Abstract
Introduction:
Sulcotrione is a herbicidal agent belonging to the family of triketones. Sulcotrione herbicides are used for weed control in maize and flax crops. To date, no cases of human poisoning had been reported in the literature linked to different herbicidal agents in the triketone family. We report here on two cases of the voluntary ingestion of this substance in the form of the branded product MikadoTM, which were recorded by the Angers Poison Centre.
Case report:
Both cases of voluntary ingestion constituted attempted suicide, and involved two men aged 30 and 37 years. Their symptoms linked to sulcotrione were limited to vomiting, despite elevated plasma concentrations of sulcotrione. In one case, hypertyrosinemia has been demonstrated. The outcome was favourable in both patients and at follow up, no ocular disorders were observed. In the second case, hypotension and transient renal failure could be linked to the concomitant ingestion of chlorophenoxy herbicides.
Discussion:
In animal toxicity studies, sulcotrione inhibit 4-hydro-phenylpyruvate dioxygenase leading to hypertyrosinemia and corneal opacities. In both cases, no ocular disorders were observed despite hypertyrosinemia in one case. These case reports were consistent with the animal toxicology findings concerning triketones, and particularly their relative safety in mammals following acute poisoning. However it seems prudent to monitor plasma tyrosine concentrations and to screen prospectively for corneal deposits if further acute intoxication events occur.
Introduction
Sulcotrione is a herbicidal agent belonging to the triketone family. Its chemical name is 2-(2-chloro-4-methylsulfonylbenzoyl)-1,3-cyclohexanedione and its molecular formula is C14H13ClO5S. Its registration number is CAS 99105-77-8. Sulcotrione herbicides are used to control weeds in maize and flax crops. The first brand-named product is Mikado™, marketed in France since 1991.
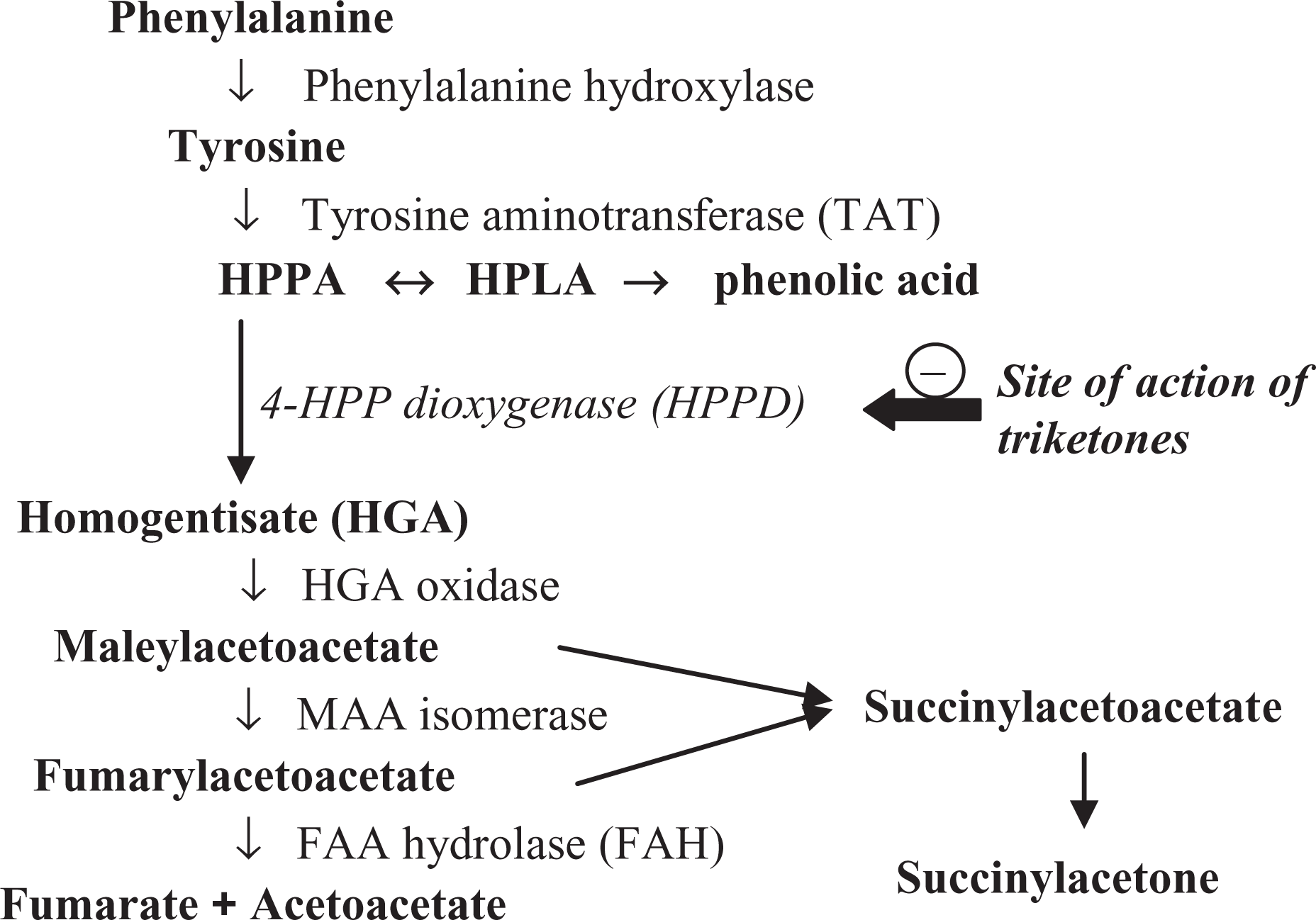
The mode of action of triketones on plants is linked to the inhibition of 4-hydroxy-phenylpyruvate dioxygenase (HPPD), an enzyme that allows the conversion of tyrosine into homogentisic acid, a precursor of chlorophyll and carotenoid synthesis. 1 HPPD inhibition thus leads to the destruction of weeds causing chlorophyll deficiency which in turn causes bleaching of the plants.
Experimental studies in mammals have demonstrated an identical mode of action involving HPPD inhibition and plasma hypertyrosinemia, sometimes complicated by ocular damage. However, to date, no cases of human poisoning have been reported in the literature involving different substances in the triketone family.
We report here on two cases of the voluntary ingestion of Mikado, herbicide containing 300 g/l sulcotrione in an aqueous solution.
Case reports
Case 1
A 30-year-old farmer ingested a quantity estimated as the equivalent of three wineglassfuls of Mikado (or about 90 g sulcotrione) and alcohol at 21:00. Vomiting occurred about 30 min later. Physical examination in the Emergency Department at 23:30 revealed normal vital parameters, with blood pressure of 130/60 mmHg, a pulse rate of 100 beats per min and a body temperature of 36.2°C. Pulse oxymetry was 99% in ambient air. Laboratory tests on samples collected at 23:55 revealed serum alcohol at 1.5 g/l, sodium (Na) 143 mmol/l, potassium (K) 3.9 mmol/l, chlorine (Cl) 109 mmol/l (N < 108), bicarbonates 20.8 mmol/l, anion gap 17 mmol/l, Ca 2.30 mmol/l, urea 5.3 mmol/l, creatinine 106 µmol/l (N < 105), total protein 77 g/l, and normal liver parameters for transaminases, alkaline phosphatase, gamma glutamyl transpeptidase (γ-GT) and bilirubin. The blood parameters were normal, with leukocytes at 9.56 giga/l and platelets at 297 giga/l. The next morning at 07:30, the parameters were as follows: Na 139 mmol/l, K 3.8 mmol/l, Cl 108 mmol/l, bicarbonates 20.1 mmol/l, normal anion gap, calcium (Ca) 2.35 mmol/l, urea 5.2 mmol/l, creatinine 108 µmol/l and protein 71 g/l. The physical examination was reassuring, and the patient was allowed to return home on D + 1 following consultation with a psychiatrist. Control laboratory tests were performed on D + 2: transaminase and γ-GT levels were normal; there was discrete hypereosinophilia at 0.411 giga/l on the blood count, with platelets at 275 giga/l, urea at 5 mmol/l, Na at 137 mmol/l, K at 4.7 mmol/l, Cl at 107 mmol/l (N < 106) and bicarbonates at 25.7 mmol/l. The patient was able to return to work. Control blood tests and eosinophil count 2 months later produced normal results. Clinical follow-up via a telephone call 3 months later to the general practitioner did not reveal any particular features, and no ocular abnormalities were found.
Plasma sulcotrione assays were performed using high performance liquid chromatography/ultraviolet detection (HPLC/UV) diode array detection, after liquid/liquid extraction under acidic pH with the following results: 530 mg/l at 23:50 (hour 3 (H3)) and 310 mg/l at 07:30 (H10).
Case 2
A 37-year-old man ingested two glasses of Mikado at 17:00. Vomiting occurred 1 hour later. Agitation was observed at his admission (20:00) to the Emergency Department. A confusion syndrome then appeared, with agitation and abnormal movements of the flapping tremor type. Transient hypotension appeared during the night, with a systolic blood pressure of 80 mmHg, which was treated with 1000 ml saline serum infusion. Anuria was observed, and a urinary catheter was inserted, which was able to confirm a full bladder. The next morning, a persistence of confusion was noted, but the situation improved during the day. An electroencephalography was performed and excluded the possibility of any epilepsy.
Initial laboratory parameters at admission during the evening were as follows: Na 138 mmol/l, K 4.8 mmol/l, Cl 102 mmol/l, CO2 T 21.6 mmol/l, protein 78 g/l (N: 60–75), urea 5.7 mmol/l, creatinine 125 µmol/l (N: 65–120) and lactates 3.80 mmol/l (N: 1–1.78). Liver parameters were not determined. The blood count was normal, apart form leukocytes at 14.5 giga/l, of which neutrophils were at 10.5 giga/l and platelets were at 311 giga/l. The prothrombin rate was 99%, and the serum ethanol level was zero.
The assessment performed in the morning of D + 1 showed: urea 9.9 mmol/l, creatinine 151 µmol/l and CO2 T 24.5 mmol/l; the remainder of the blood ionogram being normal. Liver parameters were normal, and lactates were not verified. The creatine phosphokinase (CPK) level was 180 IU/l (N: 30-172), and the blood count revealed white blood cells at 11.6 giga/l, of which neutrophils were at 9.5 giga/l and platelets at 262 giga/l. Serum ammonia was at 27 µmol/l (N < 38) and serum lithium at <0.05 mmol/l.
Laboratory assessments in the morning of D + 2 revealed: urea at 9.5 mmol/l, creatinine at 124 µmol/l, CO2 T at 23.4 mmol/l and CPK at 190 IU/l. The blood ionogram and liver parameters were normal.
Because the clinical picture that associated confusion, hypotension, renal failure and abnormal, possibly myotonic movements, could not be explained by the sulcotrione poisoning, the hypothesis of poisoning with other agents was put forward and focused in particular on chlorophenoxy herbicides. The laboratory was asked to perform appropriate assays using HPLC/UV diode array detection, after liquid/liquid extraction under acidic pH, and the results were as follows:
Plasma sample at H3: sulcotrione 550 mg/l, mecoprop 700 mg/l and dichlorprop 230 mg/l. The chromatography of amino acids was normal for phenylalanine (42 µmol/l, for an N: 45–65) and tyrosine (76 µmol/l for an N: 50–78).
Plasma sample at H18: sulcotrione 550 mg/l, mecoprop 535 mg/l and dichlorprop 230 mg/l. The chromatography of amino acids revealed elevations of tyrosine to 303 µmol/l and of phenylalanine to 70 µmol/l.
A urine specimen collected at H18: sulcotrione 1250 mg/l, mecoprop 230 mg/l and traces of dichlorprop.
The diagnosis of mixed poisoning by herbicides from two chemical families was thus confirmed.
The patient discharged himself against medical advice on D + 2 following a consultation with a psychiatrist. He returned to work (lorry driver) without any apparent problems. He never attended any of the visits scheduled for his clinical follow-up, in particular with the ophthalmologist. A follow-up telephone call at D + 30 to his home did not reveal any problems relative to specific ocular disorders, and he was never able to give us the brand name of the chlorophenoxy-herbicide involved.
Discussion
These two case reports are consistent with the animal toxicology data concerning triketones, and in particular their relative safety in mammals in the event of acute poisoning. The symptoms were limited to vomiting and hypertyrosinemia, despite high plasma concentrations of sulcotrione. Unfortunately, it was not possible to obtain longer-term data on tyrosine kinetics because of the refusal of the second patient. In addition, in this second case, poisoning with mecoprop and dichorprop, both chlorophenoxy herbicides, could alone have explained the obnubilation, hypotension and renal impairment experienced, and it is interesting to observe that this patient recovered from this poisoning event involving a high plasma chlorophenoxy herbicide concentration by means of fluid infusions alone, without alcalinization. In both the cases, the assays revealed a slow elimination of sulcotrione, but only two assays were performed in each patient, which is insufficient to obtain toxicokinetic data. In the second case, transient renal failure could have slowed elimination of the herbicides. The only pertinent element was the presence of sulcotrione in the urine, indicative of the renal excretion of at least some of this compound in an unchanged form.
Animal toxicity studies have determined an acute median lethal dose (LD50) of >5000 mg/kg for sulcotrione in the rat. The mode of action of sulcotrione on tyrosine metabolism has also been studied in the rat. 2 It inhibits hepatic activity of the HPPD enzyme, the second most important catabolic enzyme for tyrosine after tyrosine aminotransferase (Figure 1). Other herbicides in the triketone family have been studied in more detail (Figure 2), such as mesotrione and nitisinone (2-[2-nitro-4-(trifluoromethyl)benzoyl]-1,3-cyclohexanedione (NTBC)). 4 –6 Their effects on HPPD have been established by experiments in both animals and humans, data which, to our knowledge, were not available with respect to sulcotrione. With all these compounds, however, the enzymatic inhibition of HPPD results in elevated plasma and intraocular levels of tyrosine.

Chemical structure of three substances in the triketone family. 3 NTBC: nitisinone.
During chronic administration, triketones are responsible for ocular toxicity in some susceptible species such as the rat and dog. These lesions have been widely studied in the rat and are characterised by corneal effects such as opacities, sometimes accompanied by neovascularization. 7 –9 These lesions are reversible within a few weeks of discontinuing the administration. It has been demonstrated that the ocular effects of triketones are correlated in a dose-dependent manner with a prolonged elevation of plasma tyrosine levels. Corneal opacities only occur in susceptible species after repeated administrations causing a significant and prolonged elevation of plasma tyrosine levels. Mice, rabbits and rhesus monkeys do not develop ocular lesions even after the administration for long periods of high doses of triketones because they can remove the excess tyrosine from the body more effectively than rats. 10,11
The responsibility of tyrosine for the onset of ocular lesions had previously been reported in rats when
NTBC and mesotrione have been tested in healthy adult volunteers who received a single, acute oral dose 3 of 1 mg/kg NTBC and 0.1–4 mg/kg mesotrione. NTBC peak plasma levels were attained rapidly (15 min–3 h later, depending on the pharmaceutical form) and were around 8 mg/l. The elimination half-life was 54 h. The plasma tyrosine concentration rose to as much as 1100 µmol/l, but normalised within a few weeks. No data are available on the metabolism and excretion of NTBC in humans.
In humans, the administration of mesotrione in a single oral dose of 4 mg/kg caused a mean peak plasma level of 4 mg/l, reached within 6 h and with an elimination half-life of 1 h. After 12 h, mesotrione was no longer detectable in the plasma. Mesotrione was excreted via the kidneys and was found in an unchanged form in the urine. Serum tyrosine levels reached 300 µmol/l, and the normalization was achieved within 48 h. The urinary excretion of tyrosine metabolites, HPPA and HPPL, was temporarily increased.
The therapeutic development of NTBC led to marketing of the medicinal product Orfadin® (MA granted in 2005 for France). Orfadin is indicated for the treatment of patients with confirmed diagnosis of hereditary tyrosinemia type 1, in combination with a diet low in tyrosine and phenylalanine. 13
Overall, the findings regarding our two cases agreed with experimental data obtained in animals, showing a relative safety of triketones following acute exposure. Ocular complications did not occur in either of our patients, despite elevated plasma sulcotrione levels. The clinical and biological findings in the second case were very probably linked to concomitant poisoning with chlorophenoxy herbicides. The assay of plasma tyrosine levels revealed a moderate elevation of serum tyrosine, but this was lower than the 500 µmol/l threshold recommended during treatment with NTBC. This moderate elevation was comparable with that found following the experimental acute ingestion of mesotrione in humans. Finally, as with mesotrione, the excretion of sulcotrione seemed mainly to be renal and in an unchanged form. A longer-term kinetic study relative to sulcotrione and tyrosine would have provided some useful data in these cases, but could not be achieved in this emergency context.
The long-term effects of triketone derivatives in humans (with the exception of NTBC) are not known. One might question the effects of such compounds in subjects with abnormal tyrosine metabolism. In particular – and although experimental reproductive studies in the mouse are reassuring – it is relevant to question the risks of neurological damage during fetal exposure in utero, such as that observed in infants with congenital type III tyrosemia.
Finally, the outcome of these two cases of acute intoxication was reassuring despite the high plasma sulcotrione concentrations reached. However, it seems prudent to monitor plasma tyrosine concentrations and to screen prospectively for corneal deposits if further acute intoxication events occur.
Footnotes
Acknowledgment
The authors thanks the assistance of Dr L. Gamelin.
Conflict of interests
The authors declared no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
