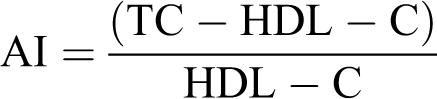Abstract
Several metals including barium (Ba) known as environmental pollutants provoke deleterious effects on human health. The present work pertains to the potential ability of selenium (Se) and/or vitamin C, used as nutritional supplements, to alleviate the toxic effects induced by barium chloride (BaCl2) in the heart of adult rats. Animals were randomly divided into seven groups of six each: group 1, serving as negative controls, received distilled water; group 2 received in their drinking water BaCl2 (67 ppm); group 3 received both Ba and Se (sodium selenite 0.5 mg kg−1 of diet); group 4 received both Ba and vitamin C (200 mg kg−1 bodyweight) via force feeding; group 5 received Ba, Se, and vitamin C; and groups 6 and 7, serving as positive controls, received either Se or vitamin C for 21 days. The exposure of rats to BaCl2 caused cardiotoxicity as monitored by an increase in malondialdehyde, hydrogen peroxide, and advanced oxidation protein product levels, a decrease in Na+-K+ adenosine triphosphatase (ATPase), Mg2+ ATPase, and acetylcholinesterase activities and in antioxidant defense system (catalase, glutathione peroxidase, superoxide dismutase, glutathione, and nonprotein thiols). Plasma lactate dehydrogenase and creatine kinase activities, total cholesterol, triglyceride, and low-density lipoprotein–cholesterol levels increased, while high-density lipoprotein–cholesterol level decreased. Coadministration of Se and/or vitamin C restored the parameters indicated above to near control values. The histopathological findings confirmed the biochemical results. Se and vitamin C may be a promising therapeutic strategy for Ba-induced heart injury.
Introduction
Contamination of drinking water with metals including barium (Ba) is an important public health problem throughout the world. This metal oxidizes readily and can react with water to emit hydrogen. Trace amounts of Ba were found in drinking water. 1 Although Ba has an important role in medical sciences, it can cause toxic effects and heart diseases. Several risk factors are associated with heart diseases like smoking, diabetes, and metal exposure. 2 After ingestion, this metal rapidly crosses the gastrointestinal mucosa, enters in the blood stream, and is distributed to soft tissues, particularly aorta, brain, kidney, spleen, pancreas, lung, and heart. 3 Moreover, McCauley and Washington 4 have found that the level of Ba in the heart was threefold higher than in other soft tissues or the whole blood after administration of an oral dose of barium chloride (BaCl2), suggesting its accumulation in this organ. Ba concentrations in drinking water are found in several countries to be ranged between 1 µg l−1 and 500 µg l−1. Daily drinking water intake of Ba (2 l) is ranged from 2 µg to 1200 µg. 5 It has been demonstrated in the previous studies that cardiovascular toxicity and diseases are associated with different doses of Ba in rats. 6 –9 Heart is a main target of mitochondrial and bioenergetic failure due to the low amount of antioxidant defenses and to high molecular oxygen content. So cardiomyocytes are easily susceptible to reactive oxygen species (ROS)-mediated oxidative tissue damage. The major sources of reactive oxygen species generation are mitochondria in the cardiac cells.
Metals may stimulate reactive oxygen species generation including superoxide anion (
To counteract oxidative stress, cells have developed antioxidant defense mechanisms including antioxidant enzymes such as catalase (CAT), superoxide dismutase (SOD), and glutathione peroxidase (GPx), 11 non enzymatic antioxidants like reduced glutathione (GSH), nonprotein thiols (NPSH), and metallothionein (MT). 12
When endogenous antioxidants fail to counteract oxidative stress, more attention has been paid to the protective effects of natural antioxidants against the harmful action of chemical-induced heart toxicities. 13
Antioxidant nutrients including dietary oligoelements and vitamins protect cells against deleterious effects of environmental xenobiotics. Selenium (Se) and vitamin C are used as the preventive elements against health disorders with oxidative stress considered as a factor in their pathophysiology. Se, a cofactor of the GPx, may prevent lipid peroxidation in mammals 14 leading to its contribution in biochemical and physiological processes like biosynthesis of coenzyme Q, controlling ion fluxes through the membrane and induction of antibodies synthesis. Vitamin C, an important component in the diet of animals and humans, 15 plays an important role against several diseases by inactivating the harmful free radicals affecting several tissues and causing their damages like brain, kidney, and heart. 16 It has been reported by Qiao et al. 17 that vitamin C enhances elastin and collagen production from aortic smooth muscle cells playing a crucial role to maintain normal arterial structure.
To our knowledge, findings concerning the effects of BaCl2 on cardiotoxicity in adult rats are scarce. Hence, the aim of the present work was to investigate the possible mechanism of cardiotoxicity induced by BaCl2 and to compare the antioxidant effects of individual and combined treatments with Se and vitamin C against cardiac toxicity.
Materials and methods
Chemicals and reagents
BaCl2 was purchased from Loba Chemie (Mumbai, India). Sodium selenite, nicotinamide adenine dinucleotide phosphate reduced form, 5,5′-dithio-bis-2-nitrobenzoic acid (DTNB) and thiobarbituric acid, GSH, nitro blue tetrazolium (NBT), and 1,1,3,3-tetraethoxypropane (TEP) were purchased from Sigma Chemical Co. (St Louis, Missouri, USA). The other ones of analytical grade were purchased from standard commercial suppliers.
Animal treatment and experimental approach
Forty-two adult rats of Wistar strain, weighing 168 ± 10 g, were used in the present study (Central Pharmacy, SIPHAT, Tunisia). They were kept in a room under temperature of 22 ± 3°C, an average relative humidity of 40%, and 12-h light/12-h dark cycle. Rats were provided daily with standard pellet diet (SNA, Sfax, Tunisia) and distilled water ad libitum. All experimental procedures were approved by the local committee of our institution and were consistent with the general guidelines for the care and use of laboratory animals. 18
Rats were randomly divided into seven groups of six each. Group 1(C): negative controls received distilled water. Group 2 (Ba): received, via drinking water, BaCl2 (67 ppm). Group 3 (Ba + Se): received both Ba (67 ppm) and Se (0.5 mg kg−1 of diet). Group 4 (Ba + vitamin C): treated via drinking water with Ba (67 ppm) and via force feeding with vitamin C (200 mg kg−1 bodyweight (bw)). Group 5 (Ba + Se + vitamin C): received both Ba (67 ppm), Se (0.5 mg kg−1 of diet) and vitamin C (200 mg kg−1 bw) by the same ways as groups 2, 3, and 4. Group 6 (Se): received Se (0.5 mg kg−1 of diet). Group 7 (vitamin C): received vitamin C (200 mg kg−1 bw) by force feeding.
The dose of BaCl2 represented 1/13 of LD50 according to Tardiff et al. 19 The dose of Se used in our study and in other findings gave high protection against stress conditions in several tissues. 20 Lower doses of Se gave less protection, while higher doses were not much effective. The daily dose of vitamin C (200 mg kg−1 bw per day) was chosen according to the previous studies. 21
At the end of the treatment period (21 days), all animals were killed by cervical dislocation to avoid stress. Trunk blood samples were drawn into heparinized tubes. After centrifugation at 2200 × g for 10 min, plasma samples were stored at −80°C until analysis. Hearts were collected and cleaned. Some samples were rinsed, homogenized in Tris hydrochloride (Tris-HCl) buffer (pH 7.4) and centrifuged. The resulting supernatants were stored at −80°C. Other samples were immediately removed, cleaned, fixed in 10% of buffered formalin solution and embedded in paraffin for histological studies.
Biochemical investigations
Protein quantification
Heart protein contents were measured according to the method of Lowry et al. 22 using bovine serum albumin as standard.
Estimation of lipid peroxidation
Lipid peroxidation in the cardiac tissue was estimated colorimetrically by measuring thiobarbituric acid reactive substances which were expressed in terms of malondialdehyde (MDA) content according to Draper and Hadley method. 23 The absorbance was measured at 532 nm. The MDA values were calculated using TEP as the standard and expressed as nanomoles of MDA per milligram of protein.
Determination of H2O2 levels
Heart H2O2 was carried out by the method of Ou and Wolff. 24 The amount of H2O2 in the heart was determined using spectrophotometer at 560 nm. Results were expressed as micromoles per milligram protein.
Determination of advanced oxidation protein product levels
Levels of advanced oxidation protein products (AOPP) were determined according to the method of Witko et al. 25 Absorbance of the reaction mixture was immediately recorded at 340 nm. The concentration of AOPP for each sample was calculated using the extinction coefficient of 261 cm−1 mM−1 and the results were expressed as nanomoles per milligram of protein.
Determination of antioxidant enzyme activities
CAT was assayed by the decomposition of H2O2 according to the method of Aebi. 26 A decrease in absorbance due to H2O2 degradation was monitored at 240 nm for 1 min and the enzyme activity was expressed as micromole of H2O2 consumed per minute per milligram of protein.
Total SOD activity was evaluated according to Beauchamp and Fridovich. 27 The developed blue color in the reaction was measured at 560 nm. Units of SOD activity were expressed as the amount of enzyme required to inhibit the reduction of NBT by 50% and the activity was expressed as units per milligram of protein.
GPx activity was measured according to Flohe and Günzler. 28 The enzyme activity was expressed as nanomoles of GSH oxidized per minute per milligram of protein.
Estimation of acetylcholinesterase activity
Acetylcholinesterase (AChE) activity was measured immediately in the heart tissue homogenates according to the method of Ellman et al., 29 using acetylthiocholine iodide as a substrate. The hydrolysis rate of acetylthiocholine iodide was measured at 412 nm. Activities were expressed as micromoles of substrate per minute per milligram of protein.
Estimation of cardiac Na+-K+ adenosine triphosphatase and Mg2+ adenosine triphosphatase
After homogenization in Tris-HCl buffer (pH 7.4), Na+-K+ adenosine triphosphatase (ATPase) and Mg2+ ATPase activities in the heart were determined according to the method of Kawamoto et al. 30 Total ATPase activity was determined by Pi assay released from hydrolyzed adenosine triphosphate (ATP) forming a complex with molybdate. ATPase activities were expressed as micromoles of Pi liberated per hour per milligram of protein.
Heart GSH levels
GSH contents were determined by Ellman’s method, 31 modified by Jollow et al. (1974) 32 based on the development of a yellow color when DTNB was added to compounds containing sulfhydryl groups. The absorbance was measured at 412 nm. Total GSH content was expressed as microgram per milligram of protein.
Heart NPSH levels
NPSH levels were determined by the method of Ellman. 31 The absorbance of colorimetric reaction was measured at 412 nm. NPSH content was expressed as nmoles per milligram of protein.
Heart MT content
MT in the heart was assayed according to the method of Viarengo et al. 33 modified by Petrovic et al. 34 The absorbance was evaluated at 412 nm. The data were expressed as micromoles of GSH per milligram of protein.
Heart biomarkers of toxicity in plasma
The activities of plasma lactate dehydrogenase (LDH) and creatine kinase (CK) were measured using the commercial diagnostic kits (Biomaghreb, Ariana, Tunisia; refs 20022 and 200063). Plasma lipid parameters such as total cholesterol (TC), triglycerides (TG), and high-density lipoprotein–cholesterol (HDL-C) levels were determined by enzymatic methods using commercial kits purchased from Biomaghreb (Tunisia; refs 20111, 20131, and 20113). The low-density lipoprotein–cholesterol (LDL-C) fraction and atherogenic index (AI) were determined according to the Friedewald equations 35
TC/HDL-C and LDL-C/HDL-C ratios were also calculated. They were considered as the pertinent indices of the incidence of cardiovascular risk. 36
Histopathological study
For the histopathological study, heart tissues were immediately fixed in buffered formalin solution (10%) and processed in a series of graded ethanol solutions, then embedded in paraffin, serially sectioned at 5 µm and stained with hematoxylin–eosin. Six slides were prepared from each heart.
Statistical analysis
The data were analyzed using the statistical package program Stat view version 5 Software for Windows (SAS Institute, Berkley, California, USA). Statistical analysis was performed using one-way analysis of variance followed by Fisher’s protected least significant difference test as a post hoc test for comparison between groups. Student unpaired t-test was also used when comparison between two groups was required. All values were expressed as means ± standard error. The 0.05 level was selected as the point of minimal statistical significance.
Results
Effects of Ba on general health and heart weight
There were no mortality during the experimental period and no significant difference in the body weight between negative and positive controls. While Ba-treated rats showed a slight reduction in their body weight (Table 1). Their absolute and relative heart weights increased significantly (p < 0.05) when compared to those of controls. Furthermore, when Se and/or vitamin C were administered to Ba-treated rats, there was a slight reduction in their heart weights as compared to that of Ba-treated rats.
Initial and final body weights, absolute and relative heart weights, daily food and water consumption in control and treated rats with BaCl2 alone or supplemented with Se and/or Vit C during 3 weeks.a

BaCl2: barium chloride; Se: selenium; Ba: barium; Vit C: vitamin C; SD: standard deviation.
aValues were expressed as means ± SD for six animals in each group.
b p < 0.05: Ba group versus control group.
c p < 0.05: (Ba + Se), (Ba + Vit C), (Ba + Se + Vit C) groups versus Ba group.
Estimation of lipid peroxidation and H2O2 levels
Our results revealed an increase of lipid peroxidation in the heart of Ba-treated group as evidenced by the enhanced MDA levels in the heart homogenates of adult rats (+87%) when compared to controls (Table 2). The supplementation of Se decreased MDA levels to reach control values, while vitamin C alleviated lipid peroxidation without reaching control values. After coadministration of both Se and vitamin C to Ba-treated rats, a total recovery of MDA levels occurred.
MDA, H2O2, AOPP, GSH, and NPSH levels in the heart of control and treated rats with BaCl2 alone or supplemented with Se and/or Vit C during 3 weeks.a

BaCl2: barium chloride; Ba: barium; Se: selenium, Vit C: vitamin C; MDA: malondialdehyde; H2O2: hydrogen peroxide; AOPP: advanced oxidation protein products; GSH: reduced glutathione; NPSH: nonprotein thiols; SD: standard deviation.
aValues were expressed as means ± SD for six animals in each group.
b p < 0.05: Ba, (Ba + Vit C) groups versus control group.
c p < 0.001: Ba, (Ba + Vit C) groups versus control group.
d p < 0.01: (Ba + Se), (Ba + Vit C), (Ba + Se + Vit C) groups versus Ba group.
e p < 0.001: (Ba + Se), (Ba + Vit C), (Ba + Se + Vit C) groups versus Ba group.
f p < 0.05: (Ba + Vit C) versus (Ba + Se).
Similarly, a remarkable rise of H2O2 levels was evident in the heart of Ba-treated group (111%), when compared to controls. When Se and/or vitamin C were supplied to Ba-treated rats, a significant reduction of H2O2 levels in the heart was observed (Table 2).
Heart AOPP levels
AOPP levels were significantly increased by 52% in the heart of Ba-treated rats when compared to controls. Se and/or vitamin C co-treatment to Ba-treated rats significantly decreased AOPP levels when compared to Ba-treated group. Se and vitamin C, administered to control rats, did not modify AOPP values when compared to negative controls (Table 2).
Heart antioxidant enzyme activities
Treatment with Ba led to a significant decrease in CAT, SOD, and GPx activities by 32%, 42%, and 23%, respectively, as compared to controls. In the group of rats treated with Ba and supplemented with Se and/or vitamin C, antioxidant enzyme activities showed a significant recovery as compared to Ba-treated group (Figure 1).
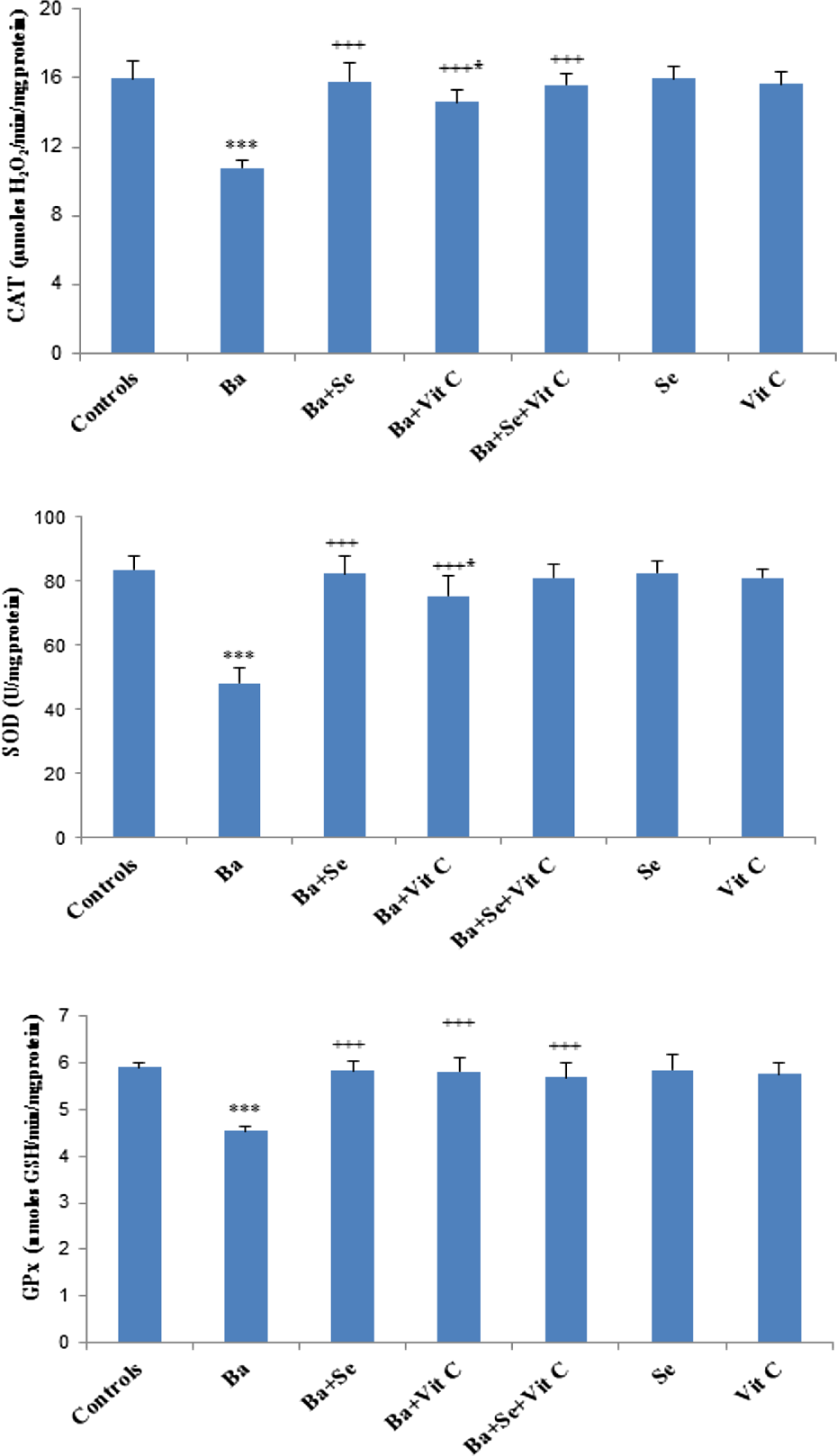
Enzymatic antioxidant activities (CAT, SOD, and GPx) in the heart of control and treated rats with BaCl2 alone or supplemented with Se and/or Vit C in their diet during 3 weeks. Values were expressed as means ± SD for six animals in each group. Ba, (Ba + Vit C) groups versus control group: *p < 0.05; ***p < 0.001. (Ba + Se), (Ba + Vit C), (Ba + Se + Vit C) groups versus Ba group: +++ p < 0.001. BaCl2: barium chloride; Se: selenium; Ba: barium; Vit C: vitamin C; SD: standard deviation; CAT: catalase; SOD: superoxide dismutase; GPx: glutathione peroxidase.
Heart AChE activity
Compared to controls, AChE activity in the heart of Ba-treated rats was reduced by 51%. When Se and/or vitamin C were supplied to Ba-treated rats, a significant increase was observed as compared to Ba-treated group (Figure 2).

AChE activity in the heart of control and treated rats with BaCl2 alone or supplemented with Se and/or Vit C during 3 weeks. Values were expressed as means ± SD for six animals in each group. *p < 0.05; ***p < 0.001: Ba, (Ba + Vit C) groups versus control group. +++ p < 0.001: (Ba + Se), (Ba + Vit C), (Ba + Se + Vit C) groups versus Ba group. $$ p < 0.01: (Ba + Se + Vit C) group versus (Ba + Vit C) group. AChE: acetylcholinesterase; BaCl2: barium chloride; Se: selenium; Ba: barium; Vit C: vitamin C; SD: standard deviation.
Effects of Ba on the Na+-K+ ATPase and Mg2+ ATPase activities
Na+-K+ ATPase activity was significantly decreased by 73% in the heart of rats treated with BaCl2 as compared to controls (Figure 3). When Se and/or vitamin C were given to Ba-treated group, Na+-K+ ATPase activity increased without reaching control values. Similarly, a significant impairment of Mg2+ ATPase activity (−44%) was observed in Ba-treated rats when compared to controls. Se and/or vitamin C coadministered to Ba-treated rats alleviated the activities of these enzymes. Heart Na+-K+ ATPase and Mg2+ ATPase activities in the positive control groups were similar to those of negative controls.

(a) Heart Na+-K+ and (b) Mg2+ ATPase activities of control and treated rats with BaCl2 alone or supplemented with Se and/or Vit C in their diet during 3 weeks. Values were expressed as means ± SD for six animals in each group. *p < 0.05; **p < 0.01; ***p < 0.001: Ba, (Ba + Se), (Ba + Vit C), (Ba + Se + Vit C) groups versus control group. +++ p < 0.001: (Ba + Se), (Ba + Vit C), (Ba + Se + Vit C) groups versus Ba group. BaCl2: barium chloride; Se: selenium; Ba: barium; Vit C: vitamin C; SD: standard deviation; ATPase: adenosine triphosphatase.
Heart nonenzymatic antioxidants
A significant decrease of GSH (−18%) and of NPSH levels (−46%) in the heart was evident in Ba-treated rats when compared to controls. Supplementation of Se alone or combined with vitamin C showed a significant recovery as compared to Ba-treated rats (Table 2).
MT levels in the heart
There was a 46% increase of MT levels in the heart of Ba-treated rats when compared to controls (Figure 4). Nevertheless, co-treatment with Se and/or vitamin C decreased MT levels to reach control values. Treatment of controls with Se and/or vitamin C alone did not cause any significant changes in MT levels when compared to negative controls.
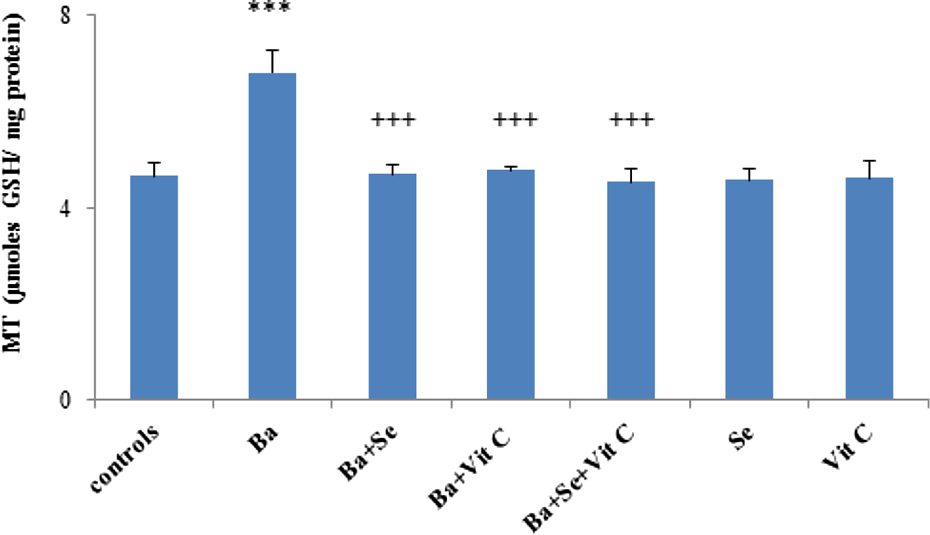
MT levels in the heart of control and treated rats with BaCl2 alone or supplemented with Se and/or Vit C in their diet during 3 weeks. Values were expressed as means ± SD for six animals in each group. ***p < 0.001: Ba group versus control group. +++ p < 0.001: (Ba + Se), (Ba + Vit C), (Ba + Se + Vit C) groups versus Ba group. MT: metallothionein; BaCl2: barium chloride; Se: selenium; Ba: barium; Vit C: vitamin C; SD: standard deviation.
Effects of Ba on LDH and CK activities
Following Ba treatment, plasma LDH and CK activities increased significantly by 55% and 50%, respectively, as compared to controls indicating the occurrence of heart injury (Figure 5). Nevertheless, a significant reduction of LDH and CK activities was observed after Se and/or vitamin C administration as compared to Ba-treated group.

LDH and CK activities in the plasma of control and treated rats with BaCl2 alone or supplemented with Se and/or Vit C during 3 weeks. Values were expressed as means ± SD for six animals in each group. *p < 0.05; **p < 0.01; ***p
Lipid profile in plasma
Plasma levels of TG and TC increased by 126% and 89%, respectively, in Ba-treated rats when compared to controls. LDL-C levels were increased by 89%, while those of HDL-C were decreased by 19%. A threefold increase in AI was occurred in Ba-treated rats. TC/HDL-C and LDL-C/HDL-C ratios were increased by 105% and 128%, respectively, when compared to controls. Upon coadministration of Se and/or vitamin C to Ba-treated rats, all the parameters indicated above were corrected as compared to those of Ba-treated group. Treatment of rats with Se or vitamin C alone did not cause any significant changes in lipid profile of rats (Table 3).
Lipid profile in plasma of control and treated rats with BaCl2 alone or supplemented with Se and/or Vit C during 3 weeks.a
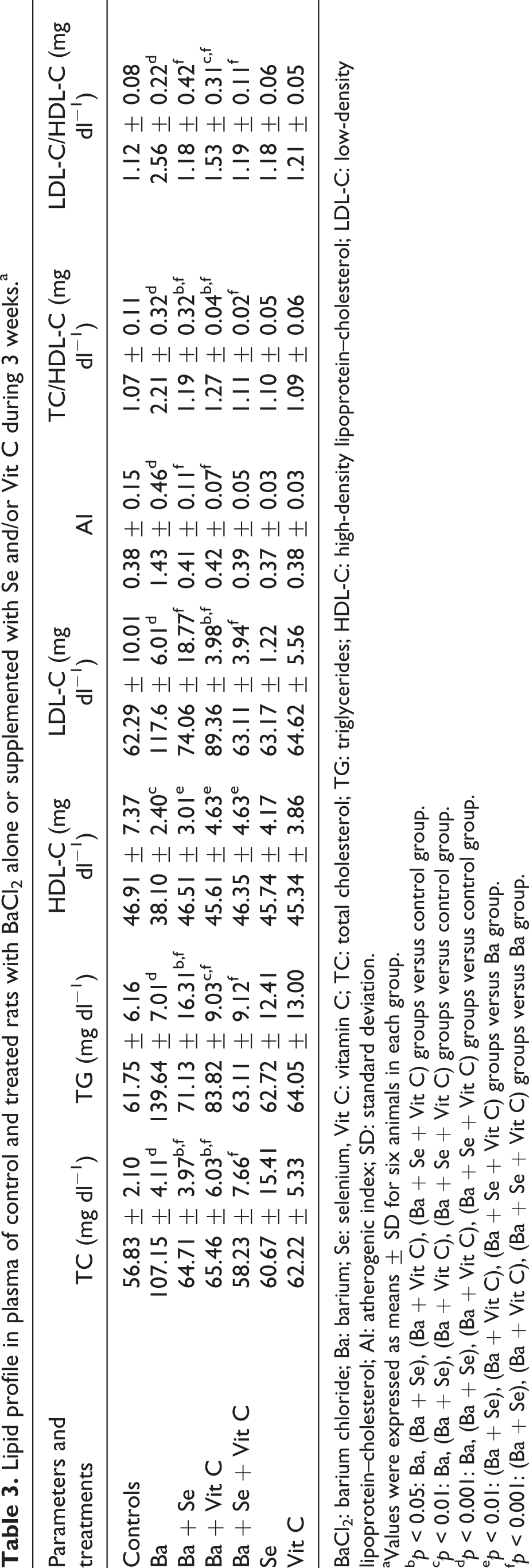
BaCl2: barium chloride; Ba: barium; Se: selenium, Vit C: vitamin C; TC: total cholesterol; TG: triglycerides; HDL-C: high-density lipoprotein–cholesterol; LDL-C: low-density lipoprotein–cholesterol; AI: atherogenic index; SD: standard deviation.
aValues were expressed as means ± SD for six animals in each group.
b p < 0.05: Ba, (Ba + Se), (Ba + Vit C), (Ba + Se + Vit C) groups versus control group.
c p < 0.01: Ba, (Ba + Se), (Ba + Vit C), (Ba + Se + Vit C) groups versus control group.
d p < 0.001: Ba, (Ba + Se), (Ba + Vit C), (Ba + Se + Vit C) groups versus control group.
e p < 0.01: (Ba + Se), (Ba + Vit C), (Ba + Se + Vit C) groups versus Ba group.
f p < 0.001: (Ba + Se), (Ba + Vit C), (Ba + Se + Vit C) groups versus Ba group.
Heart histological evaluation
In the heart of control group, there was a normal histoarchitecture (Figure 6(a)). While in Ba-treated group, there were a disruption and a loss of cardiac myofibers integrity, a marked inflammatory cells infiltration and edema (Figure 6(b) and (c)). Regeneration of cardiac myofibers was more pronounced following Se administration (Figure 6(d)) than that of vitamin C (Figure 6(e)). The Ba-treated group supplemented with both Se and vitamin C revealed a significant recovery and showed the normal histoarchitecture of cardiac muscle (Figure 6(f)). Se and vitamin C administered alone to control rats didn’t show any histological alterations in the myocardium (Figure 6(g) and (h)), respectively.

(a) Heart histological sections of control and (b and c) treated rats with BaCl2 alone, (d) Ba + Se, (e) Ba + Vit C, (f) Ba + Se + Vit C, (g) Se, and (h) Vit C showing the histopathological changes. Hematoxylin–eosin ×400. Arrows indicate:
 : leucocytes infiltration;
: leucocytes infiltration;  : edema;
: edema;  : increased interstitial space and distortion of the intercalated disc. BaCl2: barium chloride; Se: selenium; Ba: barium; Vit C: vitamin C.
: increased interstitial space and distortion of the intercalated disc. BaCl2: barium chloride; Se: selenium; Ba: barium; Vit C: vitamin C.
Discussion
Epidemiological evidences indicate that metal-induced cardiovascular toxicity may develop many disorders like myocardial dysfunction, ischemia, hypertension, QT-interval prolongation, arrhythmias, and thromboembolism. The mechanisms by which BaCl2 causes the deleterious effects have not been yet determined. The aim of the present study was to compare the antioxidant effects of individual and combined treatments with Se and vitamin C against cardiac oxidative stress in Ba-treated rats.
Exposure to metals is hazardous to physical variables based on body and organ weights. 37 Rats exposed to BaCl2 showed a slight reduction in their body weight and didn’t show modifications in food intake, while an increased absolute and relative heart weights was observed. This might be due to lipids accumulation in the heart and their increase in plasma as well as a failure of antioxidant system as reported earlier by Upadhyay 38 in patients with cardiovascular diseases. Coadministration of Se and vitamin C to the Ba-treated rats significantly improved the heart weight.
Circulating lipids and lipoproteins play a crucial role in the pathogenesis of myocardial disorders. Cardiotoxicity of Ba is associated with an increased level of circulating lipids. In fact, hypercholesterolemia and hypertriglyceridemia found in the present study were the major risk factors for the development of myocardial dysfunction which can be attributed to a decrease of cytochrome P450 enzyme activity and may also be due to an inhibition of lipase enzyme activity and plasma lipoproteins. In fact, HDL and LDL cholesterols are the important variables for coronary heart disease. In the present study, plasma levels of HDL-C decreased and those of LDL-C increased after BaCl2 treatment. Se alone or associated with vitamin C improved the levels of these parameters and helped the transport of cholesterol from peripheral tissues to the liver for catabolism and its excretion from the body.
ROS generation is a major stimulus of cellular dysfunction which increases serum marker enzyme activities in cardiovascular diseases. LDH and CK are considered as the reliable markers of cardiac damage. Thereby, an increase of their activities is a consequence of their release from the impairment membrane cells and as a result in metabolic changes of heart failure. 39 In our study, the increase of LDH and CK activities could be explained by a leakage of these enzymes from the heart to the blood indicating the myocardial membrane damage induced by Ba toxicity. Se and vitamin C prevented the leakage of LDH and CK from the heart to the extracellular compartment. This could be explained by their powerful antioxidant potential ensuring the stabilization of membrane and the reduction of cardiac tissue damage.
Furthermore, AChE activity is used as a heart biomarker of xenobiotics toxicity. The reduced AChE activity in our study might probably due to BaCl2 effects on the synaptic transmission in order to alter the release of acetylcholine, a neurotransmitter, and consequently the contraction of heart muscle. Moreover, lipid peroxidation in the cardiac tissue plays a critical role in myocardial membrane damage causing an accumulation of lipoperoxides. In fact, in the present study, Ba-intoxicated rats increased the levels of MDA and those of H2O2 causing an impairment of the cardiac membrane integrity. MDA, the breakdown product of the major chain reactions leading to polyunsaturated fatty acids oxidation, is known as a reliable marker of oxidative stress–mediated LPO in myocardial tissue. Moreover, Na+-K+ ATPase, a membrane-bound enzyme responsible to maintain Na+ and K+ gradients through cell membranes, is inhibited by ROS in soft tissues as reported by Reyes et al. 40 Free radicals generation leading to oxidative stress induced by BaCl2 provoked functional changes such as ATP depletion. In our study, the significant inhibition in the activity of heart Na+-K+ ATPase was a result of membrane depolarization allowing the influx of Na+ into the cell since Na+-K+ ATPase acted to maintain the ionic gradient for cardiac cell excitability. On the other hand, Na+-K+ ATPase activity depletion could be due to the decreased concentration of its substrate (ATP) and the increase of membrane lipid peroxidation resulting in the alteration of membranes by free radicals.
Furthermore, the increased levels of AOPP, shown in Ba-intoxicated heart tissue, were probably ascribed to free radical-mediated protein and membrane damages. A significant improvement of this parameter was found after co-treatment of Ba-treated rats with Se associated or not with vitamin C.
Antioxidant defense system is known to inhibit lipid peroxidation in mammalian tissues through reactive oxygen system disruption which plays a crucial role to initiate lipid peroxidation along with cellular damage. Enzymatic and nonenzymatic components scavenge and eliminate reactive oxygen species (ROS). In the current study, the homeostasis of antioxidant defenses was altered, as shown by a significant decrease in SOD, CAT, and GPx activities. The impairment of the enzymatic antioxidant defense system was considered as a critical event in BaCl2-induced cardiotoxicity. Supplementation of Se and/or vitamin C to Ba-treated rats prevented the failure of the antioxidant system. The combination of vitamin C and Se exhibited a synergistic effect in protecting the heart against oxidative damage.
Redox unbalance was also characterized by the depletion of nonenzymatic antioxidants including GSH and NPSH. GSH is a predominant endogenous antioxidant and is used as a substrate of GPx to remove H2O2 and lipoperoxides, then GSH is converted into Glutathione disulfide (GSSG), its oxidized form. Optimum level of GSH is an important criteria to maintain the structural integrity and physiology of cell membranes. Our results indicated that Ba treatment could facilitate free radicals production to disrupt heart function. Supplementation of Se in the diet of Ba-treated rats could protect the heart, indicating that it might play an important role in the metabolism of GSH which in turn increased the concentration of thiol groups and thus increased intracellular antioxidant power. Nevertheless, coadministration of both Se and vitamin C to Ba-treated rats was more powerful than Se or vitamin C used separately against oxidative stress. Their synergistic effects were also proven in our study by increasing the levels of nonenzymatic antioxidants, decreasing lipid peroxidation, quenching free radicals, and maintaining activities of GPx, CAT, and SOD.
In addition to the endogenous antioxidant system, MT mainly acts as regulator of metal homeostasis in tissues especially trace element metals and also acts as the potent antioxidant to protect cells from oxidative damage. 41 The possible mechanism of MT protection against oxidative injury is due to the cysteine residues which are the primary target for the reaction of oxygen free radicals with MT. 42 In our study, the impairment of heart function was accompanied by an increase of MT content after Ba exposure. According to Coyle et al, 43 the increase of MT level protects cells from the toxic insults.
Our results revealed that co-treatment with Se and/or vitamin C decreased MT levels to reach control values. The histological studies supported our biochemical data concerning the cardioprotective effects of Se and/or vitamin C against toxicity induced by BaCl2.
Histoarchitecture of the heart was altered in Ba-treated rats such as a disruption and a loss of cardiac myofibers integrity and a marked inflammatory cells infiltration. After co-treatment of Ba-treated rats with Se associated or not with vitamin C, the histological aspect of myocardiocytes was improved. Thus, the protection of the myocardium against Ba-induced heart damage was mainly due to the integrity of membrane and its stabilization by antilipoperoxidative properties of Se and vitamin C.
Conclusion
To our knowledge, the present study constitutes the first attempt to evaluate the effects of Se combined or not with vitamin C on Ba-caused cardiotoxicity in adult rats. Our results indicated that Ba caused heart damage, with a disruption of redox status, a cholinergic dysfunction, and an impairment of Na+-K+ ATPase and Mg2+ ATPase activities. A diet enriched with Se exhibited a more pronounced hypolipidemic effect than that of vitamin C. However, a marked enhancement of the antioxidant defense system was found in Ba-treated rats supplemented with both Se and vitamin C. The mechanisms which contribute to their effectiveness involve the quenching of free radicals and the antioxidant status improvement.
Footnotes
Author Contribution
Imen Ghorbel and Mariem Chaabane both the authors contributed equally to this work. The authors are indebted to Mrs Raoudha Ben Amar Abdennadher and Mr Chedli Tmar for their skillful technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministry of Higher Education and Scientific Research, Tunisia (DGRST grants, UR11/ES-70, Sciences Faculty of Sfax).