Abstract
The mitochondrial oxidative phosphorylation system was studied in liver and heart homogenates after treatment of rats with benznidazole. The drug was given by oral gavage to adult female Wistar rats for 9 consecutive days (100 mg benznidazole/kg body weight as a daily dose). The mitochondrial state 4 and state 3 respiration rates, respiratory control, efficiency of oxidative phosphorylation (ADP/O), and ATPsynthase activity were assayed. The results showed that according to all these parameters, the mitochondria in cardiac homogenates were not affected in the rats treated with benznidazole. By contrast, mitochondria in the liver homogenates of drug-treated rats were altered, showing decreased respiratory control and a lower coefficient of ADP/O as a result of an increase in the state 4 respiration rate. These data indicate the possibility of production of an uncoupling factor leading to increased proton leakage through the inner mitochondrial membrane as a result of a 9-day treatment of rats with benzonidazole. The obtained experimental data might at least partly explain the nature of benznidazole toxicity in the liver treated with benznidazole.
Introduction
Benznidazole is one of the drugs used for the treatment of Chagas’ disease, 1 a condition caused by Trypanosoma cruzi infection, which affects several million Latin Americans. 2 Side effects are a major drawback of this drug, 1,3 and the molecular/cellular mechanisms of benznidazole toxicity have not been well studied.
Mitochondria in aerobic living cells transform most of the cellular energy, supporting important physiological functions. For this reason, mitochondria play a crucial role in molecular/cellular mechanisms of many pathophysiological events, 4 including the toxicity of drugs and chemical agents.
Benznidazole is reduced, in vitro, by rat liver microsomes to a nitro anion radical and active oxygen radicals. 5,6 In addition, a triggering of nitroreductase activity has been observed, which could produce free radicals in heart microsomes of benznidazole-treated rats. 7 Enhanced oxidative stress in hepatocytes of benznidazole-treated rats has also been reported in the literature. 8 Since it is well known that isolated mitochondria are the targets, in vitro, of toxicity for free radicals and oxidative stress, 9 it has been considered to be important to evaluate whether benznidazole administration to rats could negatively impact the functional state of liver and heart mitochondria. In spite of the fact that mitochondria isolated from the heart and liver of benznidazole treated rats have been shown to retain normal bioenergetic parameters, 10 this does not exclude the possibility that mitochondrial functions in benznidazole-treated tissue homogenates might be altered through the presence of other cell components and/or benznidazole metabolic derivatives, which is not the case in isolated and washed mitochondria.
In the present article, we studied the effect of benznidazole administration to rats on the functional state of mitochondria in heart and liver homogenates, with the aim of evaluating the possibility that some cytosolic factors and/or drug derivatives might affect the mitochondrial oxidative phosphorylation system, as a possible mechanism of toxicity for benznidazole.
Methods
Benznidazole administration
Wistar female rats, weighing 150–160 g, were used in the experiments. During the study, they were provided with food and water ad libitum. Benznidazole (N-benzyl-2-nitroimidazoyl acetamide; Rochagan®, Roche) was prepared daily by trituration and suspension of the tablet in distilled water. The therapeutic scheme with benznidazole was according to that in which the drug was given by oral gavage to a group of 5 rats for 9 consecutive days (100 mg benznidazole/kg body weight (bw) as daily dose). 11 Another group of 5 animals (control group) received only distilled water by oral gavage for 9 consecutive days (the same daily volume of dosage as that for treated animals). Rats were killed 24 h after the last dose of the treatment regimen. All animals were cared for in accordance with the guide. 12
Preparation of heart and liver homogenates
In the current study, we used the biological material of the same control and treated laboratory animals that were used earlier in the experiments with isolated mitochondria. 10 All rats were subjected to an overnight fasting. Cardiac homogenate was an aliquot from the first step in the isolation of heart mitochondria prepared in accordance with Mela et al. 13 Briefly, rats were killed by cervical dislocation, and their hearts were rapidly excised, placed in cold sucrose (0.29 M) for 3 min, squeezed with a manual press (0.8 mm hole-diameter), and gently homogenized with 12 strokes using a Potter homogenizer in 3.0 ml homogenization medium/1 g of heart (homogenization medium: 225 mM mannitol, 75 mM sucrose, and 1 mM ethylene glycol tetraacetic acid (EGTA); pH 7.4); a 500 µL aliquot of this heart homogenate was used for the current study, while the remainder of the heart homogenate was used for isolation of heart mitochondria. Similarly, a 500 µL aliquot of liver homogenate (1 g of the liver in 2 ml of homogenization medium: 210 mM mannitol, 70 mM sucrose, 10 mM 2-[4-(2-hydroxyethyl)piperazin-1-yl]ethanesulfonic acid (HEPES), 1 mM EGTA, and 2.5 mM MgCl2; pH 7.2) from the first step in the isolation of liver mitochondria, prepared in accordance with Lemeshko, 14 was used to estimate the functional state of mitochondria in liver homogenates.
Assay of mitochondrial respiration and oxidative phosphorylation
Mitochondrial respiration was studied by measuring the rates of oxygen consumption using a Clark oxygen electrode, adding tissue homogenates at a final concentration of 1 mg protein/ml to a basal buffer (pH 7.2) containing (in mM) 250 sucrose, 5 KH2PO4, 10 HEPES, and 0.1 EGTA. The oxidative substrate was either 4 mM glutamate plus 1 mM malate or 5 mM succinate in the presence of 2.5 μM rotenone. The state 4 respiration rate of mitochondria (marked in the figures as state 4) corresponds to oxygen consumption in the sole presence of the oxidative substrate. The state 3 respiration rate of mitochondria (marked in the figures as state 3) corresponds to oxygen consumption in the presence of the oxidative substrate and ADP added at a final concentration of 300 µM. The respiratory control (RC) was calculated as the ratio of the state 3/state 4 respiration rates. The efficiency of oxidative phosphorylation (ADP/O) was the ratio of the quantity of added ADP (450 nmol ADP/1.5 ml) to the amount of oxygen consumed during the state 3 respiration. The mitochondrial ATPsynthase activity (marked in the figures as ATPs A) was estimated from the state 3 respiration rate and ADP/O parameters through the expression of mitochondrial ATPsynthase activity = (state 3 respiration rate) × (ADP/O). All experiments were carried out at 30°C. The protein concentration in the homogenates was determined using the biuret method. 15 All reagents were of analytical grade and were purchased from either Sigma or Merck official distributors in Colombia.
Statistical analysis
All values were means ± SEM of five independent experiments in duplicate, and their statistical significance (p < 0.05) was evaluated using Student’s t-test. Statistical analysis was performed using GraphPad PRISM version 2.0 software (San Diego, CA).
Results
Table 1 shows the mitochondrial oxidative phosphorylation parameters determined in heart homogenates with glutamate plus malate or with succinate as substrates of oxidation after a 9-day treatment of rats with benznidazole. There were no statistical differences between the drug-treated animals and the corresponding controls.
Mitochondrial oxidative phosphorylation parameters determined in heart homogenates after treating the rats with benznidazole for 9 days.
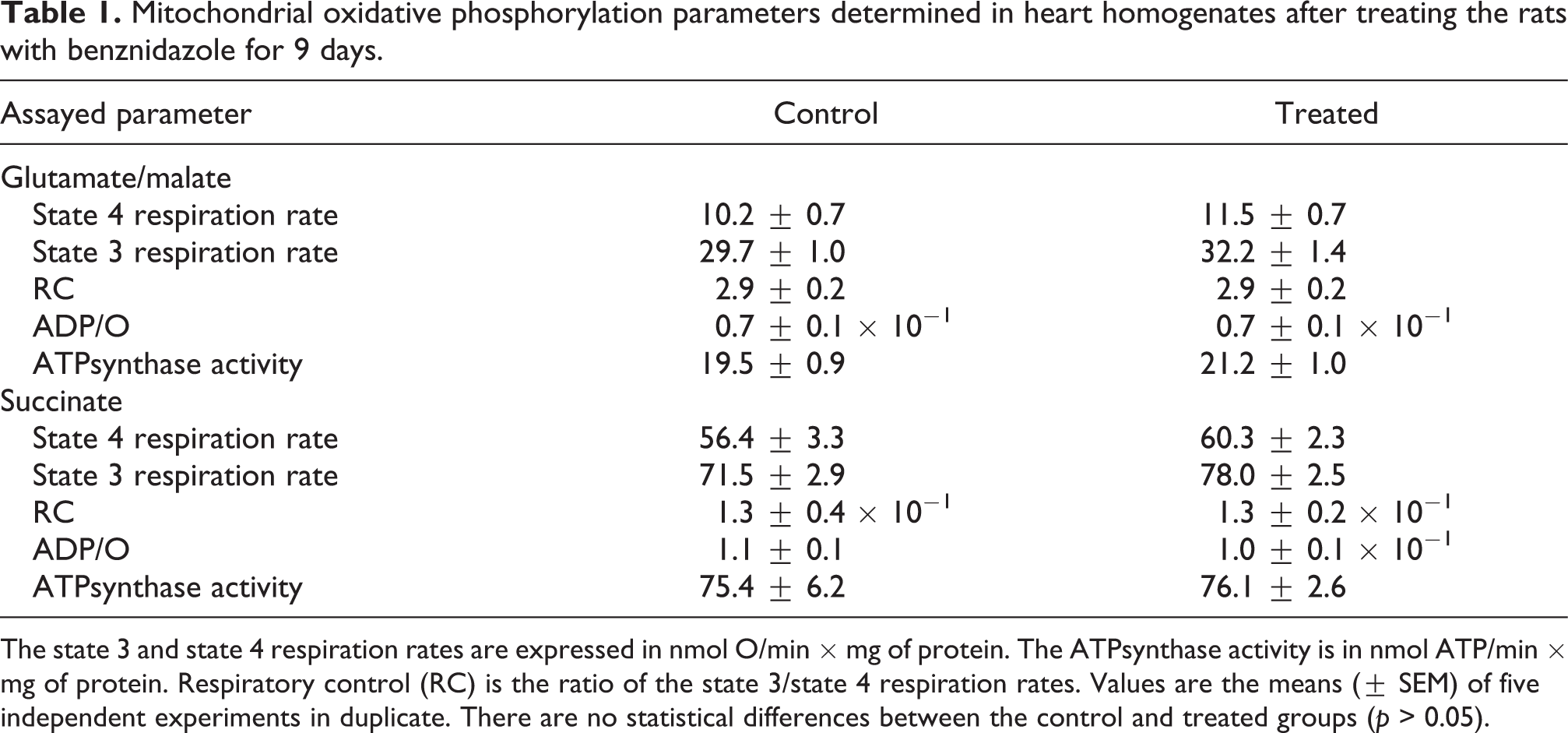
The state 3 and state 4 respiration rates are expressed in nmol O/min × mg of protein. The ATPsynthase activity is in nmol ATP/min × mg of protein. Respiratory control (RC) is the ratio of the state 3/state 4 respiration rates. Values are the means (± SEM) of five independent experiments in duplicate. There are no statistical differences between the control and treated groups (p > 0.05).
The data for mitochondria in liver homogenates, for the case of glutamate/malate as substrates of oxidation, are shown in Figure 1(a), (b), and (c). After the above-mentioned drug treatment regimen, a statistically significant increase in the state 4 respiration rate of 51.8% was determined (Figure 1(a)), while only a tendency of an increase of the state 3 respiration rate was observed (Figure 1(b)). As a result, the RC of mitochondria in liver-treated homogenates (Figure 1(c)) decreased by 16.7% with respect to the control, and this change was statistically significant.

The state 4 respiration rate ((a) and (d)), the state 3 respiration rate ((b) and (e)) and the respiratory control (RC) ((c) and (f)) of mitochondria in liver homogenates, with glutamate plus malate ((a), (b), and (c)) or succinate ((d), (e), and (f)) as substrates of oxidation, after 9-day treatment of rats with benznidazole. The state 4 respiration rate (state 4) and the state 3 respiration rate (state 3) are expressed in nmol O/min × mg of protein. RC is the ratio of the state 3/state 4 respiration rates. Control rats—white bars; treated rats—black bars. All data are means ± SEM of five independent experiments in duplicate. *Statistically significant difference (p < 0.05) in comparison to the control.
The results of the same study but with the substrate succinate are shown in Figure 1 ((d), (e), and (f)). After the drug treatment, the state 4 respiration rate (Figure 1(d)) increased by 37.8% (p < 0.05), without a statistically significant change in the state 3 respiration rate (Figure 1(e)), leading to a 6.5% decrease (p < 0.05) in RC (Figure 1(f)) with respect to the control.
Figure 2 shows the data for ADP/O and ATPsynthase activity assayed in liver homogenates using glutamate plus malate or succinate as a substrate of oxidation. The results revealed no difference in the ATPsynthase activity with glutamate plus malate (Figure 2(c)) or succinate (Figure 2(d)) between the benznidazole-treated rats and the control groups. In addition, ADP/O with glutamate plus malate (Figure 2(a)) or with succinate (Figure 2(b)) decreased by 8.2% (p < 0.05) and 8.3% (p < 0.05), respectively, with regard to the control.
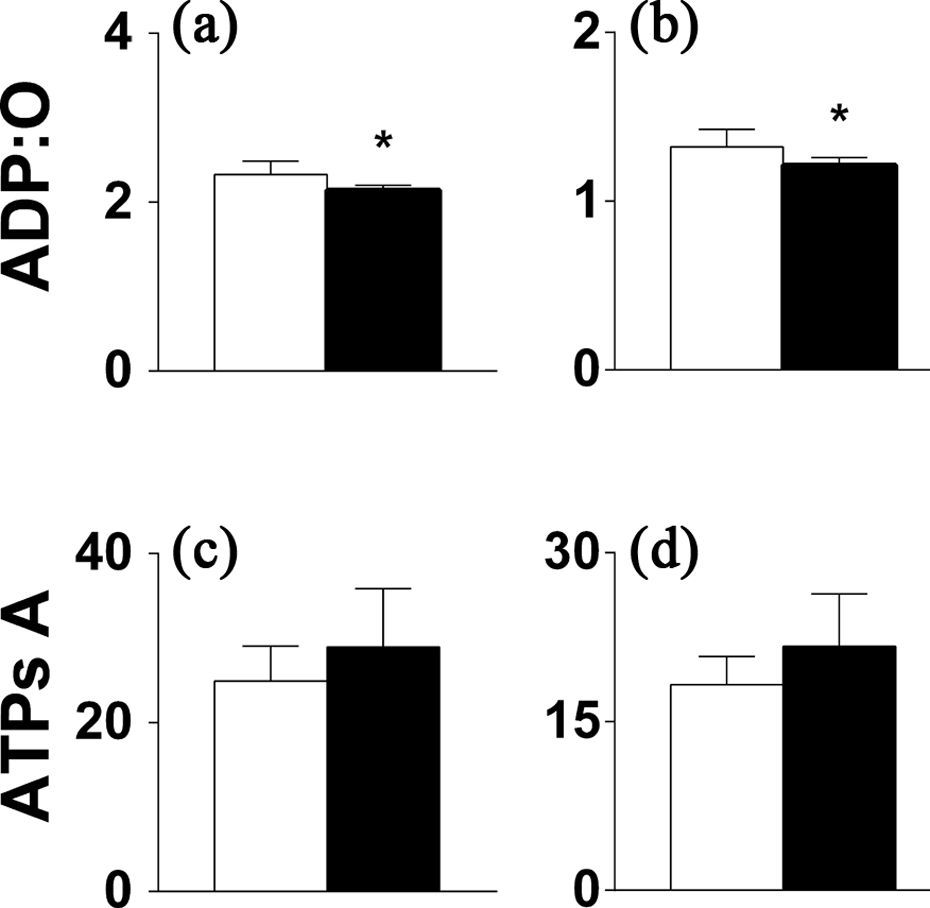
The efficiency of oxidative phosphorylation (ADP/O) ((a) and (b)) and the ATPsynthase activity ((c) and (d)) of mitochondria in liver homogenates, with glutamate plus malate ((a) and (c)) or succinate ((b) and (d)) as substrates of oxidation, after 9-day treatment of rats with benznidazole. The ATPsynthase activity (ATPs A) is expressed in nmol ATP/min × mg of protein. Control rats—white bars; treated rats—black bars. All data are means ± SEM of five independent experiments in duplicate. *Statistically significant difference (p < 0.05) in comparison to the control.
Discussion
Table 1 shows that no oxidative phosphorylation parameters of mitochondria measured in heart homogenates using glutamate plus malate or succinate as substrates of oxidation were altered after a 9-day treatment of rats with benznidazole. It is important to point out that in the author’s previous study, 10 in which the same control and benznidazole-treated laboratory animals were used, the isolated and washed heart mitochondria also were not altered through this benznidazole treatment. Taken together, all these results indicate that heart mitochondrial bioenergetics are not affected by the current benznidazole administration. There is good concordance between our results and the work of de Souza et al., 11 in which it was shown that this same 9-day benznidazole treatment regimen alone did not cause myocardium lesions, which are tested via the plasma level of cardiac creatine kinase isoenzymes.
It has been shown by de Mecca et al. 7 that the benznidazole concentration in the heart increased with time upon administration of benznidazole, reaching its maximum in the first few hours, with a subsequent significant decrease after 6 h of drug treatment. In that work, it was also reported that benznidazole did not lead to any visible structural alterations of mitochondria that were correlated with any benznidazole-induced cytosolic nitroreductive biotransformation and low intensity of benznidazole-induced microsomal nitroreductive biotransformation. Due to the fact that in the cited work rats received only a single intragastric (i.g.) dose of 100 mg /kg bw benznidazole, and in the present study the drug was applied daily for 9 days, benznidazole should be present in the heart at least 6 h daily under our experimental conditions. However, even in this last case, the drug was unable to impair the functional state of heart mitochondria, which might be explained by the very low rate of cytosolic nitroreductive biotransformation of benznidazole reported in the cited experiment.
In liver homogenates, a decrease in mitochondrial RC (Figure 1(c) and (f)) was observed with all substrates of oxidation used, as a result of a statistically significant increase in the state 4 respiration rates (Figure 1(a) and (d)) after treatment of rats with benznidazole. Nevertheless, the mitochondrial ATPsynthase activity was unchanged (Figure 2(c) and (d)), which seems to be due to a tendency of the state 3 respiration rate to increase (Figure 1(b) and (e)).
On the other hand, the decrease in RC (Figure 1(c) and (f)) resulting from the state 4 respiration increase (Figure 1(a) and (d)) indicates that mitochondria in liver homogenates were partially uncoupled, in contrast to the previously reported 10 unchanged functional state of isolated and washed liver mitochondria after an identical treatment of rats with benznidazole. All these facts indicate that when treated with benznidazole, the liver might generate some mitochondrial uncoupling factor(s) that are removed during the procedure of mitochondrial isolation. In this respect, it has been reported that benznidazole administration to rats induces oxidative stress in hepatocytes. 8
The results of the current study, together with the previously reported data, 10 indicate that the liver might be a target of potentially toxic effects of benznidazole. This suggestion supports previously reported clinical observations, in which this drug, given repeatedly, results in liver toxicity. 16
Footnotes
Acknowledgments
The author wishes to thank Dr. Armando Galeano, Coordinador de Vectores-Dirección Nacional de Salud de Antioquia, for his generous gift of the benznidazole-Rochagan® tables.
Funding
This work was supported by the financial contribution of COLCIENCIAS-Grant No 1118-04-16552 and by the National University of Colombia.
