Abstract
Background:
Abnormal activation of the Wnt/β-catenin signaling pathway increases survivin expression that is involved in hepatocarcinogenesis. Therefore, downregulation of survivin may provide an attractive strategy for treatment of hepatocellular carcinoma. In this regard, little is known about the anticancer effects of prodigiosin isolated from cell wall of Serratia marcescens on the survivin expression and induction of apoptosis in hepatocellular carcinoma cells.
Methods:
Human hepatocellular carcinoma (HepG2) cells were treated with 100-, 200-, 400-, and 600-nM prodigiosin after which morphology of cells, cell number, growth inhibition, survivin expression, caspase-3 activation, and apoptotic rate were evaluated by inverted microscope, hemocytometer, MTT assay, RT-PCR, fluorometric immunosorbent enzyme assay, and flow cytometric analysis, respectively.
Results:
Prodigiosin changed morphology of cells to apoptotic forms and disrupted cell connections. This compound significantly increased growth inhibition rate and decreased metabolic activity of HepG2 cells in a dose- and time-dependent manner. After 24-, 48-, and 72-h treatments with prodigiosin at concentrations ranging from 100 nM to 600 nM, growth inhibition rates were measured to be 1.5–10%, 24–47.5%, and 55.5–72.5%, respectively, compared to untreated cells. At the same conditions, metabolic activities were measured to be 91–83%, 74–53%, and 47–31% for indicated concentrations of prodigiosin, respectively, compared to untreated cells. We also found that treatment of HepG2 cells for 48 h decreased significantly cell number and survivin expression and increased caspase-3 activation in a dose-dependent manner. Specifically, treatment with 600-nM prodigiosin resulted in 77% decrease in cell number, 88.5% decrease in survivin messenger RNA level, and 330% increase in caspase-3 activation level compared to untreated cells. An increase in the number of apoptotic cells (late apoptosis) ranging from 36.9% to 97.4% was observed with increasing prodigiosin concentrations.
Conclusion:
From our data, prodigiosin is an attractive compound that turns the profile of high-level survivin expression in hepatocellular carcinoma cells into that of normal cells and may provide a novel approach to the hepatocellular carcinoma-targeted therapy.
Introduction
Hepatocellular carcinoma (HCC) is the fifth most common cancer in men and is the second leading cause of cancer death worldwide. 1 Currently, there are no effective therapies for HCC other than surgical resection, conventional chemotherapy, radiotherapy, or liver transplantation according to the stage of the disease. Unfortunately, these treatments have serious side effects on normal tissues, and majority of patients die within 12 months of diagnosis. In this regard, molecular directed therapy is an attractive candidate as a new therapeutic strategy. Therefore, the search for a new therapeutic agent with minimal cytotoxicity and high efficacy that targets the molecular signaling pathways that are involved in hepatocarcinogenesis is of particular interest in HCC-directed therapy. In this context, secondary metabolites of bacteria with anticancer properties are considered to be new promising agents. Prodigiosin (2-methyl-3-pentyl-6-methoxyprodiginine; Figure 1) as a secondary metabolite of Serratia marcescens is the prototype of bacterial prodiginines, a family of tripyrrole red pigments featured by its diverse bioactive effects including anticancer and antimetastatic effects. 2 –4 Prodigiosin activates apoptosis in different kinds of malignant cells with low toxicity on normal cells. 5,6 This compound has multiple mechanisms of action. At the cellular level, prodigiosin targets several vital processes that potentially contribute to its anticancer function. These include acidification of the cytoplasm by acting as H+/Cl− symporters, arrest of cell cycle progression by upregulation of p21WAF/CIP1, induction of DNA damage by inhibiting topoisomerases I and II and reduction in ATP production by uncoupling energy from mitochondrial proton gradient to ATP synthesis. 6,7

Structure of prodigiosin isolated from cell wall of Serratia marcescens.
The anticancer effect of prodigiosin has been shown in different kinds of cancer cell lines and is attributed to induction of mitochondria-mediated apoptosis. 8
Expression of survivin contributes to the apoptosis regulation in different kinds of cancer cells. Survivin is a unique member of bacluoviral inhibitor of apoptosis family that is known to negatively regulate apoptosis by inhibiting caspases. Survivin is overexpressed in nearly all human tumors but is low or absent in normal cells. This notion is clinically important, because overexpression of survivin is closely associated with tumorigenesis, poor prognosis, and drug resistance. 9 –12
It has been shown that the expression of some proapoptotic genes is decreased, and the balance between death and survival is dysregulated in HCC mainly due to overexpression of antiapoptotic genes such as survivin. 13 It has been suggested that survivin might play a pivotal role in metastasis. 14 Survivin might play an important role in progression of HCC not only by inhibiting apoptosis 15 but also by promoting cell proliferation 16 and may be positively correlated with high risk of disease recurrence and poor prognosis. 17 With these in mind, targeted survivin downregulation in HCC by a therapeutic agent may provide minimal toxicity to normal cells and may open up avenues to new therapeutic strategies for HCC-directed therapy.
The present study investigates the effects of different concentrations of prodigiosin on cell number, cell proliferation, caspase-3 activation, and downregulation of survivin gene expression in Human hepatocellular carcinoma (HepG2) cells that serve as a model for HCC.
Methods
Cell line, culture condition, and primers
HepG2 cell line as a model for HCC cells was obtained from the National Cell Bank of Iran (Tehran). The cells were grown in RPMI-1640 medium containing 10% (v/v) fetal bovine serum (FBS), 100-U/mL penicillin, 100-µg/mL streptomycin all from PAA Laboratories (Austria), and 20-mM HEPES and 2-mM
Preparation of prodigiosin and administration
Prodigiosin (Sigma, USA) was dissolved in absolute ethanol (Merck, Germany) to prepare a 20-µg/mL stock solution and stored in the dark at −80°C until use. For each experiment, the prodigiosin was freshly prepared from the stock solution at the concentrations ranging from 100 nM to 600 nM by serially diluting in culture medium. Control cells were cultured in a medium containing the same concentration of absolute ethanol (v/v) as the prodigiosin-containing media. The final ethanol concentration never exceeded 0.5% (v/v).
Cell count assay
HepG2 cells were seeded in six-well plates at a density of 5 × 105 cells/well in 1.5-mL complete medium and incubated at 37°C for 24 h. Next, the cells were treated with different concentrations of prodigiosin (100, 400, and 600 nM) for 48 h. Subsequently, the cells were harvested and the total cell numbers were determined under an inverted microscope.
Metabolic activity assay
HepG2 cells were seeded in 96-well plates at a density of 5 × 103 cells/well in complete medium. After a 24-h incubation, the cells were treated with the indicated concentrations of prodigiosin for 24–72 h after which their metabolic activities were evaluated by MTT assay. Following each incubation period, MTT solution (5 mg/mL in phosphate-buffered saline (PBS)) was added to each well and the incubation was continued for 4 h at 37°C. Thereafter, the supernatants were removed and replaced by 200 μL of dimethyl sulfoxide to dissolve the MTT formazan crystals. Finally, the absorbance in each well was measured using a microplate reader (State Fox, USA) at 492 nm.
Growth inhibition assay
HepG2 cells were seeded in 96-well plates at a density of 5 × 103 cells/well in complete medium. After a 24-h incubation, the cells were treated with various concentrations of prodigiosin for 24–72 h to determine the growth inhibition rates, and the concentrations causing either a 50% growth inhibition (IC50) or a total growth inhibition. The growth inhibition rates were calculated as 100% − (Asample − Ablank)/(Acontrol − Ablank) ×100%, and the results were expressed as percentages of the untreated control cells. To measure the total growth inhibition and the IC50 values of prodigiosin, the percentages of the proliferation rates on the y-axis were plotted against the concentrations of the drug on the x-axis. Finally, all calculations were performed using regression analysis. All experiments were repeated at least twice using triplicate assays.
RNA isolation, cDNA preparation, and reverse transcription-PCR
Total cellular RNAs were extracted from treated and untreated cells using RNA preparation kit (Sinaclon Bioscience Co., Iran) and subsequently used as a template to generate a first cDNA strand according to the manufacturer’s instructions (Fermentas, Canada). The generated fragments were subsequently used as templates for PCR amplification of double-stranded cDNA corresponding to a preselected region of the survivin coding sequence, using specific oligonucleotides Survivin-F: 5′-GCATGGGTGCCCCGACGTTG-3′ and Survivin-R: 5′-GCTCCGGCCAGAGGCCTCAA-3′ as forward and reverse primers, respectively, with the following temperature program: After an initial denaturation step at 94°C for 5 min, 35 cycles of denaturation at 94°C for 5 min, annealing at 61°C for 1 min, extension at 72°C for 1 min followed by a final extension at 72°C for 10 min were performed. In parallel, for internal control and normalization, the generated fragments were used as template for PCR amplification of a section of the human GAPDH coding sequence, using oligonucleotides GAPDH-F:5′-CAAGGTCATCCATGACAACTTTG-3′ and GAPDH-R: 5′-GTCCACCACCCTGTTGCTGTAG-3′ as forward and reverse primers, respectively. The PCR products were visualized on 1% agarose gels after ethidium bromide staining. The ratios of the survivin and GAPDH band densities were calculated and multiplied by 100 (survivin/GAPDH × 100). These ratios represent relative survivin messenger RNA (mRNA) levels.
Apoptosis assays
Caspase-3 activity assay
Caspase-3 activation was determined using a fluorometric immunosorbent enzyme assay as per the manufacturer’s instructions (Roche, Germany). Briefly, HepG2 cells were cultured in the absence or in the presence of prodigiosin at indicated concentrations for 48 h. After trypsinization, the cells were washed twice with ice-cold PBS and resuspended in cold lysis buffer for 30 min followed by centrifugation at 12,000 g for 30 min at 4°C. Thereafter, supernatants were collected and used for caspase-3 activation assessment. The principle was that caspase-3 derived from cellular lysates is captured by a monoclonal antibody. The amount of activated caspase-3 was cleaved proportionally through the addition of fluorogenic substrate in a reaction buffer. Due to proteolytic cleavage of the substrate, free fluorescent AFC (7-amino-4-trifluoromethylcoumarin) is generated and measured with excitation at 380 nm and emission at 505 nm using a fluorescence spectrophotometer.
Flow cytometric apoptosis assay
Apoptosis was analyzed by a double-staining method using Annexin V FLOUS/propidium iodide (PI) labeling solution according to the manufacturer’s instructions (Roche, Germany). Briefly, HepG2 cells were cultured in the absence or in the presence of 100, 200, and 600 nM prodigiosin for 48 h. Thereafter, the cells were harvested and washed twice with sterile cold PBS. Cell pellets were then resuspended in 100 µL of 1× binding buffer at a density of 5 × 105 cells/mL with FITC-Annexin V. Next, the cells were gently mixed and incubated in the dark at room temperature for 20 min. To differentiate cells with membrane damage, PI solution was added to the cell suspension prior to flow cytometric analysis using a fluorescence-activated cell sorter (FACScan, USA).
Statistical analyses
Data represent at least the means of two independent experiments. The results are expressed as mean ± SD. All calculations were performed using the SPSS 15 for Windows (SPSS Inc., Chicago, Illinois, USA). One-way analysis of variance was used for multiple comparisons. A value of p < 0.05 was considered to be statistically significant.
Results
Effect of prodigiosin on morphology of cells
The effects of prodigiosin on morphology of cells were observed under the inverted microscope after 48-h treatment. As shown in Figure 2, prodigiosin changed morphology of HepG2 cells to apoptotic forms and disrupted cell connections. In this regard, this compound caused cell rounding that is characteristic of nonviable cells.

Effect of prodigiosin on morphology of cells. 5 × 105 HepG2 cells/well were treated with various concentrations of prodigiosin for 48 h, and morphology of cells were observed under the inverted microscope. Magnification: ×20, C: untreated cells.
Effect of prodigiosin on cell number
After 48-h treatment with prodigiosin, the cell number was obviously decreased in a dose-dependent manner. As shown in Figure 3, prodigiosin was found to diminish the cell numbers by 65–23% at concentrations ranging from 100-nM to 600-nM prodigiosin compared to untreated cells. Based on these data, we conclude that prodigiosin is a potent agent in decreasing the HepG2 cell number at the doses used.

Effect of prodigiosin on cell number. 5 × 105 HepG2 cells/well were treated with various concentrations of prodigiosin for 48 h, and the cell number was determined under the inverted microscope. *p < 0.05 versus untreated cells (C).
Effect of prodigiosin on metabolic activity
MTT assays were used to determine the effect of prodigiosin on the metabolic activity of HepG2 cells. We found that, treatment of the cells with increasing concentrations of prodigiosin diminished their metabolic activity in a dose- and time-dependent manner, specifically after a 72-h treatment compared to untreated cells (Figure 4).

Effect of prodigiosin on metabolic activity. 5 × 103 HepG2 cells/well were treated with different concentrations of prodigiosin for 24–72 h, after which their metabolic activities were measured by MTT assay. *p < 0.05; **p < 0.01 versus untreated cells (C).
Effect of prodigiosin on growth inhibition
Treatment of HepG2 cells with increasing concentrations of prodigiosin diminished growth rate in a dose- and time-dependent manner, specifically after a 72-h treatment compared to untreated cells (Table 1). After 72-h treatment, a significant reduction in IC50 value of prodigiosin was observed and measured to be 224.4 ± 29.6 nM (Table 2).
Evaluation of prodigiosin growth inhibition rates.a

a5 × 103 HepG2 cells/well were treated with various concentrations of prodigiosin, and growth inhibition rates were measured after 24–72 h.
bp < 0.01 versus untreated control cells.
cp < 0.05 versus untreated control cells.
Evaluation of total prodigiosin growth inhibition and IC50 values.a

IC50: concentration causing a 50% growth inhibition.
a5 × 103 HepG2 cells/well were treated with prodigiosin at concentrations ranging from 100 nM to 600 nM, and growth inhibition rates were measured after 24–72 h. The results represent mean ± SD.
Effect of prodigiosin on survivin mRNA level
The role of survivin in prodigiosin-induced apoptosis has never been investigated in HCC cells. Therefore, we set out to evaluate the effect of prodigiosin treatment on the expression of survivin. To this end, HepG2 cells were treated with different concentrations of prodigiosin. As shown in Figure 5, we found that after a 48-h treatment with prodigiosin, the high level of survivin mRNA in untreated HepG2 cells decreased significantly with increasing prodigiosin concentrations. Specifically, we found that treatment with 600-nM prodigiosin resulted in 88.5% decrease in survivin mRNA level compared to untreated cells.
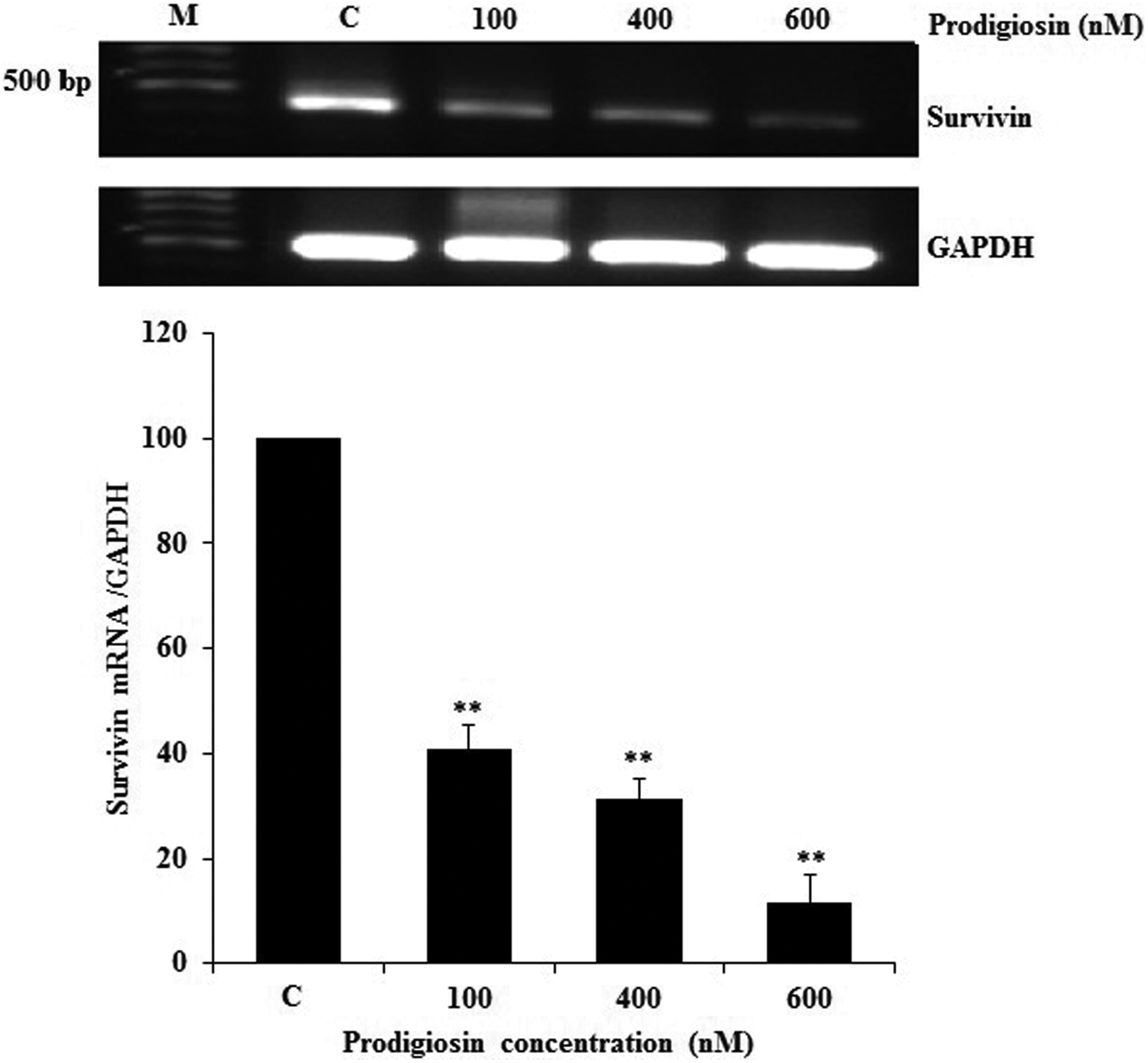
RT-PCR detection of survivin mRNA in treated and untreated tumor cells. 5 × 105 cells/well were treated with various concentrations of prodigiosin for 48 h, after which total RNAs were isolated from HepG2 cells and then subjected to a semiquantitative RT-PCR detection of survivin mRNA. RT-PCR with GAPDH-specific primers is performed to determine relative survivin mRNA levels. **p < 0.01 versus untreated cells (C).
Effect of prodigiosin on caspase-3 activation
To determine whether treatment of HepG2 cells with prodigiosin activates caspase-3 enzyme, we evaluated caspase-3 activation as a key executioner of apoptosis. As shown in Figure 6, we found that treatment with 600-nM prodigiosin resulted in 3.3-fold increase in caspase-3 activation as compared to untreated cells.

Effect of prodigiosin on caspase-3 activation. HepG2 cells were treated with various concentrations of prodigiosin for 48 h, and caspase-3 activation was measured using a fluorometric immunosorbent enzyme assay method. **p < 0.01 versus untreated control cells (C).
Effect of prodigiosin on apoptosis induction
To investigate whether the growth-inhibitory effect observed upon treatment of HepG2 cells with prodigiosin was due to the induction of apoptosis, the cells were treated with indicated concentrations of prodigiosin for 48 h, and subsequently stained with annexin V/PI and analyzed by means of flow cytometery. Treatment with different concentrations of prodigiosin induced an increase in the number of apoptotic cells (late apoptosis) ranging from 36.9% to 97.4% in a dose-dependent manner which suggests that the above observed decreases in cell number, cell proliferation, and metabolic activity may be attributed to the induction of apoptotic mechanism (Figure 7(a) and (b)).

Effect of prodigiosin on apoptosis induction. HepG2 cells were treated with various concentrations of prodigiosin for 48 h and stained with annexin V/propidium iodide and analyzed by means of flow cytometery. (Q1) Prenecrotic cells, (Q2) late apoptosis + necrosis, (Q3) living cells, and (Q4) early apoptosis cells. *p < 0.05 versus untreated control cells (C).
Associations between survivin expression, caspase-3 activation, and growth inhibition
In HepG2 cells, expression of survivin was downregulated by prodigiosin. We found that there is a significant association between survivin mRNA level and growth inhibition rate. After treatment with different concentrations of prodigiosin, the decreases in survivin mRNA levels were accompanied by increases in caspase-3 activation and growth inhibition compared to untreated cells. In this regard, a significant negative correlation was observed between survivin mRNA level and caspase-3 activation (r = −0.6). A significant negative correlation was also found between survivin mRNA level and growth inhibition (r = −0.94) whereas a significant positive correlation was found between caspase-3 activation and growth inhibition (r = 0.6).
Discussion
It has been shown that abnormal activation of the Wnt/β-catenin signaling pathway is essential in hepatocarcinogenesis. With this in mind, we evaluated the antitumor effects of prodigiosin in HepG2 cells that serve as an attractive model for HCC with abnormal activation of the Wnt/β-catenin signaling pathway in these cells. 18 The Wnt signaling pathway, via the β-catenin/Tcf4 complex, regulates the expression of multiple oncogenes, including survivin in HCC. 16 Overexpression of survivin contributes to determine the resistance of HCC cells to chemotherapy, tumorigenesis, and poor prognosis. 9 –12,17 Consequently, this would make survivin as an attractive candidate in cancer-directed therapy.
In our study, flow cytometric and caspase-3 activation assays showed that prodigiosin induces apoptosis in HepG2 cells. Interestingly, an increase in the number of necrotic cells was not observed with increasing prodigiosin concentrations, suggesting that this compound activates different molecular signaling pathways that are involved in apoptotic processes in HepG2 cells. In this regard, there was a significant negative correlation between survivin downregulation and apoptosis rate, which suggests that survivin expression may provide a novel biomarker and target in HCC cells.
Cancer cells are highly dependent on survivin to withstand the high basal levels of apoptotic stress. 19,20 Therefore, downregulation of survivin may partly explain the cytotoxicity of prodigiosin on hepatoma cells.
Our results indicate that prodigiosin decreases cell number, metabolic activity, and cell proliferation in a time- and dose-dependent manner, raising the possibility of its therapeutic use as an antitumor agent against HCC. In this regard, it has been shown that prodigiosin induces apoptosis in tumor cells in a p53 independent manner. This could mean an advantage of prodigiosin over other chemotherapeutics that need functional p53 to provoke its cytotoxic effect. 2 –6,21
Survivin expression is regulated by a complex intracellular signaling network involving the PI3K/Akt/mTOR, RAF/MAPK/ERK, NFκB, Wnt/β-catenin, and hypoxia-inducible factor-1α. 22 –24 Targeting more than one molecular and signaling pathway by a therapeutic agent may provide an attractive strategy for treatment of cancer. As survivin was found to be a direct downstream target of these pathways, 22 –24 it is possible that prodigiosin with vertical blockade of these signaling pathways prevents the upregulation of survivin. Therefore, the antitumor effects of prodigiosin may associate with inhibition of the abovementioned signaling pathways and downstream regulators, including survivin. Further experiments are needed to answer these issues.
In the present study, treatment of HepG2 cells with 600-nM prodigiosin decreased survivin mRNA level to 88.5% as compared to untreated cells, suggesting an interesting effect of prodigiosin turning the profile of high level of survivin expression in HepG2 cells into that of normal cells. Similar to our study, different researchers showed that prodigiosin dose dependently induced downregulation of survivin and triggered caspase activation in different kinds of cancer cell lines. 25,26
Prodigiosin is a DNA-interacting agent, which induces DNA single- and double-strand breaks via poisoning topoisomerases and through copper-promoted oxidative DNA damage. 27,28 Some compounds of the prodigiosin family have been reported to promote H+/Cl− symport transport and to induce neutralization of the acid compartments. 29 –31 However, the molecular mechanisms and targets responsible for these properties are not fully understood.
Our results showed the first evidence that survivin could be targeted by prodigiosin in HCC cells. In consequence, the potential of prodigiosin for inhibition of survivin may contribute to the development of more effective therapies for HCC and open up avenues to new therapeutic strategies for HCC-directed therapy.
Footnotes
Acknowledgements
The authors express their great appreciation for the financial support received for this work from the Urmia University, Iran.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
