Abstract
Cereal-based diets formulated as semi-purified diets can provide flexibility for researchers, enabling open controlled formulas, besides being cheaper than purified diets. Seeking to widen the researchers’ options in their experimental design developments, we aimed at assaying the chemical score, growth performance and protein utilization of nine semi-purified experimental chows. These diets were formulated at 17.8%, 12% and 8% protein contents, using three variations of ingredients for each one, as follows: casein (C), casein+soybean meal (C+S, 1:1 w/w protein) and casein+soybean meal+corn gluten meal (C+S+G, 1:1:1 w/w protein), without cysteine supplementation and setting casein 17.8% (AIN-93G) as reference diet. The diets C and C+S (17.8%) had the Cys as the limiting amino acid once the addition of gluten turns the Lys as second limiting. All diets had the potential for promoting growth with body mass gain, feed conversion ratio (FCR; chow consumed per body weight gain, average 3.12) and feed efficiency ratio (FER; body weight gain per chow consumed, average 0.3), except for C+S+G 8% (FCR = 6 g; FER = 0.13). These variations were mainly due to the protein amounts independently from the protein ingredient used and could characterize the C+S+G 8% as unable to support growth. For the other parameters (digestibility, net protein ratio, net protein utilization and protein efficiency ratio), there were no relevant differences between the diets. We can conclude that 17.8%, 12% and 8% chows (C and C+S) allowed a proper combination of ingredients from the point of view of palatability, nutrient availability/utilization, metabolic processes, growth performance and feed utilization parameters.
Introduction
Nutritional science research has shown that diet is a powerful environmental tool capable of changing the phenotype of an animal. 1 There is a huge variety of diets to attend animal needs and even allow performing diet-induced disease models. In these models, the diets play a key role in inducing a diseased state that reaches the altered phenotype of an animal, such as obesity, dyslipidaemia, hepatosteatosis, atherosclerosis and hypertension. 1,2 The nutrient requirement of any laboratory animal species and how to fulfil it are the critical points for the research outcome and should be carefully considered so as to guide the formulation of diets. When these critical points are associated with the interest on administering some test component, a complex net of choices among diet kinds with their modifications and administration ways turns up. Currently, the experimental designs demand for dosing animals with a bioactive substance administered through experimental diets, gavage or injections, which have significant drawbacks. Isolating the specific interests, the use of gavage and injections in medium- or long-term experiments may be more stressful to animals and their handlers, besides the time involved in these procedures. As consequence, the animals’ behaviour and metabolism are affected, this may potentially confound the endpoints assayed, in addition to the experimental mistakes committed by handlers.
This way, laboratory animal diets have long been used to vehicle the test products and fall into two main categories such as purified ingredient diets and grain-based chow diets, also named cereal-based or natural ingredient-based diets. Cereal-based diets are classified as open (composition known to the user) or closed (composition known only to the commercial manufacturer) and use either purified or non-purified ingredients. Purified diets are made of refined ingredients including isolated proteins, and the chemically defined diets are made of chemically pure sources such as amino acids, mono- or disaccharides and purified fatty acids. 3 The purified or chemically defined diets which use highly refined ingredients whose source contains only one nutrient tend to standardization so as to reduce the inherent variation in commercial chows and to facilitate interpretation of results among experiments and laboratories. 4 These formulations guarantee the absence of interferences caused by non-nutritive substances included through the mix of ingredients and allow excluding a specific compound, such as a vitamin or mineral. However, diets using purified products differ from the complexity of the alimentary matrix used by humans and animals and its interactions. These aspects affect their biotransformation by intestinal enzymes and microbiota, producing high amount of metabolites in the end of the digestive and absorptive process. Moreover, the microbiota is variable depending on animal conditions, and these different microorganisms may have diverse abilities to generate other bioactive compounds from the ones found in foods. 5 –7 Besides, sensorial characteristics of refined proteins may differ substantially from their natural source or food and has been associated with animal behaviour in order to compose a balanced diet. 8 On the other hand, these diets present as disadvantages higher costs compared to commercial laboratory diets whose retail prices range from US$40 to US$100 per kilogram, especially chemically defined diets.
Thus, many researchers adopt cereal-based diets purchased as commercial diets to feed laboratory animals and they add their test products according to the interest of each experiment. This procedure provides flexibility for researchers to add ingredients and compounds themselves, testing as many doses as necessary, even to perform a pilot assay. Notwithstanding, this procedure is impaired when the commercial chow diets are recognized as a closed formula, where the exact amount of each ingredient added is not shown by the manufacturer 9 or when this inclusion alter the nutrient proportions spoiling growth performance. The latter will be the focus of present work.
Considering the great use of rats as experimental models, with their specific National Research Council (NRC) requirements 10 and the previous considerations about experimental diets, we proposed to investigate the feasibility of using semi-purified diets as intermediate formulations between purified and closed diets. For such goal, we sought to use accessible ingredients as protein sources in order to widen the researchers’ options in their experimental design developments, providing them with the knowledge of the amino acid profile and the animals’ nutritional performance.
Materials and methods
Animals and diets
Male weanling Wistar rats, weighing 50 ± 3.0 g, were fed standard laboratory rat chow for an acclimatization period of 2 days. After this period, animals were randomly divided into 10 experimental groups of eight rats each. They were housed in individual metabolic cages, in a room maintained at 24°C ± 1°C and 50–60% relative humidity, with a 12-h light/dark cycle. The animals were handled in accordance with Brazilian College of Animal Experimentation (COBEA) (Permission: 371/2011).
Diets were formulated according to the AIN-93G diet for rat growth, 4 except for the protein content which varied in concentration and sources as shown in Table 1, corresponding to 17.8%, 12% and 8% protein. For all these concentrations, we tested the following sources of protein with their respective proportions: casein (C), casein+soybean meal (C+S, 1:1 w/w protein) and casein+soybean meal+corn gluten meal (C+S+G, 1:1:1 w/w protein). These products were purchased as follows: casein and mineral mix (Rhoster Industria e Comércio Ltda), soybean meal (Cooperativa Mista dos Produtores Rurais do Sudoeste Goiano Ltda. – COMIGO), corn gluten meal (Proteinose from Ingredion Brasil). One experimental group was fed a protein-free diet. For the preparation of the diets, ingredients were homogenized and passed through in sieve to ensure uniformity and distribution of vitamins and minerals. The diets were prepared and kept in the refrigerator. Animal experiment was carried out during 14 days as follows: Group 1 (control) received the C 17.8% diet (AIN – 93G). Group 2 received the protein-free diet. Groups 3 and 4 were fed with C+S 17.8% and C+S+G 17.8%, respectively. Groups 5, 6 and 7 were fed with C 12%, C+S 12% and C+S+G 12%, respectively. Groups 8, 9 and 10 received the same proportions as the latter with the amount of 8% of protein. Group 11 was euthanized in the first experimental day, serving as initial composition body. Feed and water were provided ad libitum. Faecal and urine collection was done for the rats during the trial between days 5 and 10. Collection of urine and faeces was done individually on a daily basis for each rat in each metabolic cage. The urine from each cubicle was collected into small urine container. During this period, the faecal collection was bulked and stored in screw capped bottles. These bottles were stored at 4°C prior to chemical analysis. At the end of the rat trial, the bulked faecal samples for each rat were weighed, dried and milled prior to laboratory analyses.
Composition of experimental diets produced (g/kg diet) in three protein levels of 8, 12 and 17.8%.

APT: no protein content; C: casein; C+S: casein+soybean; C+S+G: casein+soybean+corn gluten meal.
aBased on AIN93G, Reeves et al. (1993) except to protein content.
At the end of the study, all animals were anesthetized with ketamine and xylazine and then euthanized by withdrawing blood from the heart. At necropsy, the abdominal cavity was opened and the liver, epididymal, visceral and intercostal fat were removed and weighed. 11,12 Following this, these tissues were replaced inside the animals and the carcasses were stored under −18°C for further analysis.
Nitrogen and protein determination
Analyses of nitrogen (N) from diets, urines and faeces were determined by micro-Kjeldahl method. 13 Protein was calculated as N × 6.25.
Diets chemical composition and amino acids profile
Moisture was determined using infrared Bel Engineering® Mark M 163 (120°C, 7 min), ash content was determined by incineration at 550°C in a muffle furnace, Association of Official Analytical Chemists (AOAC) 14 and fat content by the Bligh and Dyer 15 method. Soluble and insoluble dietary fibre determination was performed by AOAC enzymatic-gravimetric method, as described by Lee and Prosky. 16 Carbohydrate concentrations were estimated by difference.
Amino acids were determined by high-performance liquid chromatography using reversed-phase Luna C18, 4.6 × 250 mm 00G-4252-EQ column and ultraviolet absorption measurements (254 nm) after protein hydrolysis using 6 N hydrochloric acid at 110°C for 24 h. 17,18 The protein chemical score was calculated according to the procedure of Khattab et al. 19 and Rakowska et al. 20 which compares the concentration ratio of the amino acids having the shortest supply (restrictive amino acid) to the concentration of these amino acids in the standard protein. 4
Calculation of growth performance and nutrient utilization parameters
All calculations were performed for each rat individually. Growth performance and nutrient utilization were assessed in such terms as follows: feed conversion ratio (FCR), feed efficiency ratio (FER), protein efficiency ratio (PER), net protein ratio (NPR), true digestibility (TD), apparent digestibility and net protein utilization (NPU). 21 –23
Statistical analyses
Experimental data were analysed using one-way analysis of variance (ANOVA) followed by Tukey’s test (significance level set to 0.05).
Results and discussion
This article intends to discuss three concentrations with three specific mixtures of protein each, taking into account common ingredients from chow retailers, so as to provide an adjusted aminogram and keep animals in a good nutritional state. For this objective, three levels of proteins were used, such as 17.8% (reference diet based on AIN-93G), 4 12% intermediate level (coincident with the protein percentage of AIN-93M) 4 and 8%. This last concentration is considered as appropriate protein level by digestibility assays, besides it avoids that high-quality protein, such as egg or casein, shows top PER values or NPU values. 24 Each of these protein concentrations contained three proportions of ingredients such as casein (C), casein+soy (C+S, 1:1) and casein+soy+gluten (C+S+G, 1:1:1). Isolating the protein as unique source of variation in the chow, all the other nutrients used in this study (Table 2) were adjusted with the values reported by Reeves et al. 4 The replacement of casein with other protein sources may turn diets cheaper besides bringing other benefits, depending on the protein source chosen. As described by Duffy et al., 25 partial or total replacing casein with soy protein has been associated with the increased survivability and delayed amyloid A-related or other pathologies. Additionally, all chows were prepared without supplementation with cysteine, in order to better observe the effects of combination of ingredients and their proportions. These formulations were supplied to the animals in powdered diets (as meals) seeking higher stability of the formula, avoiding water addition and heat generation, which are required for pelleting. Besides, the results reached are from diets with few steps to prepare, since the ingredients were mixed together without further manipulation or special equipment, and we highlight that homogenizing was a critical point performed through tamization and mixture.
Proximate composition (% wet basis ± SD) of the experimental diets under investigation.a

C+S: casein+soybean; C+S+G: casein+soybean+corn gluten meal.
aDifferent superscript letters (d to h) in each column indicate significant differences (Tukey’s test p < 0.05).
bObtained by difference.
cBased on AIN93G, Reeves et al. (1993).
The chemical score of experimental diets (Table 3) points out that the formulations in some diets do not reach the amino acid recommended levels. Chemical score compares the levels of essential amino acids between the test and standard proteins (NRC 1995) 10 and provides an estimate of the nutritive value of each protein. For maximum growth in rats, with a minimum level of protein, a balance of essential amino acids must be provided in order to fulfil the basic body requirements. The results of the chemical score of experimental diets revealed that the Cys+Met were the first limiting amino acids in all diets and the reference diet was the best one (71% of recommended) containing casein as the only source of protein (17.8%). The closest values to the experimental diets were C+S (1:1) and C+S+G (1:1:1) which reached, respectively, 60% and 55% of recommended.
Amino acid compositions profile and chemical score (parenthesis numbers) of nine experimental diets under investigation (milligrams/kilogram experimental diet).a
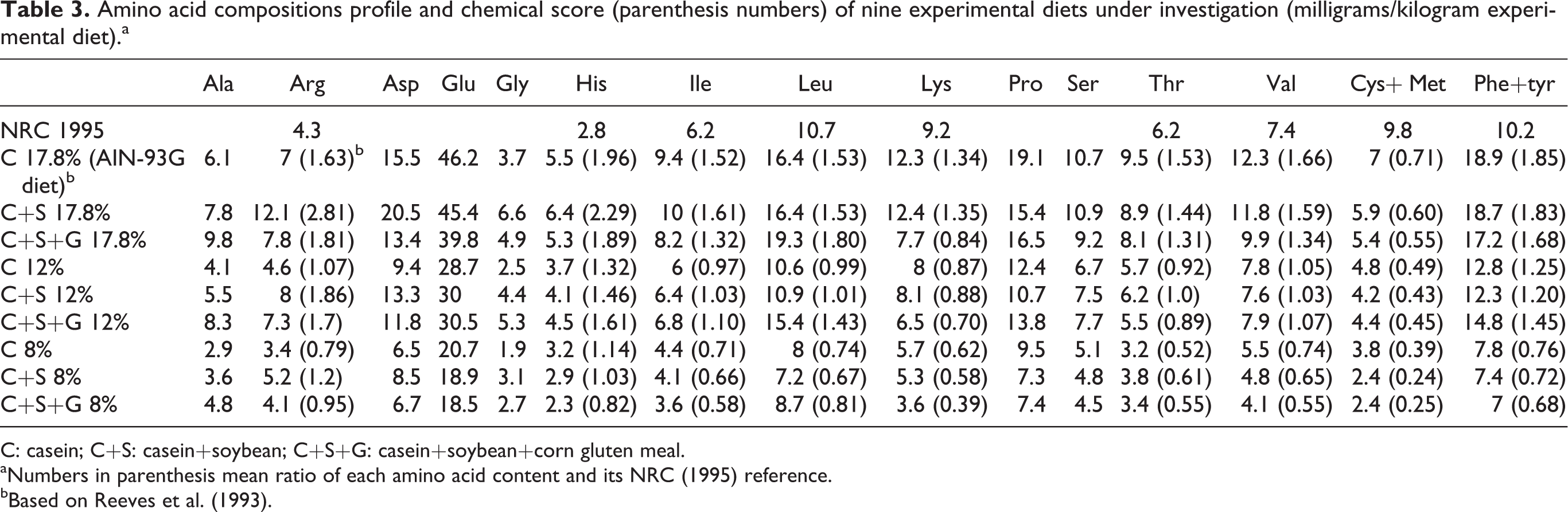
C: casein; C+S: casein+soybean; C+S+G: casein+soybean+corn gluten meal.
aNumbers in parenthesis mean ratio of each amino acid content and its NRC (1995) reference.
bBased on Reeves et al. (1993).
The diets containing 12 and 8% protein got the recommendations in the range of 49–25% and 39–25%, respectively, pointing out that the concentrations and proportions between ingredients interfered in the amino acid profile. It is remarked that these amino acids may be easily supplemented, which is already considered regular practice for purified diets, as suggested by Reeves et al. 4 with the supplementation of 3 g cystein/kg of diet. Therefore, this supplementation of amino acids may also be applied to adjust other diets, taking into account the recommendation (NRC 1995: 9.8 g sulphurated amino acid per kilogram diet). 10 We verified that the diet 17.8% C+S was complete in all the other amino acids. On the other hand, the addition of gluten to this concentration contained lysine as limiting amino acid (84%). All the 12% concentrations presented sulphur amino acid and also lysine deficiency (78–88%), which could turn them usable by supplementing with both of them. Furthermore, all 8% concentration diets presented a deficiency of five other essential amino acids. This concentration with gluten addition (C+S+G 8%) also causes inadequacy of histidine, which turns it as the worst amino acid profiled diet. In fact, this diet will present the worse values for growth performance (FER, FCR), which will be shown below, and highlights that the low amount of protein content did not allow compensating all amino acid plant deficiencies.
In the present study, the diet AIN93G for growth, pregnancy and lactation was used as reference to investigate the feasibility of semi-purified diets for young rats, as intermediate formulations between purified and closed diets. This diet was formulated by Reeves et al. 4 , which established guidelines for nutritionally adequate purified diets which could be used to standardize studies among laboratories. As the young rats have great use as experimental models because of their many growth conditions and development parameters, this diet has been extensively applied by researchers around the world. Furthermore, we used the experimental design with male rats, in order to exclude the female oestrus cycle, and period of 14 or 21 days, due to the significance of this growth window in the animal life. Furthermore, the nutrient utilization parameters consider the endogenous nitrogenous obtained from the animal group fed with a protein-free diet, which is possible to maintain the rats only for 2 weeks. 26,27
As results, during this experimental period, all tested diets had the potential for promoting growth with adequate body mass gain, in spite of some low protein contents (8% diet) and their poor amino acid profiles. Although de la Higuera 28 shows that single amino acid deficiencies lead to decreased food intake and Ene-Obong and Obizoba 29 observed that this is influenced by source of nitrogen, 30 there was no significant difference in the food intake of the rats fed with the experimental diets in relation to those fed with casein 17.8% (reference diet), indicating a good palatability and acceptability of food (Figure 1). As the weight gain of the groups of rats is due to food intake, our results also revealed that all rats gained weight and there were no significant differences among them. The same way, there was no difference among liver and fat (intercostal, epididymal and visceral) weight (data not shown) from animals fed with different diets, providing evidence about the animal well-being during the use of the diets.

Growth performance and nutrition utilization in Wistar rats fed experimental diets for 14 days. (a) Body mass gain (BMG, %) = [(final body mass (g) − initial body mass (g)/initial body mass (g)] × 100, protein and food intake; (b) true digestibility (TD) = (faecal nitrogen + faecal nitrogen from non-protein group)/nitrogen intake, and apparent protein digestibility (APD) = faecal nitrogen/nitrogen intake; (c) protein efficiency ratio (PER) = BMG (g)/protein intake (g); net protein retention (NPR) = (BMG (g) + weight loss of non-protein group/protein intake; (d) net protein utilization; (e) food efficiency ratio (FER) = BMG (g)/food intake (g); (f) food conversion ratio (FCR) = food intake (g)/BMG (g). C, casein; C+S, casein + soybean; C+S+G, casein + soybean+ corn gluten meal. Different superscript letters in each column indicate significant differences (Tukey’s test p < 0.05).
No significant differences in growth performance show that digestion and absorption of nutrients from test diets and control diet were similar. Indeed, there were no differences about apparent and true protein digestibility (APD and TD) among all chows (Figure 1) meaning excellent nutrient utilization values. However, high digestibility does not always mean high protein quality, once it is a measure of protein hydrolysis, whereas protein quality is a measure of the balance of the amino acids that are absorbed and utilized for growth and other purposes 31 so that the growth parameters and feed utilizations take important part to assay them.
FCR and FER are feed utilization parameters, where FCR means grams of chow consumed per body weight gain, while FER means body weight gain per gram of chow consumed. Many authors 32,33 have reported that FCR ranges from 1.2 to 1.5. In the present study, the FCR average was 3.12, except for the lowest concentration of protein 8% with gluten that reaches 6.33, and FER ranged from 0.4 to 0.2, being the lowest value 0.13 (C+S+G 8%). Interestingly, despite the similarity between the chow consumption and the weight gain, the variations occurred between the three levels of protein concentration were clearly observed with these two parameters. There were no variations within the groups with the same amount of protein, except for the lowest protein concentration with gluten. This points out that these variations were mainly due to the protein amounts independently from the protein ingredient used. Also, it demonstrates that low protein content chows (8%) and addition of poor protein quality ingredient (gluten) have required higher quantities of chow per gram of body mass gain (FCR = 6 g of chow) and produced lowest body mass per chow gram (FER = 0.13), which could characterize this last chow concentration as unable to support growth. This may be due to the combination with gluten, whose chow reached around 66% plant protein resulting in a diet deficient of many amino acids (Table 3). Moreover, gluten proteins are recognized by their high proline and glutamine content and gluten peptides are relatively resistant to a complete digestion due to the prolyl endopeptidasic enzyme deficiency. However, in the present study, probably there were no differences in digestibility because of the bacteria activity from the oral cavity that hydrolyses gluten peptides and from the large intestine that digests gliadin peptides. 34
Hung and Deng 35 in their studies state that an increase in diet protein level did no longer result in growth increase or improved FCR. Our results also demonstrate that chow with low amount of protein can be enough to support the growth, but in this case, the combination of protein sources should be very careful, observing the level of plant protein replacement. In our study, we used prepressed solvent extracted soybean meal without amino acid supplementation, replacing successfully up to 50% of casein protein, even in the chow with 8% of protein, which had the same behaviour as the casein reference diet. Kumar et al. 23 and Jackson et al. 36 showed that between 67% and 100% of the dietary protein can be supplied in the form of processed soybean meal as animal protein replacer in animal chow, whose inclusion level depends on a variety of different factors, including the animal species and size, soybean meal source and its processing method.
The same way as feed performance, PER, NPR and NPU showed good results for all diets with 17.8% and 12% of protein, which were similar to reference diet. Taking into account that PER means the ratio of weight gain for the amount of protein consumed 37 and NPR is an improvement over PER, 38 therefore a better predictor of protein quality, 39 we can conclude that even in lower protein concentrations, the combinations supported body weight gain of animals. The NPU is wider nutritional endpoint and reflects the ratio of the nitrogen used for tissue formation versus the amount of nitrogen digested. 38 The NPU results confirm the good digestibility and the usability of the readily digestible soy and gluten protein, inferring that the known anti-nutritional factors (lipoxygenases, protease inhibitors, haemaglutinins and cyanogenic glucosides) 40 –42 were not important to the chow nutritional utilization. Besides, even in diets with 8% protein, there was adequate nutritional performance, which may be attributable to low N intake compensated by lower faecal and urinary N excretion. 30,40 Furthermore, Yang et al. 43 and Mohanta et al. 44 observed in their studies that the decrease in NPU with increase in dietary protein is probably because of utilization of more dietary protein as energy source when high protein diets are fed to animals, which corroborates the present finding.
We have to consider that the use of these ingredients for laboratory animal chows allows flexible mixes with different protein proportions, as additional options than just purified diets. These ingredients herein used are cheap and commonly available, and the chow preparation requires no sophisticated equipment, supplying researchers with subsidies for their assay design or even new ones. Taking into account the diets formulated for the growth (14 days), our results turn evident that the ingredients tested were good protein sources in the conditions here considered, and the amino acid supplementation could be a good option to improve their chemical score. The chows with 17.8%, 12% and 8% (only for C and C+S) allowed a proper combination of ingredients from the point of view of palatability, nutrient availability/utilization, metabolic processes, growth performance and feed utilization parameters.
Footnotes
Acknowledgement
The authors thank Nutrimax for the donation of ingredients and expertise in the animal chow and Jaime Nobrega for language assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by FAPEMIG, CNPq, CAPES and LANTIN (Laboratório de Análise Nutricional e Toxicológica in vivo – UNIFAL-MG).
