Abstract
This study aimed to examine the effects of lipid emulsion on the vasodilation and cardiovascular depression induced by toxic doses of calcium channel blockers. The effects of lipid emulsion on the vasodilation induced by bepridil, verapamil, nifedipine, and diltiazem were investigated in isolated endothelium-denuded rat aortae. The effect of lipid emulsion on the comparable hemodynamic depression induced by the continuous infusion of a toxic dose of either verapamil or diltiazem was examined in an in vivo rat model. The results showed the following decreasing order for the magnitude of lipid emulsion-mediated inhibition of vasodilation: bepridil, verapamil, nifedipine, and diltiazem. Lipid emulsion (0.5–2%) reversed the vasodilation induced by a toxic dose of verapamil, whereas only a higher concentration (2%) reversed the vasodilation induced by a toxic dose of diltiazem. Pretreatment with lipid emulsion alleviated the systolic and mean blood pressure decreases induced by a toxic dose of verapamil, whereas it had no effect on the decrease induced by diltiazem. Taken together, these results suggest that lipid emulsion alleviates the severe vasodilation and systolic blood pressure decrease induced by a toxic dose of verapamil, and this alleviation appears to be associated with the relatively high lipid solubility of verapamil.
Introduction
Lipid emulsion has been used to treat systemic toxicity caused by local anesthetics and other drugs without specific antidotes. 1 –3 Calcium channel blockers including dihydropyridine and nondihydropyridine have been used to treat hypertension, angina, and arrhythmias. 4 However, the toxicity of calcium channel blockers causes serious hemodynamic depression, which leads to life-threatening complications. It has been reported that several treatments including high-dose insulin, extracorporeal life support, calcium, 4-aminopyridine, and lipid emulsion support the cardiovascular collapse induced by a toxic dose of calcium channel blockers. 5 According to a systemic review regarding poisoning by calcium channel blockers, lipid emulsion treatment, in particular, is more helpful for alleviating systemic toxicity induced by verapamil than for alleviating that induced by other calcium channel blockers. 2,5 Consistent with the lipid sink theory, which is widely accepted as one of the mechanisms relevant to lipid emulsion treatment, lipid emulsion has been reported to be more effective at attenuating the reduction in the cell viability of H9c2 rat cardiomyoblasts induced by a toxic dose of verapamil than that induced by a toxic dose of diltiazem. 6 In agreement with the lipid sink theory, which posits that lipid emulsion removes more lipid-soluble drugs from tissues more effectively than less lipid-soluble drugs, it has been reported that lipid emulsion more strongly attenuates and reverses the severe vasodilation evoked by a toxic dose of bupivacaine or levobupivacaine, which are more lipid soluble than that evoked by toxic doses of other, less lipid-soluble local anesthetics. 1,7 –10 The reported magnitude of the lipid solubility of calcium channel blockers, which was determined using oil/Ringer and octanol/Ringer, is in the following decreasing order: bepridil, verapamil, nifedipine, and diltiazem. 11 In addition, the serum concentration of verapamil during verapamil toxicity has been reported to be reduced by lipid emulsion. 12 However, the effects of lipid emulsion on both the vasodilation and comparable hemodynamic depression induced by toxic doses of calcium channel blockers remain unknown. Thus, the first goal of this study was to examine the effects of lipid emulsion on the vasodilation evoked by calcium channel blockers, including bepridil, verapamil, nifedipine, and diltiazem, in isolated rat aortae. Second, we compared the effects of lipid emulsion on the comparable hemodynamic depression induced by a toxic dose of verapamil or diltiazem in an in vivo rat model. Based on previous reports, we tested the hypothesis that lipid emulsion would attenuate calcium channel blocker-induced vasodilation in a lipid solubility–dependent manner and would more markedly alleviate the comparable hemodynamic depression induced by a toxic dose of verapamil than that induced by a toxic dose of diltiazem because the magnitude of lipid solubility of verapamil is higher than that of diltiazem. 1,5,11
Methods
All the experimental protocols were approved by the Institutional Animal Care and Use Committee of Gyeongsang National University. All the experimental procedures were performed according to the Guide for the Care and Use of Laboratory Animals approved by the National Academy of Sciences.
Preparation of isolated rat aortae and isometric tension measurements
The preparation of isolated aortic rings for tension measurements was performed as previously described in our laboratory.
8,9
Male Sprague-Dawley rats weighing between 250 g and 300 g were anesthetized by inhalation of 100% carbon dioxide. The descending thoracic aorta was separated from the surrounding fat and connective tissue under a microscope in a Krebs solution bath composed of 118 mM sodium chloride, 25 mM sodium bicarbonate, 11 mM glucose, 4.7 mM potassium chloride (KCl), 2.4 mM calcium chloride, 1.2 mM magnesium sulfate, and 1.2 mM monopotassium phosphate. The endothelium was removed from the isolated aorta by inserting a 25-gauge needle tip into the lumen of the isolated aorta and then gently rolling the isolated aorta for a few seconds. The aorta was suspended in a Grass isometric transducer (FT-03, Grass Instrument, Quincy, Massachusetts, USA). A resting tension of 3.0 g was maintained for 120 min, and the Krebs solution was exchanged with fresh Krebs solution every 30 min. After phenylephrine (10−8 M) had produced a sustained and stable contraction, endothelial denudation was verified by the observation that acetylcholine induced <15% relaxation of the phenylephrine-evoked contraction. After the Krebs solution containing phenylephrine was exchanged with fresh Krebs solution several times, the baseline resting tension was restored. Each isolated aortic ring was used to produce only one dose–response curve to characterize the calcium channel blockers or lipid emulsion. Residual endothelium remaining after endothelial denudation can affect the lipid emulsion-mediated inhibition and reversal of calcium channel blocker-induced vasodilation because the lipid emulsion itself attenuates nitric oxide-induced relaxation.
13
In addition, according to previous reports, among the calcium channel blockers, nifedipine and diltiazem increase the production of endothelial nitric oxide, whereas verapamil has no effect on its production.
14
–16
Thus, to avoid the compounding factors caused by the different effects of each calcium channel blocker (verapamil, nifedipine, and diltiazem) on the production of endothelial nitric oxide, which may lead to additive effects on the vasodilation induced by nifedipine and diltiazem, we used the endothelium-denuded aorta instead of the endothelium-intact aorta in the current study. Considering the findings of previous reports, the following experimental protocols were performed using endothelium-denuded rat aortae pretreated with the nitric oxide synthase inhibitor NW-nitro-
Experimental protocols
First, we investigated the effects of lipid emulsion on the vasodilation induced by calcium channel blockers (bepridil, verapamil, nifedipine, and diltiazem) in isolated endothelium-denuded rat aortae. Endothelium-denuded rat aortae pretreated with
Second, we examined the effect of lipid emulsion posttreatment on the vasodilation induced by a toxic dose of verapamil or diltiazem in isolated endothelium-denuded rat aortae precontracted with 60 mM KCl. After 60 mM KCl had produced a stable and sustained contraction in isolated endothelium-denuded rat aortae pretreated with
Animal and experimental preparation for the in vivo experiment
Male Sprague-Dawley rats (KOATECH, Pyeongtaek, South Korea) weighing 200–250 g were used in the in vivo study. The animals received general anesthesia with an intramuscular injection of 15 mg/kg tiletamine/zolazepam (Zoletil50®; Virbac Lab., Carros, France) and 9 mg/kg xylazine (Rompun®; Bayer, Seoul, South Korea). If tail movement (a sign of awakening) was observed during the operation, an additional injection of 5 mg/kg tiletamine/zolazepam and 3 mg/kg xylazine was used to maintain a constant level of anesthesia. The animals maintained spontaneous breathing without muscle relaxation during the experiment. Body temperature was monitored with a rectal thermometer (Sirecust 1260; Siemens Medical Electronics, Danvers, Massachusetts, USA) and was maintained at 36–38°C using an electrical heating pad. The right carotid artery was exposed after anesthesia for insertion of the catheter. Then, a 24-guage catheter (BD Insyte Autoguard; Becton-Dickinson Co., Franklin Lakes, New Jersey, USA) treated with heparin was inserted through the exposed right carotid artery for hemodynamic monitoring. A pressure transducer (Model ML 118; ADInstruments Pty Ltd, Bella Vista, Australia) was connected to a digital analysis system to measure hemodynamic variables. The hemodynamic variables, including systolic blood pressure, diastolic blood pressure, mean blood pressure, and heart rate, were measured using a computer analysis program (ChartTM5 Pro; ADInstruments Pty Ltd). Then, 24-guage catheters treated with heparin were inserted into the right and left femoral veins to infuse the pretreatment drugs (lipid emulsion or normal saline) and calcium channel blockers (verapamil or diltiazem), respectively. Each experimental group was injected with the drugs via the catheter according to the protocols below.
Experimental protocols
Two sets of experiments were performed to investigate the effects of lipid emulsion in the verapamil and diltiazem toxicity models. In total, 28 rats were randomly assigned to one of the four groups. In each set of experiments, 14 rats were divided into two groups and received a continuous infusion of normal saline or lipid emulsion. Before the administration of the pretreatment drug (normal saline or lipid emulsion), baseline hemodynamic variables, including systolic, diastolic and mean blood pressure, and heart rate, were continuously measured after a 10 min stabilization period after catheter insertion in the in vivo rat model. Either normal saline or lipid emulsion at a dose of 3 ml/kg was infused into the right femoral vein over 15–20 s. Then, normal saline or lipid emulsion at a dose of 0.17 ml/kg/min was continuously infused with an infusion pump (Model AS50, infusion pump; Baxter Healthcare Corp., Deerfield, Illinois, USA) during the entire period of the experiment. After 5 min of the continuous infusion of normal saline or lipid emulsion, verapamil or diltiazem was continuously administered. In the first set of experiments, 10 mg/kg/h of verapamil was continuously administered to two groups (normal saline or lipid emulsion) that had already received a bolus administration of normal saline or lipid emulsion, followed by the continuous infusion of normal saline or lipid emulsion into the left femoral vein using a syringe pump (Model AS50), and the hemodynamic variables were continuously measured. In the second set of experiments, 100 mg/kg/h of diltiazem was continuously administered to each group, which had already received a bolus administration of normal saline or lipid emulsion, followed by the continuous infusion of normal saline or lipid emulsion into the left femoral vein using a syringe pump, and the hemodynamic variables were continuously measured.
Materials
All the chemicals used in this study were of the highest purity commercially available. Bepridil, verapamil, nifedipine, diltiazem, phenylephrine, and
Data analysis
The data are expressed as the means ± standard deviation (SD). The vascular responses (vasodilation and vasoconstriction) produced by the calcium channel blocker or lipid emulsion were expressed as the percentage of the maximal contraction induced by phenylephrine or isotonic 60 mM KCl. The hemodynamic variables, including systolic, mean and diastolic blood pressure, and heart rate, induced by the pretreatment drug (lipid emulsion or normal saline) alone or followed by verapamil or diltiazem were expressed as a percentage of the baseline absolute hemodynamic variables, which were measured before the administration of lipid emulsion or normal saline. A two-way repeated-measures analysis of variance (ANOVA; Prism 5.0, GraphPad Software, San Diego, California, USA) was used to analyze the effects of lipid emulsion on calcium channel blocker-induced vasodilation in isolated endothelium-denuded rat aortae precontracted with phenylephrine or 60 mM KCl. In addition, the effects of lipid emulsion and normal saline on the hemodynamic variables evoked by continuous infusion of verapamil or diltiazem in an in vivo rat model were analyzed using two-way repeated-measures ANOVA. If the two-way repeated-measures ANOVA revealed a p < 0.05, Bonferroni’s post hoc test for multiple comparisons was performed. The statistical analysis of the baseline absolute hemodynamic variables of the normal saline and lipid emulsion groups was performed using an unpaired Student’s t-test. The change in heart rate evoked by the continuous infusion of verapamil or diltiazem within each group was analyzed using repeated-measures ANOVA followed by Bonferroni’s post hoc test. The logarithm (log ED50) of the concentration of calcium channel blocker that produced 50% maximal vasodilation was calculated using sigmoidal dose–response curves (Prism 5.0, GraphPad Software). The effects of lipid emulsion on the log ED50 of the vasodilation induced by bepridil, verapamil, nifedipine, and diltiazem were analyzed using one-way ANOVA followed by Bonferroni’s post hoc test. A comparison of the log ED50 values of the calcium channel blocker-induced vasodilation in isolated endothelium-denuded rat aortae without lipid emulsion was performed using one-way ANOVA followed by Tukey’s multiple comparison tests. p Values less than 0.05 were considered statistically significant.
Results
Lipid emulsion (0.25% and 1%) attenuated the bepridil-induced maximal vasodilation (p < 0.01 vs. control at 3 × 10−5 M bepridil; Figure 1(a)). Lipid emulsion attenuated the verapamil-induced vasodilation in a concentration-dependent manner (log ED50; 0.25% and 1% lipid emulsion: p < 0.05 vs. control; 1% lipid emulsion: p < 0.05 vs. 0.25% lipid emulsion; Figure (b)). Lipid emulsion (1%) attenuated the nifedipine-induced vasodilation (log ED50; 1% lipid emulsion: p < 0.01 vs. control; Figure 1(c)). Lipid emulsion did not significantly alter the diltiazem-induced vasodilation (Figure 1(d)). Lipid emulsion (0.5%, 1%, and 2%) reversed the verapamil (3 × 10−7 M)-induced vasodilation (p < 0.05 vs. time-matched control without lipid emulsion at 20–70 min; Figure 2(a)) of the 60 mM KCl-induced vasoconstriction. However, only a high concentration (2%) of lipid emulsion reversed the diltiazem (3 × 10−6 M)-induced vasodilation of the 60 mM KCl-induced vasoconstriction (p < 0.05 vs. time-matched control without lipid emulsion at 40–70 min; Figure 2(b)). The potency of the log ED50 of the calcium channel blocker-induced vasodilation was in the following decreasing order: nifedipine, verapamil, diltiazem, and bepridil (Table 1).
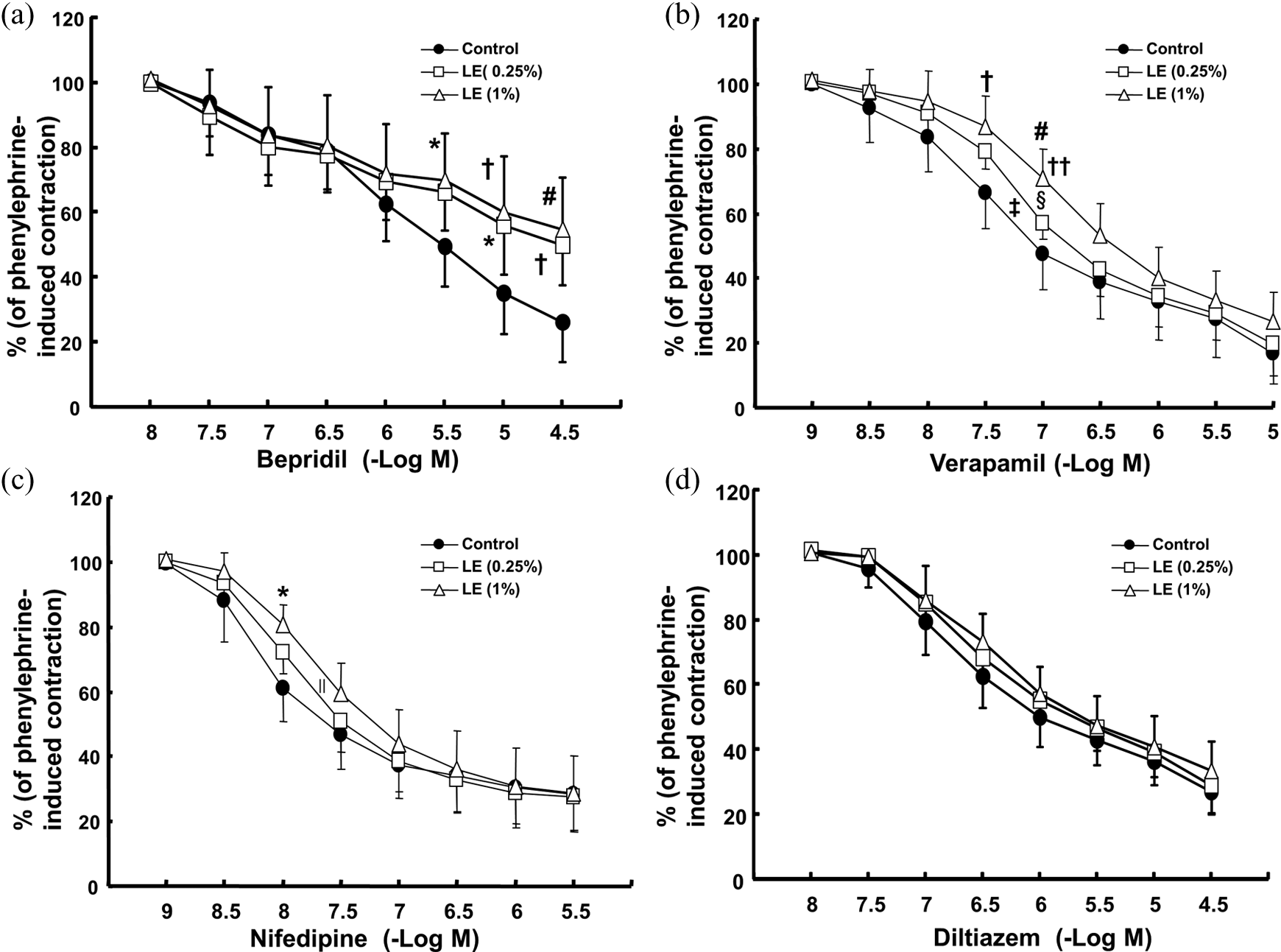
Effects of LE on the dose–response curves of cumulatively added calcium channel blockers (bepridil (a; N = 7), verapamil (b; N = 5), nifedipine (c; N = 6), and diltiazem (d; N = 5)) in isolated endothelium-denuded rat aortae precontracted with phenylephrine (10−7 M). Data are shown as the mean ± SD and are expressed as the percentage of phenylephrine-induced maximal contraction. N indicates the number of rats from which the descending thoracic aortae were derived. *p<0.05, †p<0.01, and #p<0.001 versus control. Log ED50: the logarithm of the concentration of calcium channel blocker producing 50% of the calcium channel blocker-induced maximal vasodilation. Log ED50: ‡p<0.05, ∥p<0.01, and §p<0.001 versus control; ††p<0.05 versus 0.25% LE. LE: lipid emulsion; SD: standard deviation.

Effects of LE (0.5%, 1%, and 2%) on the vasodilation induced by verapamil (3 × 10−7 M; a) and diltiazem (3 × 10−6 M; b) in isolated endothelium-denuded rat aortae precontracted with 60 mM KCl. Data are shown as the mean ± SD (N = 5) and are expressed as the percentage of 60 mM KCl-induced maximal contraction. N indicates the number of rats from which the descending thoracic aortae were derived. *p<0.05, †p<0.01, and #p<0.001 versus time-matched controls without LE. LE: lipid emulsion; SD: standard deviation; KCl: potassium chloride.
Vasodilation induced by calcium channel blockers in isolated endothelium-denuded rat aortae precontracted with phenylephrine without lipid emulsion.a

SD: standard deviation; Log ED50: the logarithm of the concentration of the calcium channel blocker that produced 50% maximal vasodilation; N: the number of rats.
aValues are the means ± SD.
b p < 0.001 versus nifedipine.
c p < 0.001 versus verapamil.
d p < 0.001 versus diltiazem.
The baseline absolute hemodynamic variables, including blood pressure and heart rate, before the administration of normal saline alone or lipid emulsion alone were not significantly different between the two groups of each experiment involving continuous infusion of verapamil or diltiazem (Tables 2 and 3). Lipid emulsion alone increased the systolic blood pressure before the administration of verapamil or diltiazem (p < 0.05 vs. normal saline; Figures 3(a) and 4(a)). Lipid emulsion attenuated the systolic and mean blood pressure decreases induced by the continuous infusion of verapamil (systolic blood pressure: p < 0.01 vs. normal saline at 2–7 min; mean blood pressure: p < 0.05 vs. normal saline at 1–7 min; Figure 3(a) and (b)). However, lipid emulsion had no effect on the systolic or mean blood pressure decreases induced by continuous infusion of diltiazem (Figure 4(a) and (b)). Lipid emulsion inhibited the diastolic blood pressure decrease only at 1 min after the infusion of verapamil (p < 0.05 vs. normal saline; Figure 3(c)), whereas lipid emulsion had no effect on the diastolic blood pressure decrease induced by the continuous infusion of diltiazem (Figure 4(c)). In addition, lipid emulsion did not significantly alter the heart rate decrease compared with normal saline during the continuous infusion of verapamil or diltiazem (Figures 3(d) and 4(d)). Verapamil decreased the heart rate in the normal saline and lipid emulsion groups (normal saline: p < 0.01 vs. baseline heart rate at 3–7 min; lipid emulsion: p < 0.05 vs. baseline heart rate at 4–7 min; Figure 3(d)). Diltiazem decreased the heart rate during the continuous infusion of lipid emulsion or normal saline (p < 0.001 vs. baseline heart rate at 2–7 min; Figure 4(d)).
Baseline blood pressure before the administration of normal saline or lipid emulsion in each group in an in vivo rat model of verapamil toxicity.a

SD: standard deviation; N: the number of rats.
aValues are the mean ± SD.
Baseline blood pressure before the administration of normal saline or lipid emulsion in each group in an in vivo rat model of diltiazem toxicity.a

SD: standard deviation; N: the number of rats.
aValues are the mean ± SD.

Effects of LE (3 ml/kg followed by 0.17 ml/kg/min) and NS (3 ml/kg followed by 0.17 ml/kg/min) on changes in SBP (a), MBP (b), DBP (c), and HR (d) induced by the continuous infusion of VRP (10 mg/kg/h) in an in vivo rat model. Data are shown as the mean ± SD (N = 7) and are expressed as the percentage of the baseline SBP, MBP, DBP, and HR. N indicates the number of rats. (a) to (c) *p<0.05, †p<0.01, and #p<0.001 versus NS. (d) #p<0.05, †p<0.01, and *p<0.001 versus the baseline HR within either the NS or LE group. LE: lipid emulsion; NS: normal saline; SBP: systolic blood pressure; MBP: mean blood pressure; DBP: diastolic blood pressure; HR: heart rate; VRP: verapamil; SD: standard deviation.

Effects of LE (3 ml/kg followed by 0.17 ml/kg/min) and NS (3 ml/kg followed by 0.17 ml/kg/min) on changes in SBP (a), MBP (b), DBP (c), and HR (d) induced by the continuous infusion of DTZ (100 mg/kg/h) in an in vivo rat model. Data are shown as the mean ± SD (N = 7) and are expressed as the percentage of the baseline SBP, MBP, DBP, and HR. N indicates the number of rats. (a) *p<0.01 versus NS. (d) †p<0.001 versus the baseline HR within either the NS or LE group. LE: lipid emulsion; NS: normal saline; SBP: systolic blood pressure; MBP: mean blood pressure; DBP: diastolic blood pressure; HR: heart rate; DTZ: diltiazem; SD: standard deviation.
Discussion
This is the first report to suggest that lipid emulsion alleviates the vasodilation and systolic blood pressure decrease induced by a toxic dose of verapamil; this alleviation appears to be associated with the relatively high lipid solubility of verapamil. The major findings of this study are as follows: (1) lipid emulsion inhibited verapamil-induced vasodilation in a concentration-dependent manner, whereas it had no effect on diltiazem-induced vasodilation; (2) lipid emulsion reversed verapamil-induced vasodilation in a concentration-dependent manner, whereas only the highest concentration of lipid emulsion slightly reversed diltiazem-induced vasodilation; and (3) lipid emulsion alleviated the systolic and mean blood pressure decreases induced by a toxic dose of verapamil, whereas it had no effect on the systolic or mean blood pressure decreases evoked by a toxic dose of diltiazem.
The reported uptake of calcium channel blockers into rat aortic vascular smooth muscle cells is in the following increasing order: diltiazem = nifedipine < verapamil < nitrendipine < bepridil. 11 In addition, the reported uptake of calcium channel blockers into cardiac and smooth muscle is in the following increasing order: diltiazem, nifedipine, verapamil, and bepridil. 17 In consideration of these previous reports, the magnitude of the uptake of calcium channel blockers into rat aortic vascular smooth and cardiac muscle appears to be approximately correlated with the lipid solubility of the calcium channel blockers. 11,17 The vasodilation induced by a toxic dose of bupivacaine or levobupivacaine has been reported to be reduced by pretreatment with lipid emulsion, and the potency of this reduction appears to be correlated with the relatively high lipid solubility of bupivacaine, supporting the lipid sink theory. 1,9,10 In the current study, the pretreated lipid emulsion-induced inhibition of vasodilation evoked by a toxic dose of calcium channel blockers was in the following decreasing order of potency: bepridil, verapamil, nifedipine, and diltiazem. Furthermore, lipid emulsion inhibited the maximal vasodilation induced by bepridil, which has the highest lipid solubility among the calcium channel blockers. 11 The potency of lipid emulsion appears to be positively correlated with the magnitude of the lipid solubility of the calcium channel blockers, supporting the lipid sink theory. 1,11 In addition, lipid emulsion attenuates the apoptosis of H9c2 cells caused by a toxic dose of verapamil or bupivacaine, which are relatively highly lipid-soluble drugs compared with other drugs of the same group. 6,18 Consistent with the results of the present study using lipid emulsion pretreatment, posttreatment with all the concentrations (0.5–2%) of lipid emulsion used in the current experiment reversed the verapamil-induced vasodilation of the 60 mM KCl-induced contraction. However, diltiazem-induced vasodilation was slightly reversed by posttreatment with 2% lipid emulsion only. Consistent with previous reports using lipid emulsion posttreatment following vasodilation induced by a toxic dose of local anesthetic, the degree of the posttreatment lipid emulsion-induced reversal of the vasodilation produced by a toxic dose of verapamil or diltiazem may be associated with the lipid solubility of verapamil and diltiazem. 7,8,11 It has been reported that the systemic vasodilation induced by nifedipine and verapamil in an in vivo experiment is greater than that induced by other calcium channel blockers. 19 In addition, the reported potency of the vasodilation induced by calcium channel blockers in an isolated vessel is in the following decreasing order: nifedipine, verapamil, and diltiazem. 20 The calcium channel blocker-induced inhibition of the contraction evoked by phenylephrine, which leads to vasodilation, is more potent with nifedipine than with verapamil. 21 Supporting these previous reports, the potency of the calcium channel blockers in the current study in producing vasodilation in isolated endothelium-denuded rat aortae was in the following decreasing order: nifedipine, verapamil, diltiazem, and bepridil (Table 1). 19 –21 However, the reported potency of calcium channel blocker-induced vasodilation in human saphenous veins is in the following decreasing order: verapamil, diltiazem, and nifedipine. 22 This difference in the potency of calcium channel blocker-induced vasodilation may be due to differences in the vessels (rat aorta vs. human saphenous vein) and contractile agonists examined (phenylephrine vs. 5-hydroxytryptamine and acetylcholine). 22
In agreement with previous reports that lipid emulsion enhances left ventricular systolic pressure via the blockade of nitric oxide release, in the present work, lipid emulsion alone increased systolic and mean blood pressure. 13,23 It has been reported that posttreatment with lipid emulsion increases survival time and improves mean arterial blood pressure following verapamil toxicity. 24,25 Consistent with these previous reports, pretreatment with lipid emulsion alleviated the systolic and mean blood pressure decreases induced by a toxic dose of verapamil. 24,25 In addition, pretreatment with lipid emulsion has been reported to inhibit the mean blood pressure decrease associated with diltiazem toxicity. 26 In contrast, pretreatment with lipid emulsion had no effect on the mean blood pressure decrease in the diltiazem toxicity model used in the current experiment. This difference may be due to the following factors. First, although pretreatment with lipid emulsion was used in both experiments, the current experiment used more diltiazem than the previous experiment because the dosage of diltiazem (100 mg/kg/h) was adjusted to produce approximately 50–55% of the baseline systolic blood pressure, which was approximately equivalent to the reduction in systolic blood pressure induced by the toxic dose of verapamil (10 mg/kg/h) used in the current study. 26 Second, different dosages of lipid emulsion pretreatment were used in the current and previous experiments (3 ml/kg followed by 0.17 ml/kg/min vs. 12.4 ml/kg). 26 The loading and continuously infused doses of lipid emulsion used in the current study were selected based on the recommendations of the American Society of Regional Anesthesia and Pain Medicine and our preliminary study results. 27 Third, different anesthetics were used in the current and previous experiments (tiletamine/zolazepam vs. ketamine), which may have led to different depths of anesthesia, contributing to hemodynamic alterations during the experiment. 26 Thus, the greater amount of diltiazem and the lower amount of lipid emulsion used in the current experiment may have contributed to the lack of change in systolic and mean blood pressure induced by lipid emulsion in the current diltiazem toxicity model. Moreover, lipid emulsion treatment reportedly attenuates the myocardial depression induced by verapamil and reduces the serum concentration of verapamil in patients with verapamil toxicity. 12,28 Considering both the tension measurements in the current study and the previous reports, smaller decreases in systolic and mean blood pressure during lipid emulsion infusion in the verapamil toxicity model appear to be associated with the relatively high lipid solubility of verapamil compared with diltiazem. 11,17,28 In addition, because it has been reported that verapamil produces more potent vasodilation and myocardial depression than diltiazem, the diltiazem dosage applied was 10 times higher than the verapamil dosage used to produce a baseline systolic blood pressure reduction of approximately the same magnitude (50–55% of the baseline systolic blood pressure) as that induced by verapamil or diltiazem in the current study. 19,20 Lipid emulsion showed a tendency to inhibit the diastolic blood pressure decrease induced by a toxic dose of verapamil, but it was not significant except at 1 min after verapamil infusion. Lipid emulsion had no effect on the diastolic blood pressure decrease induced by a toxic dose of diltiazem. The potency of the bradycardia induced by calcium channel blockers is higher with diltiazem than with verapamil. 19,29 In the present study, lipid emulsion did not significantly alter the heart rate decrease induced by verapamil or diltiazem (Figures 3(d) and 4(d)). According to a systemic review of the effects of lipid emulsion on the systemic toxicity of calcium channel blockers, verapamil-induced toxicity has been reported to respond well to lipid emulsion treatment compared with toxicity induced by other calcium channel blockers. 2,5,30 Considering these previous reports and the current results, we hypothesized that the relatively optimal response to lipid emulsion observed in the verapamil toxicity model may be partially due to the enhanced removal of verapamil from the tissues into the lipid emulsion compared with diltiazem. 2,5,11,17,30 Lipid emulsion-mediated δ-opioid receptor activation has been reported to attenuate the apoptosis induced by a toxic dose of verapamil. 6 In addition, lipid emulsion-induced glycogen synthase kinase-3β phosphorylation contributes both to the inhibition of apoptosis induced by bupivacaine in H9c2 cells and to cardioprotection. 31,32 The δ-opioid receptor is involved in reducing myocardial infarction via adenosine triphosphate-sensitive potassium channel activation. 33 Thus, further studies are needed to elucidate the roles of the δ-opioid receptor, adenosine triphosphate-sensitive potassium channel, and glycogen synthase kinase-3β phosphorylation in the lipid emulsion-mediated alleviation of cardiovascular collapse induced by a toxic dose of verapamil.
This study has several limitations. First, lipid emulsion is generally applied after the administration of a toxic dose of either a local anesthetic drug or a calcium channel blocker to treat the resultant systemic toxicity.
1,5
However, in the current study, lipid emulsion pretreatment was applied before systemic toxicity was induced by verapamil. Second, total peripheral vascular resistance, which is considered one of the main determinants of blood pressure, is mainly affected by small-resistance arterioles, such as mesenteric arteries.
34
However, we used rat aortae, which are considered conduit vessels. Third, it has been reported that lipid emulsion reduces the verapamil concentration in the blood following verapamil toxicity.
12,28
However, we did not measure the serum concentrations of verapamil before and after the administration of lipid emulsion in the current verapamil toxicity model. Fourth, the depth of the anesthesia used in the current in vivo experiment may have affected the hemodynamic results. Fifth, endothelial nitric oxide contributes to the regulation of vascular tone; however, we used endothelium-denuded aortae pretreated with
In conclusion, taken together, these results suggest that lipid emulsion alleviates the vasodilation and systolic blood pressure decrease evoked by a toxic dose of verapamil in an in vitro experiment involving isolated rat aortae and in an in vivo rat model. These effects appear to be associated with the relatively high lipid solubility of verapamil.
Footnotes
Author contribution
S-H Ok and I-W Shin contributed equally to this study as co-first authors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT & Future Planning (2015R1C1A1A01054105).
