Abstract
Formaldehyde (FA) has given positive results for the genetic damage evaluated in human peripheral blood lymphocytes. Polymorphism plays a special role in the toxicity of chemicals. DNA damage in blood cells was evaluated in workers who were occupationally exposed to FA. This study tested the association of alcohol dehydrogenase III (ADH3) and manganese-superoxide dismutase (Mn-SOD) gene polymorphism, with DNA damage of peripheral blood lymphocytes. Gene polymorphisms were evaluated in 54 workers from melamine dinnerware workshops and 34 workers from dairy production workshops. The control and exposed population were matched based on age, smoking, work history and socioeconomic status. DNA damage was evaluated by alkaline comet assay in the peripheral blood lymphocytes. ADH3 and Mn-SOD genotypes were determined in all workers using polymerase chain reaction–restriction fragment length polymorphism method. DNA damage of lymphocyte cells was significantly higher (p < 0.001) in the exposed individuals in comparison with the control population. The examinations of the exposed group had revealed significantly higher (p < 0.01) variant of Val/Val of Mn-SOD compared to control group. In workers with ADH32–2 variant, significantly higher levels of DNA damage (p < 0.01) were observed than the control population with the same genotype. It could be concluded that enzymes involved in the metabolism of FA and oxidative stress controlling are two important effective parameters for DNA damage of peripheral blood lymphocytes in exposed people. These results are regarded as a pioneer investigation for the management of health risks assessment.
Keywords
Introduction
Formaldehyde (FA) is one of the most commonly used chemicals today. 1 The National Cancer Institute studied more than 25,000 workers exposed to FA in their workplaces at 10 US plants. 2 They suggested an increased risk of lymphohematopoietic malignancies from FA exposure. Epidemiological evidence confirmed the risk of leukemia among workers exposed to FA. 3 Several mechanisms like cytotoxicity, genotoxicity, and oxidative stress have supported the carcinogenicity evidence of FA. 3,4 Some studies have expressed systemic effects of FA association with the generation of reactive oxygen species (ROS). 5,6 The genotoxicity of FA in blood cells has been reported, as seen in cases of micronucleus cells formation and DNA comet assay in occupational exposure. 7 The International Agency for Research on Cancer has classified FA as a group 1 carcinogen based on the potential causal relationship between FA exposure and hematopoietic cancers. 8
FA is metabolized by FA dehydrogenase enzymes when glutathione-dependent dehydrogenases are found in the respiratory tract. It is primarily biotransformed with alcohol dehydrogenase 5 (ADH5) to form S-hydroxymethylglutathione (SHMG). SHMG is converted to S-formylglutathione ADH3 and then further metabolized into formate. 9 It is known that polymorphisms of enzymes involved have a special role in the development of the chemical toxicity. For instance, individuals with a genetic polymorphism of a slow form of aldehyde dehydrogenase II are more sensitive to the irritant effects of acetaldehyde. 10 Several genes encoding FA metabolizing enzymes, among them ADH3 have a polymorphic gene that encodes the different forms of these isozymes. 11 ADH3 is an oxidative biotransforming enzyme incorporated in the metabolism of different chemicals including alcohol. Various studies have investigated the association between the carcinogenicity of ethanol and ADH3 polymorphism. 11 –14 Nevertheless, the relationship between the systemic effect of FA exposure and ADH3 polymorphism is less established.
Oxidative stress is an important mechanism of DNA impairment 4 and chemical toxicity. ROS generation in the body is controlled by different enzymes. Manganese-superoxide dismutase (Mn-SOD) as a mitochondrial enzyme eliminates free radicals production of cells. Genetic susceptibility is thought to be one of the strongest factors for ROS accumulation. 15 However, limited research has evaluated ADH3 and Mn-SOD polymorphisms in FA-exposed people.
In this study, we evaluated the DNA damage of peripheral blood lymphocytes in occupationally FA-exposed subjects from a number of melamine dinnerware-producing workshops. We investigated the relationship between genetic polymorphisms of ADH3 and Mn-SOD enzymes with DNA damage of peripheral blood lymphocytes to clarify the role of these genes in the carcinogenicity mechanisms of FA.
Materials and methods
Subjects
Dinnerware workshop is one of the finite jobs where workers are exposed to FA for a duration of 8 h in a day. Limited dinnerware workshops in this study are one of the limitations of our research. This study comprised 88 people: All of healthy workers (54 subjects) have at least 1 year of experience from 4 melamine dinnerware workshops and 34 workers were selected as a control group from dairy production workshops, who were not occupationally exposed to a hazardous agent. Exposed subjects within the age range of 28.41 ± 6.82 years old and work experience of 4.59 ± 6.13 years (Table 1). Exposed subjects were matched with control group by considering smoking habit (p < 0.61) and alcohol consumption (p < 0.44). Nonetheless, workers under medication were excluded from the study.
Personal information in studied subjects.

All individuals filled in a questionnaire, including their personal information, use of medications and history of occupation. Based on the questionnaire, the control and exposed population were matched in socioeconomic status and history of cigarette smoking. A volume of 3 ml of blood sample was obtained into tubes containing Ethylenediaminetetraacetic acid (EDTA) as the anticoagulant from the selected subjects. Shahid Beheshti University of Medical Sciences Institutional Review Board approved all the study procedures (number 6412).
Assessment of occupational exposure to FA
Exposure monitoring of workers from dinnerware workshops was performed according to the National Institute for Occupational Safety and Health method no. 3500. 16 Individual sampling was performed with personal pump SKC (Model 224-PC EX8,Blandford Forum, Dorset, England) with a flow of 400 ml min−1 for a period of 240 min using an Impinger with sodium bisulfate 1%. FA of air samples was determined by visible spectrophotometer (model CE 2021, Cecil, Cambridge, England) at 580 nm. The reagent contained chromotropic acid and concentrated sulfuric acids.
Genetic damage induction of FA is related to continuous exposure in the inhalation intake. In this study, the work experience in the exposed group was 4.6 ± 6.1 years. Then, subjects were subchronically 17 exposed to FA.
Subchronic inhalation intake was evaluated by average daily intake over the experience history period according to the following equation 18 :

where CII is chronic inhalation intake (ppm), C is concentration of FA in the personal air sample (ppm), ET is exposure time (8 h/day), EF is exposure frequency (240 days/year), ED is experience history (years), and AT is average lifetime (70 years).
Genotyping of enzymes gene polymorphisms
The ADH3 and Mn-SOD genotypes were determined in all the subjects. An amount of 100-μl whole blood was added in a micro tube containing 7% EDTA and then DNA was extracted using an extraction kit (CinnagGen, Iran) according to the manufacturer’s instruction. Heritability polymorphism of two enzyme genes was performed by polymerase chain reaction (PCR)–restriction fragment length polymorphism method on the extracted DNA of white cells.
The primers for amplification of genes and restriction enzymes of reactions are shown in Table 2. All samples were subjected to electrophoresis in 2.5% agarose gel, stained with ethidium bromide. 15,19 PCR multiplex for ADH3 and Mn-SOD was performed by 0.5 μl of each primer, 6-μl distilled water, 3-μl DNA template, and 10-μl Master Mix kit (CinnagGen) including Taq polymerase enzyme, magnesium chloride, deoxynucleotide, ammonium sulfate, Tris hydrochloride (HCl), and Tween-20. Cycling conditions for ADH3 were primary denaturation 95°C for 5 min, denaturation 94°C for 30 s, annealing 55°C for 30 s, extension 72°C for 30 s, and then followed by 40 cycles of a final extension 72°C for 10 min. Mn-SOD cycling conditions were primary denaturation 95°C for 5 min, denaturation 92°C for 30 s, annealing 60°C for 30 s, extension 72°C for 1 min, and then followed by 38 cycles of a final extension 72°C for 5 min.
The primers and restriction enzymes used for genotyping of ADH3 and Mn-SOD.

ADH3: alcohol dehydrogenase III; Mn-SOD: manganese-superoxide dismutase.
Comet assay
For the assessment of DNA damage, alkaline version of comet test (pH > 13) was applied in the peripheral blood lymphocytes. DNA strand breaks or comet assay is a reliable technique for the monitoring of DNA damage. 20,21 Slides were coated with 1% normal melting point agarose and kept for 10 min to solidify. An amount of 75-μl low-melting agarose (0.5%) was mixed with 10 μl of blood samples and then added to the slides. Cells kept overnight at 4°C in lysis buffer (10 mM Tris-HCl, pH 10, 2.5 M sodium chloride, 100 mM EDTA, 5% DMSO, 1% Triton X-100). Finally, slides were placed in alkaline buffer (300 mM sodium hydroxide, 1 mM EDTA, pH 13) for 30 min at 4°C. Electrophoresis was performed for 30 min at 300 mA and 23 V. The slides were washed with neutralizing buffer (0.4 MTris, pH 7.5) and stained with 2 μg ml−1 of ethidium bromide. DNA breaks were performed by a fluorescence Olympus microscope (ECLIPSE 80i microscope with WG filter, Nikon, Tokyo, Japan). The image of DNA strand breaks was evaluated by open comet software, version 1.3. At least, 50 nuclei were calculated for each sample. Tail moment, tail intensity, and tail migration were calculated as DNA damage. The results of tail intensity and tail moment are the same as comet tail lengths (data not shown).
The tail length of the comet was reported as genetic damage of blood cells for all of the workers.
Statistical analysis
The results were analyzed using SPSS statistical analysis software (version 16). Normality of all parameters was tested according to Kolmogorov–Smirnov test. The variation between the exposed and controls groups was evaluated with two samples of student t-tests in normal distribution and Mann-Whitney U test in abnormal distribution of samples. Quantitative values in abnormal distribution of samples were reported as median (min–max). The level of statistical significance was set at p value of 0.05.
Results
The distribution of data for occupational exposure to FA was abnormal. Airborne exposure concentration for FA in the exposed group ranged from 0.033 ppm to 0.14 ppm, with a median of 0.08 ppm. The median of subchronic inhalation intake was 4.73 ppm with a minimum and maximum value of 0.17 and 84.48 ppm, respectively.
Highly significant (p < 0.001) genetic damage was observed in peripheral blood lymphocytes of exposed workers compared to control. The median value of comet tail lengths of cells in exposed workers was 28.9 with a minimum of 13.9 and maximum of 81. For the control population, the median (min–max) of DNA tail lengths was 18.5 (14–71; Figure 1).

Comet assay in studied people.
A significant spearman correlation was observed (Figure 2) between a subchronic intake inhalation of FA exposure and DNA tail lengths (r = 0.4; p < 0.001).

Correlation between a subchronic intake inhalation and FA exposure. FA: formaldehyde.
Three variants of ADH3 and Mn-SOD genotypes were determined in the FA-exposed and control workers. A PCR analysis of ADH3 and Mn-SOD polymorphisms in some subjects is presented in Figure 3.

PCR analysis of ADH3 and Mn-SOD gene polymorphism. (a) Two fragments of 15 and 130 bp for ADH32–2 genotype (lane 1); three fragments of 67, 63, and 15 bp for ADH31–1 genotype (lanes 2 and 3); and four fragments of 15, 63, 67, and 130 bp for ADH31–2 genotype (lanes 4, 5, and 6). (b) One fragment of 207 bp for Ala/Ala genotype (lane 1); two fragments of 167 and 40 bp for Val/Val genotype (lane 3); and three fragments of 207, 167, and 40 bp for Ala/Val genotype (lane 2). PCR: polymerase chain reaction; ADH3: alcohol dehydrogenase III; Mn-SOD: manganese-superoxide dismutase.
Alleles distributions of Mn-SOD (V/A) and ADH3 (ADH×1/ADH×2) were in accordance with the Hardy–Weinberg equilibrium for subjects. Allele frequency of ADH3 genotype ((ADH×1/ADH×2), 0.379/0.621 vs. (ADH×1/ADH×2), 0.40/0.60; p = 0.37) and Mn-SOD genotype ((V/A), 0.518/0.482 vs. (V/A), 0.456/0.544; p = 0.42) in the FA-exposed group were not significantly different from the control subjects. Table 3 shows the genotype distribution of ADH3 and Mn-SOD polymorphisms between the exposed workers and the control group. As shown in Table 3, the frequency of genotype distribution of different polymorphisms was not significantly different between the groups.
Genotype frequency of ADH3 and Mn-SOD polymorphisms.

ADH3: alcohol dehydrogenase III; Mn-SOD: manganese-superoxide dismutase.
Table 4 presents the results of comet tail lengths of peripheral blood lymphocytes in the exposed groups between different genotype variants of ADH3 and Mn-SOD polymorphisms.
In the FA-exposed group, DNA damage was significantly higher in ADH32–2 and Val/Val variant than the control group. To concentrate the difference in genotype between the exposed group and the control group, a threshold limit value for DNA comet tail length of blood cells was further performed. The 90th percentile comet tail length of control groups was defined as a threshold limit, while individuals with levels higher than the threshold limit were considered as affected samples. Distribution of comet tail lengths for subjects with ADH32–2 genotype was normal. The results reveal that comet tail lengths in affected exposed subjects with ADH32–2 genotype are significantly (Table 4) higher than the affected individuals of control groups.
DNA damage in different genotype variants.
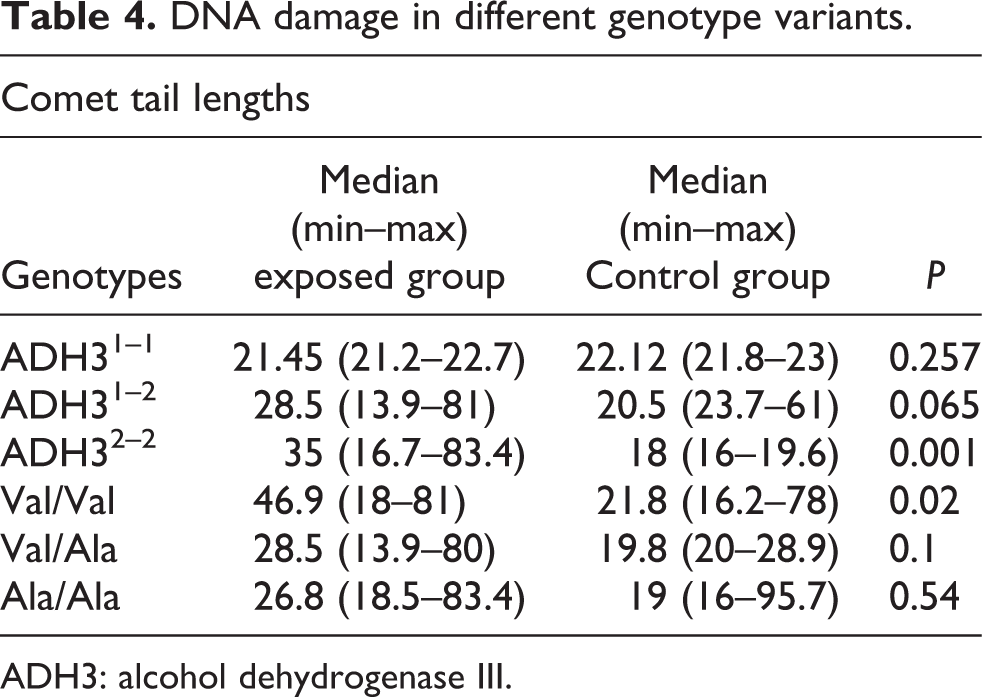
ADH3: alcohol dehydrogenase III.
Exposed workers with ADH32–2 genotype contained significantly higher levels of DNA damage (p = 0.01) than the control population with the same polymorphism (Figure 4). The mean ± standard deviation values in exposed workers and control group with ADH32–2 genotype were 38.9 ± 1.74 and 18.82 ± 0.08, respectively.

Comet assay in studied people with ADH32–2 genotype. ADH3: alcohol dehydrogenase III.
Discussion
The predicting of chemical response is an important key for risk management and prevention activity. The toxicity of chemicals depends on the individual response besides dose–effect relationship. Among different types of parameters, isoenzyme variant and genetic polymorphism are one of the strongest reasons among different types of toxicities. Herein, we analyzed the ADH3 and Mn-SOD heritability polymorphisms in FA-exposed workers and non-exposed workers and then evaluated the association between these polymorphisms with DNA damage of peripheral blood lymphocytes. The results of individual monitoring for melamine dinnerware-producing workers indicated that all exposures to FA were higher than the time-weighted average of 0.016 ppm set by National Institute for Occupational Safety and Health. Exposure to this concentration caused significant DNA damage in blood cells compared to the control group, which correlate to a subchronic inhalation intake of FA.
Oxidative stress is one of the important mechanisms of chemical toxicity. 6,22,23 Mn-SOD enzyme is one of the mitochondrial enzymes which diminishes ROS to stable components by eliminating the antioxidant capacity or by changing its shape. 5 In this study, it was found that Mn-SOD polymorphism was associated with an increased risk of DNA damage of blood cells. Different variants of Mn-SOD present various capacities of antioxidant activity 24 and can induce synergistic effects of oxidative damage in FA exposure. In our experiments, Val/Val variant of Mn-SOD in exposed subjects diminishes the comet tail length of DNA than other genotypes. These results reveal that this polymorphism reduces the scavenging of reactive oxygen radicals, subsequently leading to oxidative stress generation. Non-assessment of the oxidative stress in the studied occupationally exposed subjects is a limitation of this study. Some other studies reported increased risk of disease in Val/Val variant of Mn-SOD genotype. Petrovič et al. studied diabetic retinopathy disorder which was associated with type 2 diabetes. They introduced Val/Val polymorphism of Mn-SOD as a genetic marker for high-risk diabetic retinopathy in Caucasian populations. 25
There is some epidemiologic evidence of carcinogenicity for FA-exposed people. 8 The National Research Council in their National Toxicology Program of the 12th Report on Carcinogens 3 emphasize that the FA mechanisms of inducing cancer are not completely understood. They concluded that only few studies have addressed the details of oxidative stress for FA exposure. Our findings support the hypothesis that oxidative stress is one of the important mechanisms of FA toxicity in blood cells 22,23 and genetic variations in antioxidant enzymes may modify the risk of DNA. Several studies have evaluated ADH3 polymorphism as a potential modifier of alcohol-related cancer. Polymorphisms of ADH3 have been associated with increased risk of breast cancer, 26 esophageal and gastric adenocarcinomas, 13 and cirrhosis and chronic pancreatitis. 19 There are few studies that have evaluated FA toxicity and ADH3 polymorphism in the exposed population. In this study, we found significant relations between DNA damage of peripheral blood lymphocytes and ADH3 genotype variant.
Subjects with ADH3,1–1 ADH3,1–2 and ADH32–2 polymorphisms are classified as fast, intermediate, and slow metabolizers. We found a higher comet tail length in the peripheral blood lymphocytes of exposed individuals than heterozygote people. Thus, slow metabolizers are susceptible to DNA damage. ADH3 as a glutathione-dependent enzyme reduces FA toxicity by forming S-formyl glutathione. 27 Oxidation of FA has been reduced in ADH32–2 genotype variant, so FA toxicity was increased in slow metabolizer subjects. A percentage of 26.2% of studied subjects with ADH32–2 genotype were susceptible to DNA damage of FA exposure. In this study, the frequency of ADH3×2 allele as high-risk allele was 60% where it is similar to the frequency previously reported in other populations: 42% of European Whites, 10% of Asians, and 12% of Africans. 19,26 Sample size of occupationally exposed subjects is a limitation of this study. Design of study for evaluating DNA damage susceptibility in a larger population for other exposed employees is recommended.
In conclusion, our study presented that FA metabolism and oxidative stress are two important effective factors for DNA damage in the blood cells of exposed people. The ADH32–2 and Val/Val of Mn-SOD genotype are the risk factors for developing genetic impairment in FA-exposed people. These results present important information for risk management of occupational exposure. Our findings could be regarded as a pioneer investigation for the assessment of health risks in the recruitment tests.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
