Abstract
Cardiac arrhythmia is a common cause of mortality, and its progression may be due to abnormal sympathetic nerve activity and catecholamine release. Besides, lactate dehydrogenase (LDH) and creatine kinase (CK) downregulation and adiponectin expression play important roles in promoting coronary artery disease. The study aimed to examine the possible cardioprotective effect of members of phosphodiesterase type 5 (PDE-5) inhibitors in epinephrine-induced arrhythmia in rats. Arrhythmia was induced by cumulative boluses of epinephrine (4, 8, 16, 32, 64, and 128 mg/kg) given at 10-min intervals. Rats were randomly allocated into five groups. Group I: Normal control group received only saline. Group II: Rats injected with epinephrine and served as arrhythmia group. Groups III, IV, and V: Rats received daily oral sildenafil (0.5 mg/kg), vardenafil (3 mg/kg), and tadalafil (10 mg/kg), respectively, for 30 days prior to epinephrine injections. Injection of epinephrine to rats decreased heart rate and QTc interval but increased RR interval and duration of arrhythmia. Epinephrine group had lower serum reduced glutathione (GSH) and adiponectin levels and higher serum malondialdehyde (MDA), nitric oxide (NO), heart LDH, and CK contents. Histopathological investigations of epinephrine group provoked necrotic changes with strong positive immunoreactivity for caspases-3. While pretreatment of rats with PDE-5 inhibitors improved GSH and adiponectin contents, ameliorated serum MDA and NO levels and heart LDH and CK contents and corrected epinephrine-induced histopathological changes. PDE-5 inhibitors may delay epinephrine-induced arrhythmia through expression of adiponectin and downregulation of heart LDH and CK.
Keywords
Introduction
Cardiac arrhythmia is a common cause of mortality, and its progression may be due to abnormal sympathetic nerve activity. 1 Catecholamines as epinephrine induce arrhythmias via the activation of cyclic adenosine monophosphate/protein kinase system as a result of β-adrenergic receptor stimulation. 2 High doses of circulating epinephrine cause a transient increase in heart rate (HR) followed by a decrease in HR due to stimulation of baroreceptors and vagal-mediated slowing of the HR. 3 Moreover, arrhythmia precedes via catecholamine oxidation products, also known as aminochromes, as well as the reactive oxygen species (ROS) 4 that contribute to heart rhythm disorders, cellular alterations, and cardiac cell damage. 5 Oxidative stress has crucial roles in cardiac and vascular diseases. The deleterious effects of ROS on cardiac function are mainly due to their actions on subcellular organelles and induction of intracellular calcium overload, 6 thus decreasing the force of contraction. 7
Large doses of the catecholamines, experimentally, produce lesions similar to myocardial infarction mainly due to leakage of enzymes into the blood stream from destroyed and ruptured myocardial cells. 8 One of the reliable markers of myocardial cell damage is lactate dehydrogenase (LDH). LDH is specifically released in blood stream in myocardial damage more than other enzymes such as creatine kinase (CK). 9
Adiponectin, a circulating cytokine derived from white adipose tissue and cardiomyocytes, has been suggested to possess many cardioprotective properties, such as anti-inflammatory, antiatherogenic, and antihypertensive effects. 10
Phosphodiesterases (PDEs) are crucial regulators of the intracellular Cyclic adenosine monophosphate (cAMP), thus affecting many intracellular functions. PDEs are abundant in high concentrations in platelets, smooth muscle cells of the peripheral arterial, and venous vessels as well as coronary and pulmonary circulation. 11 PDE inhibitors antagonize the biosynthesis or actions of PDEs. PDE-5 inhibitors are used for the treatment of erectile dysfunction in men and pulmonary arterial hypertension. These vasodilator and inotropic agents have protective effects on vascular structures and myocardial muscles. 12
The relationship between cardiac changes during the progression of arrhythmia and PDE-5 inhibitors is unclear. Therefore, the aim of the current study was to investigate the possible mechanisms of PDE-5 inhibitors efficacy in epinephrine-induced arrhythmia through studying the electrocardiographic changes, biochemical cardiac markers, antioxidant parameters, anti-inflammatory cardioprotective marker (adiponectin), and apoptotic parameters along with histopathological changes.
Materials and methods
Animals
Adult male Wister albino rats weighing 120–140 g were purchased from the animal house colony of the National Research Centre (Dokki, Giza, Egypt) and were kept in the animal house under conventional laboratory conditions. Experiment was performed according to the National Regulations of Animal Welfare and Institutional Animal Ethical Committee and was approved by National Research Centre (Dokki, Giza, Egypt).
Chemicals
Epinephrine was purchased from Sigma–Aldrich (MO, USA). All other chemicals were of the highest available commercial grade.
Drugs
Sildenafil (Viagra®, Pfizer, Egypt).
Vardenafil (Levitra®, Bayer Pharma AG, Germany).
Tadalafil (Cialis®, Eli Lilly and Company, Indianapolis, USA).
Experimental design
Arrhythmia was induced by cumulative doses of intravenous (i.v.) boluses of epinephrine (4, 8, 16, 32, 64, and 128 mg/kg) given at 10-min intervals or until death of the animals. This experimental design is similar to that used by Sethi et al. 13 but these cumulative doses are based on our trials. Rats were randomly allocated into five groups; six rats each. Group I: animals received only saline solution daily for 30 days and served as normal control group. Group II: Rats injected with epinephrine and served as arrhythmia group. Groups III, IV, and V: Rats received sildenafil (0.5 mg/kg/day; per os [p.o.]), vardenafil (3 mg/kg/day; p.o.), and tadalafil (10 mg/kg/day; p.o.), respectively, for 30 days prior to epinephrine injections.
Recording of electrocardiogram pattern
On day 31, rats were anesthetized using thiopental (45 mg/kg; intraperitoneal [i.p.]) and electrocardiogram (ECG) was recorded. Thereafter, epinephrine-induced arrhythmia was conducted (baseline and every 10 min) till termination of arrhythmia(s) using ECG Powerlab module which consists of Powerlab/8sp and Animal Bio-Amplifier, Australia, in addition to Lab Chart 7 software with ECG analyzer.
The measured parameters after epinephrine-induced arrhythmia were HR, RR interval(s), QTc interval, and duration of arrhythmia(s).
Biochemical analysis of serum and heart tissue
Immediately after recording the ECG pattern, blood samples were collected from retro-orbital venous plexus of rats. Collected blood samples were allowed to stand for 10 min at room temperature, then centrifuged at 4°C using cooling centrifuge (Laborezentrifugen, 2k15; Sigma, Germany) at 3000 r/min for 10 min and sera were separated for the assessment of serum level of malondialdehyde (MDA), nitric oxide (NO) metabolites, reduced glutathione (GSH), and adiponectin content using commercially available kits.
Directly after collecting the last blood sample in the experiment, rats were killed under light diethyl ether anesthesia and their hearts were carefully isolated and homogenized (using MPW–120 homogenizer; Med instruments, Poland); the homogenate was centrifuged using a cooling centrifuge (Laborezentrifugen, 2k15; Sigma) for 10 min; the supernatant was used for the determination of heart contents, such as LDH and CK, using commercially available kits.
Histopathological examination of heart
For light microscopic examination, heart tissues from each group were fixed with 10% buffered formalin, embedded with paraffin. After routine processing, paraffin sections of each tissue were cut into 5 μm thickness and stained with hematoxylin and eosin (H&E). Photomicrographs of the stained sections were taken with the aid of a light microscope. 14
Immunohistochemistry for caspase-3
Immunohistochemical staining of anti-caspase-3 antibodies was performed by streptoavidin–biotin. Four-micrometer-thick sections were deparaffinized and incubated with fresh 0.3% hydrogen peroxide in methanol for 30 min at room temperature. The specimens were then incubated with anti-caspase-3 antibody as the primer antibody at a 1:100 dilution. The specimens were counterstained with H&E. Negative controls were prepared by substituting normal mouse serum for each primary antibody.
Data analysis
All the values are presented as means ± standard error (SE) of the means. Comparisons between different groups were carried out using one-way analysis of variance followed by Tukey–Kramer multiple comparison test. Difference was considered significant when p < 0.05. Graph Pad prism® software (version 5) was used to carry out these statistical tests.
Results
Effects of pretreatment with sildenafil, vardenafil, and tadalafil on ECG pattern
Epinephrine-injected rats showed a significant decrease in HR by 58.85% and prolonged QTc interval by 52.94% as compared to normal control rats. Moreover, epinephrine-injected rats showed a significant increase in RR interval by 213.33% as compared to normal control rats. While, sildenafil, vardenafil, and tadalafil altered epinephrine-induced changes in ECG, as they showed significant rise in HR by 145.57, 169.62, and 140.51%, respectively, and rise in QTc interval by 50, 50, and 75%, respectively, as well as decrease in RR interval by 57.45, 70.21, and 65.96%, respectively, and a decrease in the duration of arrhythmia(s) by 58.75, 60, and 60%, respectively, as compared to epinephrine-induced arrhythmia group (Table 1).
Effects of pretreatment with sildenafil (0.5 mg/kg/day, p.o.), vardenafil (3 mg/kg/day, p.o.), and tadalafil (10 mg/kg/day, p.o.) for 30 days prior to epinephrine injections (4, 8, 16, 32, 64, and 128 mg/kg, i.v., given at 10-min intervals) on ECG pattern.

ECG: electrocardiogram; ANOVA: analysis of variance; SE: standard error.
aData were expressed as mean ± SE (n = 6); Data were analyzed by one-way ANOVA followed by Tukey–Kramer multiple comparison test.
b p < 0.05: significantly different from normal control group.
c p < 0.05: significantly different from epinephrine-induced arrhythmia group.
Biochemical analysis of serum and heart tissue
Effects of pretreatment with sildenafil, vardenafil, and tadalafil on serum GSH, MDA, and NO contents
Induction of arrhythmia by epinephrine significantly decreased serum GSH content by 25.90% and increased serum MDA and NO contents by 36.25 and 21.80%, respectively, as compared to normal control group. Animals pretreated with vardenafil and tadalafil significantly increased serum GSH content by 5.88 and 10.55%, respectively. Animals pretreated with sildenafil, vardenafil, and tadalafil showed a decreased serum MDA content by 15.28, 20, and 22.83%, respectively, and decreased serum NO content by 7.65, 8.64, and 8.52%, respectively, as compared with epinephrine-induced arrhythmia group (Figure 1(a) to (c)).

Effects of pretreatment with sildenafil (0.5 mg/kg/day, p.o.), vardenafil (3 mg/kg/day, p.o.), and tadalafil (10 mg/kg/day, p.o.) for 30 days prior to epinephrine injections (4, 8, 16, 32, 64, and 128 mg/kg, i.v.; given at 10-min intervals) on serum: (a) GSH, (b) MDA, (c) NO, and (d) adiponectin contents. Data were expressed as mean ± SE (n = 6). Data were analyzed by one-way ANOVA followed by Tukey–Kramer multiple comparison test. a′ p < 0.05: significantly different from normal control group. b′ p < 0.05: significantly different from epinephrine-induced arrhythmia group. GSH: reduced glutathione; MDA: malondialdehyde; NO: nitric oxide; ANOVA: analysis of variance; SE: standard error.
Effects of pretreatment with sildenafil, vardenafil, and tadalafil on serum adiponectin content
Serum adiponectin level was lower in the epinephrine-induced arrhythmia group by 45.29% than in normal control group while higher serum adiponectin level was observed in sildenafil and tadalafil only by 20.84 and 39.58% when compared to epinephrine-induced arrhythmia group (Figure 1(d)).
Effects of pretreatment with sildenafil, vardenafil, and tadalafil on heart tissue LDH and CK contents
Epinephrine-injected rats showed a significant elevation in heart tissue LDH and CK contents by 289.32 and 291.03%, respectively, as compared to normal control rats. While, sildenafil, vardenafil, and tadalafil suppressed the elevated heart tissue LDH content by 29.28, 53.62, and 66.14%, respectively, and CK content by 33.12, 54.46, and 69.49%, respectively, as compared to epinephrine-induced arrhythmia group (Figure 2(a) and (b)).

Effects of pretreatment with sildenafil (0.5 mg/kg/day, p.o.), vardenafil (3 mg/kg/day, p.o.), and tadalafil (10 mg/kg/day, p.o.) for 30 days prior to epinephrine injections (4, 8, 16, 32, 64, and 128 mg/kg, i.v.; given at 10-min intervals) on heart tissue: (a) LDH and (b) CK contents. Data were expressed as mean ± SE (n = 6). Data were analyzed by one-way ANOVA followed by Tukey–Kramer multiple comparison test. a′ p < 0.05: significantly different from normal control group. b′ p < 0.05: Significantly different from epinephrine-induced arrhythmia group. LDH: lactate dehydrogenase; CK: creatine kinase; ANOVA: analysis of variance; SE: standard error.
Histopathological examination
Heart tissue of normal control rats showed regularly arranged myocardial fibers and muscle bundles with centrally located nuclei of cardiomyocytes (Figure 3(a)). Examination of H&E-stained sections in epinephrine-exposed heart showed loss of striations, necrotic changes, and cytoplasmic vacuolization in cardiac myocytes. Moreover, massive extended hemorrhagic areas were apparently seen. Mononuclear cellular infiltration, dilated blood vessels, and nuclei of cardiac myocytes showed changes in the form of pyknotic nuclei (Figure 3(b)). The histopathology of the heart in sildenafil-treated rat showed moderate disorganization of myocardial fibers, moderate degrees of necrosis of the cardiac muscle of rat, and congested and dilated blood vessels with pyknotic nuclei (Figure 3(c)). Vardenafil administration resulted in mild disorganization and moderate degrees of necrosis of the cardiac muscle of rat along with infiltration of inflammatory cells, congestion, and dilation of blood vessels (Figure 3(d)). Tadalafil administration resulted in diminished loss of striations and a nearly normal microscopic structure of cardiac myocytes. Mild changes in few nuclei in the form of pyknotic nuclei were observed in this tadalafil group. A decreased degree of necrosis (mild) and infiltration of inflammatory cells were also observed (Figure 3(e)).
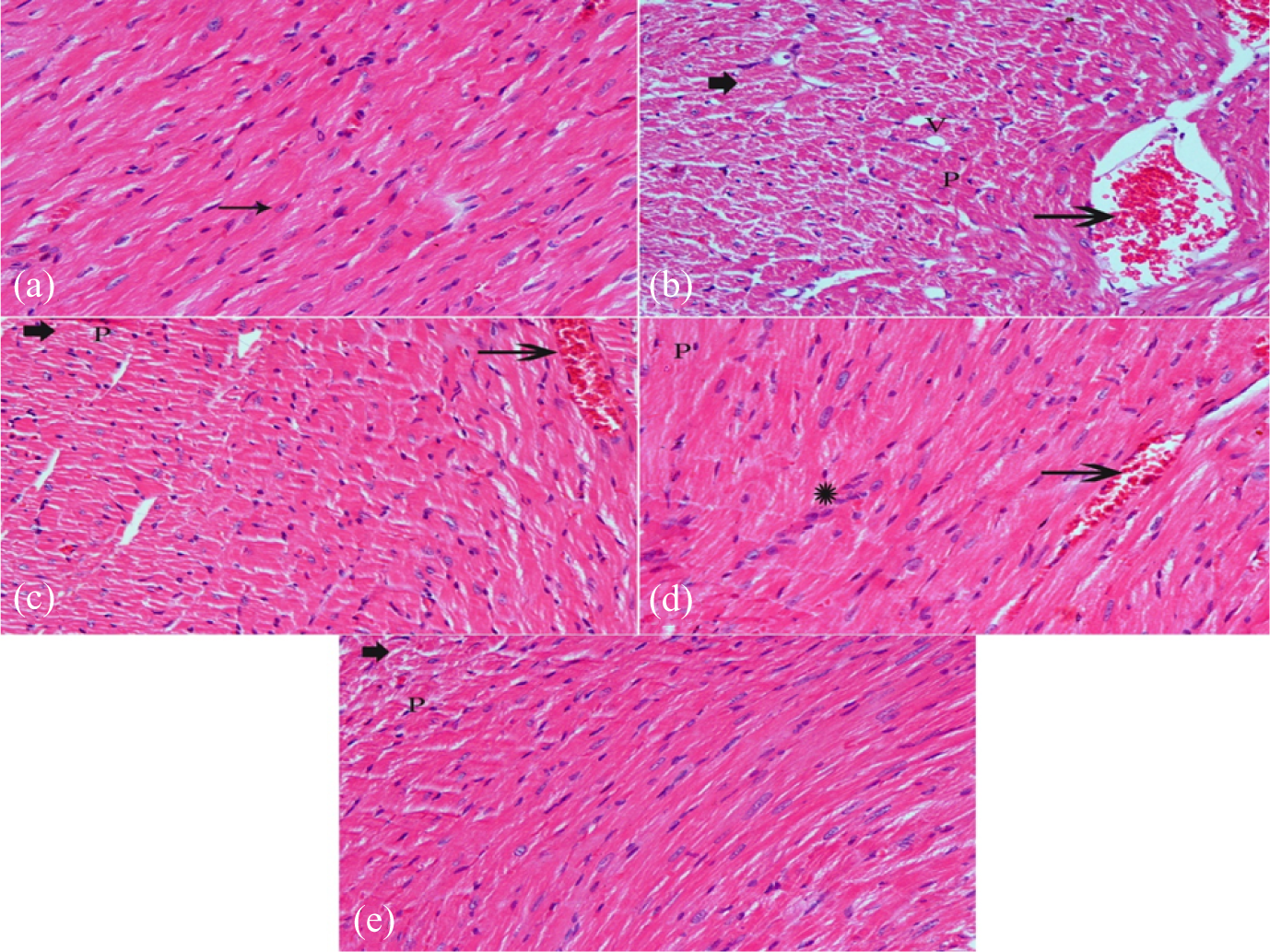
Photomicrographs of sections of the heart tissue from: (a) a normal control rat; (b) a rat with epinephrine-induced arrhythmia; (c) a rat pretreated with sildenafil (0.5 mg/kg/day, p.o.) for 30 days prior to epinephrine injections; (d) a rat pretreated with vardenafil (3 mg/kg/day, p.o.) for 30 days prior to epinephrine injections; (e) a rat pretreated with tadalafil (10 mg/kg/day, p.o.) for 30 days prior to epinephrine injections (hematoxylin and eosin, ×50).
Immunohistochemical of caspase-3
Normal control group showed no positive immunoreactivity for caspase-3 immunoreaction (Figure 4(a)). Section from the group received epinephrine showed strong positive immunoreactivity for caspases-3 in the cytoplasm of myocardial cells (Figure 4(b)). Examination of a section of the group treated with epinephrine and sildenafil showed mild positive immunoreactivity for caspases-3 in the cytoplasm of myocardial cells (Figure 4(c)). However, groups treated with epinephrine and either vardenafil or tadalafil showed week immunoreactivity for caspases-3 in the cytoplasm of myocardial cells (Figure 4(d) and (e)).
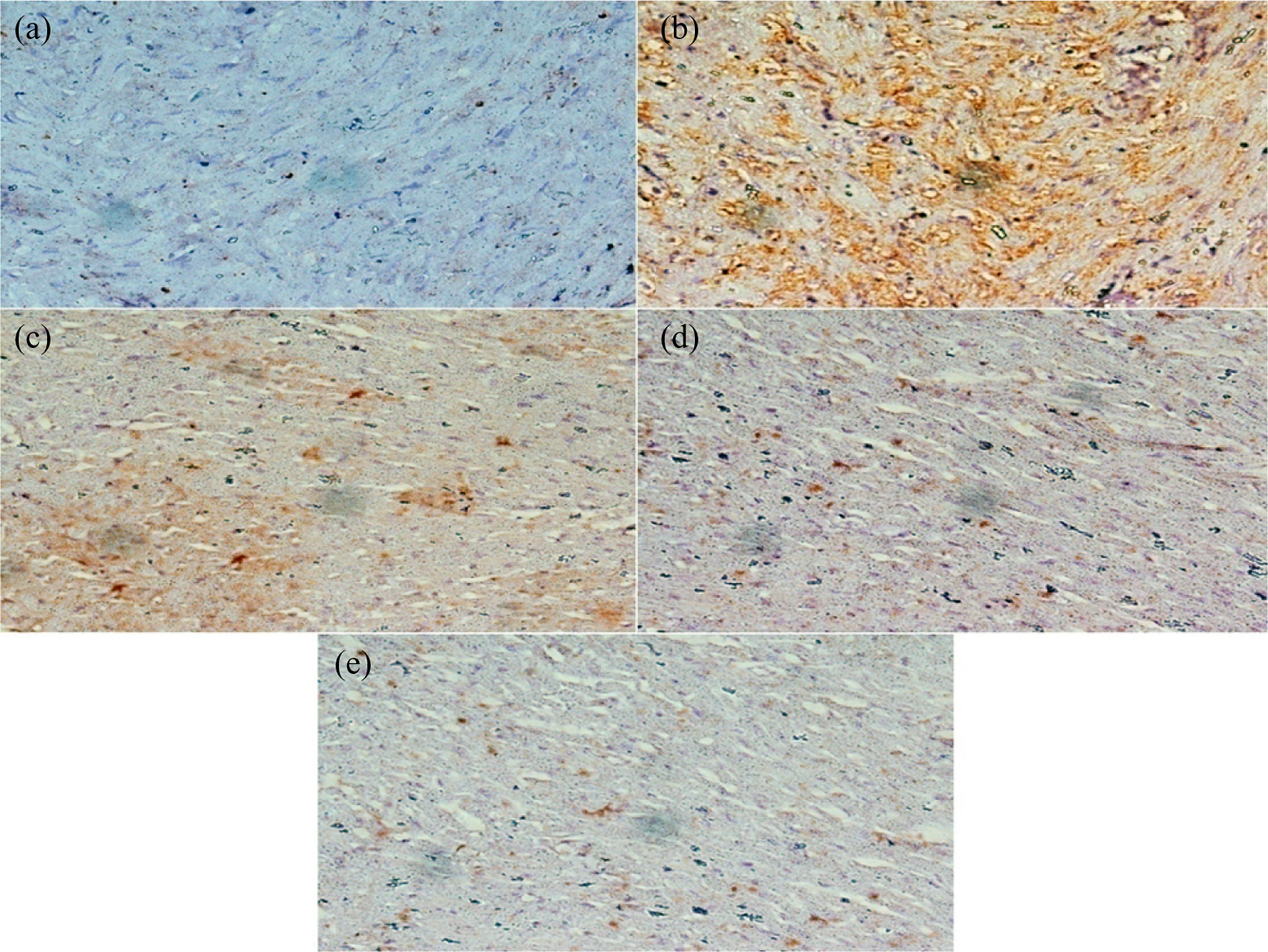
Photomicrographs of sections of the heart tissue from: (a) a normal control rat; (b) a rat with epinephrine-induced arrhythmia; (c) a rat pretreated with sildenafil (0.5 mg/kg/day, p.o.) for 30 days prior to epinephrine injections; (d) a rat pretreated with vardenafil (3 mg/kg/day, p.o.) for 30 days prior to epinephrine injections; (e) a rat pretreated with tadalafil (10 mg/kg/day, p.o.) for 30 days prior to epinephrine injections (immunostaining for caspase-3, ×400).
Discussion
The present study was designed to evaluate the effects of PDE-5 inhibitors on epinephrine-induced arrhythmia in rats. Boluses of i.v. injections of epinephrine led to prolongation of RR interval and duration of arrhythmia(s) accompanied by significant decrement of HR and prolongation of QTc interval. These results agree with Sethi et al. who observed that a bolus injection of epinephrine has short onset and long duration of arrhythmia. A bolus injection of epinephrine induced various types of arrhythmias such as sinoatrial block and missed beats. 2
PDE-5 inhibitors, in the current work, modified the myocardial sensitivity to epinephrine-induced arrhythmia, delayed the onset of arrhythmia, and decreased RR interval. Moreover, arrhythmia duration became shorter and HR and QTc intervals were significantly increased as compared to epinephrine-induced arrhythmia group. Nagy et al. reported that oral administration of sildenafil to dogs reduces arrhythmia severity. 15
Our results demonstrated an elevation in serum MDA level and a reduction in serum GSH content as a sequence of oxidative stress after boluses of i.v. epinephrine injection. These results are in agreement with Sethi et al. who observed that epinephrine produced arrhythmias as they measured the plasma levels of aminochromes, an index of catecholamine oxidation, and MDA, an index of lipid peroxidation that was about 100-fold higher on injecting epinephrine cumulative doses. 13
The current study also tested the antioxidant effects of PDE-5 inhibitors against lipid peroxidation due to injection of cumulative boluses of epinephrine and it was apparent that serum MDA level was significantly reduced in PDE-5 inhibitors group as compared to epinephrine-induced arrhythmia group. In addition, PDE-5 inhibitors elevated serum GSH content as compared to epinephrine-induced arrhythmia group. The role of oxidative stress has been extensively evaluated in the pathophysiological mechanisms of arrhythmia and the role of PDE-5 inhibitors in scavenging lipid peroxide has been investigated in this study. Sheweita et al. concluded that PDE-5 inhibitors induced both superoxide dismutase and catalase activities and decreased MDA level in rat liver. 16 Arikan et al. demonstrated that tadalafil showed significant antioxidant and protective effects against I/R injury of the rat ovary. 17
Boluses of i.v. injections of epinephrine, in the current work, showed higher serum NO content and lower serum adiponectin level when compared to normal control group. These results may be explained by Bussey et al. who stated that relaxation of pre-constricted isolated aortic rings and increased productions of the vasodilator NO by endothelial cells occur in response to adiponectin. 18 Additionally, deficiency of adiponectin exacerbates cardiac damage under various pressure overloads in in vivo experiments. 10 The current study showed that PDE-5 inhibitors resulted in lower NO content as compared to epinephrine-induced arrhythmia group. Furthermore, we tested, for the first time, the effects of PDE-5 inhibitors on adiponectin upregulation, which mainly acts as a cardioprotective cytokine. Sildenafil and tadalafil showed elevated serum adiponectin levels as compared to epinephrine-induced arrhythmia group.
Epinephrine evoked LDH and CK release reflecting the alterations in membrane integrity. These results suggested that epinephrine mediated enzyme leakage. This data was further supported by our histopathological evidence of myocardial cellular damage, which depicted loss of heart striations, necrotic changes in the cardiac myocytes, and cytoplasmic vacuolization in cardiac muscle cells. Also nuclei of cardiac myocytes showed changes in the form of pyknotic nuclei. Wheatley et al. demonstrated severe myocardial enzyme leakage in the Langendorff-perfused rat heart. 19 Osman et al. also revealed elevation in the levels of serum creatine kinase-MB (CK-MB) isoform and troponin-I in isoproterenol-induced arrhythmia group. 20
In the present study, PDE-5 inhibitors downregulated LDH and CK release as compared to epinephrine-induced arrhythmia group. This downregulation was further evidenced by our histolopathological study that depicted amelioration of heart tissue damage and necrotic changes in the cardiac myocytes of PDE-5 inhibitors group. Al-Hesayen et al. demonstrated huge evidence supporting the role of LDH and CK downregulation by acute administration of sildenafil in chronic human heart failure. 21
The present study also showed an increased caspase-3 expression in epinephrine-induced arrhythmia group. Caspase-3 is an apoptotic marker that is mainly overexpressed after epinephrine cumulative injections inducing necrotic changes in the cardiac myocytes. These changes further lead to cytoplasmic vacuolization and mononuclear cellular infiltration, thus eventually leading to cell apoptosis. Similar to our results, Sun et al. used isoproterenol to induce acute myocardial infarction and reported that isoproterenol upregulated caspase-3 activity. 22 Our study revealed that PDE-5 inhibitors ameliorated necrotic changes in the cardiac myocytes and suppressed cell apoptosis induced by epinephrine via downregulation of caspase-3 expression. Koka et al. demonstrated that tadalafil exhibited protective effects against doxorubicin-induced cardiotoxicity and attenuated myocyte apoptosis. 23
Conclusions
PDE type 5 inhibitors, such as sildenafil, vardenafil, and tadalafil, may exert significant cardioprotective effects in epinephrine-induced arrhythmia in rats. Tadalafil pretreatment was found to be more beneficial than either sildenafil or vardenafil pretreatment in amelioration of epinephrine-induced arrhythmia. PDE-5 inhibitors can reduce the inducibility of arrhythmias and apoptosis response through suppressing cardiac sympathetic nerve activity, inhibition of ROS, expression of adiponectin, downregulation of LDH and CK, and suppression of apoptosis.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
