normal cells but becomes reexpressed and upregulated in pediatric and adult ALL patients. 7,8
Wild-type (wt) p53 has been reported to be involved in regulation of metabolic adaptation of cancer cells which can negatively regulate cell growth by inhibiting PI3K/Akt/mTOR signaling pathway. 9 Inactivation of the wt-p53 expression as a tumor suppressor gene has been either by mutations or by loss of expression implicated in chemoresistance and poor prognosis in ALL. 4,10 Wt-p53 is a transcription factor that plays a key role in both cell cycle arrest and apoptosis. 11,12 Wt-p53 works through several mechanisms. It can activate DNA repair proteins when DNA has sustained damage, induce growth arrest by holding the cell cycle at the G1/S regulation point on DNA damage recognition, and initiate apoptosis if DNA damage proves to be irreversible. 13 With these in mind, high expression level of survivin in chemoresistance ALL cells along with loss of wt-p53 expression in most ALL cells would make these molecules as attractive targets in ALL-directed therapy. Therefore, simultaneously targeting wt-p53 and survivin by a natural compound may modulate apoptosis signaling pathway and induces apoptosis in ALL cells and may provide a new therapeutic approach for treatment of drug-resistant ALL.
Experimental studies have shown that fish-oil-derived eicosapentaenoic acid (EPA) as omega-3 polyunsaturated fatty acids (ω-3 PUFAs) has antiproliferative effect on different kinds of malignancies including leukemia. 14,15 In this regard, several molecular mechanisms have been considered for the activity of EPA compound against cancer cells including alteration in gene expression and modulations of cellular proliferation and apoptosis. 16,17 EPA modulates RAS/ERK/C/EBPβ signaling pathways and plays a role in regulating the growth and differentiation. 16,17 In this regard, it has been shown that EPA induces apoptosis through cytochrome c release from mitochondria and mitochondrial membrane depolarization and enhances the activities of both caspases 8 and 9. 18
Different studies have revealed an inverse relationship between different kinds of cancer risk and ω-3 PUFAs consumption, suggesting a protective role of ω-3 PUFAs against the development of different kinds of cancer. 19,20 With this in mind, we hypothesized that the physiologic levels of EPA in human plasma contribute to modulate key important apoptosis-related genes and induction of caspase-3 activation and apoptosis in ALL cells.
It has been shown that the baseline of EPA concentration in human plasma is 79 µM and consumption of 2.4 g and 4.8 g/day of ω-3 PUFAs yielded 154 and 302.5 µM of EPA in human plasma, respectively. 21 In consequence, we aimed to investigate if in vitro concentrations of EPA (50, 100, 150, and 200 µM) equal to plasma levels achievable in the human body following supplementation of the diet with different amounts of ω-3 PUFAs per day as mentioned above are able to target wt-p53 and survivin expression with induction of caspase-3 activation and apoptosis in ALL cells.
Materials and methods
Cell line, culture condition, and primers
Molt-4 cell line was purchased from the National Cell Bank of Iran (Pasture Institute of Iran, Tehran). Molt-4 cell line is wt-p53 ALL cells from a patient with recurrent disease and was grown in RPMI-1640 medium containing 10% (v/v) fetal bovine serum (FBS), penicillin (100 U/ml)/streptomycin (100 µg/ml) all from PAA (Austria), 20 mM HEPES, and 2 mM L-glutamine (Roche, Germany) at 37°C in a humidified incubator with 5% carbon dioxide (CO2). The oligonucleotides for polymerase chain reactions (PCRs; Table 1) were synthesized by Bioneer Corporation (South Korea).
List of oligonucleotides used in the present study.

PCR: polymerase chain reaction.
aAnnealing temperature in degree Celsius (number of cycles).
Chemicals, drugs, and kits
Pure (99%) cis-5, 8, 11, 14, 17-EPA (Figure 1) and trypan blue solution (0.4%) were purchased from Sigma-Aldrich Chemical Company, Germany. Caspase-3 activity, water-soluble tetrazolium salt (WST)-1 cell proliferation assay, and Annexin-V-FLUOS apoptosis assay kits were purchased from Roche. RNA isolation solution (guanidine/phenol solution) was obtained from SinaClon Bioscience Company, Iran. The kit for cDNA synthesis was obtained from Fermentas (Burlington, Ontario, Canada). Primescript™ RT reagent kit was purchased from Takara, Japan. Human survivin and p53 ELISA kits were purchased from Abcam, Cambridge, Massachussets, USA. All other general chemicals were purchased from Merck Chemical Company, Germany.

Structure of fish-oil-derived EPA is shown. EPA: eicosapentaenoic acid.
Preparation of EPA and cell treatments
EPA was dissolved in absolute ethanol to prepare a 100-mM stock solution and stored in the dark at −80°C until use. For each experiment, the EPA was freshly prepared from the stock solution at the concentrations ranging from 50 to 200 µM by serially diluting in culture medium. Control cells were cultured in the medium containing the same concentration of absolute ethanol (v/v) as the EPA containing medium. The final ethanol concentration never exceeded 0.5% (v/v).
Cell count and viability assays
Molt-4 cells were seeded at a density of 5 × 105 cells per well in 1.5 ml of complete medium in six-well plates and incubated at 37°C for 24 h. Thereafter, the cells were treated with 50, 100, 150, and 200 µM of EPA for 48 h. Following 48-h treatments, the cells were harvested and stained with trypan blue after which the numbers of viable cells and total cell numbers were determined under an inverted microscope. Cell viability after each treatment procedure was expressed as percentage of the untreated control cells.
Metabolic activity and proliferation assays
To evaluate metabolic activity and cell proliferation rates in a dose- and time-dependent manner, we evaluated these items 48- and 72-h post treatments; 5 × 103 cells per well were plated in 96-well plates. After 24-h incubation, Molt-4 cells were treated for 48 and 72 h with 50, 100, 150, and 200 µM of EPA and metabolic activities as well as proliferation rates were evaluated by performing WST proliferation assay. Following each incubation period, the WST-1 solution (10 µl) was added to each well and the cells were incubated for an additional 4 h at 37°C and 5% CO2. The absorbance in each well was then measured with a microplate reader (Biotek, Winooski, Vermont, USA) at 450 nm and a reference wavelength of 620 nm for evaluation of metabolic activities. Thereafter, proliferation rates were calculated as (A sample − A blank)/(A control − A blank) × 100%. 22 The results were expressed as the percentages of untreated control cells.
To measure the total growth inhibition and concentration causing 50% of growth inhibition (IC50) values of EPA, the percentages of the proliferation rates on the y-axis were plotted against the concentrations of the drug on the x-axis. Finally, all calculations were performed using regression analysis.
RNA isolation, cDNA preparation, and reverse transcription PCR
Total RNAs were extracted from EPA-treated and control cells using RNA preparation kit and subsequently used as a template to generate a first cDNA strand according to the manufacturer’s guidelines. The generated fragments were subsequently used as templates for the PCR amplification of the double-stranded cDNA corresponding to a preselected region of the survivin coding sequences using specific forward and reverse primers (Table 1) with the following temperature profile: After an initial denaturation step at 94°C for 5 min , 40 cycles of denaturation at 94°C for 20 s, annealing at 62°C for survivin for 30 s, and extension at 72°C for 30 s, followed by a final extension at 72°C for 10 min, were performed. In parallel, to evaluate semi-quantitation of survivin expression, the generated fragments were used as template for the PCR amplification of a section of the human GAPDH (human Glucose-6 phosphate dehydrogenase) coding sequence, using specific forward and reverse primers (Table 1). PCR products were visualized on 1% agarose gels after ethidium bromide staining. The ratios of the survivin and GAPDH band densities were calculated using gel documentation (Gel Logic Pro 212 Imaging System, Kodak, USA) and multiplied by 100 (survivin/GAPDH × 100). These ratios represent relative survivin mRNA levels.
Enzyme-linked immunosorbent assay
Survivin protein assay
Intracellular survivin protein level was assayed by the sandwich enzyme-linked immunosorbent assay (ELISA) following the procedure provided by the manufacturer (Abcam). Briefly, Molt-4 cells (5 × 105 per well) were cultured in the absence or in the presence of EPA (50, 100, 150, and 200 µM) for 48 h. Next, the cells were washed twice with ice-cold phosphate buffered saline (PBS; pH 7.4, 0.15 M) and suspended in ready to use cold lysis buffer (provided by the manufacturer) for 30 min followed by centrifugation at 12,000 × g for 15 min at 4°C. Thereafter, supernatant was then collected and used for survivin protein assay. Briefly, microtiter ELISA plate coated with the mouse antihuman survivin monoclonal antibody. Following sample application, a biotinylated detection polyclonal antibody from goat specific for human survivin is then added followed by washing with PBS buffer. Thereafter, avidin–biotin–peroxidase complex is added and unbound conjugates are washed away with PBS buffer. Tetramethyl benzidine (TMB) substrate solution was finally added and reactions were stopped after 15 min. The optical density values of the samples were recorded at 450 nm using an ELISA reader (BioTek, USA).
Wt-p53 protein assay
Intracellular wt-p53 protein level was assayed by the sandwich ELISA following the procedure provided by the manufacturer (Abcam). As mentioned above, EPA-treated tumor cell lysates were prepared and supernatant was then collected and used for wt-p53 protein assay. Briefly, microtiter ELISA plate coated with the human monoclonal antibody specific for wt-p53. Following sample application and washing, a biotinylated monoclonal antibody specific for wt-p53 was added and incubation continued for 1 h at room temperature. Next, the wells were washed and the enzyme Streptavidin-HRP that binds the biotinylated antibody was added and incubation continued for 30 min followed by washing. Thereafter, TMB substrate solution was finally added which acts on the bound enzyme to induce a colored reaction product. The optical density values of the samples were recorded at 450 nm using an ELISA reader (BioTek).
Caspase-3 activity assay
Caspase-3 activity was measured by using the caspase-3 colorimetric assay kit following the procedure provided by the manufacturer (Abcam). Briefly, treated and untreated cells were collected after 48-h treatment and resuspended in ready to use chilled lysis buffer for 10 min. Next, high-speed centrifugation was performed and the supernatants were collected and used for caspase-3 activation assay. Briefly, reaction buffer, Dithiothreitol (DTT), and DEVD-p-nitroaniline (p-NA) substrate were added to samples and plates were incubated at 37°C for 2 h. The principle was that caspase-3 derived from cellular lysate recognizes the sequence Asp-Glu-Val-Asp (DEVD). The assay is based on spectrophotometric detection of the chromophore p-NA after cleavage from the labeled substrate (DEVD-p-NA). The p-NA light emission can be quantified using a microtiter plate reader at 450 nm. Comparison of the absorbance of p-NA from an apoptotic sample with an untreated control sample allows determination of the fold increase in caspase-3 activity. Procedure measures only the functionally relevant cleaved caspase-3.
Flow cytometric analyses
Apoptosis was analyzed by a double-staining method using Annexin-V-FLOUS/propidium iodide (PI) labeling solution according to the manufacturer’s instructions (Roche). Briefly, Molt-4 cells were treated with different concentrations of EPA for 48 h. Thereafter, the cells were washed twice with sterile cold PBS and after centrifugation, cell pellets were then resuspended in 100 µl of 1 × binding buffer at a density of 5 × 105 cells/ml with FITC-Annexin V. The cells were gently mixed and incubated in the dark at room temperature for 20 min. To differentiate cells with membrane damage, PI solution was added to the cell suspension prior to the flow cytometric analysis using a fluorescence-activated cell sorter (FACScan, Franklin Lakes, NewJersy, USA).
Statistical analyses
Data represent at least means of two independent experiments with duplicate or triplicate assays. The results were shown as mean ± standard deviation. All calculations were performed using the SPSS version 20 for Windows (SPSS Inc., Chicago, Illinois, USA). Analysis of variance followed by a Tukey post hoc was used for comparisons. 23 Values of *p < 0.05 and **p < 0.01 were considered to be statistically significant and highly significant, respectively.
Results
Cell count and viability assays (trypan blue dye exclusion)
Results showed that EPA significantly decreased cell number and viability in a dose-dependent manner (Figure 2). As shown in Figure 2(a), EPA diminished cell numbers by 8.5 × 105–0.73 × 105 cells at concentrations ranging from 50 to 200 µM of EPA compared to untreated cells. Concomitantly, Molt-4 cell viabilities were measured to be 75.9–6.6% at the same concentrations ranging compared to the untreated cells (Figure 2(b)). Based on these data, EPA in the used doses can be considered as a potent agent in decreasing Molt-4 cell number and viability at the doses used.

Effect of EPA on cell number and viability; 5 × 105 Molt-4 cells were treated with various concentrations of EPA for 48 h after which (a) cell number was counted and (b) cell viability was measured. All experiments were repeated twice using triplicate assays and the results were presented as mean ± SD. *p < 0.05, **p < 0.01 versus untreated control cells (C). EPA: eicosapentaenoic acid; SD: standard deviation.
Effect of EPA on metabolic activity and cell proliferation
Treatment of cells with increasing concentrations of EPA diminished metabolic activity and cell proliferation rate in a dose- and time-dependent manner (Figure 3 and Table 2). We found that treatment for 48 and 72 h with 200 µM of EPA resulted in 2.3- and 3.8-fold decreases in the metabolic activities followed by 67.3% and 86.3% decreases in cell proliferation rates, respectively, compared to untreated control cells (Table 2). Specifically after a 72-h treatment, EPA at a concentration of 200 µM was found to be more potent in inhibiting metabolic activity and cell proliferation rate compared to other treatments at the same conditions.

Effect of EPA on metabolic activity; 5 × 103 Molt-4 cells per well were treated with different concentrations of EPA for 48 and 72 h after which metabolic activities were measured by WST-1 assay. All experiments were repeated twice using triplicate assays and the results were presented as mean ± SD. *p < 0.05, **p < 0.01 versus untreated control cells (C). EPA: eicosapentaenoic acid; SD: standard deviation; WST: water-soluble tetrazolium salt.
Evaluation of cell proliferation and growth-inhibition rates in EPA-treated cells.a

EPA: eicosapentaenoic acid; WST: water-soluble tetrazolium salt.
a5 × 103 Molt-4 cells per well were treated with different concentrations of EPA for 48 and 72 h after which cell proliferation rates were determined by WST-1 assay. IC50 and total growth inhibition values were calculated from the dose–response curves. Data represent means of two independent experiments with triplicate assays.
b p < 0.01 versus untreated control cells.
Reduction in total growth inhibition of Molt-4 cells and IC50 values of EPA was also observed after 72-h treatment compared to 48-h treatment (Table 2).
Experiments with peripheral blood mononuclear cells (PBMCs) isolated from two healthy individuals showed little to no toxicity of EPA on these cells (data not shown).
Effect of EPA on the survivin mRNA level
Survivin belongs to the IAP genes family 4 that blocks apoptosis. 5 Since the survivin mRNA level in EPA-induced apoptosis has so far not been evaluated in ALL cells, we set out to evaluate the effect of EPA treatment on the expression of survivin. To this end, Molt-4 cells were treated with different concentrations of EPA. We found that, after a 48-h treatment with EPA, the high level of survivin mRNA in untreated Molt-4 cells decreased significantly at different concentrations of EPA (Figure 4(a) and (b)). In this context, we found that treatment with 50 to 200 µM of EPA resulted in a 15.1–64.4% decrease in the survivin mRNA levels compared to untreated cells.

RT-PCR detection of survivin mRNA levels; 5 × 105 Molt-4 cells were treated with different concentrations of EPA for 48 h and total RNAs were isolated and subjected to the RT-PCR. (a) PCR products were visualized on 1% agarose gel after ethidium bromide staining. (b) Survivin mRNA levels relative to GAPDH levels in EPA-treated cells are shown. *p < 0.05, **p < 0.01 versus untreated control cells (C). RT-PCR: reverse transcription polymerase chain reaction; EPA: eicosapentaenoic acid.
Effect of EPA on the survivin and p53 protein levels
To evaluate the decrease in survivin mRNA levels results in decrease in survivin protein levels in Molt-4 cells, we evaluated the intracellular survivin protein levels in EPA-treated cells. As shown in Figure 5, treatment of Molt-4 cells with increasing concentrations of EPA decreased significantly level of survivin protein ranging from 69.5% to 45.4% compared to untreated cells. Specifically, treatment with 50 µM of EPA resulted in a maximum decrease in survivin protein level compared to untreated cells. A positive correlation was observed between survivin mRNA levels and survivin protein levels (r = 0.5).

Effect of EPA on survivin protein levels; 5 × 105 Molt-4 cells were treated with different concentrations of EPA for 48 h after which survivin protein levels were assayed in tumor cell lysates. All experiments were repeated twice using triplicate assays and the results were presented as mean ± SD. **p < 0.01 versus untreated control cells (C). EPA: eicosapentaenoic acid; SD: standard deviation.
To evaluate whether treatment with EPA induces wt-p53 accumulation, the wt-p53 protein levels were measured. In this regard, after a 48-h exposure of Molt-4 cells to EPA, a dramatic increase in the levels of wt-p53 protein was measured to be 1.9–20-fold increase for 50–200 µM of EPA compared to untreated cells (Figure 6) which reflects the effect of EPA in induction of wt-p53 accumulation within the ALL cells.

Effect of EPA on the wt-p53 protein levels; 5 × 105 Molt-4 cells were treated with different concentrations of EPA for 48 h after which wt-p53 protein levels were assayed in tumor cell lysates. All experiments were repeated twice using triplicate assays and the results were presented as mean ± SD. **p < 0.01 versus untreated control cells (C). EPA: eicosapentaenoic acid; SD: standard deviation.
Effect of EPA on caspase-3 activation
As shown in Figure 7, treatment of Molt-4 cells with EPA resulted in high levels of caspase-3 activation and increased significantly with increasing EPA concentrations. In this regard, treatment with 50–200 µM of EPA resulted in 1.3–6-fold increases in caspase-3 activation compared to untreated cells.

Effect of EPA on caspase-3 activation. Molt-4 cells were treated with various concentrations of EPA for 48 h after which caspase-3 activity was measured in tumor cell lysates using colorimetric assay. All experiments were repeated twice using triplicate assays and the results were presented as mean ± SD. *p < 0.05, **p < 0.01 versus untreated control cells (C). EPA: eicosapentaenoic acid; SD: standard deviation.
Effect of EPA on apoptosis induction
In order to investigate the growth-inhibitory effect observed upon treatment of Molt-4 cells with EPA was due to the induction of apoptosis, the cells were treated with 50, 100, 150, and 200 µM of EPA for 48 h and subsequently stained with Annexin V/PI and analyzed by means of flow cytometry techniques. With this method, the cells stained single positive for PI were considered mostly necrotic cells and cells stained single positive for Annexin V were considered mostly early apoptotic cells, but cells that were stained double positive could be either necrotic or apoptotic cells. As depicted in Figure 8 and Table 3, Molt-4 cells were treated with 50 to 200 µM of EPA displayed apoptosis in a dose-dependent manner ranging from 9.8% to 81% (total apoptosis) compared to untreated cells. Concurrently, the number of necrotic cells was increased ranging from 0.5% to 3% compared to untreated cells.

Effect of EPA on apoptosis induction. Molt-4 cells were treated with various concentrations of EPA for 48 h and subsequently, the cells were stained with Annexin V/PI and analyzed by means of flow cytometry technique. FL1: Annexin V-FITC and FL2: PI. Q1: necrotic cells, Q2: late apoptosis + necrosis, Q3: living cells, and Q4: early apoptotic cells. C: nontreated control cells. EPA: eicosapentaenoic acid; PI: propidium iodide.
Evaluation of EPA effect on apoptosis induction in Molt-4 cells.
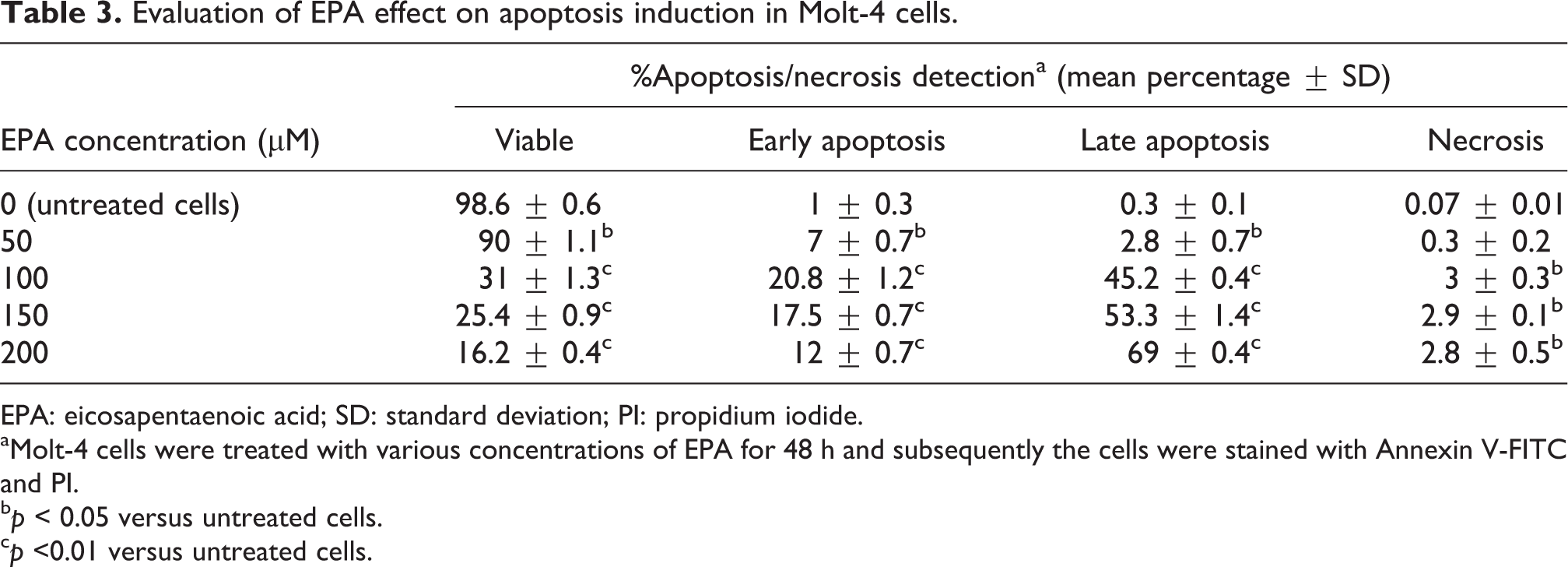
EPA: eicosapentaenoic acid; SD: standard deviation; PI: propidium iodide.
aMolt-4 cells were treated with various concentrations of EPA for 48 h and subsequently the cells were stained with Annexin V-FITC and PI.
b p < 0.05 versus untreated cells.
c p <0.01 versus untreated cells.
Effect of EPA on the p53/survivin protein and casapse-3/survivin protein ratios
The ultimate vulnerability of cells to diverse apoptotic stimuli is determined by the relative ratio of various tumor suppressor and proto-oncogene genes expression. In the present study, we determined the relative ratios of tumor suppressor wt-p53 to proto-oncogene survivin after 48-h treatment with different concentrations of EPA. In this regard, treatment with 100 µM of EPA resulted in a dramatic and maximum increase in wt-p53/survivin ratios and was measured to be 29.7-fold increase as compared to untreated cells (Table 4).
Evaluation of p53 protein/survivin protein and caspase-3 protein/survivin protein ratios in EPA-treated cells after 48-h treatment.a

EPA: eicosapentaenoic acid.
a5 × 105 Molt-4 cells per well were treated with different EPA concentrations for 48 h after which p53, survivin protein, and caspase-3 expression levels were measured. Next, p53/survivin and caspase-3/survivin protein ratios were calculated. The results represent means of two independent experiments with duplicate assays.
b p < 0.01 versus untreated control cells.
The level of proapoptotic marker such as caspase-3 versus the level of antiapoptotic marker such as survivin could be important in determining the resistance of cells to apoptosis. As shown in Table 4, treatment with EPA for 48 increased the caspase-3/survivin protein ratios. Specifically, treatment with 150 µM of EPA resulted in 5.8-fold increase in the caspase-3/survivin protein ratio compared to untreated cells.
Discussion
Outcomes are worse for patients with relapsed T-ALL. Most cases of ALL express wt-p53 with low activity and high expression level of survivin which are associated with resistant and refractory disease in ALL patients. 24 –27 Consequently, these would make wt-p53 and survivin as attractive targets in ALL treatment. Yet, no wt-p53 and survivin modulating drugs have been approved for clinical application in resistant ALL. For this purpose, we needed to explore a naturally fish-oil-derived EPA compound effect in resistant T-ALL cells with specific phenotypes. In this regard, Molt-4 cells were attractive candidate host cells. These cells have been isolated from a patient with relapsed T-ALL and carry wt-p53 with high expression level of survivin which these genes are known to play a central role in poor prognosis in ALL. 24 –27
EPA induced caspase-3 activation and apoptosis in Molt-4 cells, suggesting that the observed decreases in cell number, viability, and proliferation rates are due to the induction of apoptotic mechanisms. Concurrently, EPA compound had little to no toxicity on normal PBMCs isolated from healthy individuals. In line to our experiments, different researchers have shown that EPA has selective cytotoxicity on cancer cells with no toxicity toward normal cells. 20,28 These evidences together with our findings obtained from various concentrations of EPA (which were below the maximum tolerated dose of PUFAs per day) 29 suggest that the EPA could be an attractive safe adjuvant for treatment of ALL patients.
It is interesting to note that the ratios between necrosis and apoptosis rate remain low with increasing EPA concentrations, suggesting that this compound activates various apoptosis pathways not necrosis ones in Molt-4 cells.
The marked decreases in metabolic activities that we observed, particularly after a 72-h treatment, suggest that the EPA tested may exhibit its cytotoxic effects against mitochondria. Low metabolic activities combined with high levels of wt-p53 and caspase-3 activation were correlated with higher apoptosis rates in EPA-treated cells. It has been shown that high levels of p53 induce superoxide dismutase downregulation which increases Reactive oxygen species (ROS) concentration and promotes apoptosis. 30,31 Superoxide dismutase is found overexpressed and is in favor of mitochondrial homeostasis in cancer cells. 31 With this in mind, EPA may target superoxide dismutase through the induction of p53 expression in Molt-4 cells which results in a decline in mitochondrial function. However, mitochondrial membrane depolarization in leukemia cells by EPA as shown by different investigators should be considered. 18 Further experiments are required to highlight these questions in the present study.
In our study, EPA induced significantly high accumulation of wt-p53 protein followed by caspase-3 activation and apoptosis, suggesting that EPA is an attractive apoptosis inducer through a p53-dependent mechanism. These documents also suggest an interesting effect of EPA on the wt-p53 gain of function and a role for wt-p53 in EPA-mediated apoptosis in Molt-4 cells. In consequence, EPA may provide an attractive candidate drug to modulate wt-p53 expression in wt-p53 ALL cells. The ability of growth inhibitory effect of EPA with induction of wt-p53 accumulation is particularly interesting, as p53 exerts a key role on induction of apoptosis and cell cycle arrest. 11,12
It has been shown that survivin is a p53 target gene. Promoter of survivin gene contains a p53 response sequence and an increase in the expression of p53 inhibits survivin promoter activity, which results to the decrease in survivin protein expression. 32,33 Based on our data, low survivin protein levels combined with high levels of wt-p53 protein accumulation were associated with higher caspase-3 activation level and apoptosis rate, suggesting activity of induced wt-p53 accumulation in EPA-treated cells. However, further experiments are needed to highlight this issue in these cells.
We also found that EPA decreases the survivin mRNA and protein levels, suggesting an interesting effect of EPA turning the profile of high level of survivin expression in Molt-4 cells into that of normal cells. Survivin protein has been shown to bind specifically to and inhibit the activated form of caspase-3. 6 Induction of caspase-3 activation through inhibition of the survivin expression seems to be one of the molecular pathways leading to EPA apoptotic effect in Molt-4 cells which has been previously shown in LS174T cells. 14 Our results confirm previous data demonstrating that wt-p53 and survivin expression are regulated by PUFAs, 14,34 thus providing promising molecular targets of EPA.
Survivin is negatively regulated by wt-p53 and participates in a p53-dependent apoptotic pathway. 33 In our study, the wt-p53/survivin protein ratio was perturbed as a result of EPA treatment which correlates with higher apoptotic rate compared to untreated cells. It is possible that one of the mechanisms contributing to the EPA cytotoxicity is related to the interaction of wt-p53 with survivin promoter in Molt-4 cells similar to other studies. 32,33 In this regard, the p53/survivin protein ratio may upregulate caspase-3 activation and modulate apoptosis in treated cells compared to untreated cells. Further experiments are required to answer this question in these cells. Moreover, evaluation of this ratio could be important and may find a therapeutic index particularly when EPA as a safe adjuvant in combination with a chemotherapeutic drug is used for treatment of ALL in vivo.
In conclusion, our findings demonstrate for the first time that fish-oil-derived EPA at concentrations equal to plasma levels targets effectively wt-p53 and survivin and this compound may provide a novel therapeutic clinical application for treatment of ALL.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by Urmia University and National Cell Bank of Iran.
