Abstract
Pesticides exposure causes usually harmful effects to the environment and human health. The present study aimed to investigate the potential toxic effects of penconazole, a triazole fungicide, on the cerebrum and cerebellum of adult rats. Penconazole was administered intraperitoneally to male Wistar rats at a dose of 67 mg kg−1 body weight every 2 days during 9 days. Results showed that penconazole induced oxidative stress in rat cerebrum and cerebellum tissues. In fact, we have found a significant increase in malondialdehyde, hydrogen peroxide, and advanced oxidation protein product levels, as well as an alteration of the antioxidant status, enzymatic (superoxide dismutase and catalase) and nonenzymatic (glutathione), the cholinergic function, and membrane-bound ATPases (Na+/K+-ATPase and Mg2+-ATPase). Penconazole also provoked histological alterations marked by pyknotic and vacuolated neurons in the cerebrum and apoptosis and edema in the cerebellum Purkinje cells’ layer. Therefore, the use of this neurotoxicant fungicide must be regularly monitored in the environment.
Introduction
Triazole fungicides, a large family of broad-spectrum fungicides introduced since the 1970s, are one of the current-use pesticides in the world. 1 The widespread use of these fungicides has raised serious concerns over their potential hazards to environmental safety and human health. In fact, due to their persistence in the environment, 2 triazole fungicides have brought a severe pollution in surface water, 3 soil, 4 and food. 4,5 In a series of toxicological studies, some fungicides belonging to the triazole class have been observed to be hepatotoxic, 6 tumorigenic, 7 teratogenic, 8 endocrine disrupting, 9 genotoxic, 10 and particularly neurotoxic 11 –13 in rats. In fact, it has been reported that triazole fungicides may produce toxic effects on mammalian nervous system, like neurobehavioral deficits and neuropathology. 13 Alteration of the dopaminergic neurotransmission, via inhibiting the depolarization-evoked Ca2+ influx, has been demonstrated as a possible mechanism mediating the neurotoxic effects of triazole fungicides. 14 On the other hand, an extensive survey of available literature indicates that pesticide-induced neurotoxicity may be ascribed to their ability to generate oxidative stress through an abnormal production of reactive oxygen species (ROS). 15 In fact, because of its increased oxygen consumption, 16 relatively low levels of antioxidant defenses, 17 and its high content of polyunsaturated lipids, the brain is particularly vulnerable to the toxic insults by free radicals. Furthermore, oxidative stress has been implicated in the pathogenesis of several neurodegenerative disorders like Parkinson, 15 Alzheimer, 18 and amyotrophic lateral sclerosis 19 associated with pesticides exposure. In this regard, the involvement of oxidative stress as the underlying mechanism governing the neurotoxic effects of triazole fungicides has been previously reported in fish, 20 –22 but in mammals, the data available is quite limited 14 or even lacking in the case of some triazoles like penconazole. The latter pesticide is one of the commonly used triazole fungicides in many countries throughout the world. It displays an outstanding curative and preventive efficacy against powdery mildew. It is used on fruits, especially grapes and apples, and on vegetables such as tomato and pepper. 23 Penconazole, with a mean half-life value of 117 days, is recognized to be recalcitrant to degradation and susceptible to accumulate in soils. 24 Moreover, it has been found in excess as residues in several agricultural products. 25,26 Because of its occurrence in the environment and the possible exposure of the general population to contaminated food stuffs, penconazole may represent a risk factor for human health. Indeed, we have previously shown that this fungicide is a potent inducer of oxidative stress in hepatic and cardiac tissues of adult rats. 27,28 Thus, considering the vulnerability of mammalian brain tissue to oxidative insult, exposure to penconazole may be associated with neurotoxicity.
Therefore, the present study was conducted to elucidate, for the first time, the impact of penconazole fungicide on redox status in the cerebrum and cerebellum of adult rats. So, we have examined the extent of oxidative damage in terms of lipid peroxidation, protein oxidation, responses of enzymatic and nonenzymatic antioxidants, activities of acetylcholinesterase (AChE), a crucial enzyme involved in cholinergic neurotransmission, Na+/K+-ATPase and Mg2+-ATPase, membrane-bound enzymes essential for neuronal cell functioning and cell homeostasis, as well as histopathological aspects.
Material and methods
Chemicals
The commercial fungicide studied in the present study was Topas® purchased from Syngenta Company (Basel, Switzerland), which contained 100 g L−1 of penconazole, the active substance. Glutathione (GSH), 5,5′-dithiobis-2-nitrobenzoic acid (DTNB), thiobarbituric acid (TBA), and 1,1,3,3-tetrathoxypropane (TEP) were purchased from Sigma Chemical Co. (St Louis, Missouri, USA). All remaining chemicals of analytical grade were provided from standard commercial suppliers.
Animals and experimental design
Twelve male Wistar rats, with an average weight of 250 ± 6 g, were purchased from the Central Pharmacy (SIPHAT, Tunisia). They were maintained under standard laboratory conditions (temperature 22 ± 2°C, relative humidity 40%, and 12-h light/dark cycle) with free access to food and water. Animals were treated in accordance with the International Guidelines for Animal Care, 29 and the experimental protocol was approved by the Ethical Committee of Sfax Faculty of Sciences.
Rats were randomly assigned into two groups of six each: group 1 represented the controls and group 2 (PEN) received intraperitoneally 67 mg kg−1 body weight (bw) of penconazole every 2 days during 9 days. Previously, El-Sharkawy and El-Nisr 30 have reported that exposure of adult male rats to penconazole at two doses (50 and 100 mg kg−1 bw) induced structural and functional testicular impairment. In a preliminary study, we have tried the dose of 100 mg kg−1 bw for 9 days. This dose provoked hemorrhage and diarrhea. Therefore, we have chosen, in the present study, an intermediate dose (67 mg kg−1 bw, equivalent to 1/30 of the median lethal dose (LD50)), which provoked toxicity without mortality. During the experimental period, rats were observed for behavioral performances after penconazole administration. On the 10th day, all rats were weighed and killed by cervical dislocation to avoid stress. The cerebrum and cerebellum were immediately removed, cleaned, and weighed. Some samples were homogenized in Tris-HCl buffer (pH 7.4) using an Ultra-Turrax T25 (IKA, Germany). The homogenates were centrifuged, and the resulting supernatants were used for determining the biochemical parameters. Other samples were fixed in 10% buffered formalin solution and embedded in paraffin for histological study.
Biochemical analysis
Protein determination
Protein content was determined in cerebrum and cerebellum homogenates by the method of Lowry et al. 31 using bovine serum albumin as a standard.
Malondialdehyde measurement
The concentrations of malondialdehyde (MDA), an index of lipid peroxidation, in cerebrum and cerebellum tissues were determined according to the method of Draper and Hadley 32 by measuring TBA-reactive substances (TBARSs) formation. The MDA values were calculated using TEP as the standard and expressed as nanomoles per milligram of protein.
Hydrogen peroxide generation assay
Measurement of hydrogen peroxide (H2O2) generation in the cerebrum and cerebellum was monitored by the ferrous ion oxidation-–xylenol orange method as described by Ou and Wolff. 33 The amount of H2O2 produced was determined using the extinction coefficient of 2.67 × 105 cm−1 M−1, and the results were expressed as nanomoles per milligram of protein.
Advanced oxidation protein products determination
Advanced oxidation protein product (AOPP) levels in the cerebrum and cerebellum were determined according to the method of Kayali et al. 34 The concentration of AOPP was calculated using the extinction coefficient of 261 cm−1 mM-1, and the results were expressed as nanomoles per milligram of protein.
Determination of antioxidant enzyme activities
Catalase (CAT) activity was determined according to the method of Aebi 35 using H2O2 as the substrate. The decrease in absorbance due to H2O2 degradation was recorded at 240 nm, and CAT activity was calculated in terms of micromoles of H2O2 consumed per minute per milligram of protein.
Superoxide dismutase (SOD) activity was evaluated by monitoring the photochemical reduction of nitro blue tetrazolium (NBT) as described by Beauchamp and Fridovich. 36 One unit (U) of SOD activity corresponded to the amount of enzyme required to cause 50% inhibition of NBT reduction at 560 nm. SOD activity was expressed as units per milligram of protein.
GSH content
The cerebrum and cerebellum GSH contents were measured according to the method of Ellman 37 modified by Jollow et al. 38 This method is based on the development of a yellow color when DTNB is added to compounds containing sulfhydryl groups. The absorbance was measured at 412 nm after 10 min. The total GSH content was expressed as microgram per gram of tissue.
Determination of AChE activity
AChE activity was determined in the cerebrum and cerebellum tissues according to the method of Ellman et al. 39 using acetylthiocholine iodide as a substrate. Results were expressed as micromoles per minute per milligram of protein.
Assay of membrane-bound ATPases, Na+/K+-ATPase, and Mg2+-ATPase
Na+/K+-ATPase and Mg2+-ATPase activities in the cerebrum and cerebellum were determined using the method described by Kawamoto et al. 40 by splitting of ATP molecules by ATPase in the presence of Na+/K+ and Mg2+ ions, respectively. Phosphate reacts with ammonium molybdate in an acidic medium, forming a phosphomolybdate complex. Enzyme activity was expressed as micromoles of inorganic phosphate liberated per hour per milligram of protein.
Histological studies
Some cerebrum and cerebellum tissues, intended for histological examination, were fixed in 10% buffered formalin solution, processed using graded ethanol series, and embedded in paraffin. Sections of 3 µm thickness each were stained either with hematoxylin–eosin (H&E) or with borated toluidine blue solution for light microscopic observation. Morphological evidence of apoptosis was detected by borated toluidine blue staining according to the method described by Cohen et al. 41 Apoptotic cells appeared as darkly stained. Six slides were prepared from each tissue and for each coloration. Each cerebrum or cerebellum slide was examined and assigned for severity of changes using scores on a scale of none (−), moderate (++), and severe (+++) damages.
Statistical analysis
All values were expressed as mean ± standard deviation. Differences between the control and penconazole-treated groups were evaluated statistically using Student’s t-test. Differences were considered significant if p < 0.05.
Results
Effect of penconazole treatment on general health, animal body, cerebrum, and cerebellum weights
There were no mortalities during the experimental period and no significant differences in the body weights between control and penconazole-treated rats. Nonetheless, penconazole treatment induced behavioral changes in rats like hypoactivity and weakness, loss of equilibrium, and head bobbing. Furthermore, in PEN group, a significant increase was obtained in the absolute and relative weights of the cerebrum (+13 and +17%) and cerebellum (+15 and +13%), respectively (Table 1).
Body, absolute, and relative cerebrum and cerebellum weights of control and penconazole-treated rats.a
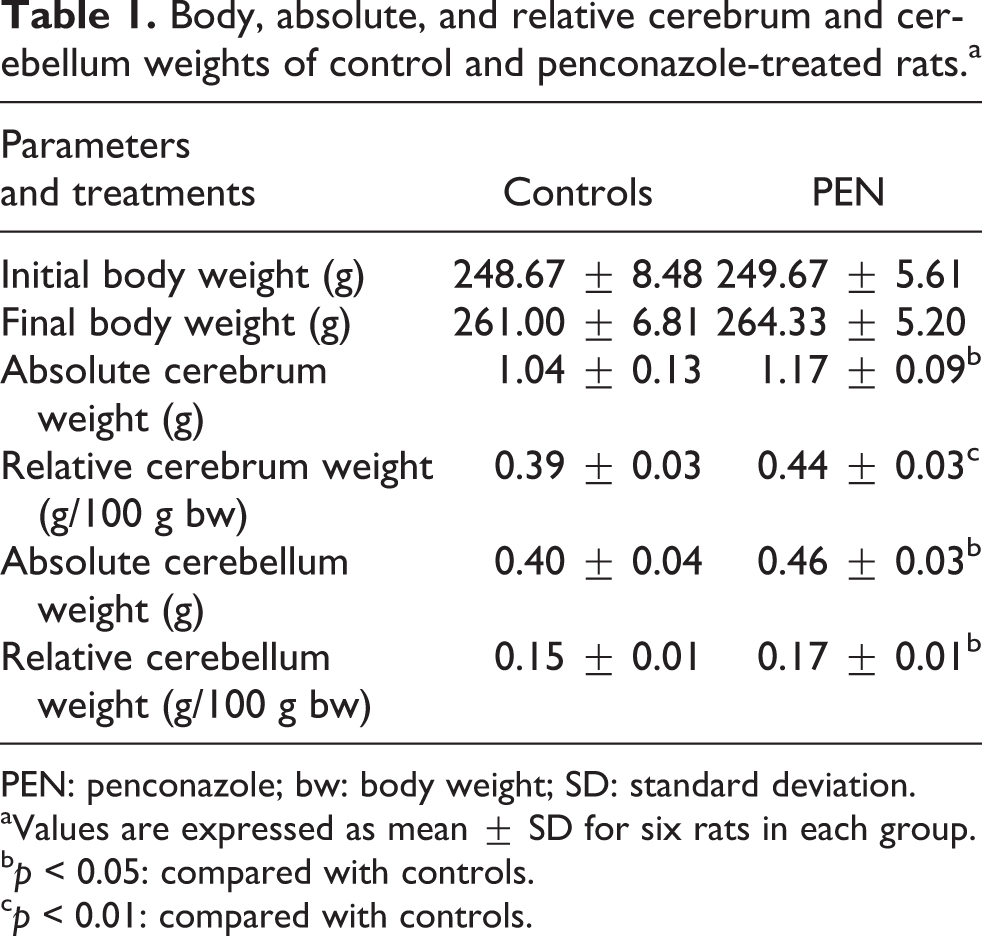
PEN: penconazole; bw: body weight; SD: standard deviation.
aValues are expressed as mean ± SD for six rats in each group.
b p < 0.05: compared with controls.
c p < 0.01: compared with controls.
Lipid peroxidation
Lipid peroxidation was assessed by measuring MDA levels. As shown in Table 2, rats exposed to penconazole exhibited a significant increase in lipid peroxidation in cerebrum (+36%) and cerebellum (+17%), as compared to the corresponding controls.
Evaluation of oxidative stress biomarkers (MDA, H2O2, AOPP) in the cerebrum and cerebellum of control and penconazole-treated rats.a

MDA: malondialdehyde; H2O2: hydrogen peroxide; AOPP: advanced oxidation protein products; PEN: penconazole; SD: standard deviation.
aValues are expressed as mean ± SD for six rats in each group.
b p < 0.001: compared with controls.
c p < 0.01: compared with controls.
d p < 0.05: compared with controls.
H2O2 generation
As shown in Table 2, higher contents of H2O2 were detected in the cerebrum (+59%) and the cerebellum (+43%) of penconazole-treated rats than those of controls.
Protein oxidative damage
The degree of protein oxidation was estimated by measuring AOPP levels. Results showed that there was a significant increase in AOPP levels in the cerebrum (+75%) and cerebellum (+16%) of penconazole-treated rats when compared to the corresponding controls (Table 2).
Enzymatic antioxidant status
Results regarding penconazole effect on SOD and CAT activities were illustrated in Table 3. CAT activity decreased significantly in cerebrum and cerebellum of penconazole-treated rats (−38 and −12%, respectively) as compared to controls. SOD activity was also slightly reduced (−8%) in the cerebrum following penconazole exposure. While in the cerebellum, penconazole treatment had no effect on SOD activity when compared to that of controls.
Enzymatic (CAT and SOD) and nonenzymatic (GSH) antioxidant status in the cerebrum and the cerebellum of control and penconazole-treated rats.a
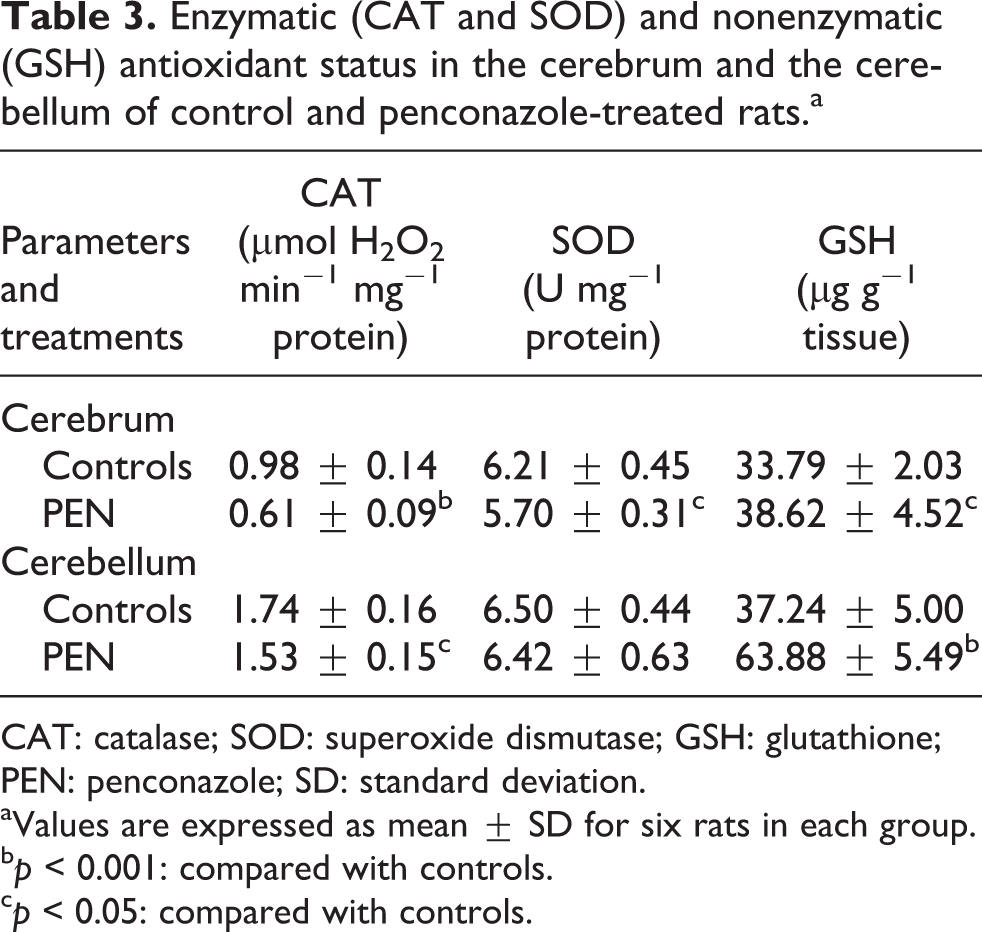
CAT: catalase; SOD: superoxide dismutase; GSH: glutathione; PEN: penconazole; SD: standard deviation.
aValues are expressed as mean ± SD for six rats in each group.
b p < 0.001: compared with controls.
c p < 0.05: compared with controls.
Nonenzymatic antioxidant status
GSH levels were significantly increased in the cerebrum (+14%) and cerebellum (+72%) of penconazole-treated rats as compared to those of controls (Table 3).
AChE activity
Our results showed that penconazole caused a significant inhibition of AChE activity in the cerebrum (−11%) and cerebellum (−25%) of adult rats (Figure 1).

AChE activities in the cerebrum (a) and cerebellum (b) of control and penconazole-treated rats. Values are expressed as mean ± SD for six rats in each group. PEN group vs. control group: *p < 0.05; ***p < 0.001. PEN: penconazole; AChE: acetylcholinesterase; SD: standard deviation.
Na+/K+-ATPase and Mg2+-ATPase activities
Na+/K+-ATPase and Mg2+-ATPase activities were found to be significantly increased in the cerebrum (93 and 23%, respectively) and cerebellum (82 and 46%, respectively) of penconazole-treated group when compared to those of control group (Figure 2).
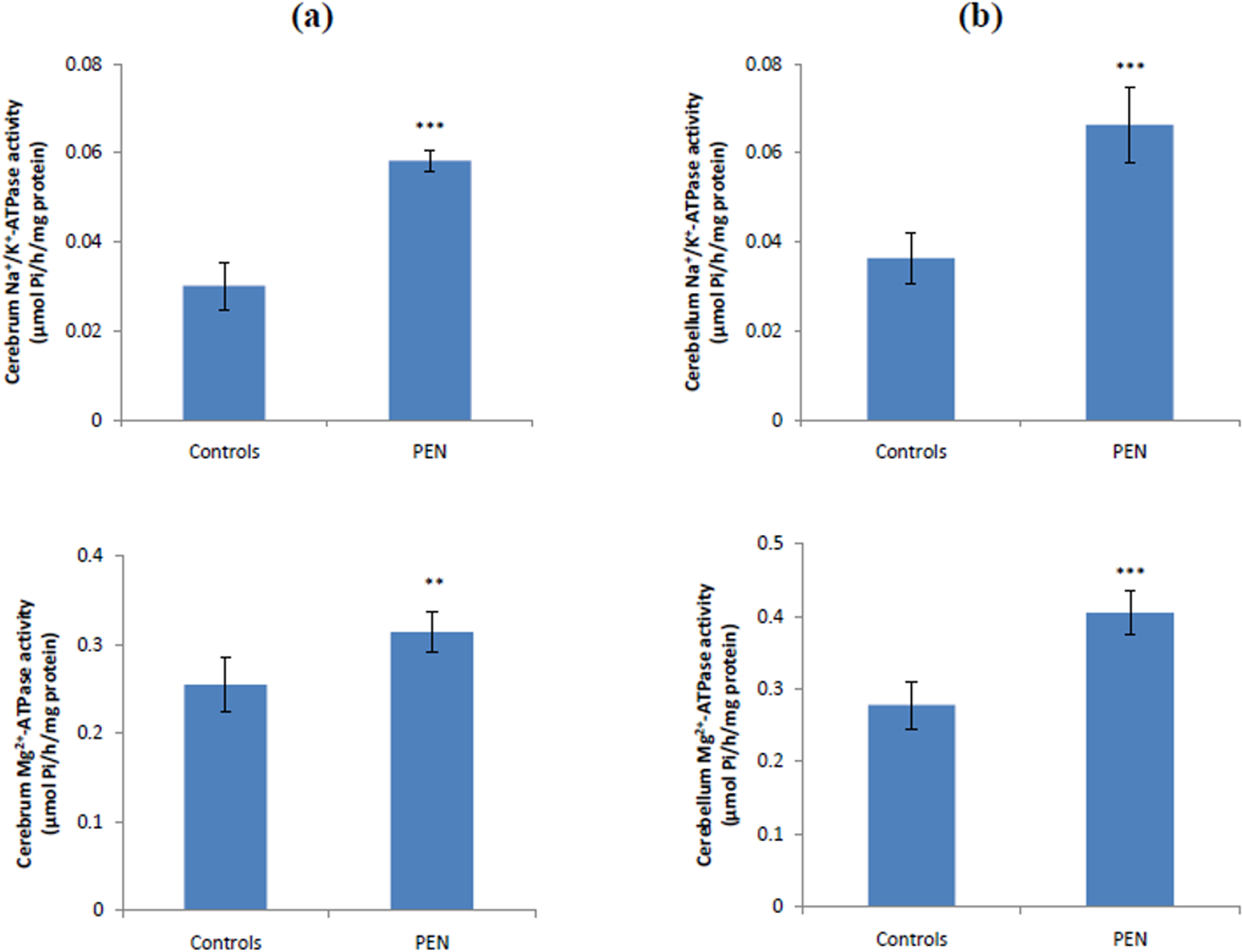
Na+/K+-ATPase and Mg2+-ATPase activities in the cerebrum (a) and cerebellum (b) of control and penconazole-treated rats. Values are expressed as mean ± SD for six rats in each group. PEN group vs. control group: **p < 0.01; ***p < 0.001. PEN: penconazole; SD: standard deviation.
Histological studies
Light microscopic examination of the cerebrum and cerebellum in control rats showed normal histological structures (Figures 3(a), 3(b), 4(a), and 4(b)). Exposure of rats to penconazole provoked histological changes in both the cerebrum and cerebellum. In fact, cerebrum sections of penconazole-treated rats, stained with H&E, showed vacuolated neurons (Figure 3(c)). In cerebellum sections, there was an edema in the Purkinje cells’ layer (Figure 4(c)). Besides, brain histological sections of penconazole-treated rats, stained with toluidine blue, showed dark pyknotic neurons in the cerebrum (Figure 3(d)) and apoptotic Purkinje cells in the cerebellum (Figure 4(d)). The histological changes in penconazole-treated rats were graded and summarized in Table 4.

Cerebrum histological sections, stained with H&E and blue toluidine, of control (a and b) and penconazole-treated rats (c and d) (×400). The arrows indicate  vacuolated neurons and
vacuolated neurons and  Pyknotic neurons.
Pyknotic neurons.

Cerebellum histological sections, stained with H&E and blue toluidine, of control (a and b) and penconazole-treated rats (c and d) (×400). The arrows indicate  Purkinje cells edema and
Purkinje cells edema and  apoptotic Purkinje cells. ML: molecular layer; PCL: Purkinje cells’ layer; IGL: internal granular layer; H&E: hematoxylin–eosin.
apoptotic Purkinje cells. ML: molecular layer; PCL: Purkinje cells’ layer; IGL: internal granular layer; H&E: hematoxylin–eosin.
Grading of the histopathological changes in the cerebrum and cerebellum sections.a

PEN: penconazole.
aScoring was done as follows: none (−), moderate (++), and severe (+++) damage.
Discussion
Owing to its high potency to reduce powdery mildew incidence in different crops, 42 penconazole, a triazole fungicide, became a pesticide of choice in many countries throughout the world. Because of its persistence in the water and the soil and its low degradation, penconazole may represent a risk factor for human health causing diseases. Hence, we aimed in the current study to evaluate the potential neurotoxicity induced by penconazole through monitoring its effects on the oxidative status in the cerebrum and cerebellum of adult rats.
In toxicological studies, body and/or organ weights are the important criteria for the evaluation of toxicity. In the present study, no significant changes were observed in body weights of penconazole-treated rats as compared to the controls. Meanwhile, a significant increase in the absolute and relative weights of the cerebrum and the cerebellum was observed following exposure of adult rats to penconazole, which could account for the edema and vacuolization shown in the histoarchitecture of the cerebellum and cerebrum, respectively. This might be the result of an increased generation of free radicals, probably generated by penconazole, and the subsequent installation of oxidative stress in the cerebellar and cerebral tissues of adult rats. Indeed, free radicals might attack polyunsaturated fatty acid (PUFA) of cell membranes causing destabilization, disintegration, fluidity alteration, and enhanced membrane permeability leading therefore to vacuolization, a primary response to cell injury. 43 The brain has a relatively poor antioxidant defense system, contains large amounts of oxidizable substrates like PUFA, and consumes 20% of the body’s oxygen. So, this soft organ is particularly susceptible to free radicals attack and, therefore, lipid peroxidation. 44 In the present study, exposure of adult rats to penconazole resulted in an enhanced lipid peroxidation, as indicated by a significant increase in MDA levels in cerebrum and cerebellum tissues. Our findings were in agreement with the previous report of Jaiswal et al. 45 who have found an increase in MDA level in the brain tissue of rats exposed to carbofuran, a carbamate pesticide.
A large number of experimental studies have indicated that ROS generation contributes to pesticide-induced neurotoxicity. 15 In general, the primary intracellular site for ROS production is mitochondria, where the electrons, possibly escaped from the electron transport chain, combine with molecular oxygen to form the superoxide anion radical (O2 • −) and other ROS. 46 Neurons have a high density of mitochondria, which explains their particular vulnerability to energy-dependent defects resulting from mitochondrial abnormalities. The capacity of these subcellular organelles to produce ROS can be generally assessed through determining the rate of H2O2 release by respiring mitochondria. Our results showed that, in line with the increase of MDA levels, there was also a significant increase in H2O2 production in the cerebrum and cerebellum of penconazole-treated rats. This finding suggested that penconazole-induced lipid peroxidation through ROS generation. Indeed, H2O2 is an oxidizing agent that can be easily converted into hydroxyl radical (OH•). The latter is the most reactive of free radical molecules, initiating lipid peroxidation by hydrogen abstraction. Moreover, the enhanced generation of ROS reflects probably a mitochondrial dysfunction induced by the fungicide. Impairment of oxidative phosphorylation has been observed following the exposure to a large number of pesticides particularly neurotoxic agents through the inhibition of a biosynthetic pathway essential for mitochondrial function or extramitochondrial generation of ROS. 47
Moreover, protein oxidant status is another index to assess oxidative stress. In fact, due to their fast reaction rates with many radicals and oxidants, proteins are the major targets for oxidative damage, leading to the loss of their specific functions. AOPP is a novel biomarker, which can be used in order to estimate the degree of protein damage. Our results showed that AOPP levels increased markedly in the cerebrum and cerebellum tissues of penconazole-treated rats when compared to controls. Hence, it can be suggested that penconazole promoted ROS generation in the brain tissue of adult rats, leading consequently to protein oxidation. Our results corroborated with previous findings of other researchers who have reported an increase in AOPP levels after intoxication by fenthion, an organophosphorus compound. 48
Furthermore, our findings regarding the effects of penconazole on brain oxidative stress were in accordance with those obtained by Li et al. 20 In fact, these authors have demonstrated that exposure to propiconazole, another triazole fungicide, induces in the brain of the fish rainbow trout the generation of TBARS, index of lipid peroxidation, and the formation of protein carbonyl, biomarker of protein oxidation.
Under physiological conditions, ROS production is largely neutralized by a protection system, which is offered by enzymatic and nonenzymatic processes. As enzymatic antioxidant systems, SOD and CAT constitute a mutually supportive defense against free radicals, converting them to the less harmful molecules. 49 SOD catalyzes the dismutation of superoxide radicals (O2 • −) into molecular oxygen (O2) and H2O2. 50 CAT can further catalyze the decomposition of H2O2 to water (H2O) and O2. 51 In the present study, we have observed a significant decrease in CAT and SOD activities in the cerebrum of adult rats following penconazole exposure. The decline of cerebral antioxidant enzyme activities might be due to an enhanced generation of superoxide radicals or to the downregulation in the synthesis of antioxidant enzymes induced by penconazole exposure. In the cerebellum, a slight decrease of SOD activity was observed following penconazole treatment. Nevertheless, exposure to this fungicide resulted in a significant reduction of CAT activity in the cerebellum of adult rats. The slight depletion in SOD activity might probably reflect the onset of an adaptive response to oxidative stress. As endogenous antioxidant enzymes act cooperatively with nonenzymatic antioxidants to scavenge free radicals, the effect of penconazole exposure on the level of GSH was investigated in the present study. GSH, the main redox equilibrium regulator, plays a key role in the protection of tissues suffering from oxidative damage. Under our experimental conditions, GSH levels increased significantly in the cerebrum and cerebellum of penconazole-treated rats. The elevation of this nonenzymatic antioxidant might be due to the induction of its synthesis, possibly to protect neuronal cells from penconazole intoxication. Alteration of the brain enzymatic and nonenzymatic antioxidant status following exposure of experimental animals to some pesticides has been earlier reported by other researchers. 45,48,52
In our present work, the generation of ROS and subsequent oxidative stress induced by penconazole mediated functional changes in the cerebrum and cerebellum of adult rats such as the alteration in the activity of membrane-bound enzymes, Na+/K+-ATPase and Mg2+-ATPase. In the brain, Na+/K+-ATPase is an enzyme responsible for the active transport of Na+ and K+ ions, maintaining the ionic gradient necessary for neuronal excitability and regulation of neuronal cell volume. 53,54 It is also implicated in other physiological functions such as metabolic energy production 55 and uptake and release of catecholamines 56,57 and serotonin. 58 Mg2+-ATPase plays an important role in maintaining high brain intracellular Mg2+, changes of which can modulate the activity of Mg2+-dependent enzymes and control the rates of protein synthesis as well as cell growth. 59 Our results showed a significant increase in the activities of Na+/K+-ATPase and Mg2+-ATPase in the cerebrum and cerebellum of adult rats following penconazole exposure. Similar results were obtained by Hazarika et al. 60 in the liver and erythrocytes of rats exposed to the insecticides malathion and anilofos. However, Li et al. 20 reported a decreased Na+/K+-ATPase activity in the brain of rainbow trout after exposure to propiconazole. Because of its lipophilic nature, it can be supposed that penconazole interacts with some neuronal membrane components, inducing changes in the conformational state of membrane-bound enzymes, consequently altering the activity of Na+/K+-ATPase and Mg2+-ATPase. Indeed, according to Silvius and McElhaney, 61 several factors such as membrane composition or the presence of lipid-soluble drugs can disrupt the lipid microenvironment, thereby changing membrane-bound enzymes activity.
The effects of penconazole exposure on brain function were also evaluated by determination of AChE activity. This enzyme is present in the neuromuscular junctions and cholinergic synapses of the central nervous system. It terminates the signal transmission by hydrolyzing acetylcholine (ACh), a neurotransmitter that conducts nerve impulses across neuromuscular junctions. 62 Our results demonstrated that penconazole exposure affected the cholinergic system as revealed by a significant decrease of AChE activity in the cerebrum and cerebellum of rats treated with this fungicide. The enhanced oxidative stress in the brain of adult rats appears to be an important reason for the disruption of the cholinergic function following penconazole exposure. Furthermore, Meyer and Cooper 63 suggest that a decrease in ACh release could be induced by the increased Na+/K+-ATPase activity. According to Sharma et al., 64 inhibition of AChE causes the accumulation of the ACh in synaptic junctions, which leads to overstimulation of postsynaptic cells and cholinergic manifestations. Consistent with our findings, the pesticide λ-cyhalothrin has been reported to elicit its toxic effects by alerting AChE activity in the brain of experimental animals. 52 In contrast, Toni et al. 22 showed an increased AChE activity in the brain of carp (Cyprinus carpio) after exposure to tebuconazole, another triazole fungicide.
In our experimental study, the inhibition of AChE activity along with the stimulation of Na+/K+-ATPase in the cerebrum and cerebellum of penconazole-treated rats could possibly explain the observed behavioral impairments like hypoactivity and weakness, loss of equilibrium and head bobbing. In fact, it is well known that AChE activity is vital to normal behavior and muscular function. 65 Cholinergic neurotransmission in the brain also plays a central role in vigilance, learning, and memory. 66 –68 Furthermore, it has been demonstrated that AChE deficit leads to muscular alterations including the contractile properties and the lack of resistance to fatigue. 69 Regarding Na+/K+-ATPase, it has been reported that this enzyme might be implicated in neuronal and synaptic plasticity 70 and therefore mediates the modulation of learning and memory. 71 The current results are in accordance with other studies, which have demonstrated that triazole fungicides cause behavioral alterations such as hypomovement and loss of equilibrium in fresh water fish 72 and head bobbing in rats. 12
In a recent study, triazole fungicides, namely flusilazole, triadimefon, tebuconazole, and cyproconazole, were found to elicit their neurotoxic effects by altering the dopaminergic transmission through inhibiting depolarization-evoked calcium influx in dopaminergic PC12 cells. 14 Based on our results, it is suggested that disruption of cholinergic transmission and membrane-bound enzymes Na+/K+-ATPase and Mg2+-ATPase is another possible mechanism mediating triazole-induced neurotoxicity.
It is well established that the enhanced oxidative stress in the brain may trigger a cascade of events leading eventually to cell death. In our histopathological study, the morphological features of neuronal death induced by penconazole exposure were reflected by the presence of dark pyknotic neurons indicating a chronic neurodegenerative process. In addition, penconazole treatment led to apoptotic Purkinje cells in the cerebellum. Apoptosis is a physiological process of cell death that plays a critical role in normal development as well as in the pathophysiology of several diseases. According to Orrenius et al., 73 it can be activated through an ROS-mediated damage to mitochondria, opening mitochondrial permeability transition pores and provoking cytochrome C release, which initiates caspase activation. So, it is suggested that penconazole, a lipophilic molecule, could easily pass through the neuronal membrane to reach mitochondria and induce ROS production, causing consequently apoptosis.
Conclusion
In summary, the results of the present study demonstrated that penconazole had the ability to induce oxidative damage in the brain of adult rats, as evidenced by an increase of lipid peroxidation and protein oxidation, in addition to the perturbations in the enzymatic and nonenzymatic antioxidant status. Exposure to this fungicide affected also the cholinergic system, activities of membrane-bound ATPases, and brain histoarchitecture. The knowledge, gained from our findings, indicates for the first time that penconazole is a potential neurotoxicant pesticide that exerts its neurotoxic effects via the generation of oxidative stress. Consequently, exposure to this fungicide should be carefully monitored.
Footnotes
Acknowledgment
The authors are thankful to Mr Chedli Tmar for maintenance of the laboratory animals and to Mrs Raoudha Ben Amar Abdennadher for her skillful technical assistance.
Authors’ Contribution
Imen Ghorbel and Awatef Elwej have contributed equally to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministry of Higher Education and Scientific Research [04/UR/09-04], Tunisia.
