Abstract
The aim of this study was to investigate the effects of vitamin C, vitamin E and selenium (Se) on the skin tissue of streptozotocin-induced diabetic rats. Swiss albino rats were divided into four groups: control, control + antioxidants, diabetic, diabetic + antioxidants groups. Diabetes was induced by intraperitoneal injection of 65 mg/kg streptozotocin. Vitamin C (250 mg/kg), vitamin E (250 mg/kg) and Se (0.2 mg/kg) were given by gavage technique to rats of one diabetic and one control group for 30 days. In the diabetic group, the levels of serum urea and creatinine, skin lipid peroxidation and nonenzymatic glycosylation levels increased, but skin glutathione levels decreased. Treatment with vitamin C, vitamin E and Se reversed these effects. The present study showed that vitamin C, vitamin E and Se exerted antioxidant effects and consequently may prevent skin damage caused by streptozotocin-induced diabetes.
Introduction
Diabetes mellitus is a group of metabolic alterations characterized by hyperglycemia due to defects in insulin secretion, insulin action or both. 1 Diabetes is associated with the generation of reactive oxygen species (ROS), causing oxidative damage particularly to various tissues. 2 Oxidative stress in diabetes leads to tissue damage, with lipid peroxidation (LPO), inactivation of protein and protein glycation as intermediate mechanisms for its complications including retinopathy, nephropathy and coronary heart disease. 3 Diabetic patients and experimental hyperglycemic animals are at the risk of oxidative stress; therefore, this situation may increase the requirement of antioxidants such as vitamins. 4,5 Damaged tissues may have altered vitamin levels and antioxidant requirements. A variety of natural antioxidants exist to scavenge oxygen-free radicals and prevent oxidative damage to biological membranes. One group of these antioxidants is enzymatic, which include glutathione peroxidase, catalase and superoxide dismutase. 6 In addition to enzymatic antioxidants, there are nonenzymatic antioxidants such as vitamin A, vitamin C (VC), vitamin E (VE) and carotenoids derived from natural sources. 7 Some of the studies have reported the existence of VC and VE deficiency in diabetic patients 8 and experimental animals. 9 The treatment with VC and VE has been found to be beneficial in preventing or reducing some complications in insulin-dependent diabetes mellitus such as cataract and nephropathy. 10 –12 The VC and VE supplementation can decrease or inhibits LPO and overcomes abnormalities in the endogenous antioxidant defense system in diabetic patients 13 and animals. 14 The other organs, such as skin, is affected from the metabolic disturbances caused by diabetes mellitus. In diabetes, increased nonenzymatic glycosylation (NEG) of skin proteins and LPO damage in skin integrity caused a decreased resistance against harmful substances. 15 –17 VC is known as a potential scavenger of ROS, and it may protect the lipids from detectable peroxidative damage. 18 VE is the primary liposoluble antioxidant, which may have an important role in scavenging free oxygen radicals, and it stabilizes the cell membranes, thus maintaining its permeability. 19 Selenium (Se) is an essential trace element, and its antioxidant effect is known to be associated with its presence in glutathione peroxidases, which protects DNA and other cellular components. 20 VC or/and VE and Se exert a synergistic effect in the prevention of biological membranes from oxidants. 21 It is likely that VC and VE act in a synergistic manner, through which VE is primarily being oxidized to the tocopheroxyl radical and then reduced back to tocopherol by VC and glutathione (GSH). 22
No studies on VC, VE and Se on skin have been found in literature. The aim of this study was to investigate the effects of VC, VE and Se in the skin tissues of normal and streptozotocin-induced diabetic rats.
Materials and methods
Animals
Female, 2–2.5 months old, clinically healthy Swiss albino rats were used. The experiments were reviewed and approved by the Institute of Animal Care and Use Committees of Istanbul University. They were maintained in standard environmental conditions and fed with laboratory pellet chow and given water ad libitum. The rats were randomly divided into four groups.
Experimental design
Group 1: control animals (
Group 2: control animals given the VC, VE and Se (
Group 3: streptozotocin (STZ)-induced diabetic animals (
Group 4: STZ-induced diabetic rats given VC, VE and Se (
Experimental animals were rendered diabetic by an intraperitoneal injection of a single dose of STZ (65 mg/kg) dissolved in a freshly prepared 0.01 M citrate buffer (pH: 4.5). 23 VC (250 mg/kg), VE (250 mg/kg) and Se (0.2 mg/kg) were given by gavage technique to the rats of one STZ-diabetic and one control group for 30 days. The animals were fasted overnight prior to experiment, but they were allowed free access to water. On day 30, the skin tissues were taken under ether anesthesia.
Biochemical assays
In this study, biochemical investigations were made in blood and skin tissues from all groups. The blood samples of rats were collected from the tail vein on days 0 and 30. At the end of the experimental period, the animals were fasted overnight and then killed. For biochemical analyses, the tissues samples of skin were washed with saline and kept frozen until the day of experiment. These tissue samples were homogenized in cold 0.9% NaCl with a glass homogenizer to make-up 10% homogenate (w/v). The homogenates were centrifuged. The supernatant fractions were removed for the determinations of the levels of GSH, LPO, NEG and protein.
Fasting blood glucose levels (after 18 h period to fasting) were determined by
Statistical analysis
Biochemical results were evaluated using an unpaired
Biochemical results
Table 1 shows the levels of blood glucose in control and experimental groups of rats. Prior to induction of diabetes, the blood glucose levels of all the groups were similar (
Mean levels of blood glucose for all the groups (mg/dl).a

VC: vitamin C; VE: vitamin E; Se: selenium.
aValues are given as Mean ± SD.
b
c
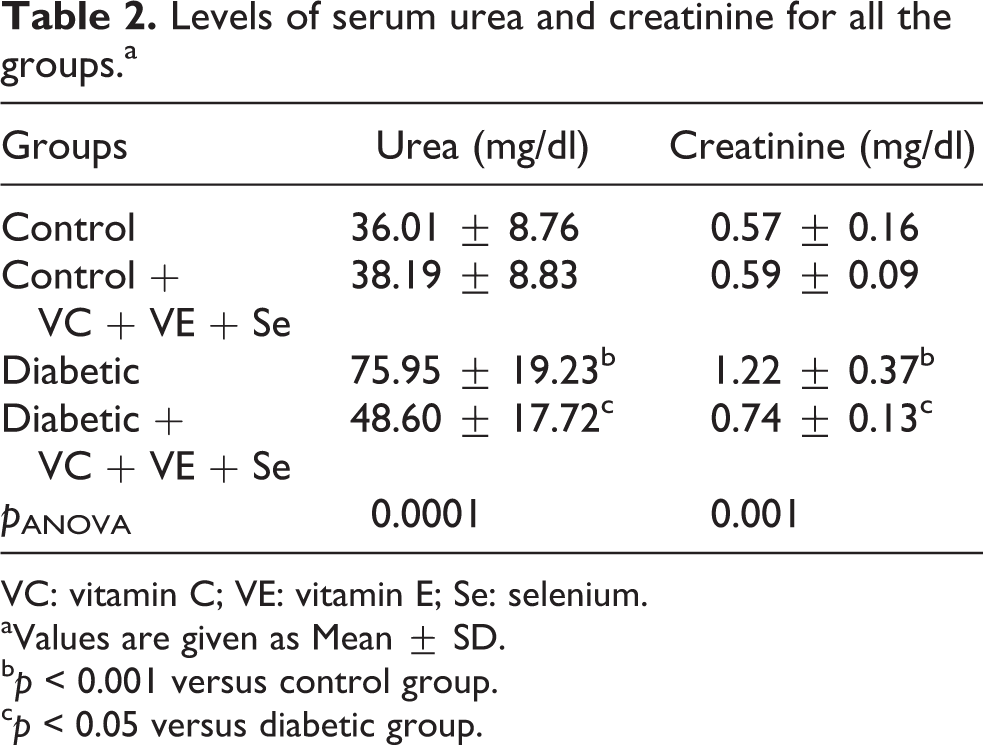
Table 2 shows the levels of serum urea and creatinine in control and experimental groups of rats. Serum urea levels were significantly increased in diabetic animals when compared with control group (
Levels of serum urea and creatinine for all the groups.a
VC: vitamin C; VE: vitamin E; Se: selenium.
aValues are given as Mean ± SD.
b
c
Table 3 shows the effects of antioxidants on skin GSH, LPO and NEG. The skin GSH levels were significantly reduced in the diabetic rats when compared with the other groups (
Levels of GSH, LPO and NEG in the skin tissues of all the groups.a

GSH: glutathione; LPO: lipid peroxidation; MDA: malondialdehyde; NEG: nonenzymatic glycosylation; VC: vitamin C; VE: vitamin E; Se: selenium.
aValues are given as Mean ± SD.
b
c
d
e
Discussion
Hyperglycemia is an important factor in the development and progression of the complications of diabetes mellitus, and good glycemic control is necessary to prevent diabetic complications. 31 In our study, the increased blood glucose concentration during the experimental period clearly indicated the persistent hyperglycemia in the STZ-induced diabetic rats. Administration of VC, VE and Se lowered the blood glucose concentration in the diabetic rats.
Elevated levels of urea and creatinine in serum are presumptive markers of diabetes. 32 In this study, the levels of serum urea and creatinine were reduced in experimental diabetic rats by the administration of VC, VE and Se. Our results are in accordance with the reports by others who used chemical antioxidant. 32
Extensive evidence has shown that chronic hyperglycemia leads to a series of biochemical events resulting in the production of high levels of ROS and eventual oxidative stress. 33 Diabetogenic effect of STZ is mainly attributed to the excess production of ROS leading to the toxicity in pancreatic cells, which reduces the synthesis and release of insulin, while affecting organs such as liver. 34 Oxidative stress in diabetes coexists with a reduction in the antioxidant capacity, which can increase the deleterious effects of free radicals. 35
GSH is an intracellular antioxidant that has a thiol group with several biological functions, including regulating cellular proliferation, transporting amino acids, synthesizing proteins and DNA, reducing disulfides and chemical groups and protecting the cell against oxidation. 36 Diabetes impairs the function of the cells antioxidant system, with lower values of GSH activity in experimental animals and diabetic patients. 37 In this study, GSH content was decreased in diabetes. Depletion of GSH levels enhances cellular damage caused by oxidative stress. Significant depletion of skin GSH levels in diabetic rats suggests its increased utilization against ROS. 38 However, with antioxidant treatment in diabetic rats, the GSH level is reversed back to normal levels. The increment in the GSH levels may protect the tissues against diabetes-associated tissue injury by reducing the susceptibility to toxic radicals. 39
Free radicals may play an important role in the causation and complications of diabetes mellitus. 40 The increased free radicals produced may react with polyunsaturated fatty acids in cell membrane leading to LPO, and it will, in turn, result in elevated free radical production. Free radicals react with lipids and cause peroxidative changes that result in enhanced LPO. 41 In our study, the LPO levels were elevated in diabetic rats as reported earlier. 42 The increase in LPO might be a reflection of a decrease in enzymatic and nonenzymatic antioxidants of defense systems. Antioxidants play an important role in protecting the human body against the damage caused by ROS. 43 In normal metabolism, there is a balance between the generation of free radicals and antioxidant defense mechanism. Nonenzymatic antioxidants such as VC and VE play an excellent role in preventing the cells from oxidative threats. These vitamins have been administered separately in studies in which they were given orally to diabetic rats, yielding a decrease in oxidative stress. 44,45
Advanced protein glycation in diabetes as a result of chronic hyperglycemia as well as oxidative stress has been postulated to play a central role in diabetic complications involving the accumulation of advanced glycation end products (AGEs) in tissue and arterial walls. 46,47 Skin AGE levels may also provide a more long-term ‘memory’ of glycaemic stress and, therefore, may be appropriate for predicting complications. 48 NEG was found to increase in diabetes mellitus, and the amount of increase is directly proportional to the fasting blood glucose level. 49 Furthermore, the level of glycation protein is considered one of the markers for the extent of oxidative stress in diabetes mellitus. In this study, the levels of NEG were found to increase in the STZ diabetic groups with respect to untreated controls. Various means of preventing this increase have been investigated in in vitro and in vivo. It has been reported that some transition metals, various plant extracts and vitamins prevent the increase of tissue NEG levels. 50 In this study, the increase in skin tissue NEG levels provoked by diabetes was significantly lowered by VE, VC and Se. This suggests that amelioration of oxidative stress which causing hyperglycemia is due to the administration of VE, VC and Se.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
