Abstract
Amanita exitialis is a lethal mushroom found in China. Knowledge regarding taxonomic characterization, toxin detection, general poisoning conditions, clinical manifestations, laboratory examinations, and clinical treatments for this species is currently lacking. We investigated three A. exitialis mushroom poisoning cohorts in Yunnan Province in 2014 and 2015, involving 10 patients. Mushroom samples were identified by morphological and molecular studies. Ultra performance liquid chromatography-electrospray ionization tandem mass spectrometry was used to detect the peptide toxins in the mushroom samples. Epidemiological information, clinical data, and results of laboratory examinations were collected and analyzed. The mushroom samples were all identified as A. exitialis. The average toxin concentration decreased from the cap to the stipe to the volva, and the average concentration of the peptide toxins decreased in the order of α-amanitin > phallacidin > β-amanitin > γ-amanitin. The latency period between ingestion and the onset of symptoms was 13.9 ± 2.1 h, and the time from ingestion to hospitalization was 49.6 ± 8.5 h. The most common symptoms were nausea and vomiting (100%). Four patients died from fulminant hepatic failure. Laboratory examinations showed that the alanine transaminase, aspartate transaminase, prothrombin time, and activated partial thromboplastin time levels peaked on the third day post-ingestion. Total bilirubin and direct bilirubin values peaked on day 7. The death group and the survival group had a similar variation trend of serological indexes, but the death group had a greater change. A. exitialis is an extremely dangerous mushroom and there is a need to educate the public to avoid picking and eating wild mushrooms that have not been definitively identified.
Introduction
More than 3800 species of mushrooms have been catalogued in China, of which approximately 435 are poisonous. 1 Statistical data from the National Management Information System of Public Health Emergencies reported by the Chinese Centre for Disease Control and Prevention (CDC) revealed 576 incidents of mushroom poisoning from 2004 to 2014, with 3701 patients and 786 deaths. 2 Mushroom poisoning has become the main cause of mortality in food poisoning incidents in China. An investigation of 102 mushroom poisoning cases in southern China from 1994 to 2012 showed that poisonous mushrooms from the genus Amanita were responsible for 70.49% of the fatalities. 3
In March 2000, eight out of nine patients died after eating wild white mushrooms in Guangdong Province. The lethal mushroom could not be identified as a known species, but was later found to be A. exitialis, a newly described lethal Amanita species, by Yang and Li. 4 Since 2000, A. exitialis has been identified in more than 6 mushroom poisoning incidents, with 33 people poisoned and 20 deaths. 3
Lethal mushrooms in the Amanita genus contain two major groups of cyclopeptides, the bicyclic octapeptide amatoxins and bicyclic heptapeptide phallotoxins. Some contain monocyclic virotoxin peptides as well. The thermostable amatoxins are 10–20 times more toxic than the others, but is the only toxin class of the three that is actually absorbed and therefore responsible for poisoning and death in humans. 5 Numerous studies have focused on detecting, quantifying, separating, and purifying toxins from A. exitialis. 6,7 The genomic diversity of amatoxins from A. exitialis has also been studied. 8 However, because of the unexpectedness and complexity of mushroom poisoning, there have been few systematic studies into A. exitialis poisonings.
In this article, the features of A. exitialis poisoning, including species identification, toxin detection, general conditions, clinical manifestation, laboratory examination, and clinical treatment, are presented and summarized.
Materials and methods
Species identification
Five specimens were collected from the area where the poisonings occurred, including four from Chuxiong Yi Autonomous Prefecture and one from Tengchong County, Yunnan Province. All specimens were processed and deposited at the National Institute of Occupational Health and Poison Control, Chinese Center for Disease Control and Prevention. Species identification was performed using morphological and molecular methods. 9 Internal transcribed spacer (ITS) DNA was amplified using the ITS5/ITS4 primer pair. 10 The polymerase chain reaction products were directly sequenced by Sangon Biotech, China, using the same primers. The GenBank database was searched for similar sequences using basic local alignment search tool (BLAST).
Detection of peptide toxins using ultra performance liquid chromatography-electrospray ionization tandem mass spectrometry
Peptide toxins, including α-amanitin (AA), β-amanitin (BA), γ-amanitin (GA), and phallacidin (PCD; Sigma-Aldrich; St. Louis, MO, USA), were used as standard references. Aliquots (0.2 g) of dried mushroom were weighed and transferred into a 15-mL polypropylene centrifuge tube in duplicate.10 ml of ultrapure water was added. After vortex-mixing for 1 min, each was sonicated for 30 min, and centrifuged at 11,200 × g for 10 min at 4 °C. The supernatants were diluted twice with ultrapure water for ultra performance liquid chromatography-electrospray ionization tandem mass spectrometry (UPLC-ESI-MS/MS) analysis.
UPLC was performed using a Waters Acquity UPLC (Waters, Milford, MA, USA) separation module equipped with a HSS T3 column (100 mm × 2.1 mm, 1.7 μm; Waters, Milford, MA, USA). The mobile phase was a mixture of 20 mM ammonium acetate in ultrapure water (A) and acetonitrile (B), with a flow rate of 0.3 mL/min under gradient conditions: 15% B held for 2.5 min, then linearly increased to 35% in the following 1.5 min, then increased to 100% in 2 min and held for 2 min, and finally returned to the initial composition in 0.1 min. The column was equilibrated for 2.9 min before the next injection. The injection volume and column oven were set at 10 μL and 40 °C, respectively.
MS/MS acquisition was conducted on a Waters XevoTM TQ-S triple quadrupole mass spectrometer (Waters, Milford, MA, USA) equipped with an ESI interface, operating in positive ion mode with multiple-reaction monitoring. The electrospray source block and desolvation temperatures were held at 150 °C and 450 °C, respectively. The capillary voltage was 3.5 kV. For each analyte, two transitions were selected, and the corresponding cone voltages and collision energies are as shown in Table 1.
MS/MS parameters for peptide toxins.

MS/MS: tandem mass spectrometry; AA: α-amanitin; BA: β-amanitin; GA: γ-amanitin; PCD: phallacidin; a: Quantificational ion pair.
The calibration curves for the four toxins, obtained using blank samples spiked with extracts before pretreatment, showed good linearity, with correlation coefficients of 0.9971–0.9998 from 0.1 mg/kg to 100 mg/kg. Samples containing concentrations of the target compounds above the calibration range were appropriately diluted and re-evaluated. The limit of detection for each toxin was 0.03 mg/kg and the limit of quantitation was 0.1 mg/kg. The recoveries, determined by spiking at 0.1 mg/kg, 0.5 mg/kg, and 1 mg/kg, were 92.7–118.8% with RSD <11.9%.
Epidemiologic survey
After obtaining informed consent, the patients and their relatives were interviewed. Medical records were collected from Chuxiong People’s Hospital and Tengchong People’s Hospital to analyze the clinical manifestations, laboratory results, and management of the patients. This study is conducted in accordance with according to the World Medical Association Declaration of Helsinki and has been approved by the regional ethical review board (No. 2017008).
Results
Species identification
Morphological analysis
Basidiocarp: small to medium-sized. Pileus: 4–8 cm in diameter; convex to applanate, sometimes slightly depressed at the center; glabrous; white, but cream-colored over the disc. Lamellae: free; white to whitish; crowed. Stipe: 7–9 cm high; subcylindrical or slightly tapering upward, with the apex slightly expanded; white to whitish; glabrous, or sometimes with fibrillose squamules. Volva: shortly limbate; thin; membranous; free limbs 1–2 cm in height; both surfaces white. Annulus: apical to subapical; thin; membranous; white; persistent or torn from the stipe during expansion of the pileus (Figure 1).

Basidiocarps of Amanita exitialis (a) from Tengchong and (b) from Chuxiong.
Basidia: 27–55 μm × 10–15 μm; 2-spored (Figure 2(b)). Basidiospores: 9.5–12.0 μm × 9.0–11.5 μm [Q = 1.0−1.15(−1.17), Q = 1.08 ± 0.04] (Figure 2(a)); globose to subglobose; amyloid.

Representative photomicrographs of Amanita exitialis (100×). (a) Basidiospore of Amanita exitialis in cotton blue and (b) basidia in 5% KOH. KOH: potassium hydroxide.
Remarks: A. exitialis is characterized by its white basidiocarps and 2-spored basidia.
Molecular phylogeny
All five samples were identified as A. exitialis (the ITS GenBank accession numbers are shown in Table 2).
ITS GenBank accession numbers of Amanita exitialis.

ITS: internal transcribed spacer.
Peptide toxin detection
The UPLC retention times for AA, BA, GA, and PCD were 2.07, 1.57, 3.34, and 3.92 min, respectively. The UPLC chromatogram of the peptide toxin samples is given in Figure 3.

UPLC chromatogram of toxin analysis for the mushroom samples. UPLC: ultra performance liquid chromatography-electrospray ionization tandem mass spectrometry.
The concentration of the toxins was different between specimens and tissues. The total peptide toxin concentration was highest in the cap (3874.6–8423.6 mg/kg dry weight) and lowest in the volva (73.0–296 mg/kg dry weight). The average concentration of the different toxins decreased as follows: α-amanitin > phallacidin > β-amanitin > γ-amanitin (Table 3).
Toxin concentrations in different tissues of Amanita exitialis from the three outbreaks (mg/kg dry weight).

AA: α-amanitin; BA: β-amanitin; GA: γ-amanitin; PCD: phallacidin; ND: not detected.
General conditions
Ten patients were involved in the three outbreaks, including six females. They ate the mushrooms after collecting them from mountain areas close to their homes. Patients from each outbreak were family members. The patient ages ranged from 4 years to 72 years, with a mean of 37.5 ± 21.4 years.
The amount of mushroom ingested by history ranged from 20 g to 300 g fresh weight. Patients 1 and 2 also consumed 30 ml of mushroom soup. The latency, defined as the time from ingestion to the onset of symptoms, ranged from 10 h to 16 h, with a mean of 13.9 ± 2.1 h. The time from ingestion to hospitalization ranged from 35 h to 65 h, with a mean of 49.6 ± 8.5 h. Four patients (patients 1, 7, 8, and 9) died due to fulminant hepatic failure. Information regarding the three outbreaks is presented in Table 4.
General conditions of the three outbreaks of Amanita exitialis poisoning.
Clinical manifestations
The most common initial symptoms were nausea and vomiting (100%; Table 5). Patients 1 and 9 developed jaundice 4 and 5 days post-ingestion, respectively. Patient 9 became anemic on day 5 and developed hepatic coma on day 15. The clinical course of each patient who died was as follows:
First symptoms of poisoning.

Patient 1 ate 300 g wild mushroom and 30 ml soup and presented with severe nausea, vomiting, and diarrhea 14 h after ingestion. On presentation at a local emergency department 58 h after ingestion, he had a normal mental status and normal vital signs. Physical examination revealed a well-developed, 72-kg, 52-years man. His vital signs were temperature of 37.0 °C; pulse, 74 beats per minute; blood pressure, 94/60 mm Hg; and respiratory rate, 19 breaths per minute. There were no cutaneous stigmata of chronic liver disease such as spider angiomata or palmar erythema. Scleras were anicteric. His abdomen was soft, flat, nontender, and without hepatosplenomegaly. His mental status was normal, and there was no asterixis. Initial laboratory studies at 60 h after ingestion were prothrombin time (PT) of 35.9 s; activated partial thromboplastin time (APTT) of 72.5 s; alanine transaminase (ALT), 3084 U/L; aspartate transaminase (AST), 1506 U/L; total bilirubin (TBIL), 0.9 mg/dL; hemoglobin 148 g/L; creatinine 75.7 µmol/L; BUN 7.04 mmol/L. Laboratory studies on day 3 after ingestion were PT of 76.4 s; APTT of 118.9 s; ALT, 4127 U/L; AST, 1849 U/L; TBIL, 134.1 mg/dL; hemoglobin 158 g/L; creatinine 64.1 μmol/L; BUN 6.09 mmol/L; patient developed jaundice on day 4 after ingestion. Laboratory studies on day 4 after ingestion were PT of 70.8 s; APTT of 92.3 s; ALT, 3881 U/L; AST, 1207 U/L; TBIL, 186.9 mg/dL. Laboratory studies on day 5 after ingestion were PT of 70.6 s; APTT of 88.5 s; ALT, 1847 U/L; AST, 392 U/L; TBIL, 204 mg/dL; hemoglobin 144 g/L; creatinine 76.7 μmol/L; BUN 5.39 mmol/L; patients and their relatives refused to continue treatment due to economic reasons and discharged on the day 5 after ingestion. Supportive therapy included intravenous hydration and polyene phosphatidylcholine, glutathione, and lipoic acid. Hemodialysis and hemoperfusion were used to reduce the level of bilirubin. Telephone follow-up was taken 1 month after the patients discharged. The patient died on the day 7 after ingestion.
Patient 7 ate 80 g wild mushroom and presented with severe nausea, vomiting, and diarrhea 12 h after ingestion. On presentation at a local emergency department 35 h after ingestion, she had a normal mental status and normal vital signs. Physical examination revealed a well-developed, 12-kg, 4-years girl. Her vital signs were temperature of 37.0 °C; pulse, 100 beats per minute; and respiratory rate, 25 breaths per minute. There were no cutaneous stigmata of chronic liver disease such as spider angiomata or palmar erythema. Scleras were anicteric. Her abdomen was soft, flat, nontender, and without hepatosplenomegaly. Her mental status was normal, and there was no asterixis. Initial laboratory studies at 44 h after ingestion were PT of 44.5 s; APTT of 68.9 s; ALT, 300 U/L; AST, 471 U/L; TBIL, 20.1 mg/dL; creatinine 35 µmol/L; BUN 6.4 mmol/L. Laboratory studies on day 3 after ingestion were PT of 90 s; APTT of 120 s; ALT, 1387 U/L; AST, 1966 U/L; TBIL, 27.7 mg/dL; creatinine 28 µmol/L; BUN 4.3 mmol/L. Patients and their relatives refused to continue treatment due to economic reasons and discharged on the day 3 after ingestion. Supportive therapy included intravenous hydration and polyene phosphatidylcholine, glutathione, and lipoic acid. Telephone follow-up was taken 1 month after the patients discharged. The patient died on the day 3 after ingestion.
Patient 8 ate 70 g wild mushroom and presented with severe nausea, vomiting, and diarrhea 13 h after ingestion. On presentation at a local emergency department 46 h after ingestion, she had a normal mental status and normal vital signs. Physical examination revealed a well-developed, 72-years woman. Her vital signs were temperature of 36.5 °C; pulse, 106 beats per minute; blood pressure, 100/60 mm Hg; and respiratory rate, 20 breaths per minute. There were no cutaneous stigmata of chronic liver disease such as spider angiomata or palmar erythema. Scleras were anicteric. Her abdomen was soft, flat, nontender, and without hepatosplenomegaly. Her mental status was normal, and there was no asterixis. Initial laboratory studies at 48 h after ingestion were PT of 24.5 s; APTT of 51.9 s; ALT, 208 U/L; AST, 337 U/L; TBIL, 26.9 mg/dL; creatinine 46 µmol/L; BUN 6.2 mmol/L. Laboratory studies on day 3 after ingestion were PT of 64.2 s; APTT of 76.2 s; ALT, 1418 U/L; AST, 2503 U/L; TBIL, 48.4 mg/dL; creatinine 50 μmol/L; BUN 7.4 mmol/L. Patients and their relatives refused to continue treatment due to economic reasons and discharged on the day 4 after ingestion. Supportive therapy included intravenous hydration and polyene phosphatidylcholine, glutathione, and lipoic acid. Telephone follow-up was taken 1 month after the patients discharged. The patient died on the day 4 after ingestion.
Patient 9 ate 30 g wild mushroom and presented with severe nausea, vomiting, and diarrhea 10 h after ingestion. On presentation at a local emergency department 65 h after ingestion, she had a normal mental status and normal vital signs. Physical examination revealed a well-developed, 52-years woman. Her vital signs were temperature of 36.2 °C; pulse, 55 beats per minute; blood pressure, 115/82 mm Hg; and respiratory rate, 21 breaths per minute. There were no cutaneous stigmata of chronic liver disease such as spider angiomata or palmar erythema. Scleras were anicteric. Her abdomen was soft, flat, nontender, and without hepatosplenomegaly. Her mental status was normal, and there was no asterixis. Initial laboratory studies at 65 h after ingestion were PT of 90 s; APTT of 120 s; ALT, 2320 U/L; AST, 2057 U/L; TBIL, 115.3 mg/dL; creatinine 44 µmol/L; BUN 2.9 mmol/L. Laboratory studies on day 4 after ingestion were PT of 78.2 s; APTT of 90.6 s; ALT, 2653 U/L; AST, 2115 U/L; TBIL, 121 mg/dL; creatinine 51 µmol/L; BUN 1.7 mmol/L. Laboratory studies on day 5 after ingestion were PT of 50.3 s; APTT of 82.7 s; ALT, 3446 U/L; AST, 1594 U/L; TBIL, 134.9 mg/dL; creatinine 48 µmol/L; BUN 2.2 mmol/L. Patient developed jaundice (cutaneous or sclera icterus) on day 4 after ingestion. Laboratory studies on day 6 after ingestion were PT of 37.4 s; APTT of 60.5 s; ALT, 2507 U/L; AST, 987 U/L; TBIL, 209.4 mg/dL. Laboratory studies on day 7 after ingestion were PT of 29.4 s; APTT of 40.7 s; ALT, 1563 U/L; AST, 191 U/L; TBIL, 230.4 mg/dL; creatinine 58 µmol/L; BUN 3.1 mmol/L. Laboratory studies on day 10 after ingestion were PT of 24.1 s; APTT of 39.8 s; ALT, 403 U/L; AST, 62 U/L; TBIL, 187.1 mg/dL; creatinine 63 μmol/L; BUN 3.5 mmol/L. Laboratory studies on day 14 after ingestion were PT of 27.7 s; APTT of 40.5 s; ALT, 190 U/L; AST, 62 U/L; TBIL, 167.1 mg/dL. Patient developed hepatic coma on day 15 after ingestion. Patients and their relatives refused to continue treatment due to economic reasons and discharged on the day 15 after ingestion. Supportive therapy included intravenous hydration and polyene phosphatidylcholine, glutathione, and lipoic acid. Hemodialysis and hemoperfusion were used to reduce the level of bilirubin. Telephone follow-up was taken 1 month after the patients discharged. The patient died on the day 27 after ingestion.
Laboratory examination
The ALT and AST levels of the patients began to rise dramatically (ALT 653 U/L; AST 510 U/L) 2 days post-ingestion, and peaked (ALT 1019 U/L; AST 903 U/L) on day 3. Then, the patient’s transaminases levels entered a general declining trend. In the death group, the ALT level increased from day 2 (ALT, 1342 IU/L) and reached a peak value on day 4 (ALT 3267 U/L). The AST level of the death group reached a peak on day 3 (AST 2094 U/L). After reaching a peak, both ALT and AST levels of the death group got a dramatic decline, and then descended gradually on day 10. In the survivor group, the ALT and AST levels, respectively, reached a peak on day 4 (ALT, 156 U/L) and day 3 (AST 110 U/L), and then entered a general declining trend. On day 14, AST and ALT values were at normal levels (Figures 4 and 5).

Dynamic changes in the ALT levels of patients who had ingested Amanita exitialis. ALT: alanine transaminase.

Dynamic changes in the AST levels of patients who had ingested Amanita exitialis. AST: aspartate transaminase.
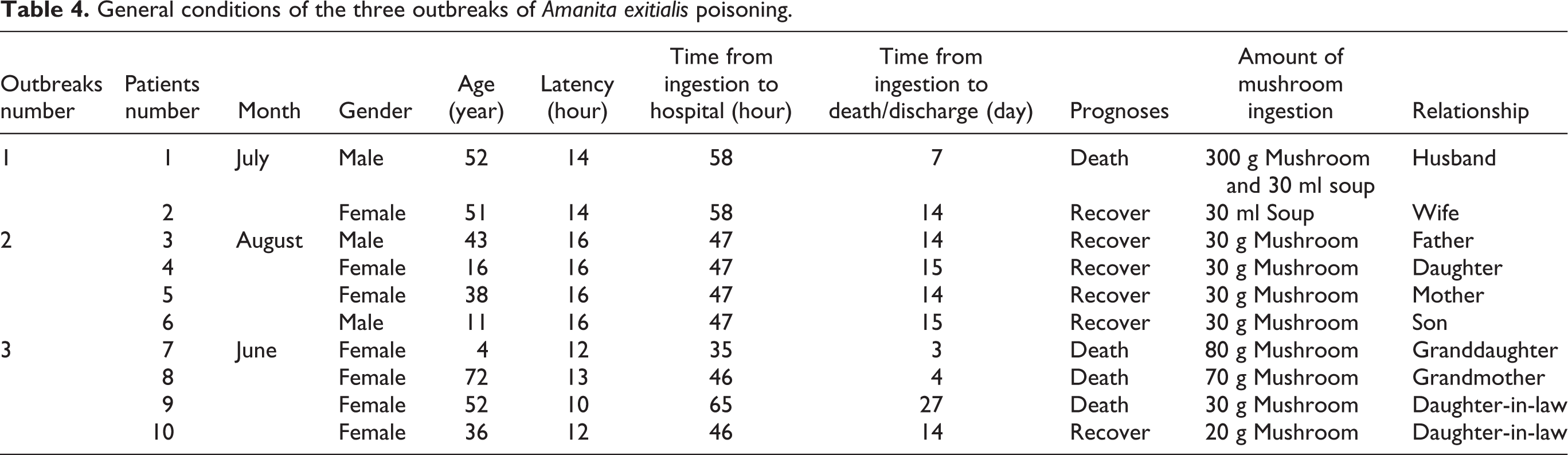
On day 2 post-ingestion, the TBIL and direct bilirubin (DBIL) values were 44 μmol/L and 23 μmol/L. These values increased, before peaking on day 7 (TBIL 89 μmol/L; DBIL 83 μmol/L). In the death group, TB and DB values, respectively, rose from 81 μmol/L and 56 μmol/L to 230 μmol/L and 189 μmol/L during day 2 and day 7. From day 7 to day 14, the bilirubin level kept a gently decreased trend. TB and DB values of patients in the survivor group were 23.1 μmol/L and 9.4 μmol/L, respectively, on day 2. Then, the bilirubin level entered a general declining trend, and returned to normal level on day 14 (Figures 6 and 7).
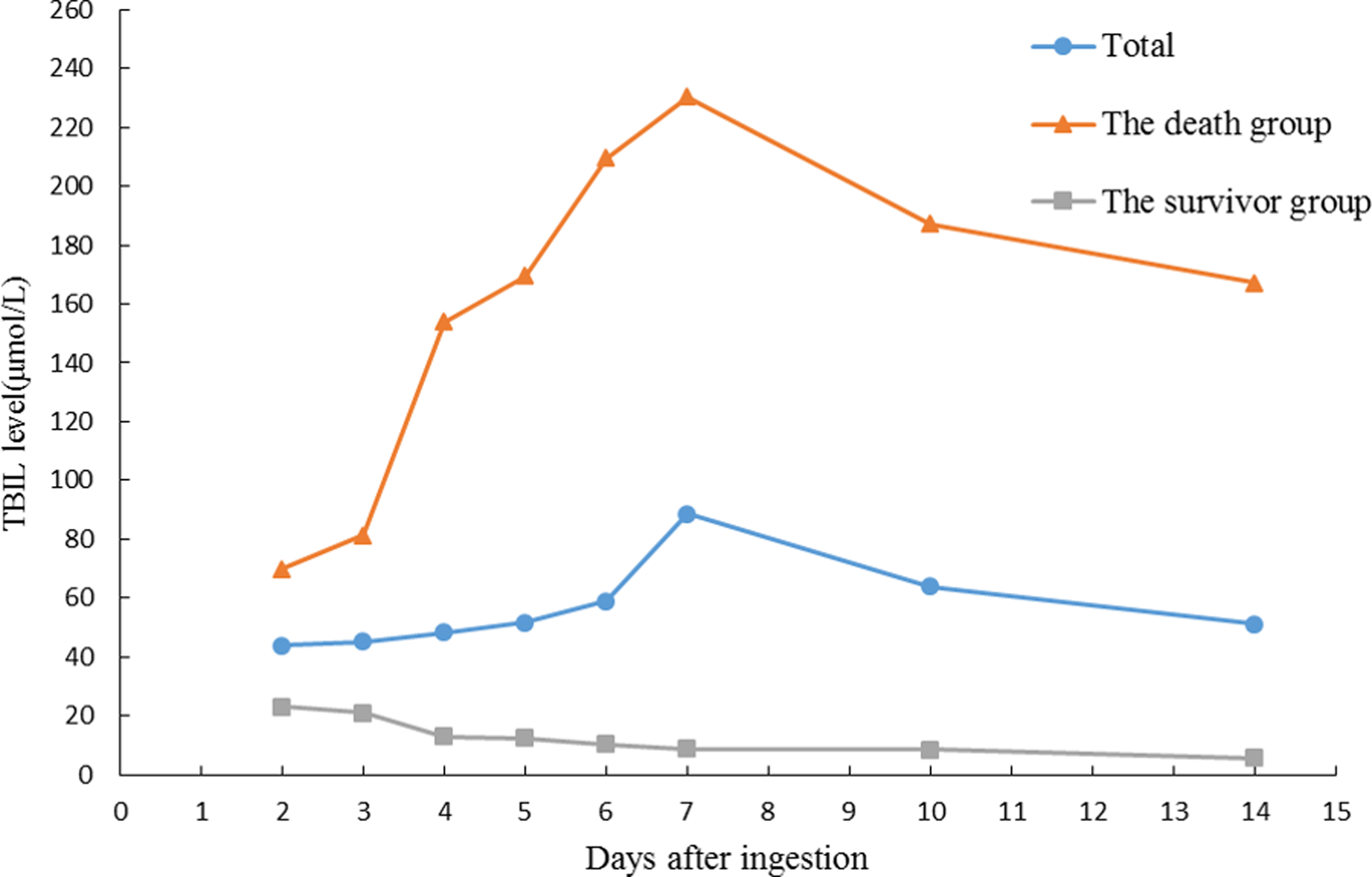
Dynamic changes in the TBIL levels of patients who had ingested Amanita exitialis. TBIL: total bilirubin.

Dynamic changes in the DBIL levels of patients who had ingested Amanita exitialis. DBIL: direct bilirubin.
The PT and APTT reached 79.8 s and 49.8 s on day 3, but then decreased and returned to normal on day 7 (PT 29.2 s; APTT 16.3 s). In the death group, their PT and APTT values got their peaks on day 3 (PT, 108.8 s; APTT, 80.0 s), and entered a dramatically declining trend during the following 4 days. PT and APTT values were 40.7 s and 29.4 s on day 7. Then, they entered a relative steady phase during the next week. PT and APTT values were 39.8 s and 24.1 s on day 14. In the survivor group, the PT and APTT values reached 60.4 s and 29.5 s on day 3 and returned to normal level on day 5 (Figures 8 and 9).

Dynamic changes in the PT levels of patients who had ingested Amanita exitialis. PT: prothrombin time.
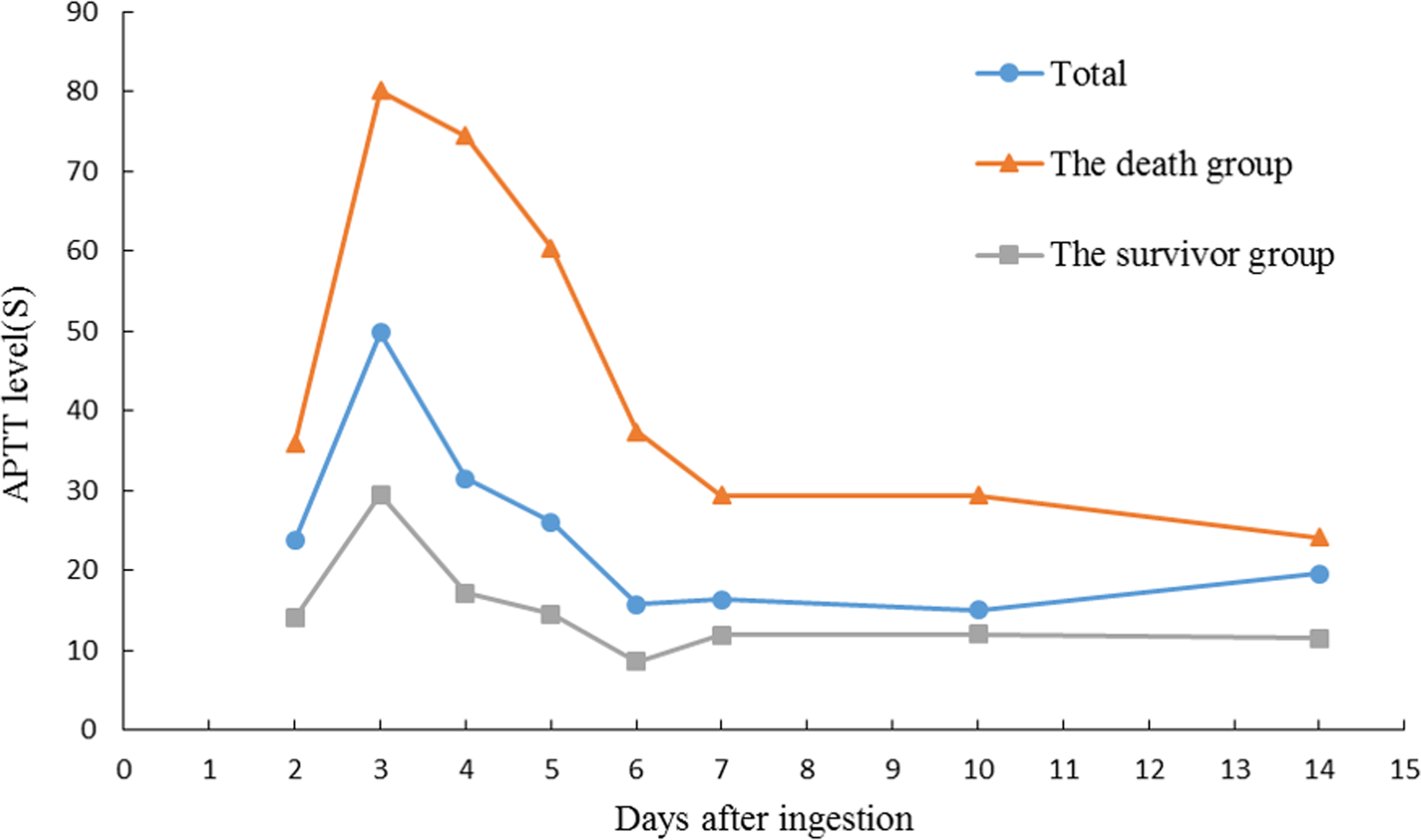
Dynamic changes in the APTT levels of patients who had ingested Amanita exitialis. APTT: activated partial thromboplastin time.
The creatinine and blood urea nitrogen values remained normal; no apparent renal injury was observed. The peak ALT, AST, PT, APTT, TBIL, and DBIL values of each patient were shown in Table 6.
The peak ALT, AST, TBIL, DBIL, PT, and APTT values of each patient.

ALT: alanine transaminase; APTT: activated partial thromboplastin time; AST: aspartate transaminase; DBIL: direct bilirubin; PT: prothrombin time; TBIL: total bilirubin.
Clinical treatment
All received supportive care, including intravenous hydration, polyene phosphatidylcholine, glutathione, and lipoic acid. Patients 1 and 9 underwent extracorporeal purification, hemodialysis, and hemoperfusion.
Discussion
Mushroom poisoning incidents occur every year, but the responsible mushroom species are rarely identified. The American Association of Poison Control Centre reported 6600 cases of poisonous mushrooms in 2012, but in only 17% were the mushrooms identified. 11 The CDC reported mushroom poisoning 576 cohorts from 2004 to 2014, but species identification occurred in less than 10%. 2 Different mushroom toxins cause different injuries; therefore, species identification is crucial for proper treatment. Immediate species identification is usually impossible, because it requires specialist expertise. The ingested mushrooms are often unavailable and any remaining cooked mushrooms are likely to be unidentifiable.
The main mushroom species causing death in Europe and North America are Amanita phalloides, Amanita bisporigera, and Amanita virosa. 12,13 These species are not native to China. However, other Amanita species from the section Phalloideae, including A. exitialis, Amanita subjunquillea var. alba, Amanita pallidorosea, Amanita fuligineoides, and Amanita rimosa are native to China and cause over 70% of Chinese fatalities. 3 The ITS sequences of our samples matched A. exitialis.
A. exitialis is morphologically similar to North American A. bisporigera, which also has white basidiocarps and two-spored basidia. However, A. exitialis has larger basidiospores, an apical to sub-apical annulus, and less inflated cells in its volva. A. phalloides may also have white basidiocarps and two-spored basidia, but it has distinctly smaller basidiospores than A. exitialis. 14 In East Asia, A. subjunquillea var. alba, A. rimosa, and Amanita oberwinklerana also resemble A. exitialis, but these species have four-spored basidia. 14 A. exitialis was previously reported in Guangdong Province only; however, our results confirm that A. exitialis can also be found in Yunnan Province.
Lethal Amanita species contain various peptide toxins including amatoxins, phallotoxins, and virotoxins. Amatoxin alone is responsible for human morbidity death. A lethal dose of α-amanitin is just 0.1 mg/kg, easily found in a single mushroom. 15 Different mushrooms contain concentrations of these peptide toxins. Even within the same species, the toxin concentration can be different. 16 Table 7 presents some of the toxin concentrations reported for Amanita species. 16 –19
Toxin concentrations in Amanita species taken from the literature (mg/kg dry weight).

AA: α-amanitin; BA: β-amanitin; GA: γ-amanitin; PCD: phallacidin; PHN: phalloin.
aToxin not detected.
This study demonstrated that toxin concentration decreased from cap > stipe > volva. The average concentration of the different peptide toxins decreased from α-amanitin > phallacidin > β-amanitin > γ-amanitin, consistent with the previous reports. 6,20,21 Enjalbert et al. 22 reported that in A. phalloides, there was more β-amanitin than α-amanitin in all tissues except the bulb. However, α-amanitin is the major toxin in other species, including A. exitialis.
Because the growth of wild mushrooms depends on geographical location and specific weather conditions, the incidence of mushroom poisoning is both seasonal and unpredictable. Mushroom poisoning most frequently occurs from June to November. An Iranian study concluded that mushroom poisoning is most frequent from September to October. 23 In Turkey, hospital visits due to mushroom poisoning were most frequent in spring and autumn. 24 In our study, the three cohorts of A. exitialis poisoning occurred from June to August. In Yunnan Province, the rainy season is also June to August; mushroom collection as well as poisonings is common during these months. There is seasonal variation in A. exitialis occurrence in different areas of China. A recent study investigated six incidents of A. exitialis poisoning in Guangdong province occurring in March and April, 3 three months earlier than the Yunnan Province outbreak. The incidents coincided with the earlier March to May rainy season of Guangdong Province.
In the course of reviewing over 28,000 cases of mushroom poisoning, Diaz 25 devised a new syndromic classification system based on the timing of symptom onset. Cyclopeptide mushrooms cause long latency (6–24 h) hepatotoxic syndrome. The latency period ranges from 6 h to 40 h, with an average of about 10 h. 15 Previous studies on A. phalloides poisonings have revealed ingestion to admission times ranging from 27.8 h to 35.7 h. 26 In our study, the mean latency and mean time from ingestion to hospitalization were 13.9 h and 49.6 h, respectively.
After the latency period, patients enter a gastrointestinal phase. The most common initial symptoms are nausea, vomiting, abdominal pain, and diarrhea. 23,25,27 This stage typically lasts 12–24 h. 13,28 –30 Liver function tests may be normal at presentation. Early presenting patients may be erroneously diagnosed with gastroenteritis and discharged. Because of the long latency, patients may not initially ascribe their symptoms to mushrooms consumed so many hours before.
By the time patients were admitted to the hospital, they usually had evidence of liver injury, which generally begins by 36–48 h post-ingestion. Diagnostic liver injury is characterized by rapidly rising serum transaminases. 31
Acute liver failure can lead to jaundice, hepatic coma, bilirubinemia, coagulopathy, and hypoglycemia. Death may occur within 3–7 days of ingestion. Patients with a favorable prognosis recover within 2–3 weeks. 15
It is known that amatoxins interact with the enzyme RNA polymerase II in eukaryotic cells, forming a tight complex. Thereby, the content of mRNA is progressively decreasing, resulting in deficient protein synthesis and cell death. 32 Damage will in the first place strike cells providing a transport device, and the main target organs are the hepatocytes of the live. So in patients of poisoning with amatoxins, biochemical signs of liver damage appear with an increase of liver transaminases, PT, APTT, and bilirubin in serum. Yilmaz et al. 33 showed that the ALT and AST levels of patients who had ingested A. phalloides began to increase after 48 h (AST, 294 U/L; ALT, 284 U/L), and peaked after the 72nd h (ALT 2496 U/L; AST 1777 U/L). Erden et al. 34 reported that the TBIL and PT values in a patient poisoned with A. phalloides peaked at 5 days post-ingestion (TBIL, 229 μmol/L; PT, 100.6 s). Our results agree with the previous reports.
Jones 35 suggested adverse prognostic criteria including PT, TB, creatinine, jaundice to encephalopathy time, and age in acute liver failure. It was reported a significant relationship among acute liver injury patients between mortality and ALT and AST levels. Therefore, ALT and AST levels can be used to judge prognosis of patients intoxicated with mushroom as important and practical parameters. 36 In our study, the death group and the survival group had a similar variation trend of serological indexes (including ALT, AST, TB, DB, PT, APTT), but the death group had a greater change.
There is currently no specific antidote for amatoxin mushroom poisoning. The current treatment methods usually include detoxification, symptomatic treatment, chemotherapies, and ultimately liver transplantation.
Kinetic studies indicate that the amatoxin cyclopeptides are completely absorbed within 6 h of ingestion; the toxins are undetectable in blood after 24 h, but urine tends to be positive for up to 4 days. 26 In our study, patients 1 and 9 underwent extracorporeal purification, hemodialysis, and hemoperfusion. It was ineffective to remove amatoxin because the patients were admitted more than 2 days post-ingestion.
Early symptomatic and supportive care is targeted at dehydration and correction of electrolyte abnormalities. 37 All of the study, patients were given symptomatic treatment, including IV rehydration, polyene phosphatidylcholine, glutathione, and lipoic acid. The treatment on these patients may be ineffective because these measurements cannot remove amatoxins from the body or prevent amatoxins damaging the liver.
Many drugs have been used to treat amatoxin poisoning. N-acetyl cysteine, alpha lipoic acid, and cimetidine have been used for purported antioxidant effects. Dexamethasone and penicillin G have been used for anti-inflammatory actions. 38 –41 None of these drugs has proven to be effective by rigorous designed clinical trials.
Cyclopeptide poisoning causes acute liver failure, and eventually death. Liver transplants are performed in North America and Europe, 42 but none have been reported in China.
This study systematically reports on three mushroom poisoning cohorts suggesting characteristics of A. exitialis as the source mushroom, including species identification, toxin detection, general conditions, clinical manifestations, laboratory examinations, and clinical treatments. We acknowledge several limitations. We present only 10 patients from three mushroom poisoning cohorts. The concentration of peptide toxins in biological samples (blood, urine, and liver) was not measured.
Conclusion
A. exitialis is now recognized as an extremely dangerous mushroom. This study reveals that poisoning by A. exitialis is characterized by the long latency before the onset of (6–24 h post-ingestion) GI symptoms and subsequent liver damage characteristic of amatoxin poisoning. A. exitialis is particularly prevalent during the rainy season in subtropical Asia. The clinical syndrome it produces is identical to other cyclopeptide mushrooms. At present, there are no guidelines for the management of mushroom poisoning in Chinese hospitals. Diagnosis and treatment is based on the history of ingestion and the associated clinical presentation. Species identification and peptide toxin detection may help confirm our clinical diagnosis. Furthermore, there is a need to educate the public to avoid picking and eating wild mushrooms that have not been definitively identified.
Footnotes
Acknowledgments
Special thanks are due to Doctor Cheng-Min Yu (Chuxiong People’s Hospital, China) for assistance on field trips. We express our gratitude to Yu-Min Niu (BJCDC, China) for toxin detection.
Author contribution
Hai-Jiao Li and Jian Sun contributed equally to this work and should be considered co-first authors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research was financed by National Nature Science Foundation of China Youth Science Foundation (31501814).
