Abstract
The use of Lucilia sericata larvae on the healing of wounds in diabetics has been reported. However, the role of the excretion/secretion (ES) products of the larvae in treatment of diabetic wounds remains unknown. This study investigated whether application of the ES products of L. sericata on the wound surface could improve the impaired wound healing in streptozotocin-induced diabetic rats. Additional analysis was performed to understand proteome content of L. sericata secretome to understand ES contribution at the molecular level. For this purpose, full-thickness skin wounds were created on the backs of diabetic and control rats. A study was conducted to assess the levels of the ES-induced collagen I/III expression and to assay nuclear factor κB (NF-κB) (p65) activity in wound biopsies and ES-treated wounds of diabetic rat skin in comparison to the controls. The expression levels of collagen I/III and NF-κB (p65) activity were determined at days 3, 7, and 14 after wounding using immunohistological analyses and enzyme-linked immunosorbent assay technique. The results indicated that treatment with the ES extract increased collagen I expressions of the wound control and diabetic tissue. But the increase in collagen I expression in the controls was higher than the one in the diabetics. NF-κB (p65) activity was also increased in diabetic wounds compared to the controls, whereas it was decreased in third and seventh days upon ES treatment. The results indicated that ES products of L. sericata may enhance the process of wound healing by influencing phases such as inflammation, NF-κB (p65) activity, collagen synthesis, and wound contraction. These findings may provide new insights into understanding of therapeutic potential of ES in wound healing in diabetics.
Introduction
Diabetes mellitus is known to be associated with a variety of alterations in connective tissue metabolism, as a result of which diabetic face the problem of poor wound healing. Loss of collagen observed in diabetes may be due to decreased levels of synthesis or enhanced catabolism of newly synthesized collagen or both. 1 Impaired wound healing is associated with hyperglycemia in diabetes. 2 In addition, diabetic wounds are characterized by a severe inflammatory response which probably contributes to the collagen distribution and inhibition of healing progression. 3
Recent studies determined that the nuclear factor κB (NF-κB) gene is activated during the healing process which in turn may play a detrimental role in wound tissue remodeling and regeneration. NF-κB is also known to stimulate the production of collagen. Collagen fiber is the main component of the extracellular matrix of tissues and is the most abundant protein in skin tissue, and optimum deposition of collagen type I and III with their proper ratio is essential for connective tissue formation and scarring prevention. 4 During the wound healing process, the imbalance of collagen synthesis and degradation resulting in excess accumulation of dermal collagen can lead to the scar complications. 5 Collagen I is responsible for higher tensile strength, while collagen III is mainly found in early wound healing stages. 6 Expression of the type I collagen gene increases remarkably at later periods of in vivo wound healing and is proportional to that of the NF-κB gene. 7
Lucilia sericata larvae have been successfully used as medical maggots in the healing of wounds nowadays and help the healing process by the proteolytic digestion of necrotic tissue disinfection and stimulation of granulation tissue formation. 8 –12 On the other hand, the excretion/secretion (ES) treatment has been shown to efficiently debride wounds and help the healing process. However, the mechanisms underlying ES treatment-induced wound healing are not yet completely understood. Our study will provide a new approach with ES application on the process of healing in diabetic wounds. The present study, for the first time, determines the effect of ES treatment on collagen expression and its modulatory role on expression patterns of NF-κB activity, which are significant factors contributing to normal healing process. Additional analysis was performed to observe the proteome content of L. sericata secretome to understand ES contribution at the molecular level. We propose that this research fills a critical gap in the literature by presenting the first study that examines the molecular mechanism of wound healing by ES treatment in diabetic wound.
Materials and methods
Experimental protocol, wound creation, and application of treatment
Twenty healthy male Wistar rats aged 5–6 weeks and weighing 300–350 g were purchased from the Experimental Animal Center of Istanbul University (approval No. 2013/39). Rats were randomly divided into four groups with five rats in each group. First group consisted of control animals, second group was the streptozotocin (STZ)-induced diabetic control, and third and fourth groups consisted of diabetic and nondiabetic animals treated with topical ES. For formation of diabetic group, a single dose of 60 mg/kg STZ (Sigma, St. Louis, Missouri, USA) prepared in freshly dissolved in 0.9% saline solution was injected intraperitoneally (within 10 min) to induce diabetes. Three days later, blood samples were drawn from the tail of these rats to determine fasting blood glucose level. Plasma glucose levels more than 220 mg/dl were accepted diabetic and were included for the experiment. Three full-thickness excision wounds were made on the dorsum of each rat with sterile punch biopsy needles (12 mm2). The wounds were treated with ES according to their respective groups topically once daily for a period of 14 days for treatment groups. Control groups were treated with normal saline. The wounded areas were photographed and collected after wounding days 0, 3, 7, and 14. A half of wound was stored at −80°C for activation assay. The other side of wound was excised in 10% formalin for immunohistochemistry. After 14 days, the rats were killed by cervical dislocation under anesthesia.
Preparation of ES (maggot secretion)
ES was extracted in milli-O ultrapure water from sterile second and third instar larvae of L. sericata. ES was collected after incubating 2000 larvae/sterile beaker in 1 mL water for 60 min in ambient temperature. Then, 4 mL of water was added at 1-h intervals for 4 h with the larvae left to their secretions in water. 13
Protein identification and bioinformatic analysis
Protein concentration was measured using Bradford protein assay (Bio-Rad, California, USA). To prepare the sample for two-dimensional (2DE) gel electrophoresis, ReadyPrep 2-D Cleanup Kit was used (Bio-Rad, USA) as recommended by the manufacturer. The precipitate was resuspended in 300 µL 2DE rehydration buffer.
2DE gel electrophoresis
Three biologic replicates were used for 2DE gel electrophoresis which was performed as described previously. 14 In brief, approximately 600 µg of protein was loaded onto an immobilized pH gradient strips (17 cm, pH 3–10, nonlinear) (Bio-Rad, USA) via passive rehydration at 22°C for 16 h. The strip was run through a stepwise incremental voltage program (250 V for 20 min (linear), 10.000 V for 2 h (linear) and 55.000 V/h (rapid)) using Protean IEF system (Bio-Rad, USA). The strip was then subjected to a two-step equilibration in buffers containing 6 mol/L urea, 2% sodium dodecyl sulfate (SDS), 0.375 mol/L Tris.Cl pH 8.8, 20% glycerol, and 2% dithiothreitol (DTT) for the first step and the same buffer without DTT but with iodoacetamide (2.5%) for the second step. The strip was then subjected to second dimension separation using SDS polyacrylamide gel electrophoresis (12%). The gel was stained with Colloidal Coomassie stain and visualized with VersaDoc4000MP (Bio-Rad, USA). For automated spot detection, parameters used were sensitivity (13.8), spot size scale (3), and minimum peak intensity (258).
Image analysis and spot cutting
The image was analyzed with PD Quest Advanced software (Bio-Rad, USA). The outside edges of the image were cropped using the automated crop tool of PD Quest Advance Software. Stain speckles were filtered, the standardized areas of interest from all gels were matched and warped, and the quantity of each spot was normalized using the total valid spot intensity. Total spot numbers and volumes within the normalized area were determined by the automated analyses. A manual editing tool was used to inspect the determined protein spots detected by the software. The spots were cut using automated spot cutting tool, ExQuest spot cutter (Bio-Rad, USA), and disposed into a 96-well plate for protein identification.
In-gel tryptic digestion and bioinformatic analysis
In-gel tryptic digestion of the proteins was performed using an in-gel digestion kit following the recommended protocol by the manufacturer (Pierce Biotechnology, Rockford, lL, USA). Protein identification experiments were performed at Kocaeli University DEKART proteomics laboratory (http://kabiproteomics.kocaeli.edu.tr/) using ABSCIEX MALDI-TOF/TOF 5800 system. Before deposition onto an matrix-assisted laser desorption/ionization (MALDI) plate, all samples were desalted and concentrated with a 10-µL ZipTipC18 following the recommended protocol (Millipore, California, USA). Peptides were eluted in a volume of 1 µL using a concentrated solution of α-cyano-4-hydroxycinnamic acid (α-CHCA) in 50% acetonitrile and 0.1% trifluoroacetic acid in water and spotted onto the MALDI target plate. The time of flight (TOF) spectra were recorded in the positive ion reflector mode with a mass range from 400 Da to 2000 Da. Each spectrum was the cumulative average of 200 laser shots. The spectra were calibrated with the trypsin autodigestion ion peaks (m/z 842.510 and 2211.1046) as internal standards. Ten of the strongest peaks of the TOF spectra per sample were chosen for mass spectrometry (MS)/MS analysis. All of the peptide mass fingerprints were searched in the MASCOT version 2.5 (Matrix Science) using a streamline software, ProteinPilot (ABSCIEX, Framingham, MA, USA). A database was created using the entries in NCBI-NR databank (https://www.ncbi.nlm.nih.gov/refseq/about/nonredundantproteins/) and implemented into the MASCOT search engine. The search criteria included enzyme of trypsin; at least five independent peptides matched; at most one missed cleavage site; MS tolerance set to ±50 ppm and MS/MS tolerance set to ±0.4 Da; fixed modification being international carbamidomethyl (Cys) and variable modification being oxidation (Met); and peptide charge of 1+ and being monoisotopic. Only significant hits, as defined by the MASCOT probability analysis (p < 0.05), were accepted. Protein score is −10*Log(p), where is the p is the probability that the observed match is a random event. Protein score, which has a value of p < 0.05, is considered significant hit. Protein scores are derived from ion scores as a nonprobabilistic basis for ranking protein hits.
Measurement of NF-κB (p65) activity by enzyme-linked immunosorbent assay (ELISA)
TransAM assays were performed according to the manufacturer’s instructions (Active Motif, Carlsbad, California, USA).
Immunohistochemical analysis of collagens I and III
The formalin-fixed tissues were routinely processed by standard procedures and serial sections of 4-μm were cut. Immunohistochemical analysis was performed as described previously. 15 Immunoperoxidase staining was performed using Zymed-Histostain-Plus Bulk Kits (85-9043, Zymed Laboratories, S. San Francisco, CA, USA), including rabbit polyclonal collagen I antibody (Novus Bio. NB600-408, 1:100 dilution) and collagen III antibody (Novus Bio. NBP2-15946, 1:100 dilution). To determine the specificity of immunostaining, sections were incubated following the abovementioned procedure, except for omission of incubation with the primary antibody. Instead of primary antibody, control serum was used as negative control.
Semiquantitation of immunoperoxidase staining
Immunostaining was evaluated using a Leica DM2500 light microscope (Leica Microsystems, Wetzlar, Germany). Collagen I and collagen III immunostainings were analyzed in the immunoperoxidase-stained wound tissues in all fields of sections using a 40× objective. Immunperoxidase staining of wound tissues was analyzed and scored from 1+ to 3+ (1+ refers to weak, while 3+ to strong immunopositivity). This analysis was performed in a blind and randomized fashion of all collagens I and III stained sections.
Statistical analysis
All data were subjected to statistical analysis using GraphPad Prism 5 software (San Diego, California, USA) statistical package. The data were expressed as mean ± standard deviation (SD). p value of less than 0.05 was considered statistically significant.
Results
The rate of contraction of the control and diabetic wounds is shown in Figure 1. Wound contraction in diabetic rats was delayed compared to that in normal rats. After ES-treatment, the treated groups showed faster wound contraction compared to untreated groups.

Photographic representation of contraction rate on different days of control and ES-treated control, diabetic and ES-treated diabetic wounds. ES: excretion/secretion.
NF-κB (p65) activity
Nuclear fractions were isolated, and NF-κB (p65) levels were analyzed by ELISA. In this study, we compared the degree of NF-κB (p65) activity with wound healing between control and diabetic groups from 0 to 14 days prior to and after treatment with ES (Figure 2). Figure 2(a) shows that NF-κB (p65) activity was significantly decreased in ES-treated control wounds on the 3rd day (201.04 ± 51.02) compared to untreated control (233.7 ± 42.7), whereas it increased on 7th (162.01 ± 55.1) and 14th days (130.9 ± 24.7) compared to untreated control (respectively; 71.8 ± 24.5 and 50.8 ± 11.6).

(a, b) NF-κB (p65) activity in the skin wound healing of untreated and treated control and diabetic groups according to days. Data are given as mean ± SD. Control and ES-treated control groups. a p<0.001 versus day 0, b p<0.05 versus day 0, c p<0.001 versus day 3, d p<0.001 versus day 3 (a). Diabetic and ES-treated diabetic groups. a p<0.01 versus day 0, b p<0.05 versus day 3, c p<0.05 versus day 0, d p<0.001 versus day 7 (b). ES: excretion/secretion; NF- κB, nuclear factor κB.
In untreated diabetic group, NF-κB (p65) activity increased from day 3 (142.3 ± 13.9) to day 7 (185.9 ± 59.2) due to high level of inflammation. NF-κB (p65) activity decreased on third and seventh days after treatment with ES (respectively; 71.3 ± 22.2 and 151.6 ± 51.01) (Figure 2(b)).
Immunohistological analyses of collagens I and III
In this study, we compared the expression of collagens I and III associated with wound healing between untreated control and diabetic wounds before and after treatment of ES (on the 3rd, 7th, 14th days; Figures 3 and 4).

Immunohistochemical analysis of collagen I in wound healing process on third, fourth, and seventh days. (a, b, c) Untreated controls, (d, e, f) ES-treated controls, (g, h, i) untreated diabetics, and (j, k, l) ES-treated diabetics. E: Epidermis; sg: Sebaceous gland; hy: hypodermis; →: St.Granulosum; sw: sweat gland; ES: excretion/secretion.
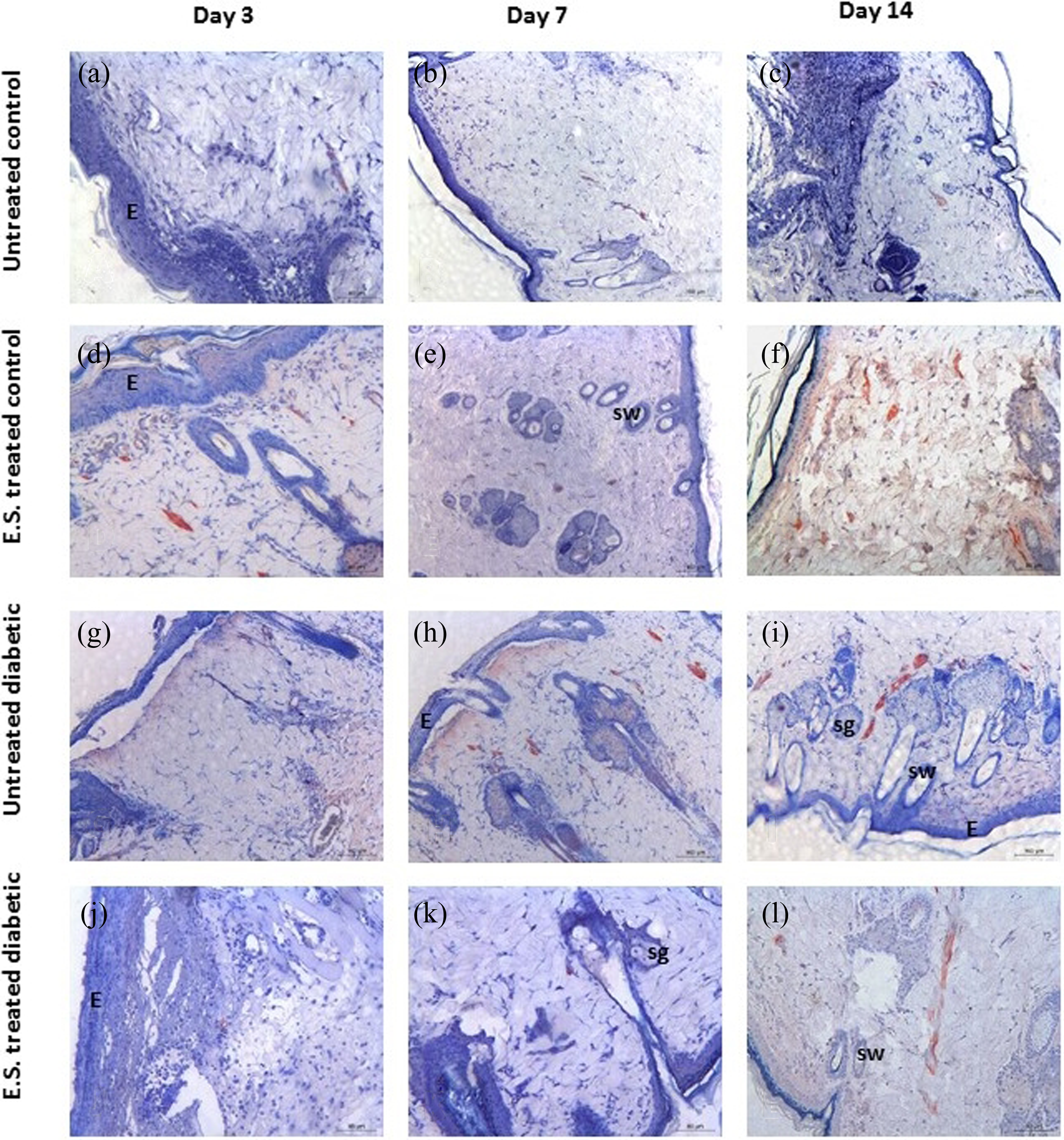
Immunohistochemical analysis of collagen III in wound healing process on 3rd, 4th, and 7th days. (a, b, c) Untreated controls, (d, e, f) ES-treated controls, (g, h, i) untreated diabetics, and (j, k, l) ES-treated diabetics. E: Epidermis; sg: Sebaceous gland; hy: hypodermis; →: St.Granulosum; sw: sweat gland; ES: excretion/secretion.
Collagen I immunohistochemistry
An increase in collagen I expression was detected on 7th and 14th days in the wound of control tissue (Figure 3(a) to (c)). The expressions of collagen I in ES-treated control wounds were linearly increased from day 0 to day 14. After treatment with ES, collagen I expression level in control wounds was the highest when compared to the untreated controls on the 3rd, 7th, and 14th days (respectively; p < 0.05; p < 0.001; p < 0.001) (Figure 3(d) to (f)) (Table 1).
Evaluation of collagen I immunopositivity.a

aData are given as mean ± SD.
b p < 0.05 versus untreated diabetic day 3, 7, 14.
c p < 0.05 versus untreated control day 3.
d p < 0.001 versus untreated diabetic, untreated control, ES-treated diabetic day 7.
e p < 0.001 versus untreated diabetic, untreated control, ES-treated diabetic day 14.
In untreated diabetic group, collagen I expression was increased on the 14th day (Figure 3(g) to (i)). In the case of ES-treated diabetic wounds, collagen I expression levels are higher than untreated diabetic groups on the 3rd, 7th, and 14th days (p < 0.05) (Figure 3(j) to (l)) (Table 1).
Collagen III immunohistochemistry
In the wound of control tissue, an increase in collagen III expression was detected on the third day while there was a decrease in expression level on the seventh day (Table 2) (Figure 4(a) to (c)). In ES-treated control wounds, collagen III expressions were decreased on the third day and increased on seventh day compared to untreated control groups (respectively; p < 0.001, p < 0.01) (Figure 4(d) to (f)).
Evaluation of collagen III immunopositivity.a

aData are given as mean ± SD.
b p < 0.001 versus untreated diabetic day 3.
c p < 0.001 versus untreated control day 3.
d p < 0.001 versus untreated diabetic day 7.
e p < 0.01 versus untreated control day 7.
In untreated diabetic group, collagen III expression was lower than that of day zero control. In untreated diabetic group, decrease in collagen III expression was observed on the third and seventh days (Table 2) (Figure 4(g) to (i)). After treatment with ES in diabetic wounds, collagen III expressions were decreased on the third and seventh days when compared to untreated diabetic group (p < 0.001) (Table 2) (Figure 4(j) to (l)). There were no statistically significant differences in the expression level of collagen III among the four groups on day 14 (Table 2).
2DE gel electrophoresis
2DE gel electrophoresis is a powerful tool that can be used for partial or complete profiling of the protein content of ES (Figure 5). Our analysis of the ES using large size gels within a pH range of 3–10 revealed the presence of at least 400 protein spots. Interestingly, the majority of separated protein spots fell into the focusing region of high pH (pH > 8.0). Some of these protein spots were expressed less and failed to be identified by MALDI-TOF/TOF analysis. However, we were able to excise the major protein bands from the gels and identify them with confidence. Here we report the protein hits, which produced peptides with ion scores (Table 3). There were five proteins that were named after the MASCOT search.

2DE gel electrophoresis of ES. 2DE: two-dimensional gel electrophoresis; ES: excretion/secretion.
The protein hits, which produced peptides with ion scores.a

aThere were five proteins that were named after the MASCOT search.
Discussion
In present study, we examined the parameters such as expression of collagens I and III and NF-κB activity in wound healing impairment in STZ diabetic rats. It is known that collagen is the predominant extracellular protein in the granulation tissue of a healing wound, and there is a rapid increase in the synthesis of this protein in the wound area soon after injury. 16 Analyses of the collagens I and/or III in wounds or in experimentally induced granulation tissue in diabetic rats were first reported in 1975. 17,18 –20 Collagen III level is known to be increased during early stages of healing. 17 Clore et al. determined that the synthesis of type III protocollagen begins as early as 10 h after wounding. 21 This finding could be an explanation for the large proportion of type III collagen detected during days 1 and 2 but transient decrease in its proportion later during days 3 and 4 during normal wound healing in human. 22 Similarly, we observed that the expression of collagen III increase on third day and decreased healing of wounds of untreated control and diabetic rats. These results show that during early stages of healing, type III collagen is synthesized which will then be replaced by type I collagen during later stage of wound repair. 23 The data in the present study show that collagen I expression is increasing linearly in all groups from day 0 to day 14. However, in this time period, increase in the collagen I expression in untreated diabetic group is less than that in normal control. These results show that ES induce collagen type I expression but inhibit collagen type III expression, accompanied by improved wound healing in STZ diabetes. According to our results, wound contraction in diabetic rats was delayed compared to that in normal rats, whereas treatment with ES resulted in much faster wound healing in untreated control and diabetics.
ES was shown to augment the process of dermal wound healing by its ability to increase collagen I synthesis. The improved collagen synthesis in diabetes is a consequence of early events during the repair process. 24 According to our results, it may be assumed that the high expression of collagen I by ES treatment may contribute to a more accelerated wound healing in diabetic rats. We investigated the influence of ES on the collagens I and III expression in granulation tissues of healing wounds in untreated diabetic and control rats. After treatment with ES, in the diabetic wounds, expression of collagen I increased in a time-dependent manner (from 3rd to 14th day) when compared to that of untreated diabetics. Type III expression in STZ-diabetic wounds is specifically decreased from 3rd to 14th days after treatment with ES. These results show that ES enhances the process of wound healing by influencing the collagen expression in STZ-induced diabetic rats.
Recent studies showed that several medical plants have a positive influence on the healing of dermal wounds in diabetic rats. Aloe vera treatment of wounds in diabetic rats enhances the process of wound healing by influencing collagen synthesis, inflammation, and wound contraction. 16 Acalypha indica extract also enhances the process of dermal wound healing by increasing collagen synthesis in rats. 25 According to Kwon et al., B glucan from Sparassis crispa increases type I collagen synthesis in dermal fibroblasts and promotes wound healing in STZ-induced diabetic rats. 26 Wei et al. showed that glucagon treatment stimulates both type I and type III collagen gene expression in a time- and dose-dependent manner. They explained that glucagon-induced fibroblast collagen biosynthesis is mediated through NF-κB-dependent mechanism. 27 In addition, hyperglycemia and FFA-induced oxidative stress lead to the activation of stress-sensitive signaling pathways, including NF-κB. 28 These data indicate that activation of NF-κB is an initial signaling event that leads to cellular dysfunction and damage. 29 It was shown that exposure to hyperglycemia initially activated NF-κB. 30 In this study, we showed that NF-κB activity was specifically increased on 7th and 14th days of treated diabetic groups. In addition, NF-κB (p65) activity was increased on 3rd, 7th, and 14th days in treated control groups. The results demonstrate that ES treatment may have a beneficial influence on the various phases of wound healing like collagen synthesis, NF-κB (p65) activity, and contraction resulting in faster healing.
Larval therapies have been used in the treatment of unhealed wounds for many years. 31,32 The larva act by both biochemical and mechanical means to debride necrotic tissue, reduce inflammation, and stimulate granulation tissue in wounds. The most commonly used species is L. sericata. 33 L. sericata larvae are used in maggot debridement therapy which is a traditional approach that has recently been accepted. Disinfection, debridement, and the acceleration of wound healing are promoted by maggot excretion products which include many different proteases. According to Pöppel et al., 34 L. sericata chymotrypsin-like serine protease seems to be an ideal candidate for the development of topical drugs for wound healing applications.
Research into the debridement mechanisms underlying maggot therapy has revealed that maggots secrete digestive enzymes, such as leucine aminopeptidase, carboxypeptidases A and B, 35 collagenase, 36,37 and serine proteases (trypsin-like and chymotrypsin-like enzymes). 35,38 Four proteolytic enzymes, including two serine proteases, a metalloproteinase, and an aspartyl proteinase, were identified. A chymotrypsin-like serine proteinase exhibited excellent degradation of extracellular matrix (ECM) components laminin, fibronectin, and collagen types I and III 39 and may therefore play an important role in the digestion of wound matrix and effective debridement. Proteinase activity may also have an effect on wound area without necrotic tissue remodeling processes. Previous reports have shown that the larvae of L. sericata have been used as medical maggots in the healing of wounds in diabetics. 8,9 Therefore, elucidation of the protein content of ES may be important to understand the molecular mechanisms that trigger the wound healing. The attribution of a name or a function to each identified protein spot was not possible, since many of the spots identified were from the open reading frames of Lucilia cuprina genome-annotation study. 35 They were annotated as hypothetical proteins without a known function. In this study, we have demonstrated that at least some of those annotated open reading frames are not only expressed at a high level but also secreted as a part of the secretome. One of the proteins identified was fatty acyl-CoA reductase. This protein is extensively characterized in humans and several other organisms. 36 –39 It is an enzyme that catalyzes the reduction in saturated fatty acyl-CoA using NADP+ and is generally involved in lipid metabolism. In humans, this enzyme is predicted to be part of an integral component of the membrane and is not known to be a secreted protein. 40 We observed two protein spots on 2DE gels that belonged to this protein. However, both spots failed to match the expected molecular weight (MW: 57,000 Da). Since fatty acyl-CoA reductase is not expected to be a secreted protein and is a transmembrane protein, it was surprising to see the presence of this protein in ES. Until the structural and enzymatic properties of this enzyme in L. cuprina are elucidated, we cannot provide a logical explanation for the presence of this enzyme in ES. It may very well be secreted as a part of a large protein complex or may simply leak as a part of a lipid remnant.
The second protein identified was 60 S ribosomal protein large subunit (RPL22). This is a cytoplasmic protein and has a role in translation elongation. However, recent evidence suggests that ribosomal proteins are also capable of performing regulatory functions that impact normal and pathological conditions. 41 The challenge of understanding the presence of RPL in ES is a conundrum that needs attention.
The third protein identified was peritrophin, a 38-kDa larval protein. 42,43 Peritrophin is a membrane protein that may have a role in the maintenance of peritrophic membrane structure and determination of the porosity of the peritrophic membrane. 44 Peritrophic membrane is a semipermeable noncellular structure and helps intactness of the peritrophic matrix which functions in the digestion of the available food. The presence of peritrophin in ES has not been reported. Whether or not its presence affect wound healing is a matter of investigation. Purified peritrophin extracts may be tested to elucidate the contribution of this protein in wound healing.
The observation of conserved oligomeric Golgi complex subunit-2 (COG-2) in ES was surprising, since this protein is supposed to be part of a Golgi complex which is required for Golgi morphology and function. 45 As a transmembrane protein, COG-2 is expected to be embedded into the Golgi membrane and should not be secreted. However, it is also possible that during high levels of protein trafficking, COG-2 may unwillingly leak into ES as part of a secretory vesicle.
The last protein identified was Nipped-B. This protein possesses a role in sister chromatid cohesion and aids enhancement of promoter–enhancer communication. 46 Overall, Nipped-B plays a role in the developmental regulation process. The localization of Nipped-B is supposed to be nucleus because Nipped-B contains a bipartite nuclear localization signal. Why we observe Nipped-B in ES and how we should explain its presence in ES is a matter that needs to be explored.
Overall, two key points may be underlined from the preliminary proteome analysis of this organism: (i) there is noticeable number of hypothetical proteins present in ES and (ii) some of the identified proteins are not part of the secretome, although they are present in ES. Future work will help us to understand why these proteins are present/observed in ES.
In conclusion, the present results indicate that ES products of L. sericata increased NF-κB activity, collagen synthesis, and wound contraction. Therefore, the use of ES may be extended to the clinical setting and proves an effective marker of wound healing in patients with diabetes, in which skin lesions often slow down the healing.
Footnotes
Acknowledgements
This study was supported by the Research Fund of The University of Istanbul (Project Number 35048). We appreciate this support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
