Abstract
Dementia is a syndrome of progressive nature, affects wide range of cognitive abilities like memory, language, calculation and so on, neuropsychiatric and social deficits to impair the routine social functions. The present study was designed to assess the effect of curcumin against colchicine-induced cognitive dysfunction and oxidative stress in rats and compare it with rivastigmine. Colchicine (15 µg/5µl) was administered to male Wistar rats intracerebroventricularly (i.c.v.) by stereotaxic apparatus to induce cognitive dysfunction. Administration of colchicine caused poor retention of memory in elevated plus maze, passive avoidance apparatus and Morris water maze paradigms. Chronic treatment with curcumin (100, 200 and 400 mg/kg, p.o.) twice daily and rivastigmine (2.5 mg/kg, p.o.) daily for a period of 28 days beginning 7 days prior to colchicine injection significantly improved colchicine-induced cognitive impairment. Biochemical assessment revealed that i.c.v. colchicine injection significantly increased lipid peroxidation, depleted reduced glutathione levels and decreased acetyl cholinesterase (AChE) activity in rat brains. Chronic administration of curcumin significantly reduced the elevated lipid peroxidation, restored the reduced glutathione levels and AChE activity; however, rivastigmine failed to prevent oxidative stress. The results of the current study indicate that curcumin (100, 200 and 400 mg/kg, p.o.) twice daily has a protective role against colchicine-induced cognitive impairment and associated oxidative stress.
Introduction
Over the past few decades, the dramatic increase in life expectancy has resulted in increased incidence of age-related disorders like dementia. 1 Dementia is a syndrome with deterioration in cognitive abilities that impairs the successful performance of activities of daily life. Memory is the most common cognitive ability lost with dementia along with language, visuospatial ability, calculation, judgment and problem solving. Alzheimer’s disease (AD) is the leading cause of dementia. 2 The pathological hallmarks of AD are the formation of senile plaques and neurofibrillary tangles containing hyperphosphorylated tau protein. 3 Several findings have revealed the role of oxidative stress in the pathogenesis of AD. 4 –6 Studies have also focused on the relationship between progression of dementia with the increase in oxidative stress biomarkers. 5,7 Oxidative stress in AD leads to neuronal injury in various brain regions like hippocampus, limbic system, neocortex, entorhinal cortex and other regions of cortex associated with cognition. 8,9
Various animal models of neurodegenerative diseases, particularly AD, have been developed and characterized. These animal models give important information regarding the pathophysiology of neurodegeneration in humans. Central administration of colchicine-induced cognitive dysfunction is an animal model of sporadic dementia of Alzheimer type (SDAT). It produces marked destruction of hippocampal granule cells, mossy fibers and septohippocampal pathways. 10 Colchicine binds to tubulin and causes neurofibrillary degeneration associated with cholinergic loss and also decrease in acetylcholine transferase. 11 In addition, central administration of colchicine has been shown to cause increase in free radical generation and oxidative damage leading to cognitive dysfunction. 9,12 Thus, administration of colchicine results in cell death along with cognitive impairment, which mimics the microtubule dysfunction in AD. 13 So, intracerebroventricular (i.c.v.) administration of colchicine is a suitable model to investigate the disease pathogenesis and to study new therapeutic modalities in the treatment of AD.
Curcumin (diferulolyl methane) is the main coloring pigment in the rhizomes of Curcuma longa. It is a dietary spice and is used for various ailments such as biliary disorders, anorexia, cough, diabetic wounds, hepatic disorders, rheumatism and sinusitis. 14 Various animal and clinical studies have demonstrated anti-inflammatory, 15 anticancer, 16 cardioprotective, 17 immunomodulatory, 18 antidepressant, 19 neuroprotective 20,21 and antioxidant activity 22 of curcumin. Curcumin is reported to be a potent antioxidant and is more potent than vitamin E as a free radical scavenger. 23 Inspite of a number of studies on the effect of curcumin in various animal models of neurological disorders, there is a dearth in literature regarding its effects on AD. Hence, the present study was designed to investigate the effects of curcumin in colchicine-induced model of SDAT and its comparison with the currently used standard drug rivastigmine.
Materials and methods
Animals
Male Wistar rats weighing 250–350 g (12–14 weeks) were used in the study. Animals were procured from Central Animal House, University College of Medical Sciences, Delhi. The animals were acclimatized to laboratory conditions prior to experimentation. After the i.c.v. administration of colchicine, the animals were kept under standard conditions of light and dark cycle with food and water ad libitum in groups of two per polypropylene cage (38 × 22 × 14 cm) with soft bedding. The study was approved by the Institutional Animal Ethics Committee, University College of Medical Sciences, Delhi. Care of animals was taken as per the guidelines of Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA). All experiments were performed between 9.00 and 15.00 h.
Surgery and i.c.v. administration of colchicine
Surgery was performed according to a previously described protocol. 10 All animals were anesthetized with chloral hydrate (350 mg/kg, intraperitoneally (i.p.)) and positioned in a stereotaxis apparatus. The head was positioned in a frame and a midline sagittal incision was made in the scalp. Two holes were drilled through the skull for the placement of injection cannula into the lateral cerebral ventricle. Coordinates for the i.c.v. cannula implantation were 0.8 mm posterior to bregma, 1.8 mm lateral to the saggital suture and 3.6 mm beneath the cortical surface according to the atlas of Paxinos and Watson. 24 The scalp was then closed with suture. After surgery, all animals received gentamicin (5 mg/kg, i.p.) for 3 consecutive days. Rats were infused i.c.v. with either artificial cerebrospinal fluid (ACSF; in mmol/L: 147 NaCl, 2.9 KCl, 1.6 MgCl2, 1.7 CaCl2 and 2.2 dextrose) or 15 µg colchicine dissolved in ACSF. The solution (5 µl) was injected using a Hamilton microsyringe positioned in the injection cannula. To promote diffusion, the microsyringe was left in place for a period of 2 min following injection. The animal was kept in a separate cage for 1 week for postoperative recovery and then returned to home cage. Special care of the animals was taken during the postoperative period.
Drugs and treatment schedule
Colchicine (Sigma Chemicals Co., St Louis, Missouri, USA) was freshly prepared at the beginning of each experiment in ACSF and given in a dose of 15 µg/5 µl, i.c.v. Rivastigmine (Rivamer, Sun Pharma, India) was given in a dose of 2.5 mg/kg p.o., daily. It was purchased from the market and suspended in 1% carboxymethylcellulose (CMC) in double distilled water and administered at 0.5 ml/100 g of body weight. Curcumin powder (Curcumaxa) was purchased from Kancor Flavors & Extracts Ltd, India. Literature given by the manufacturer indicates that the curcumin powder was manufactured using selected grades of turmeric. Curcuminoids—the main substances in turmeric extract—are complex pigments consisting of curcumin, demethoxy curcumin, bis-demethoxy curcumin and traces of dihydrocurcumin. Curcuminoids were isolated from turmeric extract, using completely safe, nonchlorinated solvent under stringent good manufacturing practice conditions. Curcumin powder was standardized to contain 95–97% curcuminoids by high-performance liquid chromatography, including pure curcumin content at levels over 70%. It has minimal residues of heavy metals, solvents and is free from harmful microorganisms. Curcumin was given in doses of 100, 200 and 400 mg/kg twice daily orally suspended in 1% CMC in double distilled water. It was administered at 1 ml/100 g of body weight. The doses of curcumin and rivastigmine were selected on the basis of previous studies conducted in laboratory and those reported in literature. 12,25
Animals were randomly divided into 13 groups having 8 animals per group (Figure 1):

Animal groups and their treatments. ACSF: artificial cerebrospinal fluid; CMC: carboxymethylcellulose; i.c.v.: intracerebroventricular; COL: colchicine; RIV: rivastigmine; CUR: curcumin.
Group 1: ACSF (5 µl i.c.v.) + CMC (vehicle for curcumin);
Group 2: Colchicine (15 µg/5 µl, i.c.v.) + CMC (vehicle for curcumin);
Group 3: ACSF (5 µl i.c.v.) + rivastigmine (2.5 mg/kg p.o. daily) for 28 days;
Group 4: Colchicine + rivastigmine (2.5 mg/kg, p.o.) for 28 days started 7 days before colchicine injection;
Group 5: ACSF (5 µl i.c.v.) + curcumin (100 mg/kg, twice daily p.o.) for 28 days;
Group 6: ACSF (5 µl i.c.v.) + curcumin (200 mg/kg, twice daily p.o.) for 28 days;
Group 7: ACSF (5 µl i.c.v.) + curcumin (400 mg/kg, twice daily p.o.) for 28 days;
Group 8: Colchicine + curcumin (100 mg/kg, twice daily p.o.) for 28 days started 7 days before colchicine injection;
Group 9: Colchicine + curcumin (200 mg/kg, twice daily p.o.) for 28 days started 7 days before colchicine injection;
Group 10: Colchicine + curcumin (400 mg/kg, twice daily p.o.) for 28 days started 7 days before colchicine injection;
Group 11: Colchicine + curcumin (100 mg/kg, twice daily p.o.) for 7 days before colchicine injection;
Group 12: Colchicine + curcumin (200 mg/kg, twice daily p.o.) for 7 days before colchicine injection;
Group 13: Colchicine + curcumin (400 mg/kg, twice daily p.o.) for 7 days before colchicine injection.
Behavioral assessment
Transfer latency on elevated plus maze paradigm
An elevated plus maze consisting of two open arms (50 × 10 cm) crossed with two closed arms (50 × 10 × 40 cm) with an open roof was used. Acquisition of memory (initial transfer latency (ITL)) was tested on day 13 after colchicine administration. Rats were placed individually at either ends of the open arms and allowed to enter either of the closed arms. A cutoff time of 90 s was chosen and for the animal which did not enter the enclosed arm within 90 s, it was pushed into one of the enclosed arm and the ITL was given as 90 s. The procedure was repeated on day 14 and day 21 to check retention latency and termed as the first retention transfer latency (1st RTL) and second retention transfer latency (2nd RTL), respectively. 26
Step-down latency (SDL) in passive avoidance apparatus
On day 14, the animals were placed on a centrally located elevated wooden platform in passive avoidance apparatus, and on stepping down from the platform the animals were given an electric shock (20 V). The animals were given 3 trainings at an interval of 1 h. The acquisition SDL in seconds was recorded after 1 h of third training session without giving shock. The experiment was repeated on day 15 and day 21 without shock and the time taken for the rat to step down was measured. This is known as first and second retention latency. A cutoff time of 5 min was chosen and for the animal which did not step down in this period, SDL was taken as 5 min. 27–29
Spatial navigation task
To examine the acquisition and retention of animals on spatial navigation task, the animals received a training session consisting of 4 trials on day 13 in Morris water maze.
The latency to find the escape platform was recorded to a maximum of 3 min. If the rat did not escape on to the platform within this time, it was guided to the platform and was allowed to remain there for 30 s. The time taken by rat to reach the platform was taken as the initial acquisition latency (IAL). Following 24 h (day 14) and 8 days (day 21) after IAL, rats were tested for retention of response. The time taken to find the hidden platform on day 14 and day 21 following central administration of colchicine was recorded and termed as first retention latency (1st RL) and second retention latency (2nd RL), respectively. 30
Biochemical tests
Biochemical tests were carried out 24 h after the last behavioral test on day 21 following colchicine injection, that is on day 22. Animals were killed by decapitation under deep ether anesthesia and the brains were removed and rinsed with ice-cold isotonic saline. Brain tissue samples were then homogenized in a 10-fold (w/v) amount of ice-cold 0.1 M phosphate buffer (pH 7.4). The homogenate was centrifuged at 10,000 g for 15 min and aliquots of supernatant were separated and used for biochemical estimation.
Measurement of lipid peroxidation
Malondialdehyde (MDA) which is a measure of lipid peroxidation was measured as described by Okhawa et al. 31
Measurement of reduced glutathione (GSH)
GSH was measured according to the method of Ellman. 32
Measurement of catalase (CAT)
Brain CAT activity was determined by the colorimetric method described by Clairborne. 33
Measurement of superoxide dismutase (SOD)
SOD activity was determined by the pyrogallol auto-oxidation method described by Marklund. 34
Acetyl cholinesterase (AChE) activity
AChE is a marker of extensive loss of cholinergic neurons in the forebrain. The AChE activity was assessed by the method of Ellman et al. 35 The protein content was estimated by the Biuret method 35 using bovine serum albumin as a standard.
Statistical analysis
Values are expressed as mean ± standard error of mean (SEM). The behavioral assessment was analyzed by a repeated measures two-way analysis of variance (ANOVA). The biochemical estimations were separately analyzed by one way ANOVA. Post hoc comparisons between groups were made using Tukey’s test. p < 0.05 was considered significant.
Results
Effect of curcumin and rivastigmine on memory performance in elevated plus maze paradigm in colchicine-injected rats
In the present study, the mean ITL values on day 13 of rats of all groups, irrespective of the treatment received, were relatively stable and showed no significant differences, that is all the rats entered the closed arm within 90 s (Table 1). After training the rats treated with ACSF, rivastigmine (2.5 mg/kg, p.o., daily) or curcumin (100, 200 and 400 mg/kg, p.o., twice daily) entered the closed arm quickly and the mean retention transfer latencies (1st RTL and 2nd RTL) to enter the closed arm on days 14 and 21, respectively, were shorter as compared to the ITL on day 13 (training session) for each group. However, the difference between the ITL values of days 13 and 14 was not found to be statistically significant. In the colchicine-injected rats there was no change in the 1st RTL and 2nd RTL on days 14 and 21, respectively, as compared to the pretraining, that is ITL on day 13, demonstrating that colchicine produced marked memory impairment. Chronic administration of rivastigmine (2.5 mg/kg, p.o., daily) and curcumin (100, 200 and 400 mg/kg, p.o., twice daily) prior to colchicine injection significantly decreased the mean RTL on days 14 and 21 of colchicine injection (p < 0.05 compared to the i.c.v. colchicine alone group). However, curcumin (100, 200 and 400 mg/kg, p.o., twice daily) for only 7 days before colchicine injection did not show any significant results (data not shown).
Effect of curcumin and rivastigmine on memory performance in the elevated plus maze paradigm in intracerebroventricular colchicine-injected rats
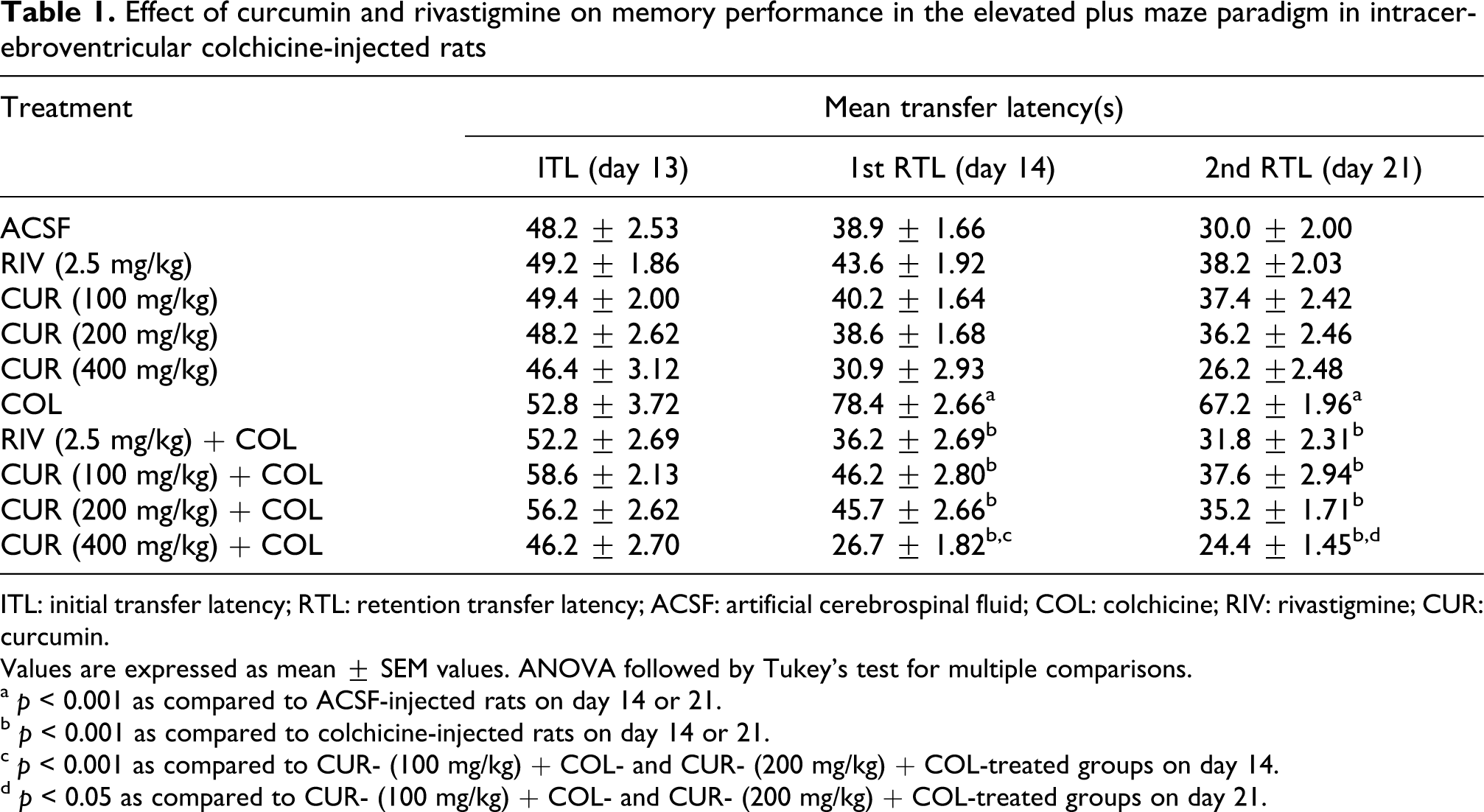
ITL: initial transfer latency; RTL: retention transfer latency; ACSF: artificial cerebrospinal fluid; COL: colchicine; RIV: rivastigmine; CUR: curcumin.
Values are expressed as mean ± SEM values. ANOVA followed by Tukey’s test for multiple comparisons.
a p < 0.001 as compared to ACSF-injected rats on day 14 or 21.
b p < 0.001 as compared to colchicine-injected rats on day 14 or 21.
c p < 0.001 as compared to CUR- (100 mg/kg) + COL- and CUR- (200 mg/kg) + COL-treated groups on day 14.
d p < 0.05 as compared to CUR- (100 mg/kg) + COL- and CUR- (200 mg/kg) + COL-treated groups on day 21.
Effect of curcumin and rivastigmine on SDL in passive avoidance apparatus
In the current study, the mean IAL values on day 14 of rats of all groups, irrespective of the treatment received, were relatively stable and showed no significant differences (Table 2). After training, the rats treated with ACSF, rivastigmine (2.5 mg/kg, p.o., daily) or curcumin (100, 200 and 400 mg/kg, p.o., twice daily) showed a significant (p < 0.05) prolongation in the step-down retention latencies on days 15 and 21, respectively, as compared to acquisition latency on day 14 (training session). On the other hand, the colchicine-injected rats showed no change in the step-down retention latencies on days 15 and 21, respectively, as compared to acquisition latency on day 14, which indicates that colchicine produced marked memory impairment. Chronic administration of rivastigmine (2.5 mg/kg, p.o., daily) and curcumin (100, 200 and 400 mg/kg, p.o., twice daily) prior to colchicine injection significantly prolonged the mean RL on days 15 and 21 of colchicine injection (p < 0.05 compared to the i.c.v. colchicine alone group). However, curcumin (100, 200 and 400 mg/kg, p.o., twice daily) for only 7 days before colchicine injection did not show any significant results (data not shown).
Effect of curcumin and rivastigmine on step-down latency in passive avoidance paradigm in intracerebroventricular colchicine-injected rats

IAL: initial acquisition latency; RL: retention latency; ACSF: artificial cerebrospinal fluid; COL: colchicine; RIV: rivastigmine; CUR: curcumin.
Values are expressed as mean ± SEM values. ANOVA followed by Tukey’s test for multiple comparisons.
a p < 0.001 as compared to ACSF-injected rats on day 14 or 21.
b p < 0.001 as compared to colchicine-injected rats on day 14 or 21.
c p < 0.001 as compared to CUR- (100 mg/kg) + COL-treated group on day 14.
Effect of curcumin and rivastigmine on spatial navigation task in colchicine-injected rats
ACSF injected, rivastigmine (2.5 mg/kg, p.o., daily) or curcumin (100, 200 and 400 mg/kg, p.o., twice daily) groups of animals quickly learned to swim directly to the hidden platform in the Morris water maze on day 13. Colchicine-injected rats showed an increase in escape latency, which decreased with continuous training during the acquisition of a spatial navigation task on day 13. Rivastigmine and curcumin treatment caused similar acquisition of a spatial navigation task on day 13 (compared with the ACSF-injected group). There was a significant difference in the mean IAL of the colchicine-injected group as compared to the ACSF-injected group on day 13, indicating colchicine-induced impairment in this spatial navigation task (p < 0.05). Administration of rivastigmine (2.5 mg/kg, p.o., daily) and curcumin (100, 200 and 400 mg/kg, p.o., twice daily) prior to colchicine injection significantly decreased the IAL to reach the platform in the pretrained rats as compared to colchicine-injected rats on day 13 (p < 0.05; Table 3).
Effect of curcumin and rivastigmine on spatial navigation task paradigm in intracerebroventricular colchicine-injected rats

ITL: initial transfer latency; RTL: retention transfer latency; ACSF: artificial cerebrospinal fluid; COL: colchicine; RIV: rivastigmine; CUR: curcumin.
Values are expressed as mean ± SEM values. ANOVA followed by Tukey’s test for multiple comparisons.
a p < 0.001 as compared to ACSF-injected rats on day 14 or 21.
b p < 0.001 as compared to colchicine-injected rats on day 14 or 21.
c p < 0.01 as compared to CUR- (100 mg/kg) + COL- and CUR- (200 mg/kg) + COL-treated groups on day 14.
Following training, the mean retention latencies (1st RL and 2nd RL) to escape onto the hidden platform was significantly decreased in ACSF-injected rats on days 14 and 21 as compared to the IAL on day 13 following colchicine injection (p < 0.05). Rivastigmine and curcumin alone treatment did not show any alteration in the 1st and 2nd RL on days 14 and 21, respectively (compared to the ACSF-injected group). In contrast, the performance of the colchicine-injected rats was changed after the initial training in the water maze on days 14 and 21, with significantly higher mean RL compared to the ACSF-injected group (p < 0.05). These results suggest that colchicine caused significant cognitive impairment. On the other hand, chronic treatment with rivastigmine (2.5 mg/kg p.o., daily) starting before colchicine injection produced a decline in the 1st RL and 2nd RL on days 14 and 21, as compared to colchicine-treated group. Similarly chronic administration of curcumin (100, 200 and 400 mg/kg p.o., twice daily) beginning prior to colchicine injection caused a dose-dependent decline in the 1st and 2nd RL on days 14 and 21. However, curcumin (100, 200 and 400 mg/kg, p.o., twice daily) for 7 days before colchicine injection did not show any significant results (data not shown).
Effect of curcumin and rivastigmine on brain lipid peroxidation and GSH levels in colchicine-injected rats
Intracerebroventricular administration of ACSF had no effect on brain MDA levels and GSH. Central colchicine administration caused a marked increase in free radical generation and lipid peroxidation as depicted by a significant rise in brain MDA levels and depletion of GSH levels as compared to ACSF-injected rats (p < 0.05). Further, curcumin and rivastigmine treatment did not cause any alteration in the brain MDA levels and GSH activity compared to ACSF-injected rats. Chronic curcumin (100, 200 and 400 mg/kg p.o., twice daily) administration prior to colchicine injection significantly and dose dependently prevented the increase in MDA levels and reduced GSH depletion (p < 0.05). Daily administration of rivastigmine (2.5 mg/kg/day, p.o.) beginning prior to colchicine injection produced no alteration in the brain MDA levels and GSH levels as compared to colchicine-treated group (Figures 2 and 3). However, curcumin (100, 200 and 400 mg/kg, p.o., twice daily) administered for only 7 days before colchicine injection did not show any significant results (data not shown).

Effect of curcumin and rivastigmine on brain lipid peroxidation levels in i.c.v. colchicine-injected rats. Values are expressed as mean ± SEM. ACSF: artificial cerebrospinal fluid; COL: colchicine; RIV: rivastigmine; CUR: curcumin. Statistics were calculated by repeated measures two-way analysis of variance (ANOVA) followed by Tukey’s test for multiple comparisons: a p < 0.05 as compared to ACSF-injected rats; b p < 0.05 as compared to colchicine-injected rats; c p < 0.05 as compared to CUR- (100 mg/kg) + COL-treated group.
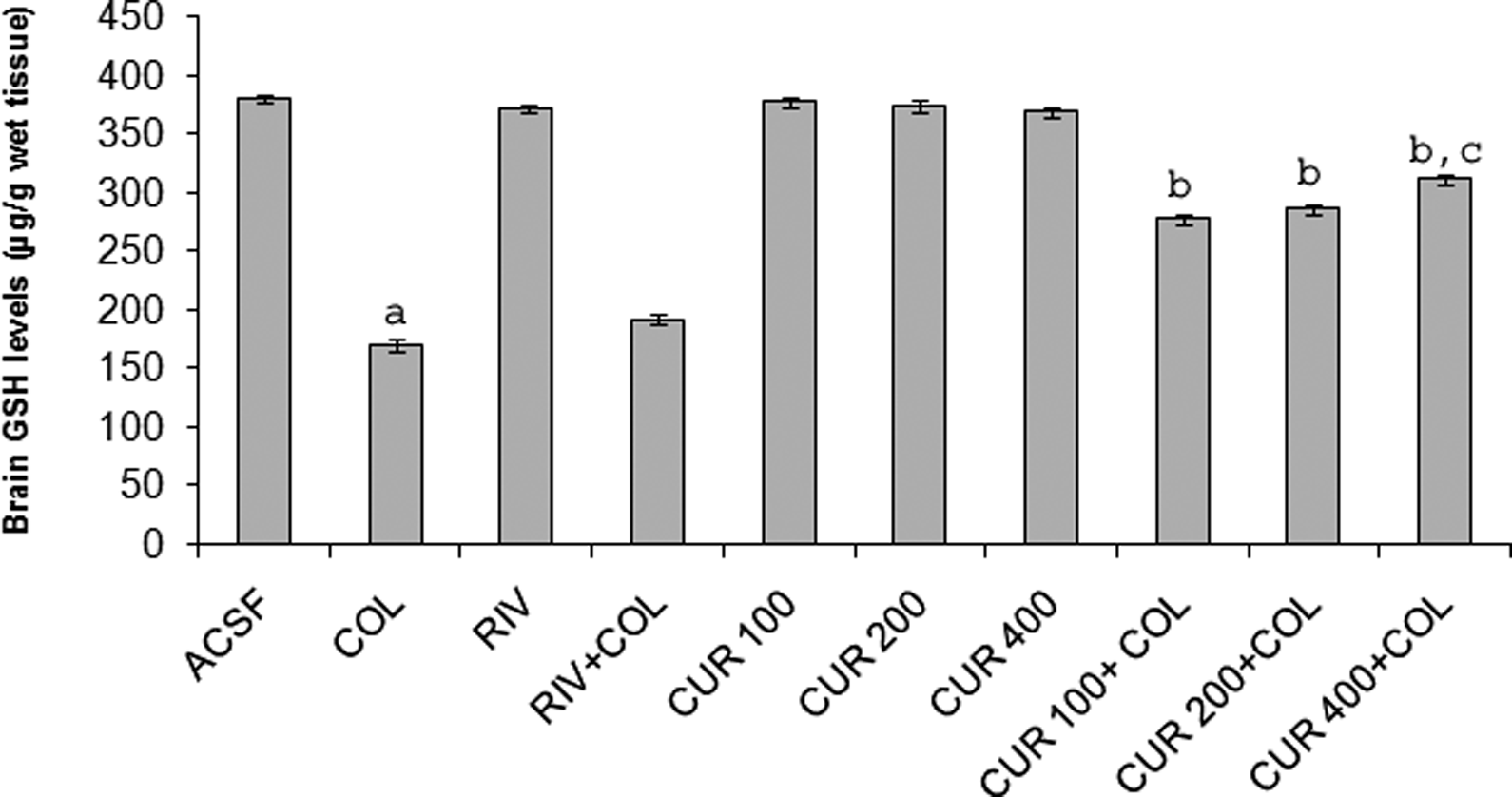
Effect of curcumin and rivastigmine on brain GSH levels in i.c.v. colchicine-injected rats. Values are expressed as mean ± SEM. ACSF: artificial cerebrospinal fluid; COL: colchicine; RIV: rivastigmine; CUR: curcumin; GSH: glutathione. Statistics were calculated by repeated-measures two-way analysis of variance (ANOVA) followed by Tukey’s test for multiple comparisons: a p < 0.05 as compared to ACSF-injected rats; b p < 0.05 as compared to colchicine-injected rats; c p < 0.05 as compared to CUR- (100 mg/kg) + COL-treated group.
Effect of curcumin and rivastigmine on SOD and CAT levels in colchicine-injected rats
Results of SOD and CAT enzyme activity did not show any change in all the groups (data not shown).
Effect of curcumin and rivastigmine on brain AChE levels in colchicine-injected rats
Intracerebroventricular administration of ACSF had no effect on brain AChE levels. In contrast, central colchicine injection produced a significant decrease in brain AChE activity compared to ACSF-injected rats (p < 0.05). Curcumin and rivastigmine treatment caused no change in brain AChE activity compared to ACSF-injected rats. However, chronic administration of rivastigmine (2.5 mg/kg, p.o., daily) and curcumin (100, 200 and 400 mg/kg, p.o., twice daily) prior to colchicine injection caused significant reduction in AChE levels as compared to colchicine-injected group (p < 0.05; Figure 4).

Effect of curcumin and rivastigmine on brain AChE levels in i.c.v. colchicine-injected rats. Values are expressed as mean ± SEM. ACSF; artificial cerebrospinal fluid; COL: colchicine; RIV: rivastigmine; CUR: curcumin. Statistics were calculated by repeated measures two-way analysis of variance (ANOVA) followed by Tukey’s test for multiple comparisons: a p < 0.05 as compared to ACSF-injected rats; b p < 0.05 as compared to colchicine-injected rats; c p < 0.05 as compared to CUR- (100 mg/kg) + COL-treated group.
Discussion
In the current study, i.c.v. colchicine induced significant memory impairment as evidenced by increased relevant retention latencies in elevated plus maze and Morris water maze paradigms and decreased latency in passive avoidance apparatus. It has also been shown in the present study that central administration of colchicine causes an increase in free radical generation leading to oxidative stress which may be responsible for cognitive impairment. Earlier studies indicate that centrally administered colchicine induced cognitive dysfunction. 36,37
Central administration of colchicine causes microtubule dysfunction and decrease in cholinergic turnover associated with the loss of cholinergic neurons in brain due to inhibition of fast axoplasmic flow or direct toxic effect on cholinergic terminals. 10 Microtubule destabilization including memory impairment might be a key factor. 13 It produces marked destruction of hippocampal granule cells, mossy fibers and septohippocampal pathways. 38 Experimentally, it has been shown that colchicine elevates glutamate/γ-aminobutyric acid ratio in the cortex of mice brain, 39 and it exerts neurotoxic effect by generating the hydroxyl radicals. 40 Alteration in glucose/energy metabolism in brain disturbs the endogenous antioxidant defence mechanism and causes neuronal damage. 41,42 This leads to oxidative stress which in turn initiates the changes in macromolecules, lipid membranes, enhances the release of glutamate and thus completes a vicious cycle that leads to free radical-induced toxicity. 43
Curcumin significantly improved cognitive dysfunction and attenuated colchicine-induced oxidative stress which is in accordance with the earlier study of Kumar et al. 44 However, alone it had no effect on various cognitive parameters and oxidative stress markers in the brain of control animals. The protective effect of curcumin against colchicine-induced cognitive impairment and oxidative stress may be ascribed to its antioxidant property. Earlier studies have also emphasized on the neuroprotective properties of curcumin in various diseases such as myocardial infarction, rheumatoid arthritis, diabetes and various skin diseases which are associated with oxidative stress. These findings may be attributed to its antioxidant property. 45 –48 Studies have shown that curcumin crosses the blood–brain barrier and produces neuroprotection directly. 49 The antioxidant property of curcumin may be attributed to its (a) nitric oxide scavenging ability 50 ; (b) presence of two electrophilic α, β-unsaturated carbonyl groups, which reacts with nucleophiles; (c) metal-chelating property; (d) ability to inhibit various oxidases like xanthine oxidase 46,51 ; (e) inhibit lipoxygenases and phospholipase D 52 –54 ; (f) free radical scavenging property as well as inhibition on the upregulation of cyclooxygenase 2 and inducible NO synthase expression. 55,56
Central administration of colchicine caused a marked decrease in AChE activity in our study and similar decrease was reported in earlier studies also. 57 Curcumin alone had no effect on cognitive performance and basal AChE levels in control animals but attenuated the reduced AChE activity when it was administered in colchicine-treated rats. This may be another mechanism for the improvement in cognitive impairment induced by colchicine.
It has been observed in the present study that rivastigmine is effective in improving the cognitive function as observed in elevated plus maze, passive avoidance and Morris water maze paradigms. However, it failed to prevent oxidative stress suggesting that besides free radical generation other mechanisms might influence cholinergic neuronal function. Experimentally rivastigmine has been found to be effective in ameliorating memory deficits in various models of amnesia. 58,59
Increased oxidative damage stimulates various intracellular signals that lead to apoptosis followed by neurodegeneration and colchicine is reported to trigger it both in vitro and in vivo. 13,60 Various studies indicate that curcumin suppresses apoptosis of cells indirectly by inhibition of ROS-induced protein kinase C-mediated activation of pathways involving the transcription factors nuclear factor-κβ activator protein-1, signal transducer and activator of transcription 3, c-Jun N-terminal kinase, Bcl-2 and p38 by its antioxidant effect 60 and directly inhibiting caspase and transcription factors. 48 It has been shown in various studies that colchicine induces neurofibrillary degeneration which is prevented by curcumin due to its antiamyloidic and fibril-destabilizing effects. 13,49,55,61 Thus, it is likely that a wide spectrum of new molecular targets may be concerned in the protective effect of curcumin in the colchicine-induced cognitive dysfunction mimicking SDAT.
In summary, the multiple effects of curcumin bring to light the search for possible protective mechanisms involved in improving the cognitive dysfunction. Hence, curcumin may find the place as potential therapeutic option for the prevention or treatment of AD and other neurodegenerative diseases of aging. To conclude, the results of the present study suggest that chronic administration of curcumin prevents colchicine-induced cognitive impairment and oxidative damage in comparison to rivastigmine which improves cognitive dysfunction but has no beneficial effects on oxidative stress. Furthermore, the use of curcumin as dietary supplement should be encouraged to ward off age-associated disorders with free radical generation and cognitive impairment like AD.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Declaration of Conflict Interest
The authors declared no conflicts of interest.
