Abstract
Hexavalent chromium (Cr (VI)) is a common environmental pollutant. Cr (VI) exposure can lead to severe damage to the liver, but the preventive measures to diminish Cr (VI)-induced hepatotoxicity need further study. S-allyl cysteine (SAC) is a constituent of garlic (
Introduction
Acute and chronic liver diseases constitute a global concern, but medical treatments for these diseases are often difficult to handle and have limited efficacy. Severe hepatotoxicity induces inflammation, necrosis, and oxidative stress of hepatocytes and results in a worldwide health problem in human. 1 In the last few years, it has been suggested that in most cases hepatocellular injury is not only due to the damaging agent itself but also due to the inflammatory cells that have been attacked by the stressed hepatocytes. Chromium is ubiquitous in the environment and can be found in pigments, chrome-plated metals, tanned shoe leather, cement, detergents, and industrial chromium waste dumps. Chromium has several oxidation states, including Cr (II), Cr (III), Cr (IV), Cr (V), and hexavalent chromium (Cr (VI)), but only Cr (III) and Cr (VI) are stable. 2 Liver is an important organ to be considered when the effects of pollutants are investigated, since this organ plays a central role in the metabolism and detoxification of biological substances. Also, most of the substances absorbed by the intestine passes first through the liver where toxins and heavy metals may accumulate. Several studies have demonstrated that the liver is capable of being injured by Cr (VI), and the hepatotoxicity is associated with increased reactive oxygen species (ROS) levels, lipid peroxidation (LPO), DNA damage, reduction of the activity of the antioxidant enzymes, and apoptosis. 2 –4
The nuclear transcription factor nuclear factor-kappa B (NF-κB) controls a number of genes involved in inflammatory responses, cell cycle progression, inhibition of apoptosis, and cell adhesion and thus plays a critical role in promoting inflammation, carcinogenesis, and cancer progression. NF-κB activity is attributed to homodimers or heterodimers of the Rel transcription factor family. 5 This protein exists in the cytoplasm in an inactive form; NF-κB is stabilized by an inhibitory subunit IκB, which inhibits its DNA-binding activity. The stimulation of the cells with a wide variety of pathogens and inflammatory cytokines dissociates IκB from NF-κB, allowing free NF-κB to migrate to the nucleus and bind to specific DNA elements and then transmit various signals from the cytosol to the nucleus. This directly alters the expression of different proinflammatory cytokines. 6 Tumor necrosis factor α (TNF-α) is a proinflammatory cytokine released by a number of different cells in the liver. The production of TNF-α is known to be one of the earliest events in the hepatic inflammatory response and is capable of stimulating the production and secretion of several other cytokines. TNF-α is important in modulating hepatocyte proliferation and tissue repair in liver injury. 7
Nitric oxide (NO) is produced from a guanidine group of the amino acid
Garlic (
Materials and methods
Chemicals
K2Cr2O7 was purchased from Ranbaxy Laboratories (Mumbai, India). SAC was obtained as a gift from Wakunaga Pharmaceutical Co. Ltd. (Hiroshima, Japan). TNF-α, NF-κB, and iNOS primary antibodies were purchased from Santa Cruz Biotechnology (Santa Cruz, California, USA). Propidium iodide (PI) was purchased from Vector Laboratories (UK). All other reagents used were of analytical grade.
Animals
Male albino rats of Wistar strain weighing approximately 200–240 g obtained from the Laboratory Animal Maintenance Unit, Tamil Nadu Animal Science and Veterinary University, Madhavaram, India, were used throughout this study. Before the commencement of the experiment, the animals were acclimatized to the laboratory conditions for 14 days. They were maintained at an ambient temperature of 25°C ± 2°C and 12/12 h of light–dark cycle. Animals were given standard rat chow (Hindustan Lever Ltd., Bangalore) and water ad libitum and housed under standard environmental conditions until treatment or being sacrificed. The experiments were conducted according to the ethical norms approved by Ministry of Social Justice and Empowerment, Government of India and Institutional Animal Ethics Committee Guidelines (approval no. 360/01/a/CPCSEA).
Experimental design
The rats were divided into four groups (six rats in each group) as follows: Group I animals served as control; group II animals received a single dose of K2Cr2O7 (17 mg/kg body weight) subcutaneously as described by Kalayarasan et al.
18
; group III animals were treated with SAC intraperitoneally at a dose of 100 mg/kg body weight (based on effective dosage fixation study) as described by Kalayarasan et al.
18
and administered for 3 days at 24-h time intervals along with K2Cr2O7 at the dose mentioned in group II; group IV animals received SAC alone with 100 mg/kg body weight. The experiment was terminated at the end of the seventh day, and the rats were anesthetized and killed by cervical decapitation. Blood was collected from the dorsal aorta and was allowed to clot for 10 min at room temperature and then centrifuged at 1200
Biochemical analysis and enzyme assays
Diagnostic marker enzymes
The collected serum was used to estimate aspartate transaminase (AST) and alanine transaminase (ALT) by the method of King, 19 and the activity was expressed as micromoles of pyruvate liberated per hour per milligram of protein. Lactate dehydrogenase (LDH) was measured by the method of King 20 and expressed as micromoles of pyruvate liberated per hour per milligram of protein. Alkaline phosphatase (ALP) activity was assayed by the method of King, 21 and the activity of ALP was expressed as King Armstrong Unit (KAU) per liter in serum.
Lipid peroxidation
Lipid peroxides were estimated in the cerebellum using the method of Ohkawa et al. 22 in which the malondialdehyde (MDA) released served as the index of LPO. 1,1,3,3-tetraethoxypropane MDA bis(diethyl acetal) was used as the standard. The level of lipid peroxides was expressed as micromoles of MDA released per milligram of protein. Hydroxy radical production was measured by the method of Cederbaum and Cohen. 23 OH• level was expressed as nanograms per milligram of protein. The estimation of protein was assayed by Lowry’s method using crystalline bovine serum albumin (BSA) as the standard and expressed as g/dL for serum. 24
Assay for enzymatic antioxidants
The activity of superoxide dismutase (SOD) was measured by the method of Misra and Fridovich, 25 which is based on the oxidation of epinephrine to adrenochrome transition by the enzyme, and the enzyme activity was expressed as 50% inhibition of epinephrine autoxidation and expressed as units per minute per milligram of protein. Catalase (CAT) was assayed by measuring the rate of decomposition of H2O2 26 and the enzyme activity was expressed as micromoles of H2O2 consumed per minute per milligram of protein. Glutathione peroxidase (GPx) activity is coupled to Nicotinamide Adenine Dinucleotide Phosphate (NADPH) utilization and the production of NADP was measured by the method described by Pagila and Valentine. 27 The activity of GPx was expressed as micromoles of glutathione (GSH) oxidized per minute per milligram of protein. Reduced GSH was assessed by the method described by Moron et al. 28 using Dithiobis 2-nitrobenzoic acid (DTNB) as substrate, and the amount of GSH was expressed as micromoles of GSH per milligram of protein. Glutathione reductase (GR) was analyzed by the method of Staal et al. 29 The activity is expressed as micrograms of NADPH oxidized per minute per milligram of protein.
Evaluating the activities of lysosomal enzymes
Briefly, a 10% (w/v) liver homogenate was prepared in 0.05 M Tris–KCl buffer, pH 7.4, containing 0.25 M sucrose at 4°C in a glass homogenizer. The homogenate was subjected to differential centrifugation and centrifuged at 600
β-
Acid phosphatase activity was assayed by the method of King. 21 Briefly, the incubation mixture of final volume (3.0 mL) contained 1.5 mL of buffer, 1.0 mL of substrate, and required amount of the enzyme source. The tubes were incubated at 37°C for 15 min. The reaction was arrested by the addition of 1.0 mL Folin’s phenol reagent. To the control tubes, enzyme was added after arresting the reaction. The contents were centrifuged, and 1.0 mL of 15% sodium carbonate was added to the supernatant. The mixture was incubated for 15 min at 37°C, and the color was read at 640 nm in a Shimadzu UV spectrophotometer and acid phosphatase activity was expressed as micromoles of phenol liberated per hour per milligram of protein.
Cathepsin D activity was determined by the method of Etherington. 32 Briefly, known aliquots (0.2 mL) of the enzyme source were incubated with 1.5% hemoglobin in 0.1 M acetate buffer, pH 3.0, for 2 h. The enzyme activity was arrested by the addition of 10% Trichloro Acetic Acid (TCA), and the liberated TCA soluble products were filtered and neutralized with 1 N NaOH. The tyrosine content of the filtrate was determined using Folin’s phenol reagent and employing the procedure of Lowry et al. 24 The blue color developed was read at 660 nm and cathepsin D activity was expressed as micromoles of tyrosine liberated per hour per milligram of protein.
Histopathological analysis of liver
Liver samples were fixed in 10% buffered formaldehyde solution, processed by the paraffin slice technique. Sections about 4-µm thickness were stained with hematoxylin and eosin (H&E) staining for routine histology. 18 For H&E staining, sections were stained with hematoxylin for 3 min, washed, and stained with 0.5% eosin for additional 3 min. After an additional washing step with water, the slides were dehydrated in 70%, 95%, and 100% ethanol and cleared in xylene. The sections were mounted with DPX and pictures were taken using a Nikon FDX microscope (Tokyo, Japan).
Immunohistochemical analysis of NF-κB and TNF-α
Immunohistochemical analysis of p65-NF-κB and TNF-α was performed by the method described by Pandurangan et al. 33 Paraffin-embedded tissue sections of 4-µm thickness were rehydrated first in xylene and then in graded ethanol solutions. The sections were placed in 3% hydrogen peroxide for 15 min to quench endogenous peroxidase activity. Sections were heated in 0.1 M citrate buffer, pH 6.0, in a microwave oven for 6 min and rinsed twice, 5 min each wash, with phosphate-buffered saline containing 0.05% Triton X-100 (PBST). After blocking nonspecific binding sites with 5% BSA for 1 h, sections were incubated with a rabbit polyclonal antibody against p65-NF-κB and TNF-α (Santa Cruz Biotechnology; 1:500) for overnight; negative control sections incubated with PBST only. Sections were rinsed with PBST, incubated with secondary antibody conjugated with horseradish peroxidase at a 1:500 dilution (Bangalore Genei, Bangalore, India) for 90 min. Diaminobenzidine was used as chromogen, followed by counterstaining with hematoxylin. Each step was performed at room temperature, except where otherwise stated. The slides were visualized under a light microscope (NikonTE-1B). To measure the relative intensity, scoring was done as arbitrary units 4 as intensely stained, 3 as moderately stained, 2 as mild staining, and 1 as poorly stained in control and experimental groups.
Immunofluorescence analysis of iNOS
Paraffin-embedded tissue sections of 4-µm thickness were rehydrated first in xylene and then in graded ethanol solutions. The slides were then blocked with 3% BSA in TBS for 2 h. The sections were then immunostained with primary antibody (mouse monoclonal IgG to rat iNOS; Santa Cruz Biotechnology) diluted 1:200 with 3% BSA in TBS) and incubated overnight at 4°C. After washing the sections thrice with TBS, the slides were then incubated with their corresponding Fluorescein Isothiocyanate FITC-conjugated secondary antibody diluted 1:40 in TBS and incubated in dark for 2 h at room temperature. Sections were then washed in TBS and incubated with the nucleus-specific counter-PI to highlight cell nuclei. Slides were cover slipped and were visualized under a fluorescent microscope (Nikon TE300). For quantification of iNOS, cell population examination of 25 grid field (each field measuring 0.245 mm2) was counted and the mean score per liver was calculated. FITC-positive cells were analyzed from six locations per rat liver tissue section, with six rats per group, and analyzed for statistical significance.
Statistical analysis
All the grouped data were statistically evaluated with SPSS version 10.0 software. Hypothesis testing methods included one-way analysis of variance followed by least significant difference test;
Results
Effect of SAC on biochemical markers
During liver damage, increased levels of liver marker enzymes are pronounced. To study the effect of SAC in liver enzyme status, activities of AST, ALT, LDH, and ALP in normal and experimental rats were determined. The hepatoprotective profile of SAC on Cr (VI)-induced toxicity is described in Figure 1. The level of the marker enzymes such as AST, ALT, LDH, and ALP was elevated in Cr (VI)-induced rats (group II) when compared to group I. These elevated activities were brought back to near normal in groups treated with SAC and Cr (VI)-induced rats (group III). These observations demonstrate that SAC treatment renders protective effect against Cr (VI)-induced hepatotoxicity.
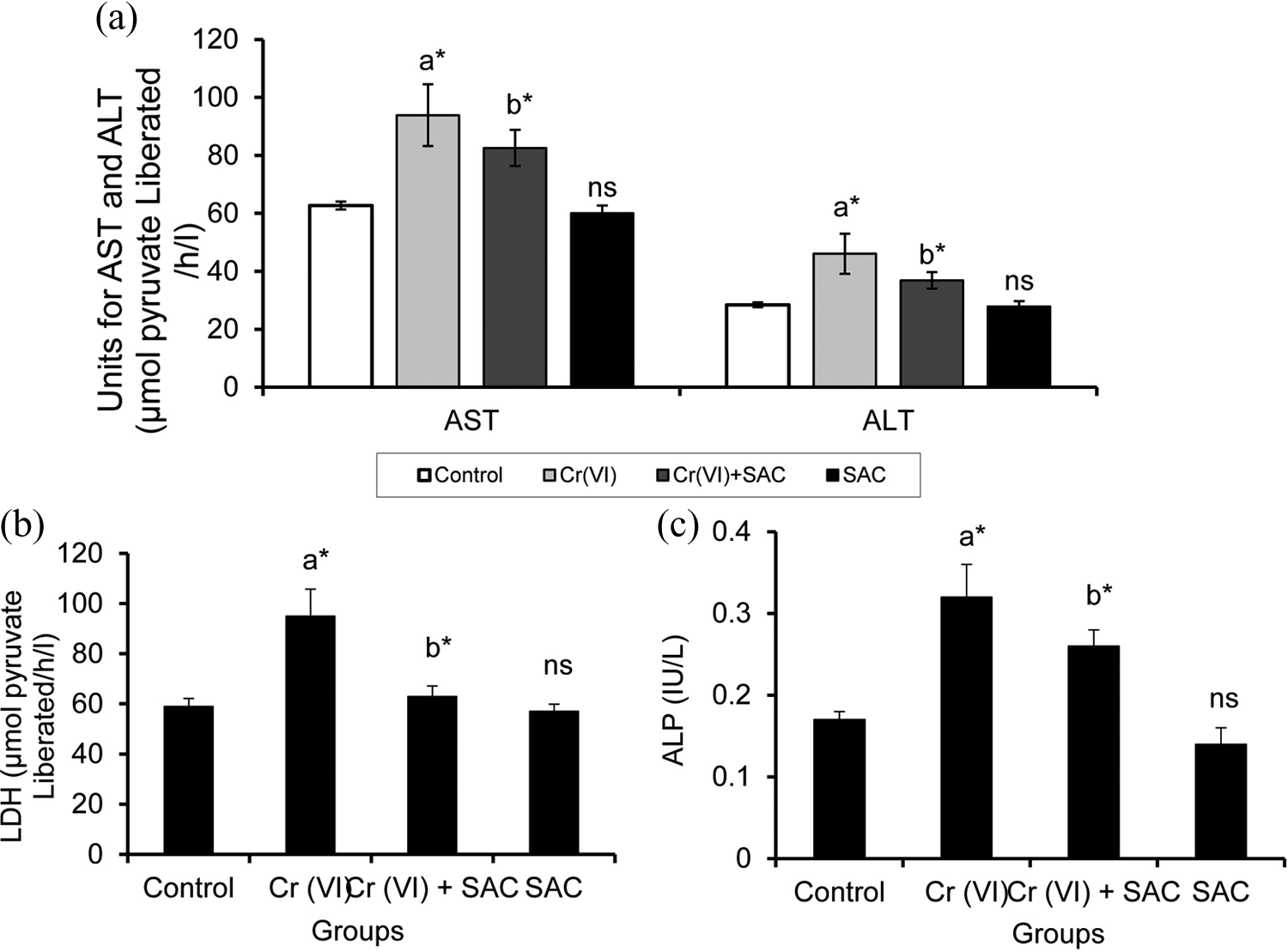
Effect of SAC on the levels of liver marker enzymes in control and experimental groups of animals. Values are given as mean ± SD for groups of six rats each. Comparisons: (a′) Control versus Cr (VI) and SAC, (b′) Cr (VI) versus Cr (VI) + SAC. *
Histological analysis
The purpose of histological staining methods by H&E is to visualize and differentiate between tissue components in normal and pathological conditions. The control liver shows the normal architecture of hepatocytes with visible central vein, nuclei, and regular arrangement of hepatocytes, as shown in Figure 2(a). Cr (VI)-induced rats show more necrotic changes around the central vein of the liver with dilation around the central axis, the centrilobular and periportal areas of rat liver showing vacuolation, as well as pronounced degenerative changes in hepatocyte was observed, as shown in Figure 2(b). Cr (VI)-administered and SAC-treated liver shows architecture with more regenerative areas of hepatocytes and very few necrotic regions visible, as shown in Figure 2(c). SAC-alone-treated rat liver shows protection of necrosis and regular arrangement of hepatocytes, as shown in Figure 2(d).

Effect of SAC on histopathological analysis in control and experimental groups of animals. Histopathological observation of liver tissue in chromium- and SAC-treated experimental groups of rat. (a) Tissue of normal rat. (b) Cr (VI)-induced rats (arrows indicate the vacuolation, as well as pronounced degenerative changes). (c) Cr (VI)-administered and SAC-treated rat liver (arrows indicate the regenerative areas of hepatocytes and very few necrotic regions visible).(d) SAC-alone treated rats. Magnifications ×20. SAC: S-allyl cysteine.
Effect of SAC on oxidants
Figure 3 depicts the levels of LPO and OH• in control and experimental groups of rats. The elevated level of LPO and OH• was observed in Cr (VI)-induced (group II) rats when compared to (group I). The LPO and OH• levels were found to be significantly reduced in SAC and Cr (VI)-induced rats (group III). No significant alterations were observed in SAC-alone-treated rats (group IV).

Effect of SAC on the status of oxidants during Cr (VI)-induced hepatotoxicity in control and experimental groups of animals. Values are expressed as mean ± SD. Comparisons: (a′) Control versus Cr (VI) and SAC, (b′) Cr (VI) versus Cr (VI) + SAC. *
Effect of SAC on enzymic antioxidants
Table 1 shows the antioxidant enzymatic profile of SAC. The levels of SOD, CAT, GPx, and GR were determined as a state to study the scavenging role of SAC, and these enzymic antioxidants are responsible for scavenging the toxic metabolites generated by free radicals. As shown in Table 1, there is significant reduction in the activities of antioxidant enzymes in the Cr (VI)-induced group when compared to group I, whereas the SAC- and Cr (VI)-induced rats (group III) had values restored to close to normal levels. The levels of these GSH-dependent enzymes were found to be significantly decreased in group II when compared to group I animals. Administration of SAC significantly elevates the levels of antioxidants in group III.
Effect of SAC on the status of enzymic and nonenzymic antioxidants in control and experimental groups of rats.

SAC: S-allyl cysteine; SOD: superoxide dismutase; CAT: catalase; GPx: glutathione peroxidase; GR: glutathione reductase; GSH: glutathione; ns: nonsignificant. Units: SOD activity is expressed as units per minute per milligram of protein, CAT as micromoles of H2O2 consumed per minute per milligram of protein, GPx as micromoles of GSH oxidized per minute per milligram of protein, GR as micrograms of NADPH oxidized per minute per milligram of protein, GSH is expressed as micromoles of GSH per milligram of protein. Values are expressed as mean ± SD for six rats in each group.
aComparison: Control versus Cr (VI).
b
cComparison: Cr (VI) versus Cr (VI) + SAC.
Effect of SAC on lysosomal enzymes
The activities of lysosomal enzymes (β-glucuronidase, β-

Effect of SAC on the status of lysosomal enzymes in control and experimental groups of animals. Results are expressed as mean ± SD for six rats in each group. Statistical significance at *
Immunohistochemical expression of TNF-α and NF-κB
The identification of NF-κB, a key player in the pathogenesis of inflammation, is highly activated at sites of inflammation in diverse disease and can induce a transcription of proinflammatory cytokines, Matrix Metalloproteinases (MMPs), and iNOS.
34
Immunohistochemical analyses of TNF-α and NF-κB (p65) in the control and Cr (VI)-induced animals are represented in Figures 5 and 6, respectively. Induction with Cr (VI) (group II) increased the expression of TNF-α and NF-κB (p65), which is shown in dense brown color (Figures 5b and 6b) Administration of SAC (group III) extensively abridged the expression of TNF-α and NF-κB (p65; Figures 5c and 6c). Control and SAC-treated rats showed a few positive expression of TNF-α and NF-κB (p65). The quantification of TNF-α and NF-κB (p65) is summarized in Figures 5(e) and 6(e), respectively. In Cr (VI)-induced rats, there was significant (

Effect of SAC on the expression of TNF-α in control and experimental groups of animals. (a) Control: normal expression of TNF-α, (b) Cr (VI)-induced animals: high expression of TNF-α was observed, (c) Cr (VI) + SAC: lesser degree of expression of TNF-α was noticed, (d) SAC: expression was similar to that of control. → shows the expression of TNF-α. (e) To measure the relative intensity, scoring was done as arbitrary units 4 as intensely stained, 3 as moderately stained, 2 as mild staining, and 1 as poorly stained in control and experimental groups. Values are expressed as mean ± SD for six rats in each group. Comparisons: (a′) Control versus Cr (VI), (b′) Cr (VI) versus Cr (VI) + SAC. *

Effect of SAC on the expression of p65-NF-κB in control and experimental groups of animals. (a) Control: normal expression of NF-κB, (b) Cr (VI)-induced animals: high expression of NF-κB was observed, (c) Cr (VI) + SAC: lesser degree of expression of NF-κB was noticed, (d) SAC: expression was similar to that of control. → shows the expression of NF-κB. (e) To measure the relative intensity, scoring was done as arbitrary units 4 as intensely stained, 3 as moderately stained, 2 as mild staining, and 1 as poorly stained in control and experimental groups. Values are expressed as mean ± SD for six rats in each group. Comparisons: (a′) Control versus Cr (VI), (b′) Cr (VI) versus Cr (VI) + SAC. *
Immunofluorescence expression of iNOS
The effect of SAC in the regulation of iNOS expression in liver was confirmed by immunofluorescence (Figure 7), counterstained with PI. Control cells exhibited few positive expressions of iNOS when compared to Cr (VI)-induced tissue section, which exhibited increased number of green fluorescence indicating enhanced expression of iNOS (Figure 7b). The expression of iNOS was decreased to a degree upon treatment with SAC and Cr (VI)-induced rats, which was evident from Figure 7(c). Similar expression pattern was observed in SAC-alone-treated group and the control (Figure 7d). Figure 7(e) shows the quantification of iNOS expressing cells per square millimeter.

Effect of SAC on immunofluorescent expression of iNOS in control and experimental groups of animals. Tissue sections were immunostained with the anti-iNOS antibody and FITC-conjugated secondary antibody (green) and counterstained with PI (red) for nuclear staining. (a) Control, (b) Cr (VI)-induced rats showing increased green fluorescence, (c) Cr (VI) + SAC-treated group shows reduced green fluorescence, (d) SAC-alone-treated group closely resembles as control. (e) Quantification of iNOS: the result was quantified per 0.245 mm2 of iNOS positively expressed cell. Hypothesis testing method included ANOVA followed by LSD test. Values are expressed as mean ± SD for six rats in each group. Comparisons: (a′) Control versus chromium, (b′) chromium versus chromium + SAC. *
Discussion
The present study demonstrated that administration of SAC could ameliorate hepatic oxidative stress and attenuate the histopathological damages induced by K2Cr2O7 in the rat liver. In addition, it clearly demonstrates the protective effect of SAC on liver marker enzymes, antioxidants, lysosomal enzymes, and inflammatory proteins. The oxidation state and solubility of chromium compounds determine their toxicity. In contrast to Cr (III), which is a naturally occurring form and an essential trace element for humans and others mammals, overdoses of Cr (VI) compounds are considered highly toxic. K2Cr2O7 is a hexavalent form of chromium and has been demonstrated to induce oxidative stress and is carcinogenic in nature.
Serum enzyme levels are significant markers to determine the severity of damage to particular tissue. AST and ALT are the most sensitive biomarker enzymes used in evaluation of the function and integrity of liver cells. Both enzymes are present mainly in the cytoplasm of hepatocytes. 35 Administration of Cr (VI) significantly increased the levels of these enzymes indicative of cell damage. An elevated level of LDH in the serum is a possible marker of necrotic lesions in the hepatocytes. 36 Oxidative damage caused by Cr (VI) administration may be the reason for increased release of LDH into the systemic circulation. A growing body of evidence indicates that transition metals act as catalysts in the oxidative deterioration of biological macromolecules; therefore, toxicity associated with these metals may be due to oxidative damage. 37 Administration of genotoxic Cr (VI) leads to the production of free radicals, excessive production of ROS via Cr (VI) reducing pathways may lead to cellular injury and results in LPO, via the oxidative catabolism of polyunsaturated fatty acids, which is a widely accepted mechanism of cellular injury and death. 38 LPO and free radical generation are the complex and deleterious process that are closely related to toxicity. 39 Increased LPO and decreased antioxidants status, an index of oxidative stress, were observed after Cr (VI) treatment. The increase in LPO could be due to the formation of hydroxyl radical (OH•) through Haber–Weiss type reaction catalyzed by Cr (VI). 40 Extensive LPO in biological membranes causes loss of fluidity, fall in membrane potential, increased permeability to H+ and other ions, and eventual rupture leading to a release of cell and organelle contents. 41 In our present study, the levels of LPO and OH• were elevated in the Cr (VI)-induced rats, while the levels of LPO and OH• were decreased in SAC-treated rats. SAC reduces the extent of LPO rather than inhibiting and significantly enhances antioxidant properties both in vivo and in vitro. 42,43 In addition, the compounds such as zinc and melatonin prevent LPO in the liver of diabetic rats subjected to forced exercise. 44,45 Moreover, SAC prevents the increase in MDA, probably in part by scavenging the very reactive hydroxyl and peroxy radical. 17 GSH is a metabolic regulator and putative indicator of health and known to protect the cellular system against the toxic effects of LPO and important in maintaining cellular redox status 46 and depletion of GSH is considered as a marker of oxidative stress. We observed a decreased level of GSH in Cr (VI)-induced rats. Retrieval of GSH levels in SAC-treated rats might be due to their strong antioxidant nature. Histological studies revealed that the liver displays significant histological lesions following Cr (VI) exposure, which might be due to the excessive free radical production. The increase in necrotic changes around the central vein, centrilobular, and periportal areas of rat liver showing vacuolation, were significantly attenuated by treatment with SAC.
The enzymes SOD, CAT, and the GSH system play a crucial role in the cellular defense against free radical damage.
47
SOD catalyzes the dismutation of superoxide into oxygen and hydroperoxides, thereby protecting the cells from detrimental superoxide anion. CAT is a heme-containing ubiquitous enzyme that removes the toxic-free radicals in vivo. CAT reduces hydrogen peroxide to water and molecular oxygen, thus protecting the cells from oxidative damage. GPx, a selenium containing enzyme, plays a central role in the catabolism of H2O2 and the detoxification of endogenous metabolic peroxides and hydroperoxides to their corresponding alcohols and free hydrogen peroxide to water, eventually protecting the cells from oxidative damage. GR plays a significant role in regenerating GSH from GSSG (oxidized glutathione), thus maintaining the balance between the redox couple. In the present study, decreased SOD activity observed in Cr (VI)-induced rats is due to the production of superoxide anions, and this dominates enzymatic activity, which leads to a fall in its concentration in liver tissues. SAC treatment seems to counteract the Cr (VI)-induced decline in SOD activity in liver tissues by scavenging the superoxide radicals. The decrease in the activities of CAT and GPx levels in Cr (VI)-induced rats in our study may be due to the intracellular accumulation of ROS with subsequent development of liver injury. Upon treatment with SAC, these enzymic antioxidants were recovered to normal levels, suggesting that oxidative stress elicited by Cr (VI) intoxication has been reversed due to their antioxidant activity. SOD accomplishes their antioxidant functions by its catalytic dismutation of O2•
− into H2O2, and CAT or GPx can degrade the resulting H2O2. Further studies reported that GPx principally degrades H2O2 in normal conditions; however, when H2O2 concentration increases, as in severe oxidative conditions, the contribution of CAT for degradation concomitantly increases and also chromium has been suggested as an active inhibitor of SOD activity in vitro.
48
Moreover, the LPO-mediated damage to Cu and Zn-SOD may result in the perturbation of cellular antioxidant defense mechanisms. SAC was administered to Cr (VI)-induced animals to counteract the Cr (VI)-induced decline in antioxidant activity in liver tissues, presumably by scavenging the superoxide radicals.
18
Previous studies on SAC supported our findings that it enhances the antioxidant enzymes in the circulation.
49,50
Lysosomes are membrane-bounded cytoplasmic organelles, which possess an acidic interior that contains many hydrolytic enzymes. Lysosomal glycosides are widely distributed in tissues and circulating blood cells and are responsible for intracellular breakdown of complex macromolecules. Elevated lysosomal enzymes in the extracellular fluid occur as a result of decreased lysosomal membrane stability.
51
The elevation in the activities of lysosomal enzymes might result in the increased liberation of glycoconjugates from the tissue. The activities of β-glucuronidase and acid phosphatase are associated with cell proliferation and hence used an index for the examination of the integrity of inflammatory process and the release of lysosomal enzymes into the extracellular compartment. Cathepsins that belong to the proteases families are among the most studied lysosomal hydrolases. An essential function of cathepsins outside the lysosomal compartment includes degradation of the extracellular matrix or induction of fibroblast invasive growth when secreted into the extracellular space.
52
The increased activities of β-glucuronidase, β-
TNF-α is an important endogenous mediator of hepatotoxicity in several experimental liver injuries through its direct cytotoxicity, NO production, and the triggering of an inflammatory cascade mediated by means of a wide variety of proinflammatory cytokine generated mainly through NF-κB activation. 53,54 It has been well established that both oxidative stress and abnormal production of cytokines, especially TNF-α, play important etiological roles in the pathogenesis of liver diseases. In our present study, the immunohistochemical expression of TNF-α was increased during induction with Cr (VI). Treatment with SAC showed a decreased expression of TNF-α in rats. TNF-α has been shown to activate NF-κB by stimulating production of superoxide anions and H2O2 and causing a rapid depletion of GSH levels in in vitro and animal experiments. 55,56
NF-κB is considered as primary oxidative stress response transcription factors that function to enhance the transcription of the variety of genes. ROS are involved in signal transduction leading to NF-κB activation, which has been an implication in the regulation of gene transcription. 57 In previous studies, OH• has been shown to activate NF-κB, which stimulates the inflammatory process. Inhibition of the expression of NF-κB in hepatotoxicity of curcumin leads to reduce the risk. 58 In this study, production of cytokines and inflammatory mediators by NF-κB response genes may reflect the degree of inflammation and damage in Cr (VI)-induced rats. Our result showed that SAC and Cr (VI)-induced rats significantly reduced the expression of NF-κB as compared with Cr (VI)-induced rats. It might be possible that SAC has anti-inflammatory effect through inhibition of NF-κB activation and reduction of TNF-α expression.
NO has a series of effects on a variety of biological processes, and it is uncertain whether it is beneficial or detrimental in the liver injury induced by hepatotoxins. NO is also an important moderator implicated in diverse functions associated with cytoprotection or injury in the liver due to its pathophysiological property.
59
NO, which is produced by iNOS, is an extremely reactive oxidant that plays a role in some physiological processes such as vasodilation, neurotransmission, and nonspecific host defense.
60
NO is produced by parenchymal and nonparenchymal liver cells from
The results obtained from this study clearly demonstrated that SAC potentially scavenges free radicals generated by the induction of Cr (VI) and also restored the enzymic antioxidants, histological features. On the other hand, it is capable of reducing the inflammation via decreasing the expressions of NF-κB, TNF-α, and iNOS. SAC reduced the incidence of apoptosis, induced by Cr (VI) in the hepatocytes. Hence, it can act as a protective agent against chromium-induced toxicity, and more studies are warranted in other hepatotoxicants.
Footnotes
Acknowledgement
The authors thank Wakunga Pharmaceutics, Japan, for the generous gift of SAC used in this study.
Author contribution
Suresh Kumar Anandasadagopan and Chitra Sundaramoorthy contributed equally to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
