Abstract
Several mechanisms for the pathogenesis of diabetic complications have been proposed, one of which is abnormal zinc (Zn) homeostasis. Zn is necessary for proper liver function since it has important antioxidant, anti-inflammatory, and antiapoptotic properties. We aimed to investigate whether or not Zn has morphologically protective effect on diabetes-induced liver damage in rats. In addition, we have investigated the role of Zn supplementation on apoptosis, lipid peroxidation levels, and the distribution of metallothionein (MT) in diabetic liver tissue. Wistar albino rats were divided into four groups: control, Zn, diabetic, and Zn-diabetic group. Experimental diabetes was induced by a single-dose streptozotocin intraperitoneally and Zn was administrated via gastric gavage tube for 6 weeks. MT expressions were showed with immunohistochemical staining and terminal deoxynucleotidyl transferase-mediated dUTP nick end labeling assay was used for apoptosis. Also, Zn, MT, and malondialdehyde (MDA) levels were determined in liver of rats. MDA levels of the Zn-supplemented diabetic group was less than the diabetic group though MT levels were increased. The number of apoptotic cells per unit area was found to be significantly decreased in this group. In the Zn-supplemented diabetic group, fibrotic tissue density and the collagen tissue density were observed less than the diabetic group. MT immunoreactivity was observed less in Zn-supplemented diabetic group. In conculusion, the present study indicated that Zn has a potential in preventing or even repairing effect against diabetic damage of the liver cells by increasing expression of MT and by reducing the apoptotic cell death and the oxidative stress.
Introduction
Diabetes is a multi-organ disease that can cause serious complications in many organs. In diabetic patients, disability and death are seen frequently in unsuitable conditions. 1 It has been reported that the liver is an organ that is seriously damaged in diabetes, so hepatic damage is major complication of diabetes. 2,3 One of the various mechanisms suggested for the pathogenesis of diabetic complications is the disruption of zinc (Zn) homeostasis. 4 Also, it has been reported that Zn is abnormally metabolized in diabetes. 5 However, the regulation of Zn deficiency in diabetic patients leads to balanced oxidant/antioxidant levels. 6 The liver is the main organ responsible for the Zn metabolism which can be affected by liver function. 7 Zn is required for normal liver function because of its antiapoptotic, anti-inflammatory, and antioxidant properties. 8 It has been reported that lack of Zn causes oxidative liver damage and/or signal transduction disorders in liver. 9,10 Its deficiency affects cell function and proliferation and survial against the disease via changing the redox state of cell and oxidant-sensitive transcription factors. Zn has been found to be protective against oxidative liver injury. 11
The role of Zn on the apoptotic process is to reduce oxidative stress and increase expression of metallothionein (MT). 12 Truong-Tran et al. 13 reported that the normal levels of Zn inhibit apoptosis, whereas unbalanced homeostasis of Zn induce apoptosis. Nutritional Zn imbalance has been suggested to cause apoptosis in liver, kidney, testis, and many cell types. 14 Zn deficiency induces apoptosis with caspase activation and increased oxidative stress. 13
Zn effectively ameliorates diabetes-related complications in various animal models. 15 It is also an effective inducer of gene- and protein expression of MT, a potent antioxidant. 16 MTs are a group of proteins that are rich in cysteine which regulates the homeostasis of essential metal ions such as copper and Zn, has 6–7 kDa molecular weight. 17 It is also an efficient free radical scavenger. 18
Liver is the basic organ for MT synthesis in the body. However, pancreatic islets and adipose tissue also secrete certain amount of MT. 19,20
Induction of systemic MT synthesis by Zn supplementation may be a potential approach in preventing diabetes-induced complications in multiple organs and the subject should be clarified in experimental animal model. Hepatoprotective properties of Zn has not been fully determined. Zn supplementation might provide protection against acute and chronic liver injury in experimental animal models.
Thus, in the present study we aimed to investigate whether or not Zn has morphologically protective effect on diabetes-induced liver damage in rats. In this study, we also investigated the effects of Zn administration on apoptosis, MT and lipid peroxidation in liver tissue of diabetic rats. The obtained results are discussed with the intent of explaining the mechanism of action of Zn on diabetic hepatic injury.
Materials and Methods
Animals
Wistar-type albino male rats weighing approximately 250–290 g were purchased from Istanbul University, Institute of Experimental Medicine (DETAE). The animals had free access to standard rat chow and drinking water. The animals were maintained and used in accordance with the Animal Welfare Act and the Guide for the Care and Use of Laboratory Animals from Istanbul University.
Induction of diabetes
The animals were made diabetic with an intraperitoneal injection of streptozotocin (STZ; Sigma-Aldrich, St Louis, Missouri, USA) at a dose of 60 mg/kg body weight freshly dissolved in 0.9% sodium chloride. To confirm the diabetic state, blood glucose concentration was measured 3 days after STZ injection. The rats with blood glucose 350 mg/dL were considered to be diabetic.
Experimental groups
A total of 32 rats were assigned into four experimental groups (n = 8 in each group), control group: non-diabetic, non-supplemented rats receiving 0.9% w/v saline via gastric gavage tube; Zn group: rats were administered Zn (30 mg/kg/day) via gastric gavage tube; diabetic group: diabetes was induced in the rats by a single-dose (60 mg/kg) intraperitoneal injection of STZ; Zn–diabetic group: rats were supplemented with Zn (same as Zn group) after STZ application.
During the experimental period (6 weeks), blood glucose levels and body weights were measured. Blood samples were obtained from the tail vein of each rat. The blood glucose levels were measured on the 1st, 21st, and 42nd days of the experiment using glucose test reagent strips and a glucometer from Accu-Chek Active (Roche, Germany). After 42 days of treatment, the animals were killed under ether anesthesia, and liver tissue samples were removed for histological examinations.
Histological analyses
Livers from the normal and experimental animals were fixed in 10% buffered formalin and were processed for paraffin sectioning. Five micrometer thick paraffin-embedded liver tissues were placed onto gelatin-coated slides, dewaxed and then rehydrated. Sections were stained with Masson and sirius red for liver collagen to evaluate the pathophysiological changes under light microscope using a 40× objective (Leica DM2500 light microscope; Leica Microsystems, Wetzlar, Germany).
Immunohistochemistry
Immunohistochemical analysis was performed as previously described. 21 Immunoperoxidase staining was performed using Ultra Vision Antibody Detection System (LabVision Corporation, Fremont, California, USA) kits, including mouse monoclonal MT antibody (Abcam; ab12228, 1:100 dilution). Immunostaining procedures were carried out following the manufacturer’s instructions. To determine the specificity of immunostaining, sections were incubated following the above procedure, except for omission of incubation with the primary antibody. Instead of primary antibody, control serum was used as negative control. Immunostaining was evaluated using a Leica DM2500 light microscope (Leica Microsystems, Wetzlar, Germany).
TUNEL assay
Terminal deoxynucleotidyl transferase-mediated dUTP nick end labeling (TUNEL) assay, a technique, in which DNA fragmentation is detected by labeling the terminal end of nucleic acids, has also been performed to confirm the apoptosis of liver tissues. For TUNEL staining, slides were stained with the reagents supplied by ApopTag Peroxidase in situ Apoptosis Detection Kit (ApopTag Plus, In Situ Apoptosis Detection Peroxidase kit, S7101-KIT, Chemicon, Japan). TUNEL assay was performed as described previously. 21
Apoptotic index
Marked apoptotic cells were counted under high-powerfields (40×) with a light microscope (Leica DM2500 light microscope, Leica Microsystems). Nuclei stained with methyl green were evaluated as healthy while cells with brown nuclear staining were considered TUNEL positive. All TUNEL-positive cells in 12 different unit areas were counted on the cross-sections by a blinded researcher. These 12 fields were randomly selected from liver slides. Average cell per unit area number for each set of specimens in each group was calculated and compared. 21
Tissue preparation
Approximately 1 g of liver was homogenized in 2 mL pH 7.4 phosphate-buffered saline. The liver homogenates were then centrifuged at 10,000 × g for 15 min at 4°C, and the resultant supernatant was used for the determination of malondialdehyde (MDA) levels. 5
Assessment of lipid peroxidation (MDA)
Lipid peroxidation was established by measuring the MDA levels in liver homogenates by means of the thiobarbituric acid reactivity assay using a method previously described using an extinction coefficient ε = 1.56 × 105 M−1 cm−1 at a wavelength λ = 532 nm. 22,23 The tissue MDA levels were expressed as micromole per gram protein. The protein content in liver was measured by method of Lowry et al. 24 with bovine serum albumin as the standard.
Measurement of MT and Zn Levels in Liver
The level of MT (ng/g) was measured with the enzyme-linked immunosorbent assay using an MT Kit (Cusabio Biotech, CSB-E11315r; China). The liver tissue Zn levels were determined with an inductively coupled plasma optical emission spectrometer (ICP-OES, Thermo Fisher Scientific, Waltham, Massachusetts, USA) after wet ashing with a mixture of nitric and perchloric acids. Approximately 0.5 g of liver tissue was obtained from each animal for this purpose. The tissue concentrations of Zn are expressed as microgram per gram wet weight. 23
Statistical analysis
Values were expressed as mean ± SD. The SPSS statistical program (v 10.0, SPSS Inc. Chicago, Illinois, USA) was used for the statistical treatment of the data. The Mann–Whitney U test was used to establish the significance of differences among the four groups. The value of p < 0.05 was considered as statistically significant.
Results
Blood glucose levels and body weight changes
Blood glucose levels in diabetic group was significantly higher than control group during the experiment (p < 0.001). A significant reduction compared to the diabetic group was seen in Zn-diabetic group (p < 0.05; Table 1). The body weight of diabetic group and Zn-diabetic group decreased significantly compared to the control group (p < 0.05).
Final blood glucose levels and body weights of groups.a

Zn: zinc.
aValues are mean ± SD.
b p < 0.001 versus controls.
c p < 0.05 versus diabetic group.
Histopatholgic findings
In diabetic group dilatation in sinusoids (thick arrow), hepatocellular vesicular vacuolization (asterisk), and local fibrotic areas (thin arrow) among the hepatocytes was observed with Masson dye (Figure 1(c)) compared to the control groups (Figure 1(a) and (b)). Less sinusoidal dilatations (thin arrow) and vesicular vacuolization were observed in Zn-diabetic group than the diabetic group (Figure 1(d)).
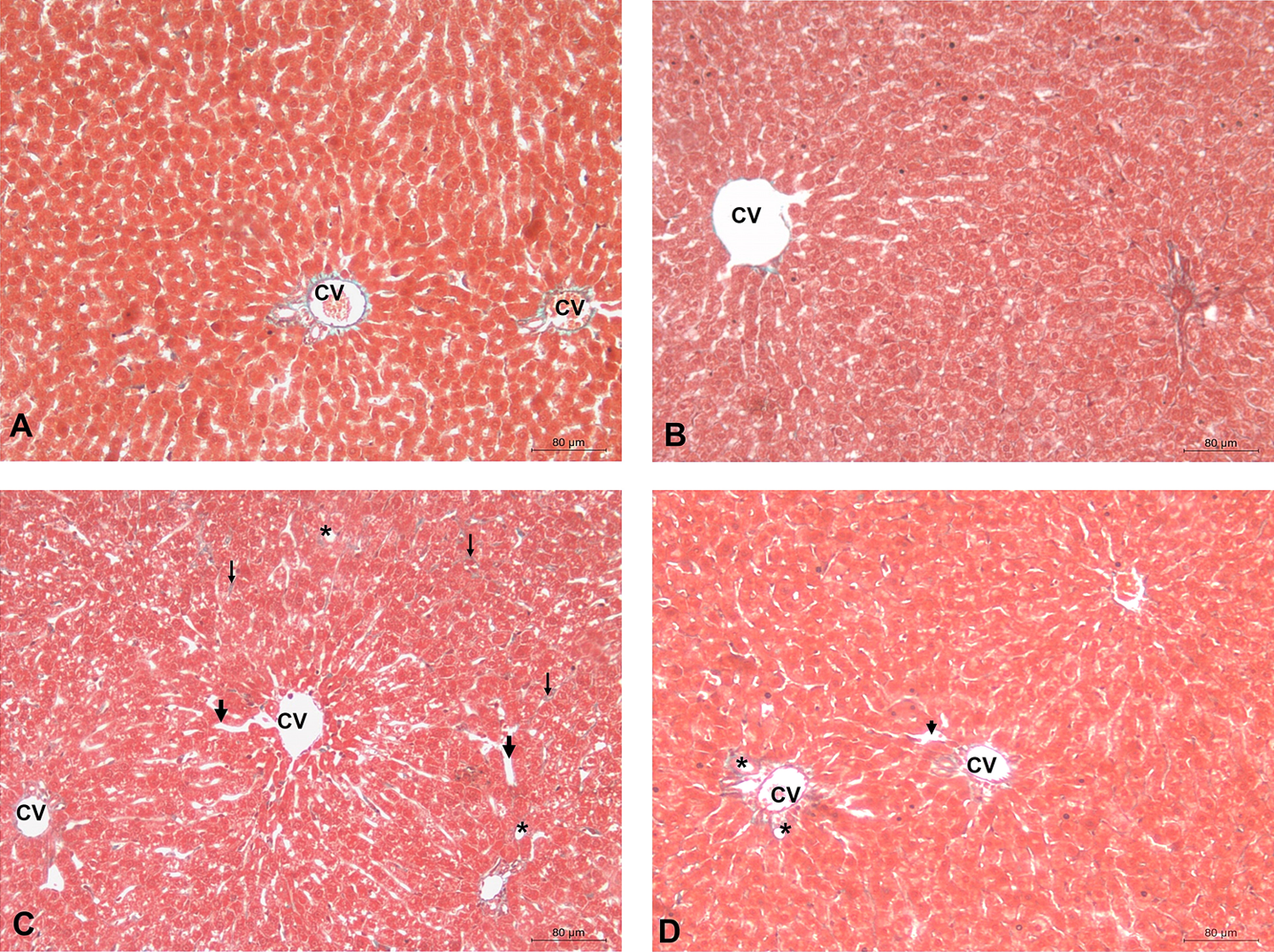
Light photomicrographs of Masson-stained sections of liver. Control (a) and Zn control (b) groups. In the diabetic group dilatation in sinusoidal (thick arrow), hepatocellular vesicular vacuolization (asterisks) and local fibrotic areas (thin arrow) among the hepatocytes were seen (c). In the Zn-diabetic group, sinusoid dilatations (thin arrow) and vesicular vacuolization were less visible (d). Bar: 80 μm. Zn: zinc.
Sirius red staining showed more intense red staining, a sign of collagen deposition, and fibrosis in diabetic rats. The increase in collagen accumulations (thin arrow) among the hepatocytes, dilate sinusoids (thick arrow), vesicular vacuolations (asterisk) and damage in the liver cells was seen in the diabetic group compared to the control group (Figure 2(c)). In Zn-diabetic group the local hepatocyte damage was observed but collagen accumulation wasn’t seen compared to the diabetic group (Figure 2(d)).

Light photomicrographs of sirius red-stained sections of liver. Control (a) and Zn control (b) groups. The increase in collagen accumulations (thin arrow) among the hepatocytes, sinusoidal dilatation (thick arrow), vesicular vacuolations (asterisks), and damage in the liver cells were seen in the diabetic group (c). In Zn-diabetic group the local hepatocyte damage was seen but collagen accumulation wasn’t seen compared to the diabetic group (d). Bar: 80 μm. Zn: zinc.
MT immunoreaction
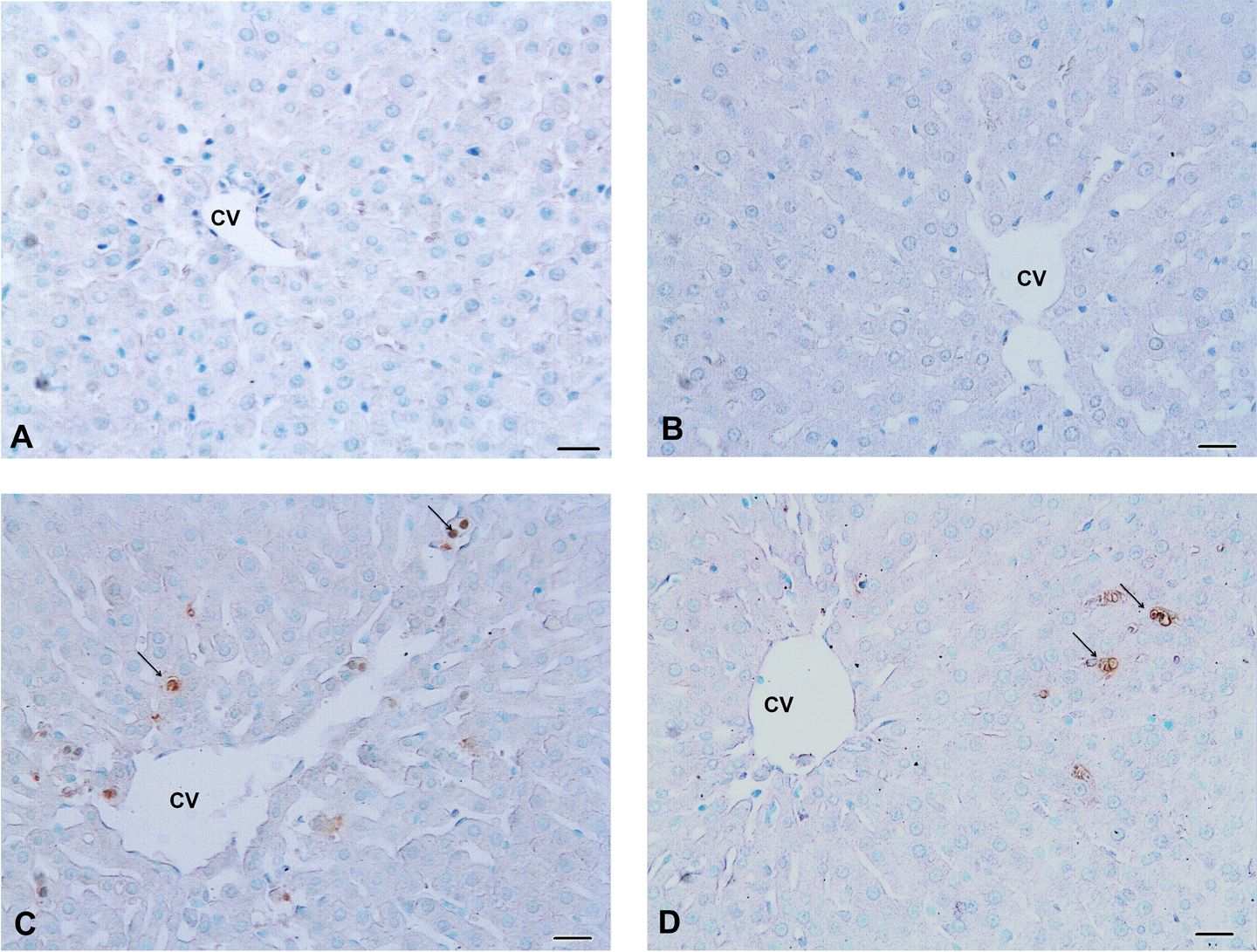
The MT immunoreactivity is shown in Figure 3. In the control groups it was observed in the cytoplasm of few hepatocyte (Figure 3(a) and (b)). It was lower in the hepatocytes of the diabetic rats relative to controls (Figure 3(c)). In the Zn-diabetic group, intense immunostaining of MT appeared in the cytoplasm of hepatocytes and sinusoids compared to the diabetic group (Figure 3(d)).

MT immunostaining (Brown stained) of liver sections. In the control (a) and Zn control (b) groups were observed a few immunopositive hepatocyte. The Zn-diabetic group (d) showed increased MT-immunoreactivity in the hepatocytes compared to diabetic rats (c). Bar: 40 μm. MT: Masson’s trichrome; Zn: zinc.
TUNEL assay
TUNEL-positive apoptotic hepatocytes in diabetic group (Figure 4(c)) and the number of apoptotic cells per unit area was found to be increased significantly (p < 0.01) compared to the control group (Figure 4(a) and Table 2). However, the number of apoptotic cells per unit area compared to the diabetic group revealed a significant decrease (p < 0.01) in the Zn-diabetic group (Figure 4(d) and Table 2).
TUNEL-stained liver sections. Cells with nuclei that stained dark brown were considered to be TUNEL positive (arrows). Control (a) and Zn control (b) groups. In the hepatocytes of diabetic group increased apoptotic cell numbers were observed compared to control groups (c). In the Zn-diabetic groups, decreased apoptotic cell number was seen compared to the diabetic group (d). Bar: 40 μm. TUNEL: terminal deoxynucleotidyl transferase-mediated dUTP nick end labeling assay; Zn: zinc.
Immunohistochemical distribution of metallothionein and apoptotic cell count in the liver sections.a

Zn: zinc; MT: metallothionein. −: non-staining; +: weak staining; ++: moderate staining; +++: strong staining.
aValues are mean ± SD.
b p < 0.001 versus control.
c p < 0.01 versus diabetic group.
Liver Zn and MT concentrations
In the diabetic group Zn (5.34 ± 0.57) and MT (26.05 ± 5.12) concentrations were significantly decreased compared to control group (Zn: 6.60 ± 0.70 and MT: 104.62 ± 14.74; p < 0.01 and p < 0.001, respectively). In the Zn-diabetic group, Zn (6.87 ± 1.35) and MT (70.01 ± 19.62) concentrations were significantly increased compared to diabetic group (p < 0.05; Figures 5 and 6).

Liver tissue Zn levels in the four study groups (mean ± SD, n = 8). Letter “a” represents p < 0.01 versus control group; b represents p < 0.05 versus diabetic group. Zn: zinc.
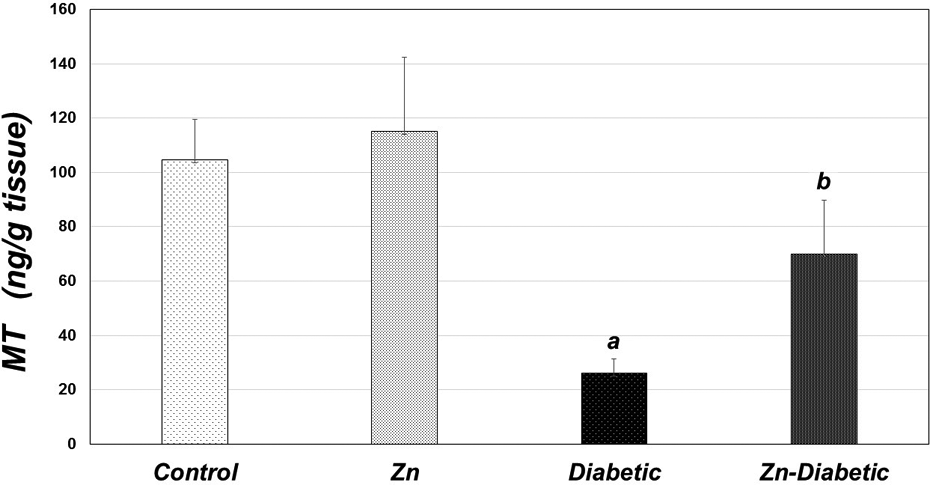
Liver tissue MT levels in the four study groups (mean ± SD, n = 8). Letter “a” represents p < 0.001 versus control group; b represents p < 0.05 versus diabetic group. MT: metallothionein.
Lipid peroxidation Levels (MDA)
In the diabetic group (14.60 ± 0.57), MDA levels were significantly increased compared to control group (11.17 ± 1.34; p < 0.01), whereas decreased significantly in Zn-diabetic group (12.63 ± 0.83; p < 0.05; Figure 7).

Liver tissue lipid peroxidation (MDA) levels in the four study groups. (mean ± SD, n = 8). Letter “a” represents p < 0.01 versus control group; b represents p < 0.05 versus diabetic group. MDA: malondialdehyde.
Discussion
Hepatic damage in diabetes also noted major secondary complications as well as hyperglycemia, hyperlipidemia, cardiovascular complications, nephropathy, and retinal damage. 25 The present study investigated the effects of Zn supplementation on apoptosis, lipid peroxidation levels, and MT expressions in liver of diabetic rats. Furthermore, the study also investigated the protective role of Zn supplementation on diabetic-induced liver damage in rats. We detected weight loss and an increase in serum glucose levels in the diabetic group compared to the control group. Our results showed that serum glucose levels in diabetic rats were decreased after treatment with Zn. This finding was consistent with the results of previous studies. 26,27
Fibrosis is considered as an excessive accumulation of connective tissue in parenchymal organs. 28 Liver fibrosis occurs in many of the chronic liver diseases which are characterized with the excessive accumulation of extracellular matrix proteins including collagen. 29 In the present study, in the diabetic group, sinusoidal dilatations and macrovesicular vacuolizations in the hepatocytes were observed, less macrovesicular vacuolizations, sinusoidal dilatations, and much more normal parenchymal liver tissue were observed in the control groups. In the Zn-diabetic group, less sinusoidal dilatations and macrovesicular vacuolizations were observed than the diabetic group. In the diabetic group, fibrosis was observed around the central vein and the sinusoidal areas were observed in the liver tissue. In the Zn- diabetic group, these findings were less than the other groups.
Induction of oxidative stress stimulates hyperglycemia and intrinsic pathways-mediated apoptosis in diabetic rats. 3 Excessive increase in lipids in hepatocytes leads to hepatocyte apoptosis and liver damage and then hepatic steatosis development has been reported. 30 Qiang et al., 31 reported that increased lipid levels related with hyperglycemia and hyperlipidemia caused hepatocyte apoptosis. In the present study, high blood glucose levels were observed in the diabetic group. In same group, the number of apoptotic cells per unit area of liver sections by TUNEL method was significantly increased compared to the control groups. This increase in liver was consistent with morphological disruption.
Antioxidant treatment is useful in diabetes and -disease related with oxidative stress. 32 Zn is an immunoregulator, antioxidant, cytoprotectant, antiapoptotic, anti-inflammatory, and growth cofactor. 33 Zn can directly eliminate hydroxyl radicals, hydrogen peroxide, peroxyl radicals, and free oxygen radicals such as hypochlorous acid. The induction of MT synthesis and Cu/Zn superoxide dismutase stabilization are long-term antioxidant effects of Zn. 19
Supplementation of Zn regulated the levels of lipid peroxidation in diabetic tissues, suggesting that Zn plays an essential biochemical function that retards the oxidative processes and it serves as a potential antioxidant. 10,23 Zn supplementation may enhance the antioxidant capacity of diabetic subjects or prevent diabetes-induced oxidative damage either directly through antioxidant action or indirectly by binding to the cell membrane. 34 In a clinical study, 11 it was found that in healthy individuals Zn supplementation reduced the lipid peroxidation caused by oxidative stress. In this study, as a marker of liver oxidative damage, the levels of MDA in liver of diabetic group were significantly higher than the levels in control group, whereas significantly decreased in diabetic group treated with Zn. Our results are in agreement with those of recent studies 35 that report an increase in liver tissue lipid peroxidation by hyperglycemia-induced glucose autoxidation and glycation of proteins. It has been reported that Zn also improves the oxidative stress in diabetic patients by reducing chronic hyperglycemia. 36 Zhu et al. 27 showed that Zn could protect the liver from oxidative damage and improve the liver function of diabetic rats. In addition, authors suggested that Zn may reactivate the antioxidant system, thus exerting antidiabetic activity.
In unbalanced Zn hemostasis, either too high or too low, are associated with induction of apoptosis, while sufficient Zn levels inhibit apoptosis. It has been suggested that Zn deficiency promotes apoptosis by increasing oxidative stress and activation of caspase. 37 Liver plays an important role in Zn hemostasis. Zn has different concentrations in various tissues. Serum Zn levels are a small percentage of the total body Zn concentration (only 0.1%). Muscles, bones, prostate, liver, and kidney contain a large amount of Zn content in the body. 38 Reduction in hepatic Zn levels has been associated with hepatic dysfunction. Zn regulates the MT production via binding directly and reversible to the Zn finger domain of metal response element-binding transcription factor-1. This protein adopts a DNA-binding authentication and translocates to the nucleus, where it binds to the metal response elements in gene promoters leading to enhance transcription. 39
It has been suggested that decreased MT activity makes people more sensitive against oxidative damage and hyperglycemia. 40 During diabetes, MT which is the free radical scavenging such as glutathione reported to decrease. Ayaz et al. 41 found decreased liver MT protein levels in STZ-diabetic group compared to healthy control group during the 5-week diabetic period. This kind of diabetes-induced decrease makes the liver tissue more sensitive to oxidative stress. MT immunoreactivity has been found mainly in cytoplasm and some nucleus of hepatocytes in perilobular area of livers in healthy animals. 32 In the present study, MT immunoreactivity has been seen moderate in few hepatocytes of control group. We detected that MT immunoreactivity was significantly decreased in the diabetic group compared to all of the other groups, and Zn-diabetic group had significant increase in the liver relative to the diabetic group. Intensive MT immunoreactivity in sinusoidal area of hepatocytes was observed in Zn-diabetic group. In parallel with the positive effects in MT, the changes in diabetic rat liver tissue morphology was improved by Zn supplementation. This is consistent with a previous studies that the expression of MT was increased in liver after Zn supplementation. 42 The synthesis of MT proteins has been proven to be induced by Zn, probably both MT and Zn could play important roles to prevent some diabetic complications. 43 Wang et al. 35 showed that oxidative stress marker MDA levels in liver were significantly reduced by Zn treatment, which was associated with an increase in liver Zn levels and MTs protein expressions.
The results showed that in the liver, Zn and MT were significantly decreased in diabetic group compared with the healthy control group. The concentrations of Zn and MT were significantly increased in Zn-diabetic group than the diabetic group. Consistent with these findings the number of apoptotic cells per unit area and MDA levels were significantly decreased in Zn-diabetic group than diabetic group. We demonstrated in an experimental animal model that Zn supplementation prevented liver damage and ameliorated pathological and MT changes in the liver of diabetic rats, resulting in retardation of the progression of diabetic liver damage. Histological and MT immunostaining assessment revealed that damage in liver caused by STZ was markedly decreased by administration of Zn.
Conclusion
Consequently, our study suggests that Zn supplementation may have beneficial effect against the liver tissue damage induced with hyperglycemia in diabetic rats by increasing expression of MT and by reducing the apoptotic cell death and the oxidative stress. Besides, the present study shows that Zn has the repair effect or even it provides prevention against diabetic damage of the liver tissue.
Footnotes
Acknowledgments
DÖ and MT formulated the present hypothesis. DÖ, SPE, and MT were responsible for analysis of the data. DÖ and MT were responsible for writing the report.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors thank the Scientific Research Projects Coordination Unit of Istanbul University for the support of the present study (project numbers; 3987 and UDP-56842).
