Abstract
The aim of the study was to compare the effect of sodium arsenate (AsV) on two different cell types: 158N murine oligodendrocytes and HepG2 human hepatoma cells. Exposure of 158N cells to AsV (0.1–400 µM; 48 h) induced a biphasic cytoxic effect defined as hormesis. Thus, low concentrations of AsV stimulate cell proliferation, as shown by phase-contrast microscopy, cell counting with trypan blue, and crystal violet assay, whereas high concentrations induce cell death associated with a loss of cell adhesion. These side effects were confirmed by staining with propidium iodide and cell cycle analysis, characterized by the presence of a subG1 peak, a criterion of apoptosis. The effects of AsV on mitochondrial function, as determined by the 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium (MTT) assay, the measurement of mitochondrial transmembrane potential with 3,3′-dihexyloxacarbocyanine iodide, and the rate of mitochondrial adenosine triphosphate confirm the impact of AsV on the mitochondria. In contrast to 158N cells, HepG2 cells were susceptible to all AsV concentrations as shown by microscopic observations, by counting with trypan blue. However, no alteration is noted in the cell membrane integrity, which indicated an apoptotic mode of cell death, and this side effect is confirmed by the cycle analysis, which revealed a subG1 peak. Of note, there was a loss of MTT, suggesting that AsV induces mitochondrial complex II dysfunction. Altogether, our data show that the cytotoxic characteristics of AsV depend on the cell type considered.
Keywords
Introduction
Exposure to arsenic (As) is known to cause a number of toxic effects involving multiple organ systems (liver, lung, kidney, nervous system, and so on). The carcinogenic potential of As has also been recognized. 1 st mammals metabolize inorganic As (iAs) via methylation to methylarsonic acid (MMA) and dimethylarsinic acid (DMA). The methylation occurs via alternating the reduction of pentavalent to trivalent As and the addition of a methyl group. S-adenosylmethionine, which occurs principally in the liver, is the main methyl donor in As methylation. 2 A previous study demonstrated that As metabolism differs from one patient to another. Wang et al. 3 reported interindividual differences in excretion profiles and the relative concentrations of major As species in urine in four Chinese patients with acute promyelocytic leukemia undergoing As trioxide (As2O3) treatment. It is important to note that genetic polymorphisms in As metabolism genes, such as the human arsenic methyltransferase gene and glutathione S-transferase gene, are thought to be related to interindividual variations in As metabolism. 4,5 Also, Sohini and Rana 1 demonstrated that male rats accumulated higher concentrations of As in the most important target organs (liver and kidney) than did female rats. On the other hand, the relative ratios of the three As metabolites (iAs, MMA, and DMA) were variable depending on the organ, soluble/nonsoluble proteins, and the time after the administration. 6 Recent data have demonstrated that differences in the kinetic behavior of cells affect the uptake and retention of arsenicals. 7,8 In previous studies, it was demonstrated that peripheral nerves contained higher concentrations of arsenicals than did the central nervous system, with a predominance of the dimethylarsenic metabolite of iAs. 9 The brain shows distinct vulnerability to this metalloid. 9,10 It has also been reported that As preferentially accumulates in red blood cells than in bone marrow and plasma. 11 Many studies also reported that exposure to arsenicals (As2O3 sodium arsenate (AsV) and AsIII) is marked by their preferential accumulation in the kidney. 1,12 –14 However, the increased vulnerability of the liver in comparison with the kidney is probably due to the greater uptake of AsIII than AsV in hepatocytes. 15 Indeed, different toxicity and metabolism mechanisms of different As species have been documented. 1,7
As it seems that sensitivity to AsV varies from one cell type to another, especially between liver cells and nerve cells, we conducted a comparative study of the effect of AsV on HepG2 human hepatoma cells and 158N murine oligodendrocytes. The effects were characterized at various cellular levels especially at the mitochondrial level.
Materials and methods
Cells and treatments
HepG2 cells (hepatoma cells) were seeded at 7.5 × 104 cells per well in 12-well microplates containing 1 mL of culture medium composed of Dulbecco’s Modified Eagle’s medium (DMEM) supplemented with 10% (v/v) heat-inactivated fetal calf serum (FCS; Pan Biotech, Germany), while 158N murine oligodendrocytes were seeded at 10 × 104 cells per well in 12-well microplates containing 1 mL of culture medium composed of DMEM supplemented with 5% (v/v) heat-inactivated FCS (Pan Biotech) and 1% antibiotics (100-U/mL penicillin and 100-mg/mL streptomycin; Pan Biotech). All cell lines were incubated at 37°C in a humidified atmosphere containing 5% carbon dioxide, and the conditions of treatment with AsV were as follows: after plating different cells for 24 h, the cells were further treated for 48 h with various AsV concentrations (0.1, 1, 10, 100, 200, and 400 μM) in their respective culture medium. The 158N and HepG2cells were also treated with 7-ketocholesterol (7KC) at a concentration of 50 µM used as a positive control for cell death induction associated with mitochondrial dysfunctions. 16
Cell counting with trypan blue
After 48 h of treatment with AsV (0.1, 1, 10, 100, 200, and 400 μM), cells were trypsinized with a 0.05% trypsin and 0.02% ethylene diamine tetra-acetic acid editic acid (EDTA), centrifuged (900 g, 5 min), and resuspended in their respective culture medium. The total number of viable cells was determined by the presence of trypan blue, which stains dead cells blue. Dead cells were counted using an automatic counter (TC20 automated cell counter; Bio-Rad, USA). The measurements were made in triplicate.
Study of cell morphology by phase-contrast microscopy
The morphology of 158N and HepG2 cells was observed after 48 h of culture in the absence or the presence of AsV (0.1, 1, 10, 100, 200, and 400 μM) under an inverted phase-contrast microscope (Axiovert 40 CFL; Zeiss, Germany). Digitalized images were obtained with a camera (Axiocam ICm1; Zeiss).
Crystal violet staining procedure
Adherent cells were quantified by staining with crystal violet. 17 Cells were seeded in triplicate in 24-well plates and cultured without or with AsV (0.1, 1, 10, 100, 200, and 400 µM) for 48 h. At the end of treatment, cells were washed with phosphate buffer saline (PBS), stained with crystal violet (Sigma–Aldrich, USA; 5 min), and rinsed with water. Absorbance was read at 570 nm with a microplate reader (Tecan, Swiss) after the extraction of the dye with 0.1-mol/L sodium citrate in 50% ethanol.
Flow cytometric quantification of cell permeability to propidium iodide
As previously described by Nury et al., 18 adherent and nonadherent cells were pooled and stained with propidium iodide (PI; 5 µg/mL), which only enters dead cells or cells with damaged cytoplasmic membranes. 19 The cells were then immediately analyzed with a Galaxy flow cytometer (Partec, USA). Red fluorescence of PI was detected through a 625 ± 20-nm bandpass filter, and fluorescent signals were measured on a logarithmic scale. For each sample, 10,000 cells were acquired and data were analyzed with Flowmax software (Partec) or FlowJo software (Tree Star Inc, USA).
Colorimetric MTT assay
The MTT assay was carried out as previously described 17 on HepG2 and 158N cells plated in 24-well flat-bottom culture plates after 48 h of treatment with AsV (0.1, 1, 10, 100, 200, and 400 μM). The MTT assay was used to evaluate the effects of AsV on cell proliferation and/or viability. The plates were read at 570 nm with a microplate reader.
Measurement of mitochondrial transmembrane potential with DiOC6(3)
After 48 h of treatment, the mitochondrial transmembrane potential (ΔΨm) was measured with 3,3′-dihexyloxacarbocyanine iodide (DiOC6(3); Invitrogen, 40 nM, USA). 20 Mitochondrial depolarization is indicated by a decrease in the green fluorescence collected through a 520/10-nm bandpass filter. Flow cytometric analyses were performed on a Galaxy flow cytometer (Partec). Ten thousand cells were acquired for each sample. Data were analyzed with Flowmax software (Partec) or FlowJo software (Tree Star Inc, USA).
Cell cycle analysis
Cell cycle analysis was realized on nonadherent and adherent cells collected by trypsinization with a 0.05% trypsin-0.02% EDTA solution. Cells were pooled, washed with PBS, and stained with PI as previously described. 21 Briefly, cells were resuspended in 80% cold ethanol (2 h, −20°C), washed with PBS, and resuspended in 300 µL of PBS containing 80 mg/mL PI and 200 µg/mL ribonuclease RNase. After 1 h of incubation (37°C), 1–2 mL of PBS was added and flow cytometric analyses were performed on a Galaxy flow cytometer. Fluorescence of PI was collected using a 590 ± 10-nm bandpass filter and measured on a linear scale. Twenty thousand cells were acquired, and the percentage of cells in different cell cycle phases was manually estimated.
Mitochondrial ATP assay
The adenosine triphosphate (ATP) assay was performed using the ATP Bioluminescence Assay Kit CLS II (Roche Molecular Biochemicals, Germany). The ATP assay is based on the determination of a light emitted from a bioluminescent enzyme reaction that involves luciferin and firefly luciferase (provided by the kit) and ATP (present in the cell lysate). ATP acts in the oxidation of the luciferin–luciferase complex. Light emission due to oxidation is proportional to the amount of ATP. The light was measured using a Tecan plate reader. To measure mitochondrial ATP, ATP from glycolysis was eliminated by treating the cells with iodoacetate (25 µmol/L) for 1 h. Briefly, the supernatant recovered from the cell lyses was used for the mitochondrial ATP assay. A standard curve was prepared from an ATP stock solution (10.5 mg/mL) using lyophilized ATP provided by the kit. Fifty microliters of luciferase was added to 50 µL of each cell lysate and to each point of range (standard curve), and the bioluminescence of each sample was determined. The standard curve was plotted and used to calculate the concentration of mitochondrial ATP.
Western blot analysis of OXPHOS protein complexes
As previously described by Kharroubi et al., 22 158N cells were treated with AsV at 0.1, 1, 10, 100, 200, and 400 μM for 48 h. After treatment, the cells were trypsinized and incubated with radio immunoprecipitation assay (RIPA) lysis buffer (0.1% (w/v) sodium dodecyl sulfate (SDS), 1% (w/v) NP40, 0.5% (w/v) Na-desoxycholate, 150-mM sodium chloride, 2-mM EDTA, and 50-mM Tris-HCl, pH 8) containing a mixture of protease and phosphatase inhibitors (50-mM sodium fluoride, Protease Inhibitor Cocktail Tablets (Roche Molecular Biochemicals)). After 30-min incubation at 4°C in the lysis buffer, the cell debris was eliminated by centrifugation (20 min, 10,000 g), and the supernatant was collected. The protein concentrations were measured using bicinchoninic acid reagent (Sigma–Aldrich). 23 Fifty micrograms of protein per slot were diluted in loading buffer (125 mmol/L Tris-HCl, pH 6.8, 10% (w/v) mercaptoethanol, 4.6% (w/v) SDS, 20% (v/v) glycerol, and 0.003% (w/v) bromophenol blue), separated by SDS-polyacrylamide gel electrophoresis (PAGE), and transferred to a nitrocellulose membrane. After blocking nonspecific binding sites overnight by 5% nonfat milk in phosphate buffer saline +0.1% Tween 20 (TPBS) (PBS, 0.1% Tween 20), the membranes were incubated for 1 h at room temperature with the anti-OXPHOS (Oxidative phosphorylation) mixture (MS601, Abcam/Mitosciences, UK) against the structural components of all five oxidative phosphorylation complexes (complex I subunit NDUFB8 (20 kDa), complex II subunit (30 kDa), complex III subunit core 2 (47 kDa), complex IV subunit II (24 kDa), and complex V ATP synthase subunit alpha (53 kDa)) at a 1:500 dilution (TPBS with 1% nonfat milk). After three 10-min washes with TPBS, the membranes were incubated with horseradish peroxidase–conjugated secondary antibody (Santa Cruz Biotechnology, Germany) at a 1:5000 dilution (TPBS with 5% nonfat milk) for 1 h at room temperature and washed three times with TPBS for 10 min. Autoradiography of the immunoblots was performed using an enhanced chemoluminescence detection kit (Santa Cruz Biotechnology), and immunoblots were quantified with ImageJ software. The results were expressed as a fold change to untreated SK-NB-E cells. Actin was used as a loading control. The monoclonal antibodies in the cocktail were chosen because they act against subunits that are labile when the corresponding complex is not assembled. Moreover, the combination is readily resolved in SDS-PAGE when the appropriate gel conditions are used.
Statistical analysis
The data were analyzed using the Statistical Package for Social Sciences (SPSS) program, release 11.0 for windows (SPSS, Chicago, Illinois, USA). In each assay, the experimental data represent the mean of 10 independent assays ± standard deviations. The statistical significance of the differences in values for the different treatment groups was calculated by a Mann–Whitney U test. The threshold for significant differences was set at p < 0.05.
Results
Evaluation of the effect of AsV on cell growth and morphology
Incubation of 158N cells with different concentrations of AsV showed biphasic toxicity after 48 h. There was a significant increase in cell proliferation induced by low concentrations (0.1, 1, and 10 µM; p ≤ 0.05), with a decrease in the number of dead cells after treatment with 0.1 µM. However, at concentrations ≥100 µM, AsV was toxic on the 158N cells, with cytotoxic effects characterized by a decrease in viability and cell growth. This decrease was concentration dependent. AsV has cytostatic effects on this neuronal cell line (Figure 1(a)). These data were confirmed by observations by phase-contrast microscopy. Treatment with low concentrations (0.1, 1, and 10 µM) led to an increase in the number of adherent 158N cells, compared with the respective control cells. While with high concentrations (100, 200, and 400 μM), a reduced number of viable cells were found. This decrease was associated with a concentration-dependent increase in the number of floating round cells, corresponding to less adherent, dying cells (Figure 1(b)).

Evaluation of the effects of AsV on cell growth and cell morphology of 158N murine oligodendrocytes. (a) Number of cells per well of 24-well plates determined by trypan blue staining: total cells and dead cells (trypan blue–positive cells); (b) observation with phase-contrast microscopy. Data shown are mean ± standard deviation. Significant differences between control and treated cells (by AsV and 7KC) are indicated by * (Mann–Whitney U test; *p ≤ 0.05). AsV: sodium arsenate; 7KC: 7-ketocholesterol.
Treatment of HepG2 cells with AsV increased the total number of cells in a dose-dependent manner (Figure 2(a)). Similarly, the number of dead cells in the wells treated with AsV increased significantly (p ≤ 0.05) compared to control. This increase was concentration dependent (Figure 2(a)). Microscopic observation of HepG2 cells showed a concentration-dependent decrease in cell proliferation after 48 h of treatment. When the concentration of AsV was increased, the cell clumps became smaller and were associated with a loss of adhesion (Figure 2(b)). In agreement with previous reports., 18 7KC (50 µM) was toxic in 158N cells (Figure 1(a)). However, it was not toxic in HepG2 cells (Figure 2(a)).

Evaluation of the effects of AsV on cell growth and cell morphology of human hepatoma HepG2 cells. (a) Number of cells per well of 24-well plates determined by trypan blue staining: total cells and dead cells (trypan blue–positive cells); (b) observation with phase-contrast microscopy. Data shown are mean ± standard deviation. Significant differences between control and treated cells (by AsV and 7KC) are indicated by * (Mann–Whitney U test; *p ≤ 0.05). AsV: sodium arsenate; 7KC: 7-ketocholesterol.
Evaluation of the effect of AsV on cell confluence by crystal violet
The treatment of 158N cells with AsV (0.1, 1, 10, 100, 200, and 400 µM) for 48 h showed marked concentration-dependent effects on cell growth, characterized by biphasic toxicity. In 158N cells treated with AsV at 0.1, 1, and 10 µM, the crystal violet test showed a significantly increased number of adherent cells compared with control cells (p ≤ 0.01). However, an increased number of cells floating in the culture medium were observed at concentrations of 100 µM and higher (p ≤ 0.05). Treatment of cells with 7KC (50 µM) induced a significant decrease in the number of adherent cells (Figure 3(a)).

Effects of AsV on cell confluence and cytoplasmic membrane integrity. Effect of AsV on cell confluence of 158N cells by crystal violet staining (a) and on the cytoplasmic membrane integrity of 158N (b) and HepG2 cells (c) with the PI test. Data shown are mean ± standard deviation. Significant differences between control and treated cells (AsV and 7KC) are indicated by * (Mann–Whitney U test; *p ≤ 0.05). AsV: sodium arsenate; 7KC: 7-ketocholesterol; PI: propidium iodide.
Evaluation of the effects of AsV on cytoplasmic membrane integrity and/or cell death as assessed with PI
Two types of response were identified after the treatment of 158N cells with different concentrations of AsV. No effect of AsV was revealed on 158N cells (Figure 3(b)) after treatment with low concentrations (0.1, 1, and 10 µM), while at high concentrations (100, 200, and 400 µM), we observed an increase in the percentage of cells with altered cytoplasmic membranes and/or dead cells (PI+ cells). This side effect was concentration dependent and was significantly increased at concentrations ≥200 µM (Figure 3(b)). HepG2 cells incubated with AsV showed no significant increase in the proportion of PI+ cells, except at the highest concentration (at 400 µM, a significant decrease was noted; Figure3(c)). The 7KC (50 µM) significantly increased the proportion of PI+ cells (Figure 3(b) and (c)).
Evaluation of the effect of AsV on the cell cycle
Cell cycles in 158N treated with 0.1 and 1 µM of AsV were similar to those in untreated cells (control). However, from the concentration of 10 µM onwards, we observed an accumulation of cells in the S-phase with a concomitant decrease in numbers in the G1 and G2/M phases (Figure 4(a)). These changes were concentration dependent and became statistically significant at 100 µM. Treatment with 10, 100, 200, and 400 µM resulted in the appearance of a sub-G1 peak suggesting an apoptotic process. 24 HepG2 cells treated with 0.1, 1, and 10 µM showed no significant variations in the distribution of cells in the different phases of the cell cycle compared with untreated controls. The 100-µM concentration increased the sub-G1 fraction (p ≤ 0.05) and decreased the G2/M phase (p ≤ 0.05). The greatest disturbances were observed after treatment with 200 and 400 µM: an increase in the sub-G1 fraction with a decrease in the G0/G1 and G2/M phases and an arrest in the S-phase (Figure 4(b)).

Effect of AsV on the distribution of the cells in the different phases of the cell cycle. Distribution of control and AsV-treated cells in the different phases of the cell cycle: (a) 158N cells and (b) HepG2 cells were determined by flow cytometry after staining with PI. Data shown are mean ± standard deviation. Significant differences between control and treated cells (AsV and 7KC) are indicated by * (Mann–Whitney U test; *p ≤ 0.05). AsV: sodium arsenate; 7KC: 7-ketocholesterol; PI: propidium iodide.
Evaluation of the effects of AsV with the MTT assay
The MTT assay is an indicator of mitochondrial activity and evaluates cell vitality. Treating 158N cells with high concentrations (100, 200, and 400 µM) of AsV for 48 h induced a significant decrease in the percentage of positive MTT cells. This decrease in the metabolic capacity was not significant in cells treated with low concentrations (0.1, 1, and 10 µM) (Figure 5(a)). The incubation of HepG2 cells with any concentration of AsV (0.1–400 μM) for 48 h significantly decreased the metabolic capacity of HepG2 cells (Figure 5(b)). The 7KC (50 µM) significantly decreased the percentage of positive MTT cells in both 158N and HepG2 cells (Figure 5).
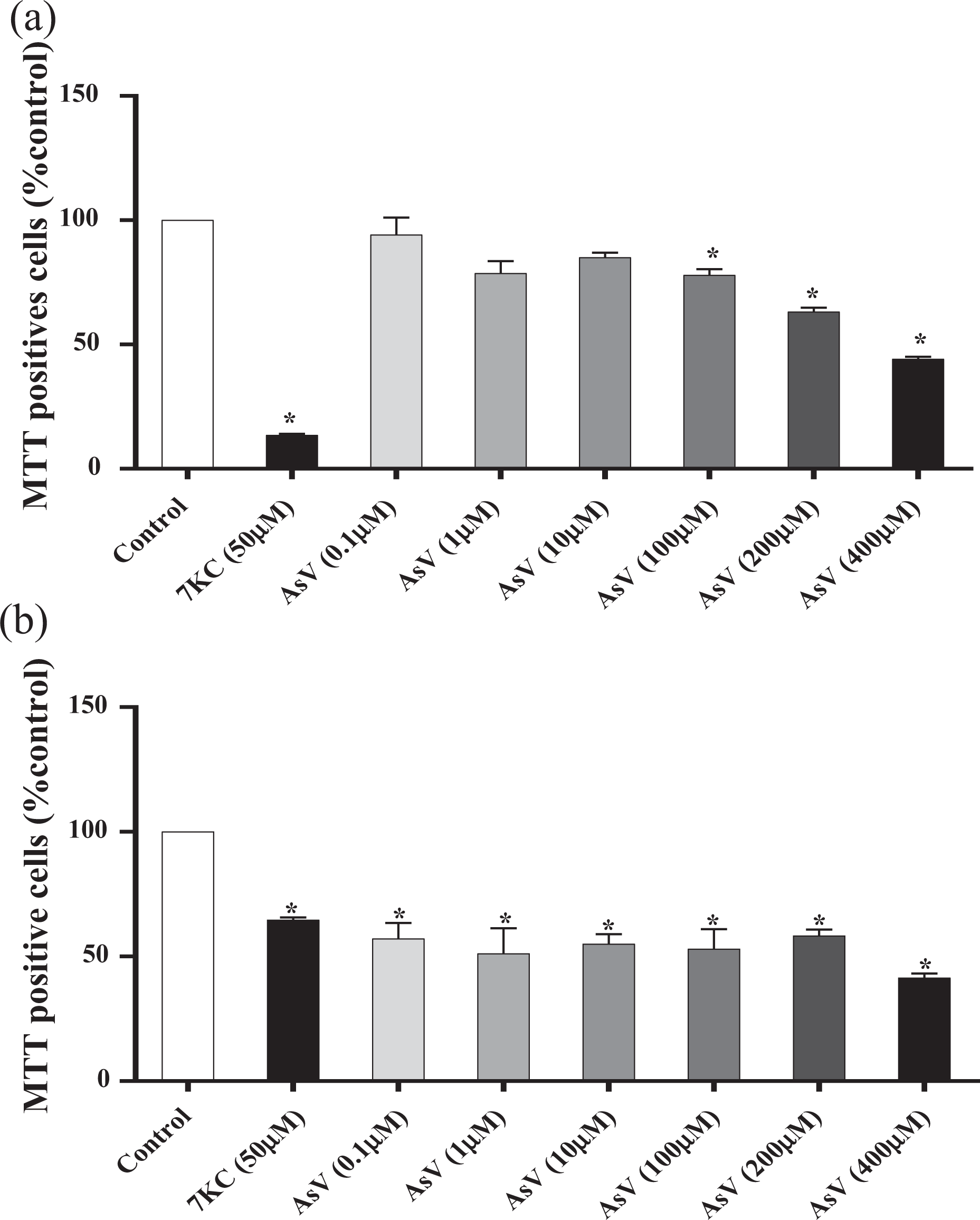
Effects of AsV on cell proliferation and mitochondrial metabolism. Effects of AsV on cell proliferation and/or mitochondrial metabolism were determined in (a) 158N cells and (b) HepG2 cells with the MTT assay. Data shown are mean ± standard deviations. Significant differences between control and treated cells (AsV and 7KC) are indicated by * (Mann–Whitney U test; *p ≤ 0.05). AsV: sodium arsenate; 7KC: 7-ketocholesterol.
Effects of AsV on ΔΨm with DiOC6(3)
In 158N cells treated at concentrations of 0.1, 1, and 10 µM compared with control cells, no significant difference was observed in the proportion of cells with depolarized mitochondria. However, at concentrations of 100, 200, and 400 μM, significant concentration-dependent increases (p ≤ 0.05) in the number of depolarized cells were observed (Figure 6(a)).

Evaluation of the effects of AsV on transmembrane mitochondrial potential. The effects of AsV on ΔΨm of (a) 158N cells and (b) HepG2 cells were determined by flow cytometry after staining with DiOC6(3). Data shown are mean ± standard deviation. Significant differences between control and treated cells (AsV and 7KC) are indicated by * (Mann–Whitney U test; *p < 0.05). AsV: sodium arsenate; 7KC: 7-ketocholesterol; ΔΨm: mitochondrial transmembrane potential; DiOC6(3): 3,3′-dihexyloxacarbocyanine iodide.
The effect of AsV on the ΔΨm of HepG2 cells varied slightly depending on the concentration. At concentrations of 0.1, 1, and 10 µM, there was a nonsignificant decrease in the proportion of cells with depolarized mitochondria, while at concentrations of 100 and 200 µM, the number of cells with depolarized mitochondria was slightly increased. This effect was much greater (p ≤ 0.05) at 400 μM (Figure 6(b)). In 158N and HepG2 cells, 7KC (50 µM) significantly increased the percentage of cells with depolarized mitochondria compared with control cells (Figure 6).
Effect of AsV on the level of mitochondrial ATP
In 158N cells, treatment with AsV for 48 h also had a biphasic effect on the mitochondrial ATP level. A slight increase in mitochondrial ATP levels was noted in 158N cells treated at 1 and 10 µM. With concentrations of 100, 200, and 400 μM, a significant decrease in ATP levels was observed (Figure 7(a)). HepG2 cells treated with 10, 100, 200, and 200 µM of AsV showed a significant decrease (p ≤ 0.05) in ATP levels, whereas no changes were detected at 0.1 and 1 µM (Figure 7(b)).

Effects of AsV on ATP level. The effect of AsV on ATP level was determined in (a) 158N cells and (b) HepG2 cells. Data shown are mean ± standard deviation. Significant differences between control and cells treated with AsV are indicated by * (Mann–Whitney U test; *p ≤ 0.05). AsV: sodium arsenate; ATP: adenosine triphosphate.
Effects of AsV on the expression of mitochondrial complex subunits
The effect of AsV on the expression of proteins belonging to the different mitochondrial complexes (complex I subunit NDUFB8 (20 kDa), complex II subunit (30 kDa), complex III subunit core 2 (47 kDa), complex IV subunit II (24 kDa), and complex V ATP synthase subunit alpha (53 kDa)) was evaluated using Western blotting, associated with quantitative densitometry analysis (Figure 8). Values of complex I–V subunits after treatment with low concentrations of AsV (1 and 10 µM) were similar to those of untreated cells (control). With AsV (0.1 µM), a slight decrease in the expression of all complex (I–V) was observed. However, concentrations of 100 and 200 µM significantly inhibited the activity of complexes III and V (Figure 8). Treatment with the highest concentration of AsV (400 µM) inhibited the expression of all complex subunits (I–V), especially that of complex I subunit, expression of which was totally suppressed (Figure 8).
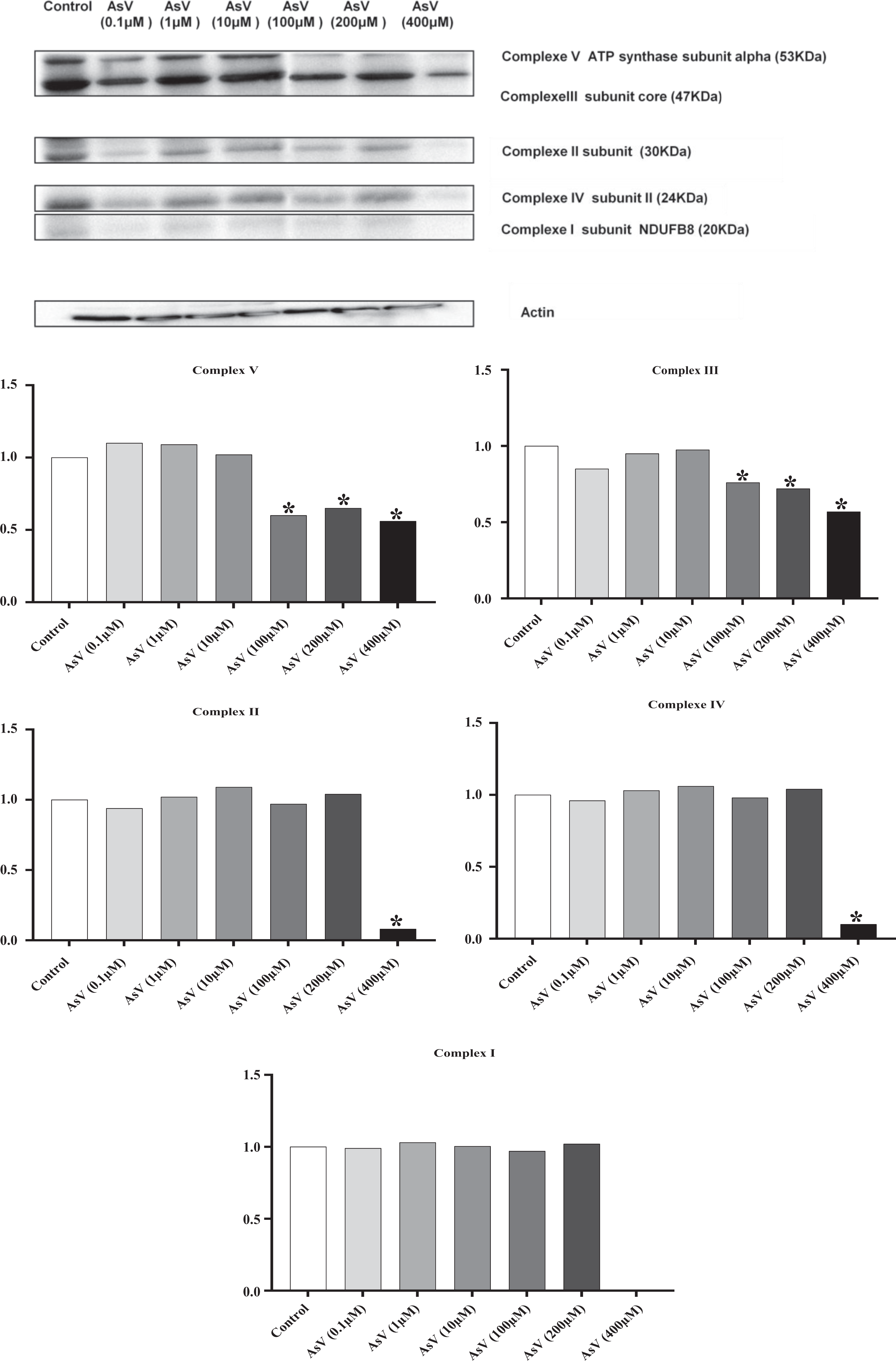
Analysis of the effects of AsV on the expression of mitochondrial complex subunits. (a) Western blots obtained with specific antibodies raised against subunits of the respiratory chain complex (MitoProfile Total OXPHOS Human WB Antibody Cocktail, Abcam/Mitosciences) and actin. (b) Quantification was carried out with ImageJ software; each complex was normalized versus actin; for each complex, data were expressed as (normalized value of the assay/normalized value of the control). Significance of the difference between control and treated cells is indicated by * (Mann–Whitney U test; *p ≤ 0.05). AsV: sodium arsenate.
Discussion
In the present study, the cytotoxic effects of AsV on oligodendrocytes (158N cells) and hepatoma cells (HepG2 cells) were analyzed. We observed that AsV induced different cytotoxic effects depending on the cell type considered.
Different concentrations of AsV (0.1–400 µM) had a biphasic effect, defined as hormesis, on 158N murine oligodendrocytes. Low concentrations 0.1, 1, and 10 µM induced an increase in cell proliferation revealed by phase-contrast microscopy, cell counting in the presence of trypan blue, and with crystal violet staining. The toxicity of AsV at 100, 200, and 400 µM was shown by a decrease in cell proliferation. These results are in agreement with our previous findings. 22 The stimulation of cell proliferation at low AsV concentrations (0.1–10 µM) could have therapeutic virtues by stimulating cell growth, or adverse side effects by stimulating carcinogenesis. In contrast, high concentrations (100–400 µM) induced a decrease in cell proliferation and cell death. Therefore, at whatever concentration, AsV can be regarded as having more adverse side effects than benefits and may thus be deleterious to human health.
Concerning HepG2 cells, AsV induced toxic effects at all of the concentrations considered (0.1–400 µM) in a concentration-dependent manner as shown by phase-contrast microscopy and cell counting with trypan blue. We previously demonstrated that AsV can lead to hepatotoxicity and a propensity toward liver cancer in rats, as confirmed by histological findings. 25 Many other studies have also reported that chronic exposure to AsV is associated with hepatomegaly, hepatoportal sclerosis, liver fibrosis, and cirrhosis with concomitant increases in serum bilirubin, alanine transaminase (ALT), aspartate transaminase (AST), and alkaline phosphatase (ALP). 26,27 The results of many other in vitro studies are consistent with ours. 28 –32
In addition, in 158N cells treated with high doses (100–400 µM) of AsV, the alteration of the cytoplasmic membrane also confirmed its cytotoxiciy: increased percentages of PI+ cells were detected. At low concentrations (0.1–10 µM), as in the control, only low percentages of PI+ cells were found. 22 The cytoplasmic membrane HepG2 cells were not altered by AsV whatever the concentration used. This observation led us to presume that AsV induces cell death by apoptosis, which was confirmed by the presence of a sub-G1 peak. 24 This finding is in agreement with previous in vitro studies, which showed that AsV induced apoptosis in a variety of cells 29,33,34 , especially in rat liver and in hepatocyte cultures. 28 In hepatocytes, proapoptotic proteins (Bad, Bax, and Bim) were upregulated, and antiapoptotic proteins (Bcl-2 and Bcl-xL) were downregulated. These proteins are involved in the control of caspase 3 and caspase 9 activation. In the liver, As also markedly increases Bax messenger ribonucleic Acid (mRNA) and reduces Bcl-2 mRNA. 28 It is widely accepted that apoptosis can be triggered by various pathways where mitochondria play key roles. 34 Moreover, in 158N cells, we noted an increase in the sub-G1 cell population, which probably corresponds to apoptotic cells. 22 These observations confirm that whatever the inorganic form of As considered and whatever the cell line used, apoptosis is the mode of cell death induced by this metalloid and probably by directly targeting the mitochondria as previously suggested by Sordet et al. 35
In a previous study conducted by Hosseini et al., 33 it was shown that the incubation of isolated liver mitochondria with arsenite led to lipid peroxidation and ATP depletion. In addition, a significant collapse of ΔΨm, mitochondrial swelling, and finally the disruption of mitochondrial outer membrane integrity occurred. It was therefore suggested that mitochondria might be the most important target of As-induced hepatotoxicity. 36 In addition, previous studies in isolated hepatocytes showed that mitochondria are involved in apoptosis and that AsV disturbs ΔΨm in hepatocytes. 33,36 –38 Many independent in vitro studies showed that As induces depolarization of the mitochondrial membrane in different cell lines. 28,29 In our study, whatever the concentration of AsV (0.1–200 µM), the ΔΨm of HepG2 cells was not affected. It is therefore supposed that in HepG2 cells, AsV does not alter complexes I, III, and IV of the mitochondrial respiratory chain, which are responsible for the proton gradient. On the other hand, AsV significantly reduced the function of complex II (succinate dehydrogenase activity) as suggested by the MTT test. 33 With complex II, under normal conditions, the free energy change is low because of the small difference in the reduction potential between electron donors and acceptors. Accordingly, complex II does not contribute to the expulsion of protons from the matrix to the inter membrane space. This could explain why inactivation of this complex does not induce mitochondrial membrane depolarization. The loss in ΔΨm after treatment with 400 µM of AsV can be explained by the fact that this concentration is so high that it disturbs all cellular mechanisms. It is therefore impossible to determine which compartment of the cell is affected first and implicated in the intoxication mechanism.
In contrast to HepG2, AsV concentrations that induced cytoxicity in 158N cells also induced mitochondrial depolarization in these cells. Several previous studies are in agreement with our results. 22,38,39 The effect of AsV on 158N with regard to the protein expression of the respiratory chain complexes, assessed by western blotting of the OX/PHOS complex, showed that high concentrations (100–200 µM) significantly decreased complexes V and III only while complex II remained intact. In contrast, a concentration of 400 µM reduced all complexes (I–V) and suppressed complex I subunit expression. However, the lowest concentration of AsV (0.1 µM), which induced an increase in cell proliferation, showed lower intensities on the bands corresponding to the OX/PHOS complex subunits as compared with controls. As it is known that oxidative phosphorylation is related to mitochondrial metabolic activity, it is suggested that low concentrations of AsV might also have an important negative impact on cell activity, especially on lipid metabolism. This could have dramatic consequences on brain and hepatic functions, as it is known that a tight regulation of lipid metabolism is required for the optimal activity of nerve cells and hepatocytes.
In our study, the decrease in the mitochondrial ATP level observed in 158N cells after 48 h of treatment with AsV (100, 200, and 400 μM) could also be the consequence of mitochondrial dysfunction. This suggests either an alteration in the expression or the activities of enzymes of the respiratory chain complexes involved in the synthesis of ATP and the maintenance of ΔΨm. 22 Indeed, one of the most important function of mitochondria is the generation of ATP by oxidative phosphorylation. 40 The depletion of mitochondrial ATP could result from inhibition of the mitochondrial respiratory chain and mitochondrial permeability transition (MPT) pore opening. 41 Opening of the MPT pore causes unlimited proton movement across the inner membrane resulting in the uncoupling of oxidative phosphorylation and a further reduction in ATP levels.
Besides, arsenate is a chemical analog of phosphate, which can disrupt at least some phosphate-dependent aspects of metabolism. It can be translocated across cellular membranes by phosphate transport proteins, leading to imbalances in phosphate supply. 42 We can therefore presume that AsV in HepG2 cells competes with phosphate during phosphorylation reactions at the level of complex V in ATP synthesis, leading to the formation of AsV-ADP (adenosine diphosphate). The formation of this complex AsV-ADP decreases the ability of cells to produce mitochondrial ATP and carry out normal metabolism. 42
Conclusion
Our data bring new evidence supporting the hypothesis that the characteristics of the cytotoxic effects of AsV depend on the nature of the cells considered. Thus, AsV induces a biphasic effect in 158N oligodendrocytes: low concentrations promote cell proliferation, while high concentrations induce cell death by altering mitochondrial activity and disrupting RedOx homeostasis. HepG2 cells were however susceptible to all AsV concentrations; in these cells, AsV impairs complex II activity. Although AsV induced different cytotoxic activities in 158N and HepG2 cells, the major side effects were observed at the mitochondrial level, thus supporting the hypothesis that the mitochondria are a major target of AsV-induced cytotoxicity.
Footnotes
Acknowledgement
The authors thank Mr Philip Bastable (Pôle Recherche—Délégation à la Recherche Clinique et Innovation—CHU Dijon, France) for English corrections.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the Université de Bourgogne (Dijon, France) and the Université de Monastir (Monastir, Tunisia).
