Abstract
Diabetes mellitus (DM) is a chronic metabolic disorder characterized by hyperglycemia due to abnormalities in either insulin secretion or action. A range of vanadium complexes have been synthesized and demonstrated to be effective in lowering hyperglycemia. Thiamine administration was also reported to prevent deterioration in fasting glucose and insulin levels, and to improve glucose tolerance in hyperglycemic patients. This study has been conducted to evaluate the ionic vanadyl(II) thiamine hydrochloride complex (VC) as a new anti-diabetic candidate. The new complex was characterized by infrared spectroscopy (FT-IR), electronic spectra, magnetic susceptibility, electron spin resonance (ESR), scanning electron microscopy (SEM), and thermogravimetric analysis (TGA). The anti-diabetic effect of VC was investigated in comparison to vanadium sulfate in streptozotocin (STZ)-induced diabetic rats. Treatment of diabetic rats with VC versus vanadyl sulfate showed a more potent effect on reducing serum glucose and cholesterol close to normal levels. VC suppressed the diabetes-induced upregulation of hepatic glucose transporter (GLUT)-2, Phosphoenol pyruvate carboxykinase (PEPCK), and hormone-sensitive lipase (HSL) more significantly than vanadyl sulfate. Either vanadyl sulfate or VC restored hepatic sterol regulatory element-binding protein transcription factor-1c (SREBP-1c) and muscle hexokinase (HK) mRNA expression that was downregulated in diabetic group. Pyruvate kinase (PK) mRNA expression was restored more significantly in VC-treated than vanadyl sulfate-treated diabetic rats. These results indicate that the newly synthesized VC could be an effective anti-diabetic candidate as the anti-diabetic activity of the ionic vanadium was enhanced after being modified with the organic ligand, thiamin. The results also suggest that VC achieves its effect most likely through modulating the transcription of energy metabolizing enzymes.
Introduction
Diabetes mellitus (DM) is a chronic metabolic disorder characterized by hyperglycemia. It results from abnormalities in insulin secretion and/or action at peripheral tissues. 1 The prevalence of diabetes and pre-diabetes is increasing worldwide because of population growth, aging, urbanization, and increasing prevalence of obesity and physical inactivity. 2 The World Health Organization (WHO) estimated that there were 30 million people who had diabetes worldwide in 1985. This number increased to 135 million by 1995 and reached 217 million in 2005. By the year, 2030 WHO predicts this number will increase to at least 366 million. 3
The increase in blood glucose level results mainly from the impairment of either liver or peripheral tissues glucose utilization and the activation of gluconeogenesis in the liver and the kidney. 4 The liver plays a central role in controlling glucose homeostasis. Glucose transport and phosphorylation are the first steps in glucose utilization. In the liver, glucose is transported by two members of the facilitative glucose transporter (GLUT) family, GLUT1 and GLUT2. 5
Hyperlipidemia is a major risk factor for cardiovascular complications in patients with type 2 diabetes, regardless of age. 6 It is characterized by elevated triglyceride levels, a low level of high-density lipoprotein cholesterol (HDL-C). 7 Hyperglycemia can result in various complications through direct and indirect effect on the vascular system of humans. These complications include macrovascular complications (coronary artery disease, peripheral arterial disease, and stroke) and microvascular complications (diabetic nephropathy, neuropathy, and retinopathy). These complications are considered the main reason of mortality in DM. 8
As DM is associated with many metabolic disturbances and can result in severe complications, numerous therapies were developed to maintain the normal level of blood glucose in diabetes patients. 9 A range of vanadium complexes 10 have been synthesized and demonstrated to be effective in lowering hyperglycemia and most importantly, these compounds decrease the side effects of inorganic vanadium salts, possibly by enhancing their rates of absorption in streptozotocin (STZ)-induced diabetic rats. 11
Patients with type 1 or type 2 diabetes had increased renal clearance and fractional excretion of thiamine compared to healthy people. 12 Oral thiamine supplements prevented the development of incipient nephropathy in STZ-induced diabetic rats. 13 Supplementation with high-dose thiamine was reported to have the ability to prevent deterioration in fasting glucose and insulin levels, and improve glucose tolerance in patients with hyperglycemia. 14 Thiamine hydrochloride is a non-toxic, inexpensive, and stable compound. It contains in its structure a pyrimidine ring and a thiazole ring linked by a methylene bridge (Figure 1a). The current study aimed to synthesize a new anti-diabetic candidate through combining thiamine with vanadayl sulfate and asses its anti-diabetic activity compared to that of vanadium sulfate.

Structure of thiamine hydrochloride (a). Suggested structure of (VO(thiamine)2).SO4 complex (b).
Materials and methods
Chemicals
Thiamine hydrochloride (thiamine) and vanadyl(II) sulfate hydrate were purchased from Sigma Aldrich Company (Sigma-Aldrich, St. Louis, MO, USA) and other chemicals and solvents used without purification.
Synthesis and characterization of vanadyl(II) thiamine complex
VOSO4.2H2O (1.20 g; 6 mmol) was dissolved in 20 mL methanol. A methanolic solution of thiamine hydrochloride (1.52 g; 12 mmol) was added to the vanadyl(II) sulfate solution with stirring. The resulting mixture was refluxed at 60ºC with a continuous stirring for 2 h and then kept in a cupboard to ensure slow evaporation. Pale blue powder was precipitated and then dried under vacuum over anhydrous calcium chloride.
The elemental analyses of carbon, hydrogen, and nitrogen contents were performed using a Perkin Elmer CHN 2400. The molar conductivity of freshly prepared 1.0 × 10−3 mol/cm3 dimethyl sulfoxide (DMSO) solution was measured using Jenway 4010 conductivity meter. The electronic absorption spectra of vanadyl(II) thiamine complex was recorded in DMSO solvent with a range of 200–900 nm using a UV2 Unicam UV/Vis spectrophotometer fitted with a quartz cell of 1.0 cm path length. The infrared spectra were recorded on a Bruker FT-IR spectrophotometer (400–4,000 cm−1). Solid reflectance spectra were measured on UV-3101 PC, Shimadzu, UV-Vis. NIR scanning spectrophotometer. Magnetic data were calculated using magnetic susceptibility balance (Sherwood Scientific, Cambridge Science Park Cambridge, UK), at a temperature of 25ºC. The electron spin resonance (ESR) spectrum for vanadyl(II) thiamine complex was performed on Jeol, JES-FE2XG, ESR-spectrometer at a frequency of 9.44 GHz. The thermal studies TG/DTG–50H were carried out on a Shimadzu thermogravimetric analyzer under static air until 800°C. Scanning electron microscopy (SEM) images were taken in Quanta FEG 250 equipment.
Animals and experimental design
Twenty adult male Wistar rats weighing 250–300 grams (age range, 75–90 days) were purchased from an experimental animal center, Faculty of Pharmacy, King Saud University, Saudi Arabia. Animals were housed in the animal facility of the College of Science, Taif University, KSA, at 24°C and 55% humidity with 12 h light/dark cycle. The rats were fed a standard pellet diet, and water ad libitum.
After 12 h of fasting, the animals received a single dose of 80 mg/kg BW intraperitoneal (i.p.) injection of streptozotocin (STZ) (Sigma Chemical Co., St. Louis, MO, USA) dissolved in 10 mM sodium citrate buffer, pH 4.5. Control (non-diabetic) animals were fasted and received equal volume of citrate buffer. After injection, rats had free access to food and 5% glucose solution overnight to avoid hypoglycemic shock. After 72 h, animals with blood glucose levels of 250 mg/dl or higher were considered diabetic, and randomly divided into three groups of five animals each; diabetic non-treated, diabetic-vanadium sulfate (100 mg/kg BW/ day orally)-treated and vanadium-thiamine complex-treated group (100 mg/kg BW/day orally). After 2 weeks of treatment, animals were anesthetized using diethyl ether, and blood samples were collected from the medial canthus of the eyes. Blood samples were allowed to clot, centrifuged, and serum was collected and stored at −80ºC until use for measuring glucose level, TG, and total cholesterol. The rats were then sacrificed by head decapitation. Immediately, the liver and muscle were collected and kept in RNA-Later (Sigma-Aldrich, St. Louis, MO, USA) at −80ºC until used for RNA extraction.
RNA extraction and cDNA synthesis
Total RNA was extracted from 100 mg of each tissue sample using QIAzol lysis reagent (QIAGEN Inc., Valencia, CA, USA) according to the manufacturer’s instructions and as detailed previously. 15 Integrity of the prepared RNA was checked by electrophoresis, while RNA concentration and purity were determined spectrophotometrically at 260 nm and 280 nm. For cDNA synthesis, 2 µg RNA were reverse transcribed with oligo-dT primer and Moloney murine leukaemia virus (M-MuLV) reverse transcriptase (SibEnzyme Ltd. AK, Novosibirsk, Russia) as previous described. 15 The resultant cDNA was preserved at −20°C until used.
Semi-quantitative polymerase chain reaction (PCR)
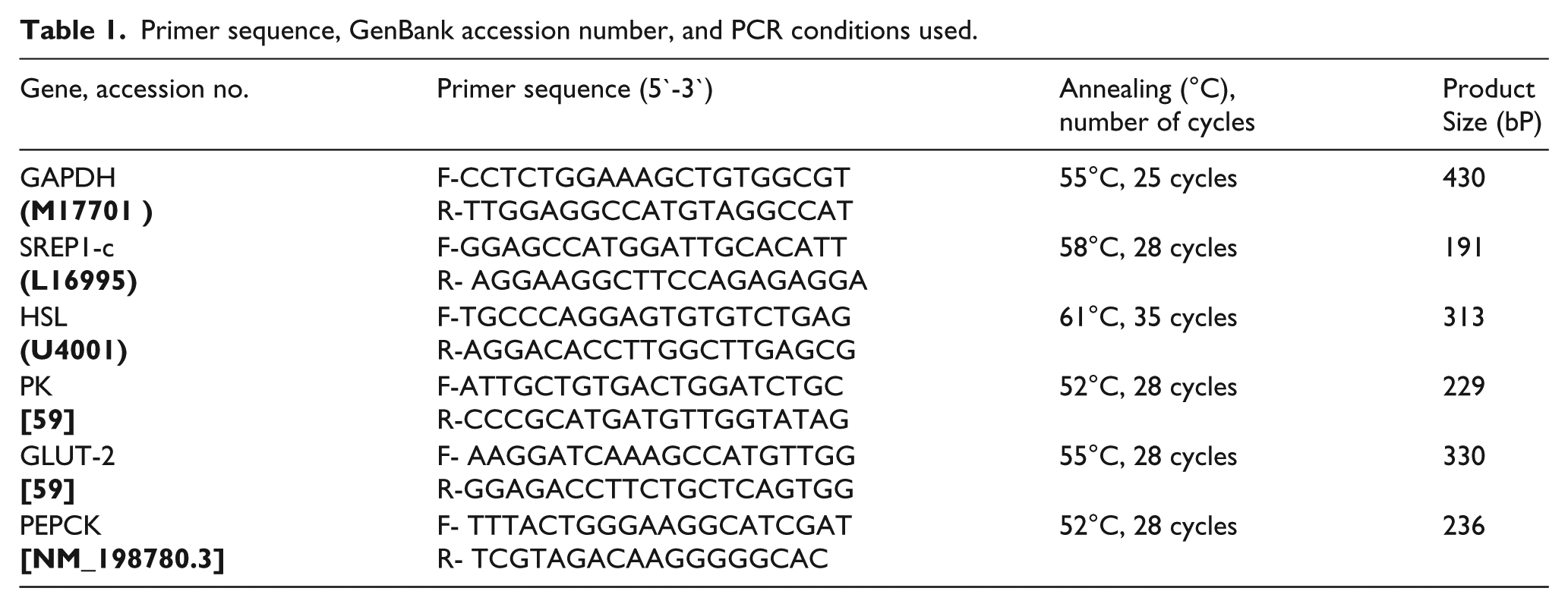
mRNA expression of some genes involved in glucose and lipid metabolism was tested by semi-quantitative PCR using corresponding specific primers of these genes (Table 1). The tested genes were pyruvate kinase (PK), phosphoenol pyruvate carboxykinase (PEPCK), glucose transporter-2 (GLUT-2), hormone sensitive lipase (HSL), and sterol regulatory element-binding protein-1c (SREBP-1c). The used primers were designed using Oligo-4 computer program and nucleotide sequence published in Genebank (Table 1) and synthesized by Macrogen (Macrogen Company, GAsa-dong, Geumcheon-gu, Republic of Korea). PCR was conducted in a final volume of 25 µL consisting of 1 µL cDNA, 1 µL (10 picomoles) of each primer (forward and reverse), and 12.5 µL PCR master mix (Promega Corporation, Madison, WI, USA). The final volume was brought to 25 µL using sterilized, nuclease-free deionized water. PCR was carried out using a PeX 0.5 thermal cycler with the cycle sequence of denaturation at 94°C for 5 min for one cycle, followed by 28–35 cycles (Table 1) each of which consisted of denaturation at 94°C for 1 min, annealing at the specific temperature corresponding to each primer (Table 1) and extension at 72°C for 1 min with an additional cycle as a final extension at 72°C for 5 min. As a reference, expression of glyceraldehyde-3-phosphate dehydrogenase (GAPDH) mRNA was tested using specific primers (Table 1). PCR products were electrophoresed on 1% agarose A (Bio BAsic INC. Konrad Cres, Markham, ON, Canada) gel in TE (Tris-EDTA) buffer at 100 volt for 30 min with ethidium bromide staining. PCR products were visualized under UV light and photographed. The intensities of the bands were quantified densitometrically using NIH image program (http://rsb.info.nih.gov/nih-image).
Primer sequence, GenBank accession number, and PCR conditions used.
Measurement of serum glucose, triacylglycerol, and total cholesterol
Serum glucose, triacylglycerol, and total cholesterol were measured spectrophotometrically using specific commercial kits (HUMAN Gesellschaft für Biochemica und Diagnosticam bH, Wiesbaden, Germany) according to manufacturer’s instructions.
Statistical analysis
Data were presented as means ± standard errors of means. Statistical analysis for the obtained results was performed using one-way analysis of variance (ANOVA) followed by the least significant difference (LSD) test for the multiple comparisons among the groups. The analysis was done by SPSS software (SPSS version 13.0, IBM, Chicago, IL, USA). Values for P <0.05 were considered statistically significant.
Results
Elemental analyses and physical data
The resulted vanadyl(II) complex was pale blue in color. The molar conductivity measurement in DMSO solution showed a higher value of 102 Ω−1cm 2 mol−1 that referring to the electrolyte 1:2 complex. 16 The increase in the electrolytic nature of the vanadyl(II) thiamine complex was due to the presence of ionic sulfate (SO4− –) outside the coordination sphere. The presence of sulfate ions in synthetic complex was checked by adding saturated solution of barium(II) chloride reagent which gave white precipitate. The elemental analyses data of the (VO (thiamine)2).SO4 complex (Figure 1b) was observed to be: Calcd. %C, 34.42; %H, 4.33; %N, 13.38; %V, 6.08. Found: %C, 34.08; %H, 4.31; %N, 13.21; %V, 6.01. The calculated molecular weight using elemental analyzer was 837.54 g mol−1.
Infrared spectra
The infrared spectra of thiamine and its vanadyl(II) thiamine complex are shown in Figure 2a and their bands assignments are discussed in Table 1. The IR spectrum of thiamine-free ligand showed characteristic bands of ethanol, amino, methyl, and aromatic (pyrimidine ring and a thiazole ring) groups. These bands are assigned in the thiamine-free chelate and can be summarized as follows. The frequencies of –OH ethanol group had three vibration motions one of them stretching vibration motion at 3,630 cm−1 and the other frequencies were present at (1,380, 1,287, 1,247) cm−1, (752, 699, 685), and 1,066 cm−1 due to in-plane, out-of-plane bending motions and stretching vibration motion of C-O (17). The primary –NH2 group attached to pyrimidine ring had four distinguished vibration motions as asymmetric stretching NH, symmetric stretching, bend of NH2 group and stretching C-N bands were assigned (3,497, 3,431) cm−1, 3,266 cm−1, 1,650 cm−1, and (1,044, 1,031) cm−1, respectively.17,18 The terminal methyl groups in both pyrimidine ring and a thiazole rings were revealed by existing four vibration motions: asymmetric stretching –CH, symmetric stretching –CH, asymmetric bending –CH3, and symmetric bending –CH3 group at (2,966, 2,926), 2,854, 1,449, and 1,362 cm−1, respectively. On the other hand, the stretching and bending vibration motions of –CH of aromatic rings had some vibration bands at (3,106, 3,078, 3,016), (1,238, 1,222), and 900, 831 cm−1 which assigned to stretching of –CH, –CH in-plane bending, and –CH out-of-plane bending, respectively.17,18 Both stretching vibration motions of C=N and C=C were present at (1,660, 1,607, 1,544) and 1,464 cm-1, respectively. 17 The stretching vibration motion of C-S in thiazole ring was assigned at 773 cm−1. The infrared spectrum of thiamine vanadyl(II) complex (Table 2 and Figure 2a) exhibited some disappeared and shifted bands as well as appearing some new bands in comparison with spectral data of free thiamine as follows. The stretching vibration bands of –OH and C–O of ethanol group at 3,630 and 1,066 cm−1 in free thiamine was disappeared. The stretching and vibration bands of –NH2 group in vanadyl(II) complex still unchanged. The stretching vibration band of C-S of thiazole ring shifted to lower wave number in vanadyl(II) complex and found at 700 cm−1 that was decreased by approximately 73 cm−1 in comparison with free thiamine. The presence of a medium band at 989 cm−1 in vanadyl(II) thiamine complex is indicative of the V=O group of vanadyl(II) ion. 17 The sulfate ion group characteristic bands were observed at 1,114, 643, and 570 cm−1 assigned to stretching and bending motions of S-O bond. The new bands observed at 400–4500 cm−1 in the spectrum of vanadyl(II) complex are indicative of M-O and M-S modes. 18 It seems that thiamine acted as a bidentate ligand through the S of the thiazole ring and the O of the ethanolic group since the C-O stretching frequency of the hydroxyl group disappeared on complexation.

Infrared spectrum of (VO(thiamine)2).SO4 complex (a), ESR spectrum of (VO(thiamine)2).SO4 complex (b).
Infrared bands assignments of thiamine and its vanadyl (II) thiamine complex.

Electronic and magnetic measurements
Thiamine-free ligand has absorption bands in the ultraviolet region 200–400 nm at 234 and 298 nm due to n→π* and π→π*. 19 On complexation, there was a sensible shift in both 234 and 298 nm bands, indicating the involvement of S of the thiazole ring and the O of the ethanolic group in complexation. New band presence in the visible region at approximately 400 nm was assigned to d-d transition absorption and charge transfer spectra from vanadyl(II) metal ions to ligand (M–L) or ligand to metal (L–M). The magnetic moment for vanadyl(II) thiamine complex was found to be 1.72 B.M. at room temperature, which is a characteristic value for mononuclear oxovanadium complexes with S=0.5 and a dxy or dx2–y2 ground state. 19 The electronic spectrum of VO(II) complex displayed three bands at 13,513, 16,529, and 22,222 cm−1 due to the 2B2→2E, 2B2→2B1, and 2B2→ 2A1 transitions, respectively, for square pyramidal geometry around V(IV) ion. The ESR experimental spectrum of vanadyl (II) thiamine is shown in Figure 2b. The g║ < g┴ relations is consistent with square pyramidal complexes with C4V symmetry with the unpaired electron in the dxy orbital. 19
Scanning electron microscopy
The microstructure, surface morphology, and chemical composition of thiamine as a free ligand and vanadyl(II) complex were studied using scanning electron microscopy (Figure 3a). The surface morphology of SEM micrograph reveals the well sintered nature of the compounds with variant grain sizes and shapes. The distribution of the grain size of vanadyl(II) thiamine complex is homogeneous (leaves) although the free thiamine ligand (Figure 3B) has small-to-medium particles with different sizes. Clear large grains were obtained with agglomerates for (VO(thiamine)2).SO4 complex. The particle size distribution of the vanadyl(II) complex was evaluated and the average particle size was found to be 2–10 μm.

SEM image of thiamine (a): and (VO(thiamine)2).SO4 complex (b).
Thermal and kinetic data
The thermal (TG/DTA) and kinetic characteristic data of the (VO(thiamine)2).SO4 complex were determined from DTG thermograms and the curves were collected in Figure 4. The DTA analysis was carried out mainly to determine the thermodynamic behavior (exothermic or endothermic process) and the decomposition temperatures. The DTA curve of vanadyl(II) thiamine complex showed a broad, sharp, and intense exothermic peak with a maximum at 510°C corresponding to the decomposition process. The vanadyl(II) complex was stable up to 200°C where decomposition started and was completed at 650°C. The mass loss 7.52% may be attributed to the decomposition of the two thiamine hydrochloride moieties ligand with liberated gaseous molecules. The obtained end product residue was close to that expected for metal carbide VC. The decomposition equation of the VO(Thy)2 compound can be suggested as follows: (VO(thiamine)2).SO4 →C23H36Cl4N8O7S3↑ +VC↓. In the present investigation, the general thermal behaviors of the (VO (thiamine)2).SO4 complex in terms of stability ranges, peak temperatures, and values of kinetic parameters have been studied and discussed. The kinetic parameters were evaluated using the Coats–Redfern equation:

This equation on integration gives;

Where

Where kB is the Boltzmann constant, h is the Plank’s constant, and Ts is the DTG peak temperature. 20 The Horowitz-Metzger equation is an illustrative of the approximation methods.

When n = 1, the LHS of equation 4 would be log[–log (1–α)]. For a first-order kinetic process the Horowitz-Metzger equation may be written in the form:
log[log(wα / wγ)] = E*θ/2.303RTs2 – log2.303. Where θ = T - Ts, wγ = wα – w, wα = mass loss at the completion of the reaction. The plot of log[log(wα / wγ)] vs θ was drawn and found to be linear from the slope of which E* was calculated. The pre-exponential factor, A, was calculated from the equation: E* /RTs2 = A/[ φ exp(-E*/RTs)].

TG/DTA curves of (VO(thiamine)2).SO4 complex.
The entropy of activation, ΔS*, was calculated from equation (3). The enthalpy activation, ΔH*, and Gibbs free energy, ΔG*, were calculated from; ΔH* = E* – RT and ΔG* = ΔH* – TΔS*, respectively.
In the present investigation, the general thermal behaviors of the (VO (thiamine)2).SO4 complex in terms of stability ranges, maximum temperature peaks, and values of kinetic parameters, are shown in Table 3. The kinetic and thermodynamic parameters were evaluated using the Coats–Redfern and Horowitz–Metzger equations. 21 The entropy of activation, ΔS*, was calculated. The enthalpy activation, ΔH*, and Gibbs free energy, ΔG*, were calculated from ΔH* = E* – RT and ΔG* = ΔH* – TΔS*, respectively.
Kinetic and thermodynamic parameters of the thermal decomposition of (VO(thiamine)2).SO4 complex.

ΔG*: Gibbs free energy, ΔH*: enthalpy of activation, ΔS*: entropy of activation, A: pre-exponential factor, r: correlation coefficient of the linear plot, CR: Coats–Redfern equation, E*: activation energy, HM: Horowitz–Metzger equation.
The thermodynamic behavior of vanadyl(II) thiamine complex was non-spontaneous (more ordered) reactions. The thermodynamic data obtained with the two methods were in harmony with each other. The correlation coefficients of the Arrhenius plots of the thermal decomposition steps were found to lie in the range of 0.98–0.99, showing a good fit with linear function. The thermograms and the calculated thermal parameters for the complexes showed that the stability of these complexes depends on the nature of the central metal ion.
Effect of vanadyl sulfate and VC on serum glucose, serum cholesterol, and triacylglycerol levels
Concerning blood glucose level, STZ-induced diabetic rats showed a significant increase in blood glucose while vanadyl-sulfate treatment showed a decrease in blood glucose toward normal levels. However, treatment with vanadayl-thiamine complex showed a further decrement in glucose level (Figure 5a). Treatment of diabetic rats with vanadyl-sulfate showed a normalization of the serum triacylglycerol but it did not show any improvement of the serum total cholesterol levels. Moreover, treatment of diabetic rats with vanadyl-thiamine complex showed a normalization of both serum triacylglycerol and total cholesterol (Figure 5b and c).
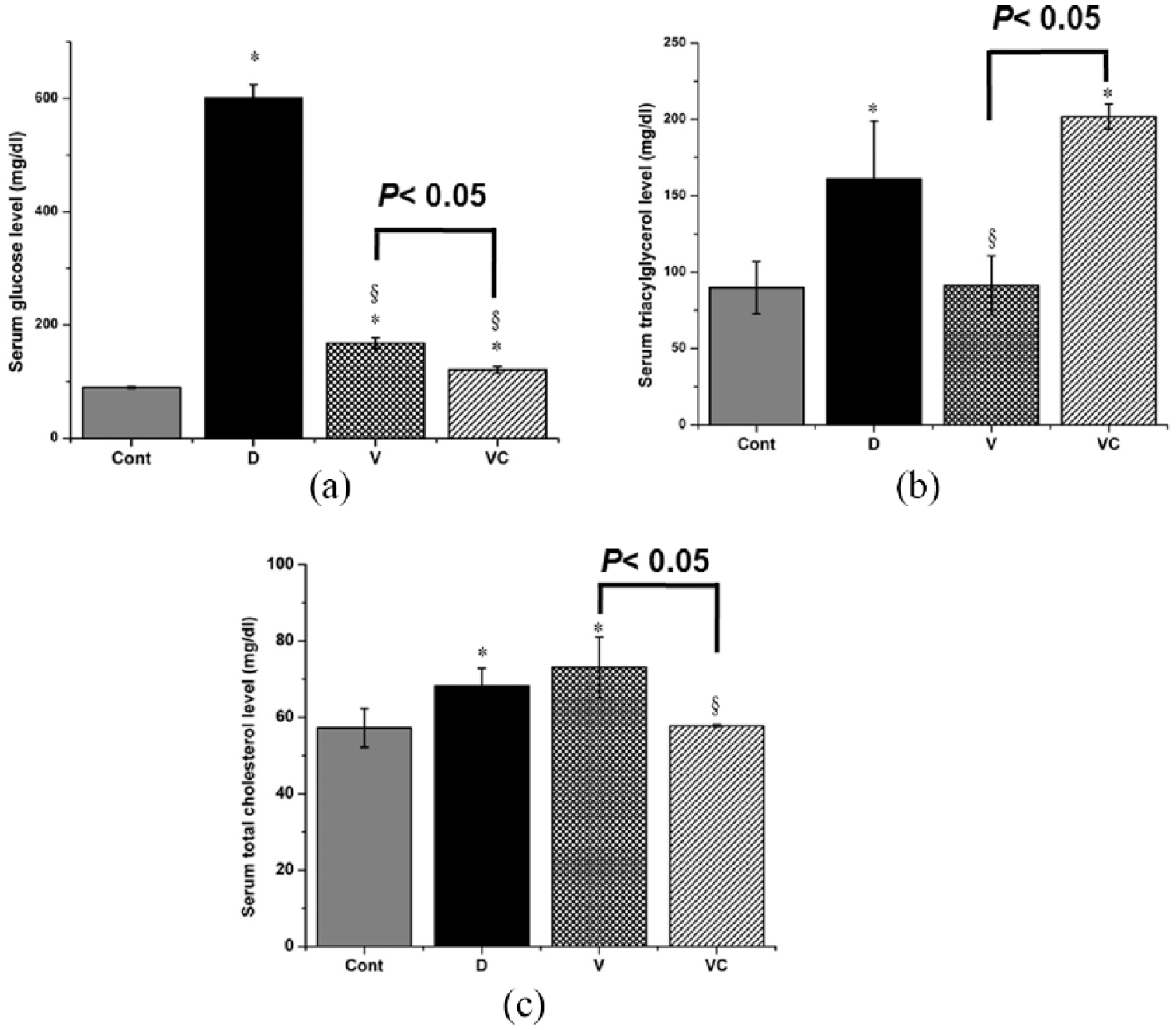
Effect of vanadyl sulfate and vanadyl-thiamine complex on serum levels of glucose (a), triacylgycerols (b), and total cholesterol (c) in STZ-induced diabetic rats. Values are means ± SE of three independent experiments. *P <0.05 vs. Control group, §P <0.05 vs. diabetic group. C: control group, D: STZ-induced diabetic group; V: vanadyl-sulfate-received diabetic group, VC: vanadyl sulfate complex-received diabetic group.
Effect of vanadyl sulfate and VC on mRNA expression of genes related to glucose metabolism in liver and muscle
STZ-induced diabetic rats showed a significant upregulation of hepatic glucose transporter-2 (GLUT-2) mRNA expression compared to control. On the contrary, diabetes downregulated the expression of muscle hexokinase II (HKII), one of glycolysis enzymes, compared to control. Treatment of diabetic rats with either vanadyl sulfate or VC significantly inhibited the diabetes-induced GLUT-2 expression. Furthermore, VC showed a more potent inhibitory effect on GLUT-2 expression compared to vanadyle sulfate. Similarly, either vanadyl sulfate or VC significantly inhibited the diabetes-induced decrease of the HKII mRNA expression which was even higher than its control level of expression (Figure 6a and b). Parallel to the effects on HKII expression, STZ-induced diabetic rats showed a significant decrease of liver pyruvate kinase (PK); mRNA expression compared to control. Either vanadyl sulfate or VC significantly inhibited the diabetes-induced decrease of the PK mRNA expression where vanadyle sulfate restored about 40% of the PK control level of expression and VC was more powerful in restoring the PK expression level (80% control level) as shown in Figure 7a. In the same context of these findings, diabetic rats showed a significant increase in hepatic mRNA expression of phosphoenol pyruvate carboxykinase (PEPCK); one of gluconeogenic enzymes, compared to control. However, either vanadyl sulfate or VC treatment significantly suppressed PEPCK mRNA expression compared to either diabetes or even control with VC showed a more potent inhibitory effect than vanadyl sulfate (Figure 7b).

Effect of vanadyl sulfate and vanadyl-thiamine complex on hepatic GLUT-2 (a) and muscle HK II (b) mRNA expressions in STZ-induced diabetic rat. Total RNA was prepared from liver and muscle tissues and the expressions of GLUT-2 HK II were analyzed by RT-PCR in liver and muscle respectively. Values are means ± SE of three independent experiments. *P <0.05 vs. Control group, §P <0.05 vs. diabetic group. C: control group, D: STZ-induced diabetic group, V: vanadyl sulfate-received diabetic group, VC, vanadyl sulfate complex-received diabetic group.

Effect of vanadyl sulfate and vanadyl-thiamine complex on hepatic PK (A) and PEPCK (B) mRNA expressions in STZ-induced diabetic rats. Total RNA was prepared from liver tissue and the expressions of PK and PEPCK were analyzed by RT-PCR. Values are means ± SE of three independent experiments. *P <0.05 vs. Control group, §P <0.05 vs. diabetic group. C: control group, D: STZ-induced diabetic group, V: vanadyl sulfate-received diabetic group, VC, vanadyl sulfate complex-received diabetic group.
Effect of vanadyl sulfate and VC on the hepatic expression of lipogenic and lipolytic genes
STZ-induced diabetic rats showed a significant upregulation of the lipolytic enzyme hormone-sensitive lipase (HSL) mRNA expression compared to control. However, they showed a downregulation of the lipogenic transcription factor SREBP-1c expression. Treatment of diabetic rats with VC significantly decreased the HSL mRNA expression compared to either diabetic (non-treated) or vanadyl-treated diabetic group. Either vanadyl or VC significantly increased the expression of SREBP-1c compared to diabetic (non-treated) group (Figure 8a and b).

Effect of vanadyl sulfate and vanadyl-thiamine complex on hepatic HSL (a) and SREP-1C (b) mRNA expressions in STZ-induced diabetic rats. Total RNA was prepared from liver tissue and the expressions of HSL and SREP-1C were analyzed by RT-PCR. Values are means ± SE of three independent experiments. *P <0.05 vs. Control group, §P <0.05 vs. diabetic group. C: control group, D: STZ-induced diabetic group, V: vanadyl sulfate-received diabetic group; VC: vanadyl sulfate complex-received diabetic group.
Discussion
In this study, we synthesized a new VC and tested its hypoglycemic effect in STZ-induced diabetic rats in comparison with vanadyle sulfate. The obtained results indicate that VC is more effective in correcting many diabetes-associated abnormalities, the cause of diabetes complications. This effect makes VC more advantageous than vanadyle sulfate. This efficient hypoglycemic effect of VC could be explained by its significant stimulatory effect on the expression of genes that favor cellular uptake and catabolism of glucose (GLUT-2, HKII, and PK) and its inhibitory effect on gluconeogenic gene (PEPCK) expression. These effects were mostly significantly higher than those of vanadyl sulfate. Pyruvate kinase (PK) is a rate-limiting glycolytic enzyme. Its activity was decreased in the liver of alloxan-induced diabetic rats 22 and increased by insulin administration to diabetic rats. 23 In the current study, the marked increase of the PK mRNA expression in diabetic rats treated with VC (80% of its control level) far higher than vanadyle sulfate-treated diabetic rats (about 40% of its control level). These results indicate that the hypoglycemic effect of VC acts mainly through increasing glycolysis as PK was described as a rate-limiting glycolytic enzyme that catalyzes the generation of pyruvate and ATP from phosphoenolpyruvate. 24 Moreover, the clear induction of PK in diabetic rats treated with VC indicates its insulin mimics effects as the PK activity was stated to be lower in diabetes and higher in the liver by the administration of insulin to diabetic rat tissues. 25
Glucose phosphorylation, the first step of glucose metabolism, is catalyzed by hexokinase in muscles. 26 A marked reduction in HKII mRNA level and HKII activity has been reported in the muscle of diabetic patients. 27 The significant upregulation of the muscle HKII in VC-treated diabetic compared to diabetic rats and the obviously higher HKII induction in VC-treated compared to vanadyle sulfate-treated diabetic rats indicate the ability of VC to stimulate glucose uptake by skeletal muscles. This may imply that its strong hypoglycemic effect may operate in part through increasing glucose uptake by muscle as insulin injection to diabetic rats was associated with hypoglycemic effect and normalization of muscle HKII levels. 28
PEPCK, one of the key enzymes of hepatic carbohydrate metabolism, catalyzes a regulatory step in gluconeogenesis. Similar to our findings, insulin deficiency is associated with an increased hepatic expression of PEPCK and hyperglycemia. 29 The reduction of PEPCK mRNA in VC-treated diabetic rats to about 50% of its levels in diabetic rats treated with vanadyle sulfate may explain in part the VC superior hypoglycemic effect. The reduction of hepatic PEPCK levels in our study may indicate VC complex as a promising therapeutic for diabetes where the restoration of elevated PEPCK expression was considered a good therapeutic target for diabetes therapy. 30 The stronger hypoglycemic effect of VC than vanadyl sulfate shown in the present study could be explained by decreased gluconeogenesis due to the stronger downregulation of PEPCK by VC which agrees to the previous study which suggested that the decreased blood glucose in high fat diet-fed mice treated with hawthorn was caused by a decreased gluconeogenesis following PEPCK reduction. 1
GLUT-2 is considered the actual glucose sensor. It is essential for the rapid glucose response in the liver. 31 In our study, the GLUT-2 upregulation in diabetic rats is in accordance with previous results which reported that liver GLUT-2 expression increased in diabetic rats, and returned to normal levels after insulin administration. 31 The stronger downregulating effect of VC than vanadyle sulfate on GLUT-2 expression assures the superior hypoglycemic effect of VC. GLUT-2 translocates from the cytoplasm to the plasma membrane in response to high levels of plasma glucose and transports plasma glucose into hepatocytes. 32 An abnormally high level of intracellular glucose could be deleterious to some types of cells. 33 Thus our results may indicate the ability VC to protect liver from the diabetic glucose load. Moreover, the GLUT-2 inhibitory effect of VC could be through its hypoglycemic effect.
HSL determines the rate-limiting step to complete lipolysis and determine the stimulated lipolysis. 34 The HSL activity is negatively regulated by insulin and upregulated by glucose. 35 Our results showed an induction of hepatic HSL mRNA in diabetic rats which could be due to lack of insulin and hyperglycemia. HSL induction may explain the increased levels of triacylglycerol and total cholesterol levels in the diabetic rats. The normalization of serum total cholesterol levels by VC treatment indicates its higher efficiency for treating diabetes and preventing diabetic complications compared to vanadyle sulfate.
Sterol regulatory element binding protein-1c (SREBP-1c), a lipogenic transcription factor, is a key regulator of hepatic lipid metabolism through controlling the gene expressions of enzymes for fatty acid synthesis and uptake and triacylglycerol synthesis.36,37 SREBP-1c gene transcription is induced by insulin 38 and downregulated in STZ-induced diabetic rats. 39 In the current study, the SREBP-1c mRNA downregulation in STZ-induced diabetic rats is in accordance with results of the previous study. 39 However, the effect of VC on SREBP-1c mRNA is comparable to that of vanadyl sulfate. In conclusion, we synthesized and characterized a new anti-diabetic candidate which is ionic vanadyl(II) thiamin complex. The current results suggest that vanadyl-thiamine complex is an effective organic vanadium anti-diabetic compound. The new compound surpassed the vanadyl sulfate in treating hyperglycemia, improving glucose and lipid metabolism as well as in alleviating other symptoms of diabetes mellitus.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Taif University, Kingdom of Saudi Arabia is acknowledged for supporting this work (grant no. 1349-432).
