Abstract
Physicochemical properties of titanium dioxide nanoparticles (TiO2 NPs) can be tuned by doping with metals or nonmetals. Copper (Cu) doping improved the photocatalytic behavior of TiO2 NPs that can be applied in various fields such as environmental remediation and nanomedicine. However, interaction of Cu-doped TiO2 NPs with human cells is scarce. This study was designed to explore the role of Cu doping in cytotoxic response of TiO2 NPs in human lung epithelial (A549) cells. Characterization data demonstrated the presence of both TiO2 and Cu in Cu-doped TiO2 NPs with high-quality lattice fringes without any distortion. The size of Cu-doped TiO2 NPs (24 nm) was lower than pure TiO2 NPs (30 nm). Biological results showed that both pure and Cu-doped TiO2 NPs induced cytotoxicity and oxidative stress in a dose-dependent manner. Low mitochondrial membrane potential and higher caspase-3 enzyme (apoptotic markers) activity were also observed in A549 cells exposed to pure and Cu-doped TiO2 NPs. We further observed that cytotoxicity caused by Cu-doped TiO2 NPs was higher than pure TiO2 NPs. Moreover, antioxidant
Introduction
Nanotechnology provides opportunity to develop nanoparticles (NPs) with unique features that have potential to be applied in several fields. The NPs are defined as materials, structures, or devices that have at least one dimension in the range of 1–100 nm. The NPs are being utilized in every aspect of human life including medical science, agriculture, and defense industries. 1,2 However, the intentional, 3,4 or unintentional, 5 exposure of these NPs may pose health hazard to the environment and human. 6 –9 Being smaller than cells and organelles, NPs can penetrate basic biological structures that may, in turn, disrupt their function. 10,11
Titanium dioxide (TiO2) NPs are now being produced in a large industrial scale and used widely due to its chemical stability and low cost. 12,13 TiO2 NPs have been attracted significant attention due to their applications in photocatalysis, antibacterial, environmental remediation, and water disinfection. 14 –17 TiO2 NPs are also being utilized in a broad range of products such as colorant for food and paints and used in sunscreens as an ultraviolet blocking agent. 18 –20 However, the environmental and human exposure of TiO2 NPs is still debatable and is a rapidly expanding area of research. Study has shown that after inhalation, NPs can be translocated into the bloodstream and distributed to different organs of the body. 21 In addition to inhalation, TiO2 NPs can also be circulated in the bloodstream via dermal contact and ingestion. 21 Several studies have shown that TiO2 NPs have the potential to induce cytotoxicity, oxidative stress, inflammation, DNA damage, and apoptosis to different cells. 13,22 –24
Recent studies suggested that doping with metals or nonmetals effectively tune the optical and electrical properties of semiconductor TiO2 NPs. 19,25,26 In semiconductors, electrons (e−) in conduction band and holes (h+) valence band have been traditionally used for photocatalytic degradation of organic and inorganic pollutants. 27 In these experiments, adequate electrons and holes were typically generated by ultraviolet irradiation. Photocatalytic behavior of TiO2 NPs can be further improved by doping. The most efficient approach to extend the photo-response of TiO2 into visible range is doping or surface modification with transition metals (e.g. copper (Cu)). 28,18 Studies have shown that Cu-doped TiO2 NPs effectively kill bacteria under visible light. 29,30 Therefore, Cu-doped TiO2 NPs have great potential to be applied in medicine and environmental remediation. 31,32
There is scarcity of studies on toxicity/biocompatibility of Cu-doped TiO2 NPs in human cells. Some studies reported that metal-doped (e.g. Ag or Cu) TiO2 NPs induce toxicity to zebra fish embryo. 33,34 However, Xu et al. 35 observed that Cu-doped TiO2 films possess better blood compatibility compared with the nondoped TiO2 film. Hence, this study was designed to investigate the role of Cu-doping in cytotoxicity mechanisms of TiO2 NPs. To achieve this objective, we assessed the cell viability, reactive oxygen species (ROS), lipid peroxidation (LPO), glutathione (GSH), mitochondrial membrane potential (MMP), and caspase-3 enzyme activity in human lung epithelial (A549) cells after exposure of pure and Cu-doped TiO2 NPs. The A549 cells, derived from human lung carcinoma, have widely been used in toxicity and biomedical research. 36,37
Materials and methods
Chemicals and reagents
Dulbecco’s modified eagle’s medium (DMEM), penicillin–streptomycin, and fetal bovine serum (FBS) were bought from Invitrogen (Carlsbad, California, USA). Titanium (IV) isopropoxide, rhodamine-123 (Rh123), 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazoliumbromide (MTT),
Synthesis of pure and Cu-doped TiO2 NPs
Pure and Cu-doped TiO2 NPs were synthesized by the sol–gel method. We have used titanium(IV) isopropoxide Ti[OCH(CH3)2]4 and copper sulfate (CuSO4·5H2O) as precursors. Briefly, 10 ml of titanium(IV) isopropoxide was dissolved in 50 ml of absolute ethanol. The mixture solution was stirred for 15 min. The resultant solution was mixed with 10 ml of distilled water and the solution was stirred for 1 h to obtain a clear solution. Now, the mixture was transformed into gel. After aging 24 h, the gel is filtered and dried. Then, prepared TiO2 samples were calcined at 450°C for 2 h. Cu-doped (3% Cu at wt%) TiO2 NP was prepared by impregnation of pure TiO2 nanopowder suspension in water, with addition of an appropriate amount of copper sulfate while continuous stirring for 3 h. The samples were then filtered, washed, and dried at 60°C for overnight. Then, prepared faint bluish Cu-doped TiO2 NPs were calcined at 450°C for 2 h.
Characterization of pure and Cu-doped TiO2 NPs
Crystal structure, phase, and purity of pure and Cu-doped TiO2 NPs were examined by X-ray diffraction (XRD) instrument (Westborough, MA, USA). XRD spectra of NPs were acquired at room temperature with the help of a PANalytical X’Pert X-ray diffractometer (Westborough, MA, USA) equipped with an Ni filter using Cu Kα (
Hydrodynamic size and zeta potential of pure and Cu-doped TiO2 NPs in water and culture medium were determined using dynamic light scattering (DLS; Nano-ZetaSizer-HT, Malvern, UK) as described by Murdock et al. 38 Briefly, pure and Cu-doped TiO2 nanopowder was suspended in deionized water and complete cell culture medium (DMEM + 10% FBS). Further, suspension of NPs was incubated at 37°C for 24 h. Then, suspension was sonicated at room temperature for 30 min at 40 W and DLS experiments were done.
Cell culture
Human lung epithelial cell line (A549) was obtained from American Type Culture Collection (ATCC; Manassas, Virginia, USA). Cells were cultured in DMEM medium supplemented with 10% FBS and 100 U/ml penicillin–streptomycin at 5% CO2 and 37°C. At 80–90% confluence, cells were harvested using 0.25% trypsin and were subcultured for nanotoxicity experiments. Cells were allowed to attach on the surface of culture flask for 24 h before the exposure of pure and Cu-doped TiO2 NPs.
Exposure of pure and Cu-doped TiO2 NPs to cells
Dry powder of pure and Cu-doped TiO2 NPs was suspended in DMEM medium at a concentration of 1 mg/ml and diluted to desired dosages (2–200 µg/ml). The various dosages of NPs were then sonicated at room temperature for 15 min at 40 W to evade agglomeration of NPs before exposure to cells. In some experiments, cells were preexposed for 1 h with 10 mM of NAC before 24 h co-exposure with or without pure and Cu-doped TiO2 NPs. In some experiments, ZnO NP was used as a positive control.
MTT assay
Cell viability was measured using MTT.
36
This assay measures the mitochondrial function by determining the ability of living cells to reduce MTT into blue formazan product. Briefly, 1 × 104 cells/well were seeded in a 96-well plate and exposed to different concentrations of pure and Cu-doped TiO2 NPs (2–200 µg/ml) for 24 h. After the completion of exposure time, culture medium was discarded from each well to avoid interference of NPs and replaced with 100 μl MTT laden media (0.5 mg MTT per ml of media) and incubated at 37°C until a purple-colored formazan product was formed (∼3 h). The resulting formazan product was dissolved in acidified isopropanol (1 µl concentrated hydrochloric acid per 1 ml of isopropanol). Then, 96-well plate was centrifuged at 2300
Neutral red uptake assay
Neutral red uptake (NRU) assay was performed following the method of Ahamed et al.
36
Briefly, 1 × 104 cells/well were seeded in a 96-well plate and treated with various concentrations of pure and Cu-doped TiO2 NPs (2–200 µg/ml) for 24 h. At the end of the exposure time, test solution was aspirated and cells were washed with phosphate buffer saline (PBS) twice before being incubated for 3 h in medium supplemented with 50 µl of neutral red dye (0.6 mg/ml in PBS). The medium was washed off rapidly with PBS. Cells were then incubated for a further 20 min at 37°C in a mixture of acetic acid (1%) and ethanol (50%) to extract the dye. The 96-well plate was then centrifuged at 2300
Cell morphology
Cells were treated with pure and Cu-doped TiO2 NPs at a concentration of 200 µg/ml for 24 h. After the completion of exposure time, the morphology of A549 cells was assessed by a phase-contrast microscope (Leica DMIL, Germany).
ROS assay
Intracellular level of ROS was measured using the DCFH-DA probe.
39
In brief, 1 × 104 cells/well were seeded in a 96-well black-bottomed culture plate and allowed to attach on the surface for 24 h in a CO2 incubator at 37°C. Then, cells were treated with pure and Cu-doped TiO2 NPs at the concentrations of 50 and 100 µg/ml for 24 h. After the completion of the exposure time, cells were washed with PBS before being incubated in 100 μl of 50 μM DCFH-DA solutions for 45 min at 37°C. Then, cells were lysed in alkaline solution and centrifuged at 2300
MMP assay
MMP level in cells following exposure to pure and Cu-doped TiO2 NPs was assessed using Rh-123 fluorescent dye. 39,40 Briefly, cells (1 × 104 cells/well) were exposed to NPs for 24 h. At the end of the exposure time, cells were washed twice with PBS. Then, cells were treated with 10 µg/ml of Rh-123 for 1 h at 37°C in dark. Cells were again washed with PBS. Then, fluorescence intensity of Rh-123 dye was measured using a microplate reader (Synergy-HT, BioTek).
Preparation of crude cell extract
Crude cell extract was prepared according to the method described in our earlier paper.
41
Briefly, cells were cultured in 75-cm2 culture flask and treated with pure and Cu-doped TiO2 NPs with or without NAC for 24 h. At the end of exposure time, cells were harvested in ice-cold PBS by scraping and washed with PBS at 4°C. Then, cell pellets were lysed in cell lysis buffer [1 × 20 mM Tris-HCl (pH 7.5), 150 mM NaCl, 1 mM Na2EDTA, 1% Triton, 2.5 mM sodium pyrophosphate]. Buffer with lysed cells was centrifuged at 15,000
Lipid peroxidation assay
LPO was estimated by measuring the formation of malondialdehyde (MDA) using the method of Ohkawa et al.
42
MDA is one of the end products of LPO. In brief, a mixture of 0.1 ml crude cell extract and 1.9 ml of 0.1 M sodium phosphate buffer (pH 7.4) was incubated at 37°C for 1 h. After incubation, mixture was precipitated with 5% TCA and centrifuged at 2300
GSH assay
Intracellular GSH level was quantified by the method of Ellman.
43
In brief, a mixture of 0.1 ml of crude cell extract and 0.9 ml of 5% trichloroacetic acid (TCA) was centrifuged at 2300
Caspase-3 enzyme activity assay
Activity of caspase-3 enzyme was assessed by Bio-Vision colorimetric assay kit (Milpitas, CA, USA). This assay was based on the principle that activated caspases in apoptotic cells cleave the synthetic substrates to release free chromophore
Protein estimation
Cellular protein content was estimated by the Bradford method 45 using bovine serum albumin as the standard.
Statistical analysis
Statistical analysis was done by one-way analysis of variance followed by Dunnett’s multiple comparison tests. Significance was ascribed at
Results
TEM characterization
Size, growth pattern, and distribution of crystallites were carried out by FETEM. Figure 1(a) and (b) represents the low-resolution images of pure and Cu-doped TiO2 NPs, respectively. The primary particle size was assessed from measuring over 75 NPs in random fields of view. The average size of pure and Cu-doped (3%) TiO2 NPs was 30 and 24 nm, respectively. These results suggest that Cu doping significantly reduces the size distribution of TiO2 NPs (Table 1). Figure 1(c) demonstrates the high-resolution TEM image of Cu-doped TiO2 NPs. This image shows the presence of both TiO2 and Cu NPs with high-quality lattice fringes without any distortion. The estimated interplanar spacing of adjacent lattice fringe of TiO2 NPs is 0.350 nm, which corresponds to the peak (101) of anatase TiO2, while a lattice fringe of Cu NPs was 0.21 nm. These lattice distances were in accordance with the XRD spectra (Figure 2).

TEM of pure and Cu-doped TiO2 NPs. (a) and (b) Low-resolution TEM images. (c) High-resolution TEM images. TEM: transmission electron microscopy; TiO2 NPs: titanium dioxide nanoparticles.
Characterization of pure and Cu-doped TiO2 NPs (mean ± SD,

TiO2 NPs: titanium dioxide nanoparticles; CDMEM: complete Dulbecco’s modified eagle media, which contains 10% FBS; DI: deionized water; FBS: fetal bovine serum.
aParticle size and zeta potential in solution were measured by ZetaSizer Nano (Malvern).
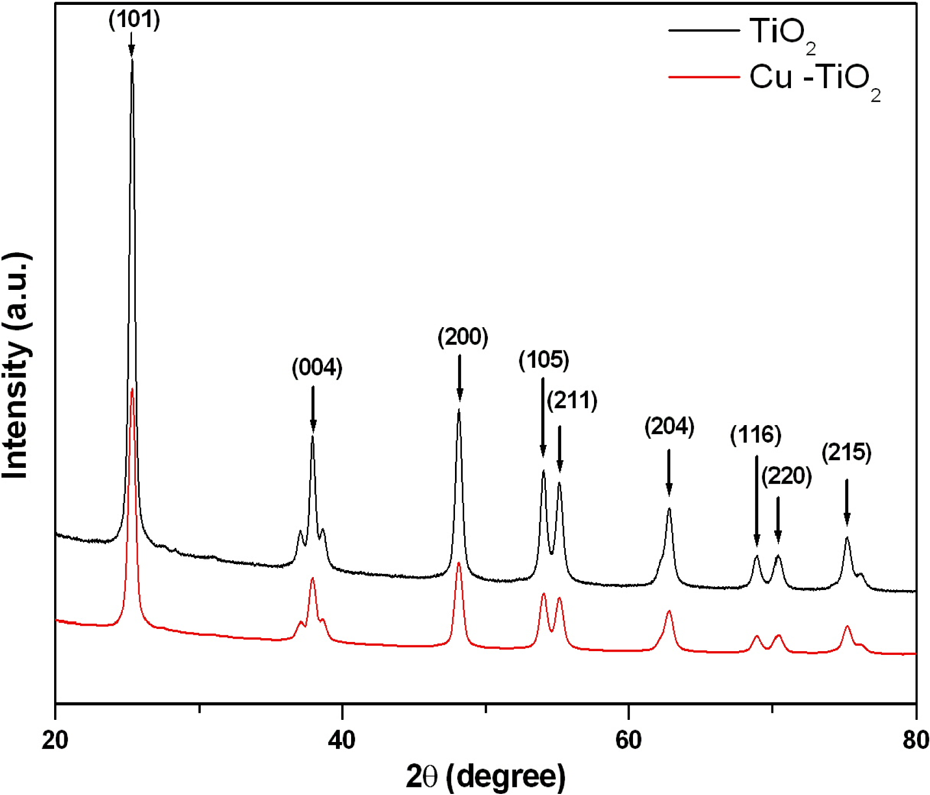
X-ray diffraction spectra of pure and Cu-doped TiO2 NPs. TiO2 NPs: titanium dioxide nanoparticles.
XRD characterization
Figure 2 shows the XRD pattern of pure and Cu-doped TiO2 NPs recorded in 2
DLS characterization
Hydrodynamic size and zeta potential of pure and Cu-doped TiO2 NPs in deionized water and culture medium are given in Table 1. Hydrodynamic size of pure and Cu-doped TiO2 NPs was 4–6 times higher as compared to the primary size (size calculated from TEM and XRD). Zeta potential measurement indicated that pure and Cu-doped TiO2 NPs dispersed in deionized water had positive surface charge. However, the surface charge of the aggregates in complete culture medium was negative (Table 1).
Cu doping enhanced the cytotoxic response of TiO2 NPs
The A549 cells were treated with various concentrations of pure and Cu-doped TiO2 NPs for 24 h and cytotoxicity was determined by MTT, NRU, and morphology assays. Results showed that cytotoxic response of Cu-doped TiO2 NPs was higher as compared to pure TiO2 NPs. MTT assay has shown that cell viability due to pure TiO2 NPs exposure was reduced to 86%, 71%, 58%, and 42%, while cell viability reduction due to Cu-doped TiO2 NPs was 77%, 63%, 51%, and 33% at the concentrations of 25, 50, 100, and 200 µg/ml, respectively (Figure 3(a)). Similar trends of cell viability reduction were observed in NRU data (Figure 3(b)).

Cytotoxicity of pure and Cu-doped TiO2 NPs. The A549 cells were treated with different concentrations of pure and Cu-doped TiO2 NPs for 24 h. At the end of exposure time, parameters were measured. (a) MTT assay and (b) NRU assay. Data represented are mean ± SD of three identical experiments made in three replicate. *Significant difference as compared to control (
Phase-contrast microscopic images (Figure 3(c)) also showed a significant lowering of cell density and rounding of cells after exposure to 200 µg/ml of pure and Cu-doped TiO2 NPs for 24 h. These results are in agreement with cell viability data.
Cu-doping increased the oxidative stress response of TiO2 NPs
We further observed that both pure and Cu-doped TiO2 NPs were found to induce oxidative stress in A549 cells evident by induction of ROS and LPO and depletion of GSH (Figure 4(a) to (c)). Moreover, oxidative stress response of Cu-doped TiO2 NPs was more severe than those of the pure one.
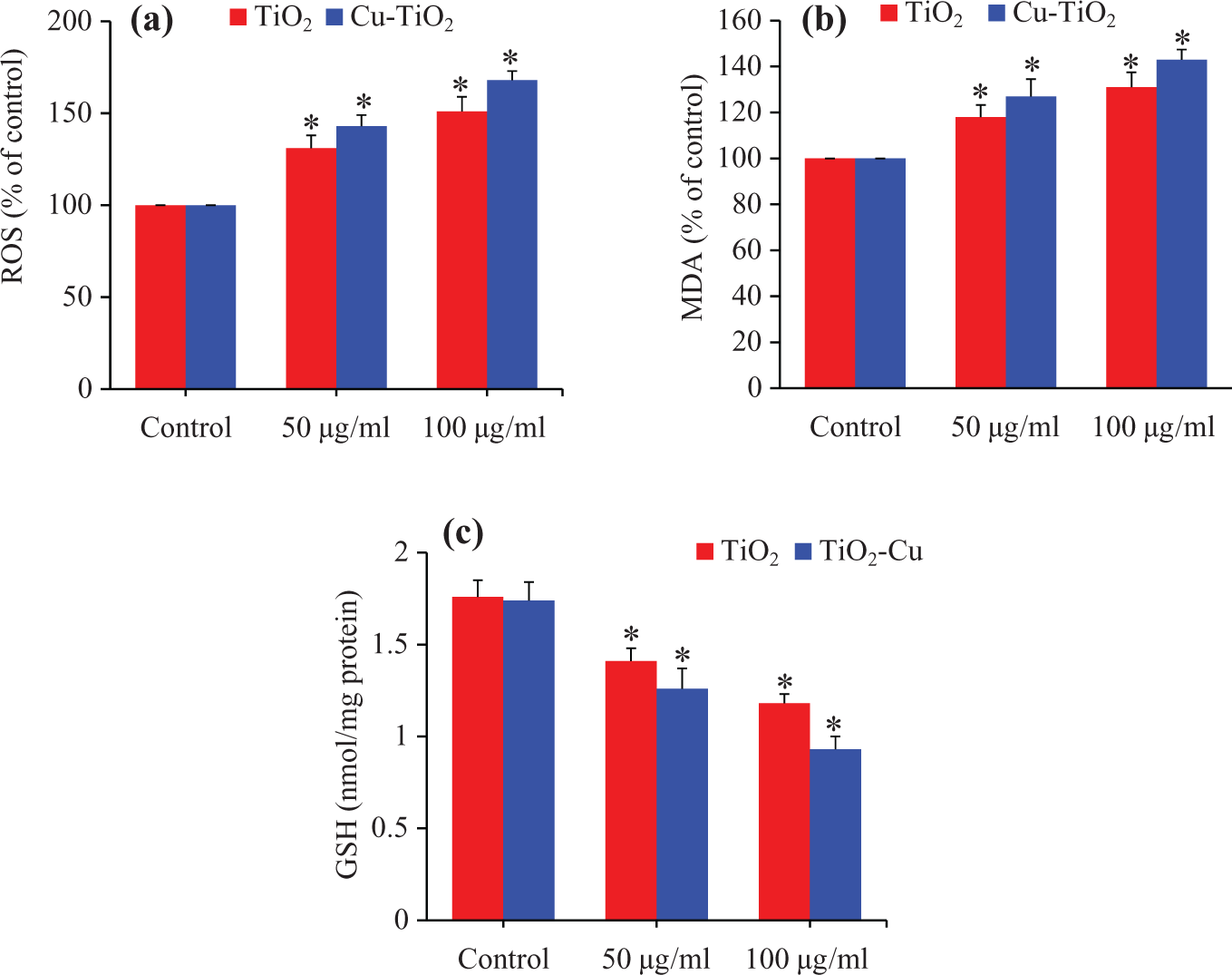
Oxidative stress response of pure and Cu-doped TiO2 NPs. The A549 cells were treated with pure and Cu-doped TiO2 NPs at the concentrations of 50 and 100 µg/ml for 24 h. At the end of exposure time, parameters were determined. (a) ROS, (b) MDA, and (c) GSH. Data represented are mean ± SD of three identical experiments made in three replicate. *Significant difference as compared to control (
Cu-doping increased the apoptotic response of TiO2 NPs
The MMP level was evaluated after pure and Cu-doped TiO2 NPs exposure using Rd123 fluorescent probe (Figure 5(a)). Results have shown that both pure and Cu-doped TiO2 NPs reduced the level of MMP dose dependently (Figure 5(a)). We further determined the activity of apoptotic enzyme caspase-3 in A549 cells treated with pure and Cu-doped TiO2 NPs at the concentrations of 50 and 100 µg/ml for 24 h. Results demonstrated that the activity of this enzyme was higher in cells after exposure to pure and Cu-doped TiO2 NPs (Figure 5(b)). Similar to oxidative stress and cytotoxicity data, caspase-3 enzyme activity along with MMP loss due to Cu-doped TiO2 NPs was higher as compared to pure TiO2 NPs.
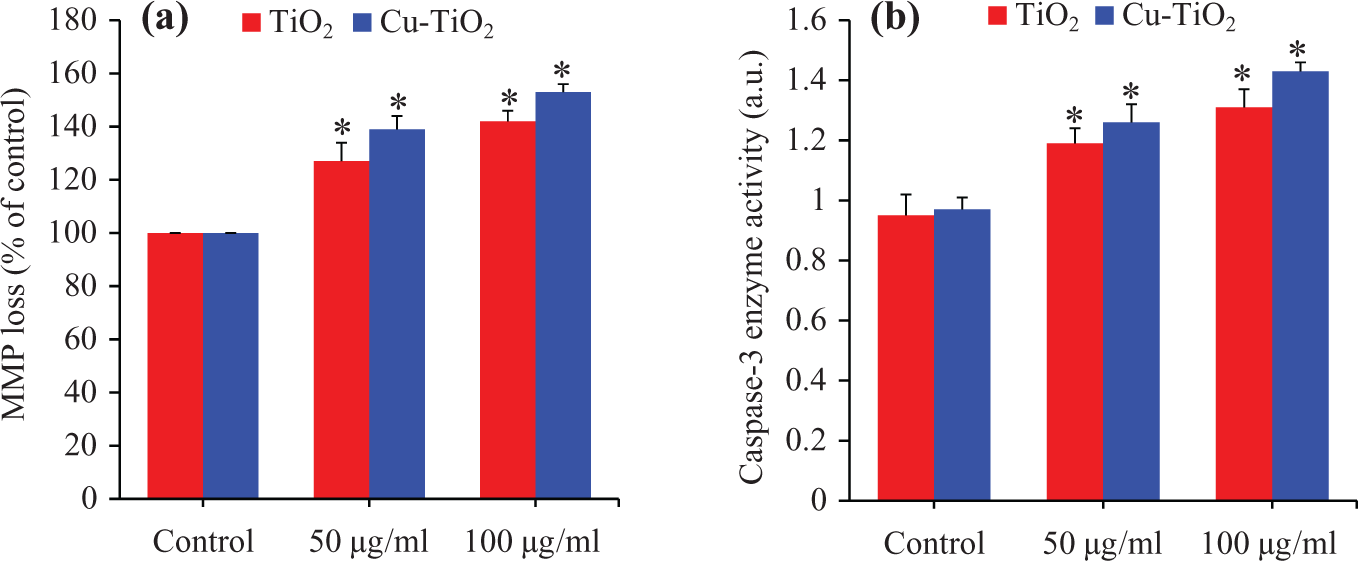
MMP and caspase-3 enzyme activity in A549 cells after exposure to pure and Cu-doped TiO2 NPs. The A549 cells were treated with pure and Cu-doped TiO2 NPs at the concentrations of 50 and 100 µg/ml for 24 h. (a) MMP and (b) Caspase-3 enzyme activity. Data represented are mean ± SD of three identical experiments made in three replicates. *Significant difference as compared to control (
Pure and Cu-doped TiO2 NPs-induced cytotoxicity was mediated through ROS
In order to investigate whether oxidative stress might play a critical role in cytotoxic response, A549 cells treated with both pure and Cu-doped TiO2 NPs in the presence or absence of ROS scavenger NAC. We have also taken ZnO NPs as a positive control. We observed that NAC effectively prevented ROS generation and GSH depletion caused by pure TiO2 NPs, Cu-doped TiO2 NPs, or ZnO NPs (Figure 6(a) and (b)). We further noticed that co-exposure of NAC, abolished almost fully the cell viability reduction caused by pure TiO2 NPs, Cu-doped TiO2 NPs, or ZnO NPs exposure (Figure 6(c)). Altogether, our data suggested that cytotoxicity in A549 cells induced by pure and Cu-doped TiO2 NPs was mediated through oxidative stress.

Pure and Cu-doped TiO2 NPs induced cytotoxicity was mediated through ROS generation. The A549 cells were treated with pure and Cu-doped TiO2 NPs at the concentration of 50 μg/ml in the presence or absence of NAC (10 mM) for 24 h. The ZnO NPs was used as a positive control. After the exposure completed, oxidative stress and cytotoxicity parameters were determined. (a) ROS, (b) GSH, and (c) MTT cell viability. Data represented are mean ± SD of three identical experiments made in three replicates. *Significant difference as compared to control (
Discussion
For the realistic interpretation of data, it is indispensable to characterize the physical and chemical properties of NPs before their biochemical or toxicity studies. 46,47 We have characterized the shape, size, crystalline nature, and aqueous behavior of pure and Cu-doped TiO2 NPs. High-resolution transmission electron microscopy image suggested the presence of both Cu and TiO2 NPs with high-quality lattice fringes without any distortion. XRD spectra indicated that both pure and Cu-doped TiO2 NPs have the same anatase TiO2 peaks and no peak shift. This is the characteristic of surface functionalization rather than Cu incorporation into the TiO2 lattice. Both TEM and XRD measurements showed that the size of Cu-doped TiO2 NPs was slightly smaller (24 nm) as compared to pure TiO2 NPs (30 nm). Reduction in the size of metal oxide NPs after doping is a very common trend and also reported in our earlier work. 26,48 In this case, investigators postulated that when Cu2+ ions doped into the TiO2 lattice, they likely to inhibit the growth of TiO2 crystals. Hydrodynamic size (secondary particle size) of pure and Cu-doped TiO2 NPs was three to five times bigger than those of primary particle size (TEM or XRD size; Table 1). The higher size of NPs was due to the tendency of particles to agglomerate in the aqueous state. The present data is in agreement with other reports 49,50 and briefly discussed in the previous work. 51 Zeta potential study indicated that both the pure and Cu-doped TiO2 NPs dispersed in deionized water had positive surface charge. However, the surface charge of the aggregates in complete culture medium was negative. Different charges on the surface of NPs may be due to coating of negatively charged proteins present in culture medium. Hence, primary size, hydrodynamic size, as well as surface charge of NPs might play important roles in biomedical and toxicity studies.
NPs exposure may get through inhalation, ingestion, or dermal contact.
9
Hence, it is critical to explore the toxicity of Cu-doped TiO2 NPs in relevant human tissues. We have chosen human lung epithelial (A549) cells as the
Cytotoxic response of pure and Cu-doped TiO2 NPs in A549 cells was examined by MTT, NRU, and morphological studies. These parameters serve as sensitive and integrated tools of cytotoxicity. 53,54 Both MTT and NRU assay showed that pure and Cu-doped TiO2 NPs reduced the cell viability in a dose-dependent manner. Microscopy data also showed a significant lowering of cell density and rounding of cells after exposure to these NPs. Moreover, cytotoxic response induced by Cu-doped TiO2 NPs was higher than those of pure TiO2 NPs. Our cytotoxicity data were in accordance with lower MMP and higher caspase-3 enzyme (apoptotic enzyme) activity of A549 cells exposed to pure and Cu-doped TiO2 NPs. Alterations in lysosomal and mitochondrial activity along with MMP loss suggested that Cu-doped TiO2 NPs could induce apoptotic cell death through the mitochondrial pathway. Mitochondrial events of apoptotic cell death involve the opening of a pore in the inner mitochondrial membrane, referred to as mitochondrial permeability transition pore (MPTP) and MMP loss. 55 Opening of the MPTP results in mitochondrial swelling and rupture of the outer mitochondrial membrane. This phenomenon consequently leads to the release of apoptogens that likely engage the components of the apoptosis machinery to further enhance the cell death. 56,57
At last, we explored the potential mechanisms of cytotoxicity caused by pure and Cu-doped TiO2 NPs in A549 cells. ROS generation and oxidative damage of cellular biomolecules have been proposed as possible mechanisms of toxicity caused by NPs. 51,58 It is well stated that ROS has potential to induce genetic damage, inflammation, and cell death. 59,60 However, investigations exploring the underlying mechanisms through which NPs generated ROS and ultimately cells death are still underway. Our recent work demonstrated that doped metal oxide NPs have potential to cause oxidative damage to human cells. 4,51 In this study, we found that ROS level and MDA level, a marker of LPO, increase with increasing the concentrations of pure and Cu-doped TiO2 NPs. Generation of ROS beyond a limit is toxic to cells because of their ability to oxidize cellular biomolecules including antioxidant GSH, which plays a vital role in preserving redox homeostasis. Pure and Cu-doped TiO2 NPs induced depletion of GSH level in a dose-dependent manner in A549 cells. Cell has a number of defense mechanisms against oxidative stress including direct interaction with antioxidant molecules. Indeed, co-exposure of an antioxidant NAC (ROS scavenger) effectively abrogated the ROS generation and GSH depletion as well as the cell viability reduction induced pure and Cu-doped TiO2 NPs in A549 cells, indicating that oxidative stress might be the responsible factor in the cytotoxicity of these NPs.
Conclusion
Characterization data demonstrated that size of Cu-doped TiO2 NPs was lower than pure TiO2 NPs (24 nm vs. 30 nm). Both pure and Cu-doped TiO2 NPs induced cytotoxicity and oxidative stress in a dose-dependent manner in A549 cells. However, the toxic response of Cu-doped TiO2 NPs was slightly higher than those of pure one. We further observed that antioxidant NAC effectively abrogated the oxidative stress and cytotoxicity caused by pure and Cu-doped TiO2 NPs. Altogether, our data suggested Cu-doped TiO2 NPs toxicity in A549 cells via ROS generation. This study warranted further investigation on the role of Cu-doping in toxicity mechanisms of TiO2 NPs in different in vitro as well as in vivo models.
Footnotes
Acknowledgement
The authors are grateful to the Deanship of Scientific Research, King Saud University for funding through Vice Deanship of Scientific Research Chairs.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
