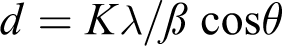Abstract
Silver nanoparticles (AgNPs)embedded poly (N-vinylpyrrolidone/dextran) hydrogels were prepared by gamma radiation. Highly stable and uniformly distributed AgNPs have been obtained within hydrogel networks as nanoreactors via in situ reduction of silver nitrate using sodium borohydride as reducing agent. The formation of AgNPs has been confirmed by Fourier transform infrared spectroscopy. The ultraviolet–visible spectroscopy measurements show a distinct characteristic absorption peaks around 420 nm, indicating the formation of AgNPs. X-ray diffraction analysis and dynamic light scattering demonstrated that the hydrogels have regulated the AgNPs size to a nanoscale. The combination of the AgNPs with doxorubicin (DOX) as a model of antitumor drug forms a new biocompatible nanodrug. Our results show that the AgNPs effectively increased survival rate and decreased tumor volume of Ehrlich ascites carcinoma–bearing mice more than the DOX-treated group and enhanced the cytotoxicity of DOX in different human cancer cell lines (HepG2, T47D, and PC3). DOX-AgNPs decreased malondialdehyde and total nitrate/nitrite levels and increased superoxide dismutase activity and glutathione content in the rat cardiac tissues compared with the DOX-treated group. In conclusion, DOX-AgNPs improved therapeutic index and reduced DOX-induced cardiotoxicity via preservation of cardiac markers, which represents a promising candidate for cancer treatment.
Introduction
Doxorubicin (DOX), an anthracycline antibiotic, is a well-known chemotherapeutic agent which is used in the treatment of a wide variety of cancers. 1 A key factor in the response of cancer cells to chemotherapeutic drugs is the activation of the apoptotic pathway. 2 DNA has multiple mechanisms of action including the production of free radicals, the intercalation of DNA, and inhibition of topoisomerase II, resulting in DNA double-strand breaks, cross-link DNA strands, and activation of the apoptotic pathway. 3,4 Because of this mechanism of action, high concentration of DOX in normal tissues can cause serious damage to healthy cells. In clinical therapy, the most serious toxicity is life-threatening cardiomyopathy, leading to heart failure. 5 Hence, improved pharmacological specificity for the DOX anticancer agent is much needed.
Metallic nanoparticles (NPs) have unique optical, electrical, and biological properties that have attracted significant attention due to their potential use in many applications, such as catalysis, biosensing, drug delivery, and nanodevice fabrication. Nanodrug delivery systems with controlled release, drug targeting, as well as significantly increase the bioavailability of drugs, which greatly overcome the weaknesses of traditional drug delivery. 6 Previous studies reported that anticancer drugs and poly-NP complexes can increase the anticancer drug accumulation in tumor cells so as to achieve a better therapeutic effect. 7,8
Silver nanoparticles (AgNPs) have many biomedical applications due to its excellent biocompatibility and antibacterial properties. AgNPs display a synergistic effect 8 and a cytotoxic effect on cell viability which have a principal role in antitumor effect. 9,10 AgNPs aid in gathering and transporting drugs into the cancer cells, 8 and they also obstruct with metabolism of cancer and tumor proliferation. 11 Previous studies reported that AgNPs can induce cell death in vitro and in vivo through a reactive oxygen species (ROS)-mediated apoptotic process. 9,10
Generally, metal NPs agglomerate due to high-active surface area. To stabilize and control the NPs structures, various surfactants, polymers, dendrimers, biological templates, and biomacromolecules were used. 12 By considering the importance of hydrogel networks and natural polymers (carbohydrates) such as dextran, starch, carboxymethylcellulose, acacia as effective carriers for nanosystems, prompted us to draw a new idea in which semi-inter penetrating network (IPN) hydrogels prepared in presence of prepolymers that would be better choice to cook metal nanoparticle in their networks. 13 Hydrogels offer large free space between the cross-linked networks in the swollen stage that can act as a nanoreactor for the nucleation and growth of the NPs. 14 For this purpose, the combination of poly (N-vinylpyrrolidone) (NVP) hydrogel and dextran systems is selected because of their more relevance for pharmaceutical and biomedical applications.
In this study, we explore the combination of the AgNPs with DOX to form a new biocompatible nanodrug to facilitate the cellular drug uptake, improve the therapeutic index, and reduce cardiotoxicity.
Materials and methods
Drugs and chemicals
NVP of purity 99% was received from Merck (Germany), and Dextran-70 GR powder was received from Nice Company for chemicals (Cairo, Egypt). Silver nitrate (AgNO3) with minimum assay 99% (Mumbai, India) was purchased from Oxford Laboratory Mumbai (India). Sodium borohydride (NaBH4) with minimum assay 98% was purchased from Titan Biotech Limited (Mumbai, India). DOX hydrochloride was purchased from Sigma-Aldrich (St. Louis, Missouri, USA). The stock solution of the drug was dissolved in phosphate buffered saline (PBS) and preserved at −20°C. The solution was diluted in PBS immediately before each experiment to the desired final concentrations. Dulbecco’s modified Eagle’s medium (DMEM), trypsin/EDTA, PBS, penicillin G and streptomycin antibiotics, acetic acid, Trizma base, and sulphorhodamine-B (SRB) were purchased from Sigma-Aldrich Co. All chemicals were used without further purification.
Preparation of P(NVP/dextran) hydrogels
Poly (NVP/dextran) hydrogel was prepared by gamma radiation polymerization. Equal volumes of NVP (50%) aqueous solution were mixed with different concentrations of dextran solution (10, 15, 20, and 25%) and continuously stirred to form homogeneous mixture after heating at 80°C for 30 min. The viscous solutions were transferred into the glass tube to irradiate by gamma source at 30 kGy. After irradiation, the cross-linked hydrogels were cut into disks, dried, and weighed. Then, the hydrogels were extracted in deionized water for 6 h at 80°C and were occasionally shaken. The insoluble part corresponding to the cross-linking sample was dried and weighed.
Preparation of AgNPs
Accurately weighed dry hydrogels were equilibrated with water for 1 day, and these hydrogels were transferred to another beaker containing 50 ml of 0.2 mol/l of AgNO3 aqueous solutions and allowed to equilibrate for another 1 day. Here, most of the silver ions are transferred from solution into hydrogel networks by anchoring through CONH2, –OH groups of hydrogel chains, and the rest of metal ions were occupied in free network spaces of hydrogels. These silver ion loaded hydrogels were finally transferred into a beaker containing 50 ml 0.4 mol/l of sodium borohydride (NaBH4) aqueous solution as reducing agent and allowed for another 1 day. The obtained hydrogel loaded AgNPs are often termed in the forthcoming sections as P(NVP/dextran)-AgNPs.
Characterization
UV–vis spectrophotometry
Characterization of the formed AgNPs in the hydrogels was carried out using Jasco V560 (Pye-Unicam, England) model ultraviolet-visible (UV–vis) spectrophotometer (Pye-Unicam, England) with a scan range of 300–600 nm. For this study, AgNPs were extracted from hydrogel–silver nanocomposites by soaking in distilled water (10 mg hydrogel/ml H2O) over a period of a week at room temperature; the supernatant was used to measure the absorption spectra. 15
Fourier transform infrared measurements
The prepared hydrogels were dried and ground to a fine powder, and then a fixed weight is mixed with a fixed weight of dried KBr and pressed to a disk for IR analysis. Fourier transform infrared (FTIR) spectra of hydrogels were measured by FTIR spectrometer (6300; JASCO, Japan) in the range from 400 cm−1 to 4000 cm−1.
X-ray diffraction
The X-ray diffraction (XRD) method was used to identify the AgNPs in the polymer nanocomposites. These measurements were carried out on Shimadzu X-ray diffractometer (XRD-6000 model) equipped with X-ray tube (Cu target, 40 kV (voltage), 30 mA (current)). The X-ray data were recorded in the range from 4° to 90° 2θ with continuous scanning mode and scanning speed 8°/min. The average particle size of the NPs was calculated from the full width at half maximum (FWHM) and the peak position of XRD line broadened according to the Scherrer equation
where d is the particle size, K = 0.89 is the Scherrer constant related to the shape and index (hkl) of the crystals, λ is the wavelength of the X-ray (Cu Ka, 1.54056 Å), θ is the diffraction angle, and β is the full width at half maximum (FWHM in radian) k is the wavelength of the X-ray (Cu Ka, 1.54056 Å), λ is the wavelength of the X-ray (Cu Ka, 1.54056 Å), θ is the diffraction angle, and β is the full width at half maximum (FWHM in radian) h is the diffraction angle, and b is the FWHM in radian. 16
Dynamic light scattering
To measure the particle size by dynamic light scattering (DLS), we use the method developed by Kumar, 17 where 0.01 g of polymer powder contains 12 mmol AgNO3 extracted in 20 ml distilled.
Animals
Adult male albino Wistar rats weighing 150–200 g and female Swiss albino mice weighing 18–20 g were obtained from animal facility, Pharmacology Unit, National Cancer Institute (NCI), Cairo University, Egypt. Animals were kept under standard conditions and were allowed free access to a standard requirement diet and water ad libitum. Animals were kept under a controlled lighting condition (light:dark, 13 h:11 h). The animals’ treatment protocol has been approved by the animal care committee of the NCI, Cairo University, Egypt.
Cells and cell cultures
Hepatocellular carcinoma cell line; HepG2, human ductal breast epithelial tumor cell line; T47D and human prostate cancer cell line; and PC3 were used in this study and were obtained frozen in liquid nitrogen (−180°C) from the American Type Culture Collection and grown as monolayer culture in RPMI-1640 medium supplemented with 10% FBS and 100 units/ml penicillin and 2 mg/ml streptomycin. They were obtained from NCI, Cairo University, Egypt. The adherent cells were grown as “monolayer culture” in DMEM supplemented with penicillin (100 IU/ml), streptomycin (100 μg/ml), and 10% fetal bovine serum. The cell lines were incubated at 37°C in 5% CO2–95% air in a high humidity atmosphere in the water-jacketed incubator (RCO 3000 TVBB; Revco, GS Laboratory Equipment, Asheville, North Carolina, USA). The tumor cell lines were maintained in the NCI, Cairo, Egypt, by serial subculturing.
Experimental design
Twenty-four male albino rats were divided randomly into four groups (n = 6). Group I served as control group received 0.5 ml normal saline intraperitoneally (i.p.). Group II was administered DOX (5 mg/kg, i.p.). Group III was injected with AgNPs (5 mg/kg, i.p.). Group IV was injected with DOX-AgNPs (5 mg/kg).
Biochemical studies
Twenty-four hours after treatment, rats were anesthetized with ether. Blood samples were obtained by heart puncture, and serum samples were separated for determination of serum creatine phosphokinase (CPK) and lactate dehydrogenase (LDH) activities. Hearts were quickly excised, washed with saline, blotted with a piece of filter paper, and homogenized using a Branson Sonifier (250, VWR Scientific, Danbury, Connecticut, USA). The homogenate was used for determination of superoxide dismutase (SOD) activity, total nitrate/nitrite NOx, malondialdehyde (MDA), and reduced glutathione (GSH) levels.
CPK and LDH were determined according to the method of Swanson and Wilkinson 18 and IFCC, 19 respectively, using ready-made kit. MDA and GSH in cardiac tissue homogenates were determined spectrophotometrically using the methods of Buege and Aust 20 and Ellman, 21 respectively. SOD and NOx were measured according to the methods of Minami and Yoshikawa 22 and Miranda et al., 23 respectively.
Antitumor activity
Percentage survival of animals and mean survival time in rats (in vivo study)
Forty female albino mice were inoculated with Ehrlich ascites carcinoma (EAC) cells (2.5 × 106 cells/0.1 ml). Each mouse was injected i.p. with EAC cells. Twenty-four hours after cell inoculation, 40 mice were classified into four groups. In group I, animals were injected with 0.2 ml of saline and served as control. In group II, animals were injected with DOX (5 mg/kg, i.p.). In group III, animals were injected with AgNPs (5 mg/kg, i.p.). In group IV, animals were injected with DOX-AgNPs (5 mg/kg, i.p.). The change in the percent survival of animals was recorded daily during a period of 45 days following the treatment
Mean survival time (MST): The MST of each group was calculated by dividing the sum of survival days of all mice in each group by the total number of mice in each group (10).
Tumor volume
Forty female albino mice were inoculated with EAC cells. A total of 2 × 103 EAC cells were transplanted subcutaneous in the right thigh of the lower limb of each mouse. Mice with a palpable tumor mass (100 mm 3 ) that developed within 7 days after implantation were divided into four groups: In group I, 10 mice were injected i.p. every other day with normal saline and used as a control group; in group II, 10 mice were injected with DOX (5 mg/kg, i.p.); in group III, 10 mice were injected with AgNPs (5 mg/kg, i.p.); in group IV, 10 mice were injected with DOX-AgNPs (5 mg/kg). The change in tumor volume was measured every other day for 4 weeks using a Vernier caliper and calculated using the following formula according to Osman et al. 24
where A and B denote the minor and major tumor axes, respectively.
Cytotoxicity assay
Cytotoxicity was measured by the SRB method according to that of Skehan et al. 25 and expressed as the survival fraction compared with untreated control cells. Hepatocellular carcinoma; HepG2, human ductal breast epithelial tumor cell line; T47D and human prostate cancer cell line; and PC3 cell lines were seeded in 96-well microtiter plates at a concentration of 5 × 103 cell/well in a fresh medium and left to attach to the plates for 24 h. After 24 h, cells were incubated for additional 48 h with various concentrations of DOX (1–100 μg/ml) and AgNPs (0.460 mg/ml), completed to total of 200 μl volume/well using fresh medium and incubated for 48 h. Control cells were treated with vehicle alone. For each drug concentration, three wells were used. The cells were fixed with 10% trichloroacetic acid for 1 h at 4°C. Wells were washed five times with distilled water and stained for 30 min at room temperature with 0.4% SRB dissolved in 1% acetic acid. The wells were then washed four times with 1% acetic acid. The plates were air-dried and the dye was solubilized with 10 mM tris-base (pH 10.5) for 5 min on a shaker (Orbital shaker OS 20; Boeco, Germany) at 1600 r/min. The optical density was measured spectrophotometrically at 564 nm with an ELISA microplate reader (Meter tech. Σ 960, Warminster, PA 18974, USA). The percentage of cell survival was calculated as follows: The IC50 values (the concentrations required to produce 50% inhibition of cell growth) were calculated from the equation of the dose response curve. The experiment was repeated three times for each treatment.
Results
Characterization of the prepared hydrogels
UV–vis spectrophotometry
The absorption peaks of P(NVP/dextran)-AgNPs hydrogel is illustrated in Figure 1. From the figure, it can be showed that the absorption peak of AgNPs was at 422 nm. Further, complete absence of peaks or tails at 560 nm is clearly observed.

UV–vis spectra of P(NVP/dextran)-AgNPs at silver ions contents 0.2 mol/l and irradiation dose 30 kGy. UV–vis: ultraviolet–visible; NVP: N-vinylpyrrolidone; AgNPs: silver nanoparticles.
FTIR spectra
The FTIR spectra of hydrogels measured absorption peaks of P(NVP/dextran) and (NVP/dextran)-Ag nanocomposites at silver ions concentrations 0.2 mol/l are illustrated in Figure 2.

FTIR spectra of P(NVP/dextran) and P(NVP/dextran)-AgNPs and irradiation dose 30 kGy. FTIR: Fourier transform infrared; NVP: N-vinylpyrrolidone; AgNPs: silver nanoparticles.
NP size measurements
XRD study of the P(NVP/dextran)-AgNPs
Figure 3(a) and (b) shows the XRD measurements to identify the P(NVP/dextran) and P(NVP/dextran)-Ag nanocomposites with Ag contents 0.2 mol/l. Figure 3(a) shows the observed peak of P(NVP/dextran) hydrogel at 2θ values of 11° and 21°. The absorption peaks of P(NVP/dextran)-Ag nanocomposites at 2θ values of 38°, 44°, 64°, and 77° are present in Figure 3(b). The Ag particle size is calculated using FWHM according to the Scherrer equation.
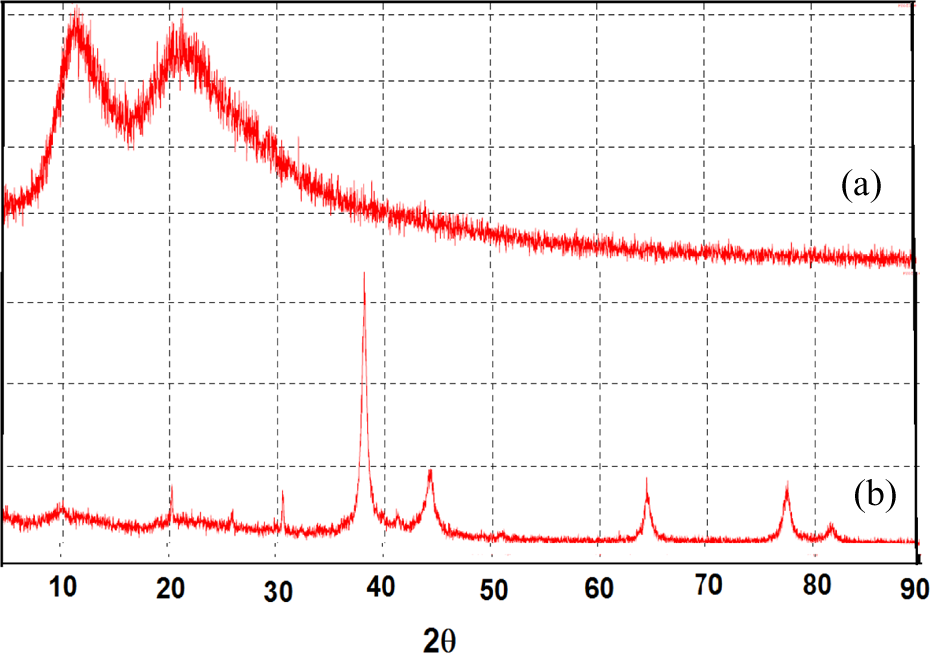
XRD measurement of (a) P(NVP/dextran) and (b) P(NVP/dextran)-AgNPs at silver ions contents 0.2 mol/l. XRD: X-ray diffraction; NVP: N-vinylpyrrolidone; AgNPs: silver nanoparticles.
Dynamic light scattering
Figure 4 shows the particle size measured by the DLS of P(NVP/dextran)-Ag nanocomposites prepared at Ag content 0.2 mol/l.

DLS measurement of AgNPs at silver ions contents 0.2 mol/l. DLS: dynamic light scattering; AgNPs: silver nanoparticles.
Percent survival
Figure 5 shows the percent survival of EAC-bearing mice treated with DOX (5 mg/kg, i.p.), AgNPs, and DOX-AgNPs (6 Gy). After 18 days of the experiment, the percentage of survival was 0% in control untreated tumor-bearing mice, while after 25 days of the experiment, the percentage of survival was 0% in AgNPs-treated group. On the other hand, after 31 days, the percentage of survival was 0% in mice treated with DOX, while the percentage of survival was 0% in mice treated with DOX-AgNPs and exposed to irradiation after 41 days.
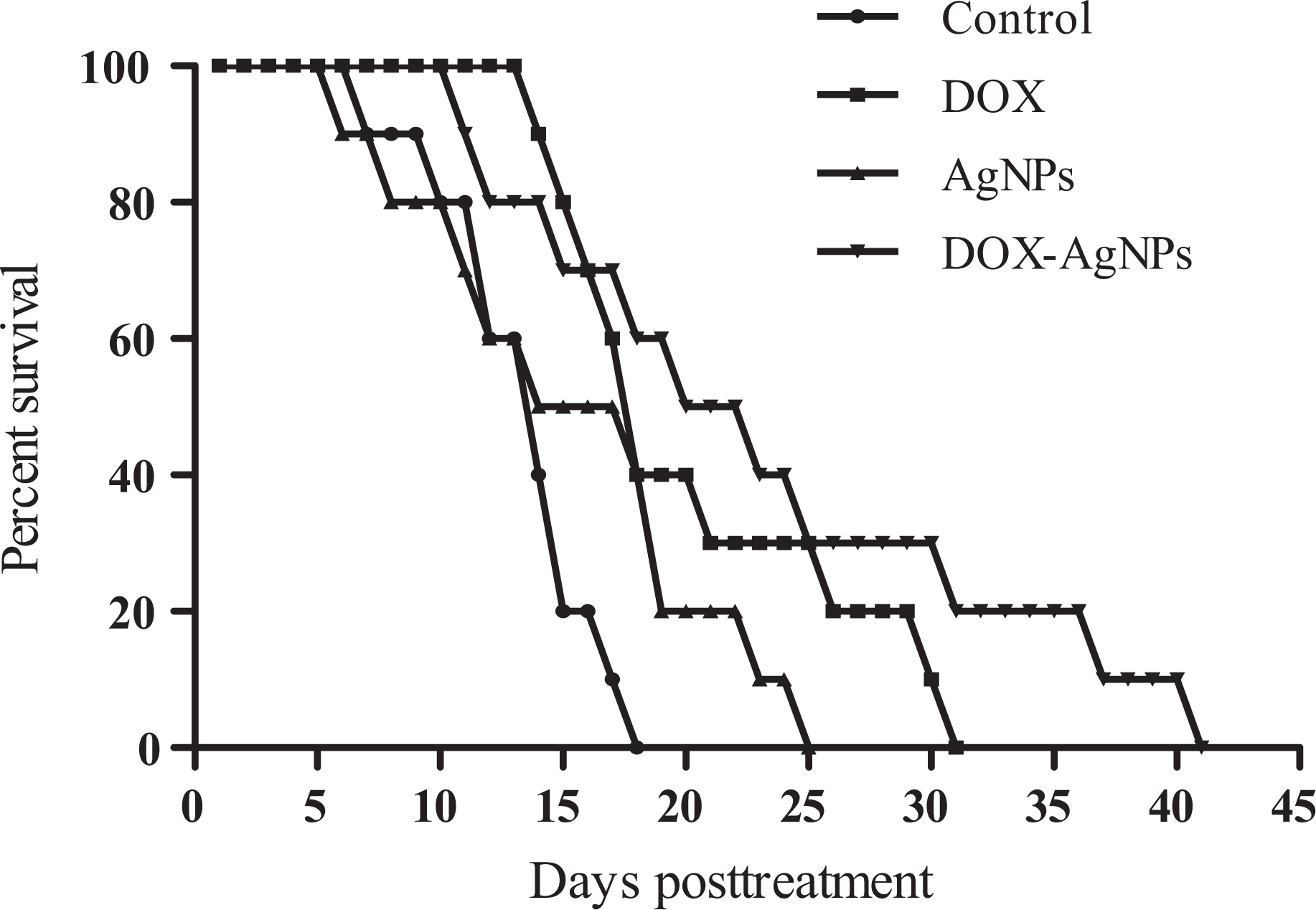
Effects of DOX (5 mg/kg), AgNPs, and DOX-AgNPs on the percent survival of EAC-bearing mice. DOX: doxorubicin; AgNPs: silver nanoparticles; EAC: Ehrlich ascites carcinoma.
Mean survival time
Figure 6 shows the effect of AgNPs on the cytotoxic activity of DOX against the growth of EAC cells inoculated in female Swiss albino mice. The control EAC-bearing mice showed an MST of 12 days. Administration of DOX (5 mg/kg, i.p.) and AgNPs (5 mg/kg) increased the MST to 19.7 and 15.2 days, respectively. Treatment with DOX-AgNPs increased the MST to 22.3 days (Figure 6).

Effects of DOX (5 mg/kg), AgNPs, and DOX-AgNPs on the MST of EAC-bearing mice. DOX: doxorubicin; AgNPs: silver nanoparticles; MST: mean survival time; EAC: Ehrlich ascites carcinoma. *indicates significant change compared with the control value at p≤ 0.05 using ANOVA followed by Tukey-Kramer as a post ANOVA test.
Tumor volume
Figure 7 shows the effect of DOX (5 mg/kg, i.p.), AgNPs, and DOX-AgNPs on the growth of solid Ehrlich carcinoma-bearing mice. The tumor volume of the control group showed progressive increase, whereas the treatment with DOX resulted in a significant reduction in tumor volume as compared to the control group. AgNPs induced significant reduction in tumor volume compared to the control group. Treatment with DOX-AgNPs resulted in a significant reduction in tumor volume compared to the control group.
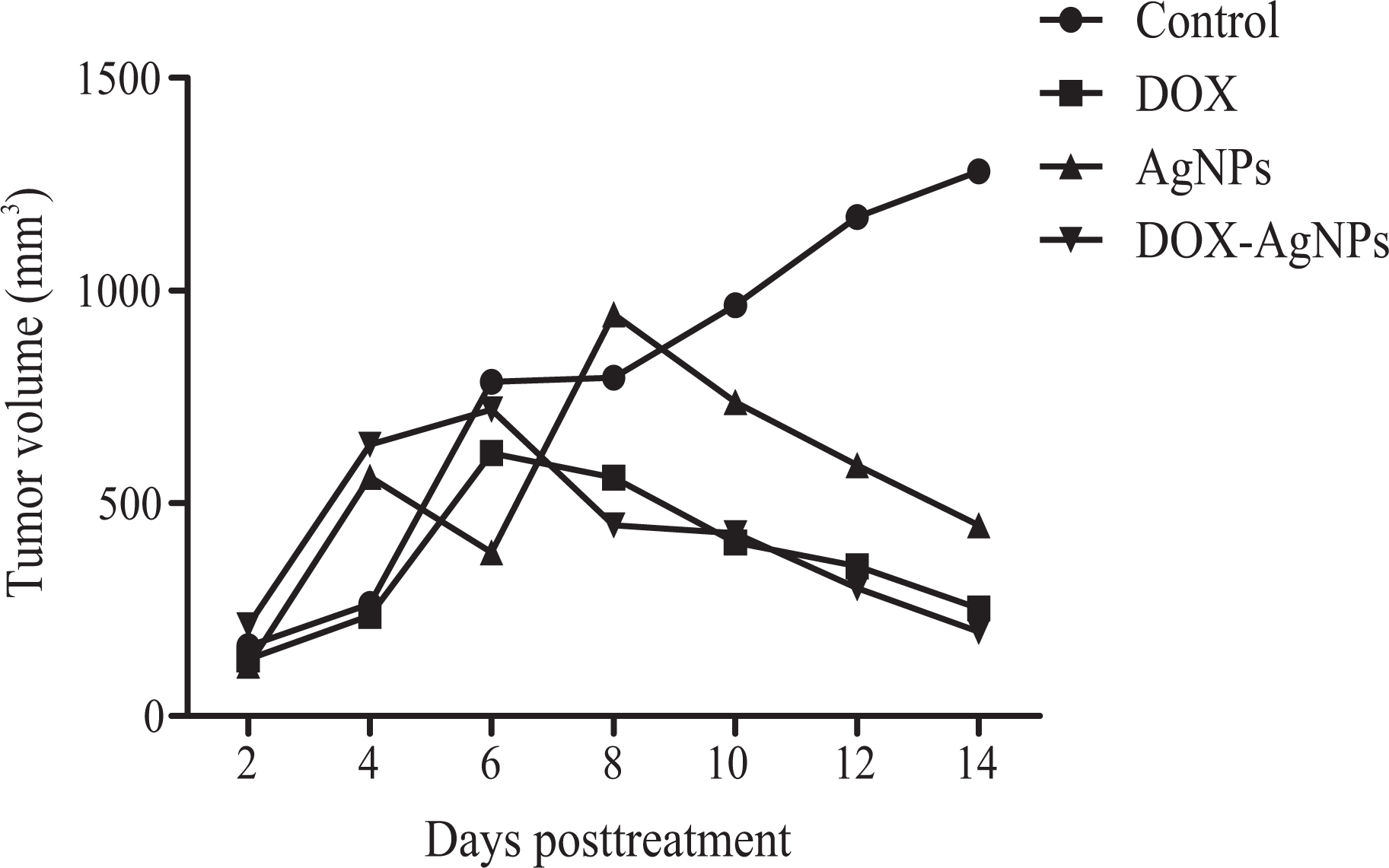
Effects of DOX (5 mg/kg), AgNPs, and DOX-AgNPs on tumor volume of solid Ehrlich carcinoma-bearing mice. DOX: doxorubicin; AgNPs: silver nanoparticles.
Cell viability of HepG2
It is observed from Figure 8 that AgNPs showed no cytotoxic activity against HepG2, T47D, and PC3 cell lines. DOX significantly inhibited the cell proliferation in HepG2, T47D, and PC3 cells in a dose-dependent manner with an IC50 value of 4.8, 4.58, and 5.78 μg/ml, respectively (Figure 8). The combination of DOX and AgNPs (DOX-AgNPs) exhibits a cytotoxic effect against HepG2, T47D, and PC3 cells over that of DOX. The IC50 values were 4.73, 4.13, and 4.43 μg/ml, respectively (Figure 8).

Surviving fraction of HepG2, T47D, and PC3 cells treated with DOX (1–100 μg/ml), AgNPs, and DOX-AgNPs after 48 h. Data are expressed as mean ± S.E from three independent experiments, each performed in triplicate. DOX: doxorubicin; AgNPs: silver nanoparticles.
Biochemical parameters
Administration of ADR in a single dose (5 mg/kg) induced a significant increase in the activities of CPK and LDH, respectively, compared to the control nontreated group. However, the activities of CPK and LDH increased in AgNPs (5 mg/kg, i.p.) treated group, but this difference was not significant (P > 0.005). Treatment with DOX-AgNPs (5 mg/kg, i.p.) showed a significant decrease in CPK and LDH activities compared to the DOX-treated group (Figure 9).

Effects of DOX (5 mg/kg), AgNPs, and DOX-AgNPs on the activities of serum CPK and LDH. a and b indicate significant changes in control and DOX, respectively, at p ≤ 0.05 using ANOVA followed by Tukey–Kramer as a post-ANOVA test. DOX: doxorubicin; AgNPs: silver nanoparticles; CPK: creatine phosphokinase; LDH: lactate dehydrogenase.
The effects of DOX, AgNPs, and their combination on the oxidative and nitrosative stress biomarkers in cardiac tissues are shown in Figure 10. Administration of DOX and AgNPS resulted in a significant increase in MDA and NOx levels and a significant decrease in GSH and SOD activities compared to the control group. On the other hand, the administration of DOX-AgNPs (5 mg/kg, i.p.) induced a significant decrease in MDA and NOx and a significant increase in GSH and SOD compared to the DOX-treated group (Figure 10).
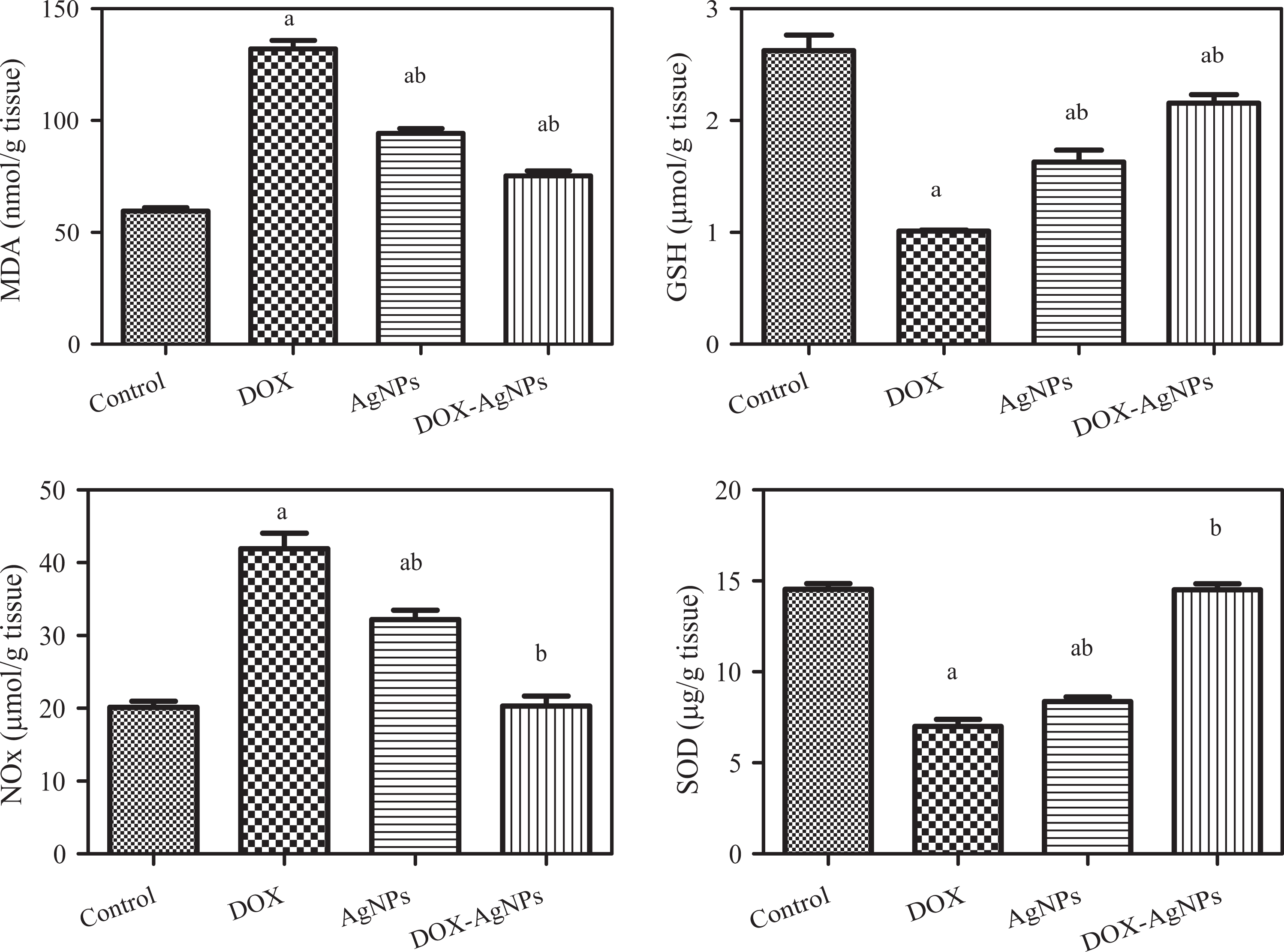
Effects of DOX (5 mg/kg), AgNPs, and DOX-AgNPs on MDA, reduced GSH, and total nitrate/nitrite (NOx) levels and SOD activity on cardiac tissues. a and b indicate significant changes in control and DOX, respectively, at p ≤ 0.05 using ANOVA followed by Tukey–Kramer as a post ANOVA test. DOX: doxorubicin; AgNPs: silver nanoparticles; MDA: malondialdehyde; GSH: glutathione; SOD: superoxide dismutase.
Discussion
Radiation technique seems promising for the preparation of hydrogels because polymers and/or monomer in aqueous solution undergo cross-linking upon irradiation to yield a gel-like material. Radiation modification of dextran with NVP will lead to formation of hydrogel matrices with many excellent properties to use as polymeric stabilizer for AgNPs.
AgNPs formed in the entire P(NVP/dextran) hydrogel networks can be confirmed because the silver salts loaded in P(NVP/dextran) hydrogels are readily reduced by NaBH4, which immediately turn into a dark brown color. The dark brownish black color of the hydrogel disks indicated the formation of AgNPs within the swollen hydrogel. 26 AgNPs were tested by UV–vis spectral analysis in the range of 300–600 nm wavelength that are assigned to AgNPs which arose from the surface plasmon resonance. 27 The absorption peak of AgNPs was at 422 nm, which confirms that there are no aggregations or cluster formation of larger AgNPs because of complete absence of peaks or tails at 560 nm. 28
In the present study, on the basis of C=O and OH band positions, the interactions between NVP and dextran can be confirmed. NVP and dextran are compatible and miscible polymers via the hydrogen bond interactions between the –CO groups of NVP and −OH groups in dextran; the C=O band has been assigned at 1660 cm−1. On the other hand, the absorption peaks 1093 cm−1 and 1176 cm−1 are assigned to C–O groups in stretching mode for NVP. The appearance of the peak around 1435 cm−1 besides the peaks 1093 cm−1 and 1176 cm−1 attributed to the CO····HO hydrogen bond in P(NVP/dextran). 29
The absorptions that appear at 1213, 1160, and 1019 cm−1 are attributed to the C–O–C stretching vibration of the hydroglucose units (a triplet peak of dextran). The bands at 2930 cm−1 are characteristic of the C–H stretching vibration and 1457 cm−1 attributed to CH2. Meanwhile, a broadband appears in the region of 3615 cm−1 corresponding to the O–H and overlaps with NH stretching vibration. The interaction of AgNPs with hydrogels results in a decrease in the broadband of OH and the absorption band of C=O. As can be seen in Figure 2, the presence of the silver in P(NVA/dextran)-AgNPs leads to the weakening of all bands measure the interaction between NVP and dextran especially the carbonyl band. The shift to lower energies should be due to the formation of coordinative bonding between NVA/dextran molecules and AgNPs. Spectral comparison indicates a red shift of a peak from 1660 cm−1 in the pure hydrogels to 1662 cm−1 in the hydrogels containing AgNPs. This band is attributed to C=O stretching of NVP structure, and its shift to lower energies should be due to the chemical interaction between NVP molecules and AgNPs surface. 13
In this study, the observed peak of P(NVP/dextran) hydrogel at 2θ values of 11° and 21° corresponds to the amorphous nature of the hydrogel (Figure 3(a)), and four peaks characterized the presence of AgNPs in the polymer at 2θ values of 38°, 44°, 64°, and 77° (Figure 3(b)). These peaks correspond to the reflection planes (111), (200), (220), and (311) of the bulk Ag, which can be assigned to Ag face cubic center structure. 30 The calculated Ag size was found to be 11.5 nm for Ag content 0.2 mol/l. 26
The particle size of P(NVP/dextran)-Ag was found to be 55.8 nm. However, the DLS results were found to be in good agreement with the result observed by XRD; the relatively greater average size measured by DLS may be due to the association and aggregation of Ag particles in H2O via Van der Waals force or hydrogen band. 31,32
The major criteria to be taken into consideration for any potential anticancer drug are its efficacy in prolongation of MST and decrease of tumor volume and viable tumor cell. 33 In the present study, DOXsignificantly increased the percent of survival time and MST and decreased the tumor volume of SEC-bearing mice in comparison to the control group. DOX produces ROS via one-electron reduction to the corresponding semiquinone free radicals, which then react rapidly with oxygen to generate superoxide radical anions. 3 Accumulation of ROS inside the cells is believed to be connected to DOX-mediated cytotoxicity. 2 The cytotoxicity of DOX has been confirmed by the induction of apoptosis and cell cycle progression. 1,34
AgNPs showed significant increase in the percent survival time and MST and significant reduction in the tumor volume of solid Ehrlich carcinoma-bearing mice in comparison to the control group. In agreement with our results, Sriram et al. 11 showed that AgNPs had a significant inhibitory effect on tumor cell proliferation and survival. Significant decrease in cell viability by AgNPs was probably a result of generation of ROS, reduction in ATP production, and damage to the mitochondrial respiratory chain, which in turn leads to DNA damage, followed by cell cycle arrest at G2/M phase. 9,10,35 The effect of AgNPs in increasing the MST and life span depends on their ability to reduce tumor cell viability and induce cytotoxicity. On the other hand, Kim and Ryu 36 reported that this effect might be due to its ability to induce apoptosis in cancer cells.
In the present study, DOX-AgNPs significantly increased the percent of survival time and MST and decreased the tumor volume of SEC-bearing mice. The combination of AgNPs and DOX was found to be more effective than the activity of the DOX individually; in case of the mixture of AgNPs and DOX, AgNPs may have damaged the cell membrane leading to disruption in permeability that may have raised the uptake of the drug into cells. The NP may affect factors that control biokinetics, DNA synthesis, or cell growth due to its ROS production. Tiwari et al. 37 reported that an increased level of ROS was found in the sera of AgNPs-treated rats. AgNPs were taken up by vascular endothelial cells and induced intracellular ROS elevated, which was closely related to disruption of the integrity of endothelial layer. 38
Osman et al. 9 reported that AgNPs enhanced the activity of chemotherapeutic drugs cyclophosphamide and busulfan. The antitumor effect of silver is the result of active physicochemical interaction of silver atoms with the functional groups of intracellular proteins, as well as with the nitrogen bases and phosphate groups in DNA. 11 AgNPs reduced cell viability, and their toxicity was associated with mitochondrial membrane depolarization, increased accumulation of ROS, raised mitochondrial oxygen consumption, and caspase-3 activation. 39
In the cytotoxicity test, the combination of AgNPs and DOX was found to be more effective than the activity of DOX individually. The NP may affect the factors that control biokinetics, DNA synthesis, or cell growth. AgNPs may have damaged the cell membrane leading to disruption in permeability that may have elevated the uptake of the drug into cells. By using AgNPs and the drugs in combination, the required amount of drug will be reduced, which may lead to decrease the side effects of the chemotherapeutic drug. 35
In contrary to the present results, previous in vitro studies revealed that AgNPs could cause strong cytotoxicity in a broad spectrum of cells, such as mesenchymal stem cells, 40 HepG2 human hepatoma cells, human gliomacells (U251), human normal bronchial epithelial (BEAS-2B) cells, and HeLa cells. 10 ,41 –43 Previous studies summarized that AgNPs induced oxidative stress (OS) and apoptosis in cultured cells and animal tissues. 37,39,44
The present study demonstrated that the activities of serum CPK and LDH and serum are significantly increased in DOX-treated group compared with the control. Increased activities of these enzymes in serum are indicative of cellular damage, loss of functional integrity, and/or permeability of cell membrane. 45,46
The results of this study showed that AgNPs increase the activities of serum LDH and CPK. These results are in agreement with previous studies. 47,48 The AgNPs exposure could induce the changes in cell shape, reduce cell viability, increase LDH release, and finally result in cell apoptosis and necrosis. 48 Moreover, internalized AgNPs can disrupt the integrity of the cell membrane, cause lysosomal swelling, and even rupture lysosomal membranes. 49 On the other hand, these results are not in agreement with the findings of Seyedalipour et al. 50 who revealed that the LDH activity was not changed significantly after exposure to different concentrations of silver NPs, which shows the safety of these particles on LDH activity.
Our results exhibited a significant increase of cardiac MDA and NOx content with concomitant decrease in SOD activity and reduced GSH content in DOX-treated rats, suggesting the primary role of OS in DOX cardiotoxicity. These data are in agreement with earlier studies. 51 –53
The ROS leads to depletion of the endogenous antioxidant system and oxidative injury. GSH peroxidase and SOD constitute the first line of cellular defense against ROS, because they can decompose superoxide anions and hydrogen peroxide before their interaction to form the more reactive hydroxyl radical. 53 MDA is an end product of lipid breakdown caused by OS, and its content in tissues is considered to be a good indicator of radical-induced lipid peroxidation. 45 The decreased activity of SOD might be attributed to its increased utilization for scavenging ROS. 46
The results of the present study revealed a significant increase of cardiac MDA and NOx content with concomitant decrease in SOD activity and reduced GSH content in AgNPs-treated rats. These results are in agreement with previous studies. 47,54 Depletion of GSH and changes in the activity of various antioxidant enzymes have been implicated in oxidative damage. 55
NPs can damage different organs and tissues as they are small enough to pass from smallest capillary vessel of body and biological membranes and be toxic through producing OS and free radicals. 47 On the other hand, AgNPs elevate OS by decreasing GSH and SOD and increasing NOx and lipid peroxidation, inflammation, DNA damage, apoptosis, and necrosis. 54
AgNPs can produce ROS and OS in vivo, as well as release Ag ions. The dissolution of AgNPs into Ag ions triggers the production of hydroxyl radicals. According to a Fenton-like reaction, dispersed AgNPs can induce ROS like hydroxyl radicals in the presence of hydrogen peroxide. Hydrogen peroxide inside a cell at a low dose can accelerate the dissolution of AgNPs and produce much stronger OS. 56 AgNPs and Ag ions initiate lipid peroxidation and increase the permeation of cell membrane systems leading to the leakage of cytoplasmic contents, such as LDH, and eventual necrosis, while the rupture of lysosomal membranes releases cathepsins into the cytoplasm and activates lysosome-mediated apoptosis.54
On the other hand, the administration of DOX-AgNPs induced a significant decrease in DX-induced cardiotoxicity via decreasing the MDA and NOx and increasing the GSH and SOD compared to the DOX-treated group. Previous researches have revealed that nanosized carrier enhances the accumulation of DOX in the tumor site and decreases the side effect. 57 –59
Conclusions
This work was aimed to prepare and characterize the P(NVP/dextran)-AgNPs to evaluate its effect on the cytotoxicity of DOX. The FTIR results represented a chemical interaction between P(NVP/dextran) molecules and AgNPs surface. The results of UV–vis, the XRD, and the DLS analysis showed a relatively narrow size of AgNPs. The formation of a new biocompatible nano cancer drug by the combination of P(NVP/dextran)-AgNPs with DOX was used to facilitate the cellular drug efficiency. The combination of AgNPs and DOX improved the therapeutic index and reduced the DOX-induced cardiotoxicity by decreasing cardiac injury via preservation of cardiac markers. DOX-loaded AgNPs represent a promising candidate for cancer treatment.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.