Abstract
This study aimed to investigate the implication of some apoptotic and lipid peroxidation markers in preeclampsia (PE). A total of 25 women with PE and 25 age- and parity-matched normal pregnant women were enrolled in this study. The malondialdehyde (MDA) level, caspase-9 activity and the percentage of DNA fragmentation were significantly higher in placental tissue of PE than in control women. The serum level of MDA was significantly elevated in women with PE having delivery by cesarean section (CS) than in women with PE having vaginal delivery. In vitro study demonstrated that the addition of 0.5 mM Fe2+ and 0.1 mM ascorbate caused increase in the production of MDA level in placental tissue with PE than normal placentas, and vitamin E (100 µM) caused lower inhibition of in vitro lipid peroxidation in placental tissue with PE when compared with normal tissue. The activity of caspase-9 and percentage of DNA fragmentation were associated with the severity of the PE and both could differentiate between PE and control women with 88% and 100% sensitivity and 96% and 100% specificity, respectively. The activities of caspase-8 and/or -9 were positively correlated with the maternal age but only caspase-8 was negatively correlated with neonatal birth weight and placental weight. In conclusion, the elevations of MDA, caspase-9 activity and the percentage of DNA fragmentation in the placentas of women with PE implicate the involvement of lipid peroxidation and apoptosis in PE. The placenta represents a considerable source of the elevated circulating MDA in PE.
Introduction
Preeclampsia (PE) is a syndrome defined by the new onset of hypertension in the second half of pregnancy that is generally, but not always, accompanied by proteinuria. It is a multisystem disorder characterized by uteroplacental and maternal endothelial dysfunction. PE complicates 3–5% of all pregnancies and continues to be a major cause of morbidity and mortality both for the mother and the infant. It is a significantly greater problem than gestational hypertension alone, as the latter by definition does not affect maternal organ systems. While in recent times the understanding and management of this condition have improved, the exact cause of PE has not been fully defined. 1,2 Increased awareness of the long-term complications of PE and its associations with vascular disease in later life, 3 –5 both for the mother and the infant of low-birth weight, is of major concern. 6,7
Oxidative stress is described as an imbalance in the production of reactive oxygen species and the ability of antioxidant defenses to scavenge them. In PE and some pathologic pregnancies, a heightened level of oxidative stress is encountered. 8 Lipid peroxide formation, a marker of oxidative stress, is increased during pregnancy and PE. The lipid peroxide concentration of microvillus membrane can be quantified by malondialdehyde (MDA) level – the most widely used lipid peroxidation marker in syncytiotrophoblast plasma membranes. The concentrations of MDA are highly correlated with those of isoprostane, another previously described specific marker for oxidative stress in PE. 9
Apoptotic DNA fragmentation is a key feature of apoptosis, a type of programmed cell death. Apoptosis is characterized by the activation of endogenous endonucleases with subsequent cleavage of chromatin DNA into internucleosomal fragments, which is roughly equal to or multiples of 180 base pairs (360, 540, etc.). DNA fragmentation is a secondary consequence, rather than an integral cause, of apoptosis. 10 Apoptosis in target cells can be evaluated by detecting the pattern of DNA fragmentation by agarose gel electrophoresis.
Apoptosis can be initiated by toxicants, drugs or perturbation of the cellular homeostasis in some disease conditions. Apoptosis has been implicated in the pathogenesis of many disease states including cancer, neurological disorders, autoimmune diseases and aging. 11 Its rate is thought to be increased in PE as a result of placental oxidative stress. 1,12 Apoptosis signaling events can roughly be grouped into four different phases: the premitochondrial initiation phase, during which a cell receives the death signal at the plasma membrane; the mitochondrial decision phase; the postmitochondrial amplification phase, which leads to activation of different caspases that act in concert to propagate the death signal; the late (degradation) phase, during which cellular proteins are proteolytically cleaved with the evidence of DNA fragmentation. 13 Caspases comprise a structurally related group of cysteine proteases that share a dominant specificity for cleaving peptide bonds following Asp residues. 14 They are roughly classified into initiator caspases, such as caspase-8 and -9, and effector caspases, such as caspase-3 and -7. 13
Therefore, the aim of the present study was to investigate the changes in the activities of caspase-8 and -9, percentage of DNA fragmentation and MDA level as markers of apoptosis and oxidative stress in preeclamptic women and the possible interplay between these markers and the maternal risk factors and perinatal outcome along with the response of the placental tissue to prooxidants (iron chloride (FeCl2) and low dose of ascorbate) and the antioxidant α-tocopherol.
Patients and methods
A total of 25 preeclamptic primigravidas and 25 randomly selected age- and parity-matched normal pregnant women, who attended the outpatient clinic of the Gynaecology Department at Kasr El-Aini Hospital, Cairo University, Cairo, Egypt, were enrolled in this study. Informed consent was obtained from all participants. The study protocol was approved by the institute ethics committee and was conformed by the ethical guidelines of the 1975 Helsinki Declaration.
The diagnosis of PE was made by strict criteria according to Chesley. 15 These criteria included onset of hypertension during the third trimester (≥140/90 mmHg on two occasions), detectable urinary protein (≥1+ by dipstick or ≥300 mg/24 h) and edema. Women with a history of hypertension before 20 weeks gestation and patients whose pregnancies were complicated by diabetes, peripheral vascular disease, chronic renal disease, multifetal gestation or major fetal anomalies were excluded from this study.
The PE group (group 1) was subdivided into two subgroups: group 1(A), which included 12 patients with mild PE, and group 1(B), which included 13 patients with severe PE. The criteria for severe PE were determined by systolic blood pressure (SBP) >160 mm Hg or diastolic blood pressure (DBP) >110 mmHg on two separate occasions: >4 h apart in the presence of repeated proteinuria (++ or more) and at least 4 h apart. 16
Materials
Maternal venous blood samples (∼5 ml) and human-term placentas were collected immediately after delivery from each woman. Placental samples were taken away from areas with infarction or calcification. Placentas from cases with prolonged rupture of membranes were avoided because of chorioamnionitis. Small pieces of placental tissues were immediately frozen at −80°C till assays.
Laboratory assays
Estimation of lipid peroxides in serum and placental tissues
Lipid peroxidation was estimated by measuring thiobarbituric acid reactive substances mainly MDA in placental tissues and serum according to the method of Esterbauer and Cheeseman. 17 The level of MDA in the serum and the placental tissue samples was expressed in micromoles per liter and nanomoles per milligram protein, respectively. The protein concentration in tissue extracts was measured by the method of Bradford. 18
In vitro lipid peroxide formation
Placental tissues were homogenized in four volumes of 0.15 M sodium chloride (NaCl)–10 mM sodium phosphate buffer, pH 7.4, and filtered through a gauze. The filtrate was diluted with 0.15 M potassium chloride (KCl)–10 mM Tris-HCl buffer, pH 7.4, so as to give a final protein concentration of 1 mg/ml. According to the method of Buege and Aust, 19 formation of lipid peroxide in relation to time (after 1, 2 and 3 h of incubation at 37°C) was examined in placental tissue with PE compared with the normal placenta. Applying the same method, lipid peroxidation response to the addition of prooxidants (0.5 mM FeCl2, 0.1 mM ascorbate) and antioxidant (100 µM α-tocopherol) was determined after 2 h of incubation at 37°C. For each placental sample, a control was run simultaneously without addition of prooxidants or antioxidants. The results were expressed as percentage stimulation or percentage inhibition calculated as follows: % stimulation (or %inhibition) = (nM MDA of test sample/nM MDA of its control) × 100.
Assessment of apoptosis in placental tissues
Determination of activities of caspase-8 and -9
The activities of both caspase-8 and -9 in placental tissues were determined using Apo Target™ colorimetric assay kits (BioSource Europe S.A., Nivelles, Belgium) according to the manufacturer’s instructions. Each kit includes substrate and optimized buffers. The substrate was composed of chromophore p-nitroaniline and a synthetic tetrapeptide IETD (ILe-Glu-Thr-Asp) or LEHD (Leu-Glu-His-Asp) in caspase-8 or -9 kits, respectively. Upon cleavage of the substrate by caspase-8 or -9, free p-nitroaniline was liberated and quantified spectrophotometrically at 405 nm.
Determination of percentage of DNA fragmentation
The percentage of DNA fragmentation in placental tissues was measured by diphenylamine (DPA) assay and confirmed by agarose gel electrophoresis according to the method of Perandones et al. 20 Placentas were mechanically dissociated in hypotonic lysis buffer. The cell lysate was centrifuged at 13,000g for 15 min, then the supernatant containing small DNA fragments was separated immediately and half the supernatant was used for gel electrophoresis. The other half as well as the pellet containing large pieces of DNA were used for the colorimetric determination by DPA assay. Briefly, 200 µl perchloric acid (0.5 M) was added to the pellet (reconstituted in 200 µl hypotonic lysis buffer) containing native DNA and to the supernatant containing fragmented DNA followed by the addition of two volumes of a solution containing 0.088 M DPA, 98% v/v glacial acetic acid, 1.5% v/v sulfuric acid and 0.5% acetaldehyde solution. The samples were kept at 4°C for 48 h. The color developed was then spectrophotometrically measured at 575 nm. The percentage of DNA fragmentation was expressed by the formula:
Percent of DNA fragmentation = absorbance of supernatant × 100/2 × (absorbance of supernatant + absorbance of pellet)
Measurement of the serum level of uric acid
The serum level of uric acid was determined by the enzymatic colorimetric method 21 using the kit provided by Randox Laboratories Ltd. (Crumlin, UK) according to the manufacturer’s instructions. The results were expressed as milligrams per deciliter.
Statistical analysis
The statistical data are reported as the mean ± SD and percentages when appropriate. Differences between the two groups were evaluated with student’s t test or chi square test, as appropriate (Table 1). A comparison of the variables between more than two groups was performed using a one-way analysis of variance (ANOVA) test followed by Duncan’s test for inter-group comparisons as in Table 2. The Pearson correlation coefficient was used to determine correlations between different variables (Table 3 and Figure 2). A receiver operating characteristic (ROC) analysis was used to determine the optimum cutoff value for the studied markers (Table 4). The accuracy is represented using the terms sensitivity and specificity. ps < 0.05 were consideredstatistically significant. All statistical calculations were performed using the computer program SPSS (Statistical Package for the Social Science; SPSS Inc., Chicago, Illinois, USA) version 15 for Microsoft Windows.

In vitro formation of lipid peroxides induced by 0.5 mM ferrous chloride or 0.1 mM ascorbate and the inhibition induced by 100 µmol α-tocopherol in normal (n = 25) and preeclamptic (n = 25) placental tissues. Incubation time = 2 h.*, ** and # indicate statistical significance from the corresponding controls at p < 0.05, 0.01 and 0.001, respectively.

Correlation between the serum levels of uric acid and MDA in the PE group.
Characteristics of the patient and control groups.

Values are presented as mean ± SD and as range (between brackets).
Proteinuria and mode of delivery are presented as %. *Statistical significance from the control group using student's t test. The values are considered statistically significant at p < 0.05. CS: cesarean section.
Comparison of different variables in the normal control, mild, and severe preeclamptic groups.
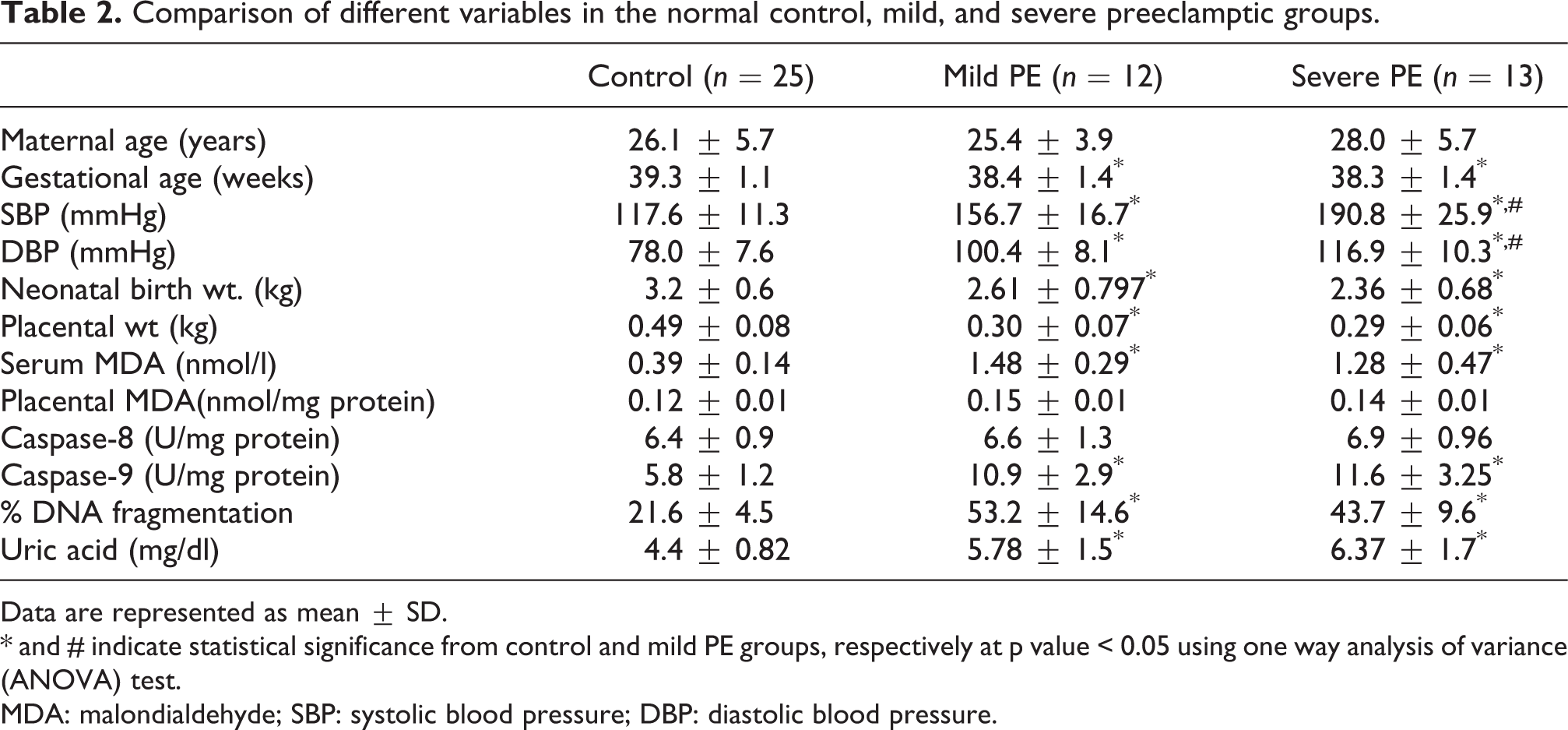
Data are represented as mean ± SD. * and # indicate statistical significance from control and mild PE groups, respectively at p value < 0.05 using one way analysis of variance (ANOVA) test. MDA: malondialdehyde; SBP: systolic blood pressure; DBP: diastolic blood pressure.
Correlation between clinical data, pregnancy outcome, and laboratory data in the preeclamptic group.

MDA: malondialdehyde; SBP: systolic blood pressure; DBP: diastolic blood pressure.
Bold characters indicate significant values at r = ±0.33 and p < 0.05. Gest., gestational; Mat., maternal.
Sensitivity and specificity of the different laboratory data in PE placentas as calculated by the ROC curve.

AUC: area under ROC curve; MDA: malondialdehyde.
Results
A total of 50 primigravidas were included in this study; 12 were with mild PE, 13 with severe PE and 25 age-matched normotensive control women. The basic characteristics of the patients and controls are shown in Table 1. In Table 1, the mean admission SBP and DBP blood pressures are significantly higher in the PE group than the control women (p < 0.001, p < 0.01, respectively). The gestational age, neonatal birth weight and placental weight are lower in the women with PE compared with the control group (p < 0.01).
With the exception of caspase-8 activity, the expression of apoptotic markers caspase-9, percentage of DNA fragmentation (each p < 0.001) and the lipid peroxidation product; serum (p < 0.001) and placental MDA (p < 0.05) and the serum level of uric acid (p < 0.05) were significantly higher in the PE group than the control group.
According to the mode of delivery, the serum level of MDA was shown to be higher in women with PE having delivery by cesarean section (CS) than in women with PE having vaginal delivery (mean ± SD = 1.4 ± 0.37 vs. 1.03 ± 0.33, respectively) at p < 0.05. Also, the serum level of MDA was higher in the women with PE having vaginal delivery than the control women who had vaginal delivery (mean ± SD= 1.03 ± 0.33 vs. 0.39 ± 0.14, respectively) at p < 0.001. Meanwhile, the mode of delivery did not cause any statistically significant change in the placental level of MDA (data not shown).
The results of the in vitro lipid peroxide formation showed higher levels of MDA formation in placental tissue with PE than in normal placentas when incubated at 37°C for 1, 2 or 3 h (data not shown). After 2 h of incubation period at 37°C in the presence and absence of prooxidants (Figure 1), the percentage stimulation by 0.5 mM FeCl2 was significantly higher in the placental tissue with PE (mean = 187.9 ± 31.68%) than in the normal placenta (mean = 139.8 ± 25.6%) at p < 0.01. Furthermore, a significant increase in percentage stimulation was induced by 0.1 mM ascorbate in the placental tissue with PE (mean = 165.2 ± 50.06%) compared with the normal placenta (mean = 130.18 ± 32.06%) at p < 0.05. Addition of 100 µM α-tocopherol caused a significantly lower percentage inhibition in the placental tissue with PE (mean = 80 ± 12.77%) than in the normal placenta (mean = 52.27 ± 10.57%) at p < 0.001.
Blood pressure, gestational age, neonatal birth weight and placental weight as well as serum MDA, caspase-9 activity, percentage of DNA fragmentation and serum level of uric acid (Table 2) were significantly different in women with mild and severe PE compared with the normal controls. Interestingly, severe PE group did not differ significantly from the mild group regarding neonatal birth weight, placental weight and other biochemical measures (Table 2).
Correlation study among clinical data, pregnancy outcomes and laboratory data (Table 3) revealed that, whereas the maternal age was positively correlated with the activities of caspase-8 (r = 0.53, p < 0.05) and caspase-9 (r = 0.36, p < 0.05) in the PE group, it was negatively correlated with the percentage of DNA fragmentation in the PE groups (r = −0.33, r = −0.43, p < 0.05, respectively) and control (data not shown). Caspase-8 was negatively correlated with neonatal birth weight (r =
In the placental tissue with PE, ROC curve analysis (Table 4) showed that the percentage of DNA fragmentation was the most sensitive (100%) and specific (100%) followed by caspase-9 activity: 88% sensitivity and 96% specificity.
Discussion
Despite intensive efforts to find mechanisms and markers that induce PE, no specific etiological factor has been identified. 12 Oxidative stress occurs when hypoxic placental tissues are reoxygenated as a result of defective remodeling of uterine spiral arteries in PE. 8 The product of attack by free radicals on polyunsaturated fatty acids can be considered as a footprint of free radical activity. Our results revealed higher placental level of MDA in the preeclamptic group compared with the control group.
It was reported previously that the mode of delivery affects the rate of lipid peroxidation in the placentas of normal pregnancy because during vaginal delivery, placental anoxia may occur as a result of uterine contractions 22 and lipid peroxides may be formed from unsaturated lipids in the course of prostaglandin synthesis, which is needed for the initiation of labor in spontaneous delivery. 23 The current results suggest that the situation is different in PE, as the placental level of MDA did not significantly differ between women having vaginal delivery (40% of our cases) and those having delivery by CS. Meanwhile, there was an increase in the serum level of MDA in women with PE having delivery by CS (60% of our cases) over women with PE having vaginal delivery. Therefore, we suggested that the hyperoxic state that occurs during delivery by CS may constitute considerable source for the increased serum level of MDA in our preeclamptic group. Zhang et al. 24 and Khaw et al. 25 reported that hyperoxia per se can generate free radicals via direct mitochondrial electron transfer.
The current study revealed increase in the level of uric acid in the preeclamptic group compared with the control group. Increase in the serum uric acid level in our PE group is in harmony with other reports. The association of uric acid with PE has been known since the late 1800s as reported by Bainbridge and Roberts. 26 Uric acid is a marker of oxidative stress, tissue injury and renal dysfunction and its raised level has been suggested to be associated with an almost doubled risk of severe complications in PE including perinatal death. 27 Uric acid is the end product of purine metabolism and is synthesized by the enzyme xanthine oxidase. Hypoxia, ischemia of the placenta and cytokines such as interferon induce the expression of xanthine oxidase and therefore increase the production of uric acid. 28 Also, a significant positive correlation between the serum levels of uric acid and MDA levels was found in our preeclamptic group. Taken together, our results suggest that the increase in serum level of MDA in the preeclamptic patients represents part of the disease process. However, a partial contribution by the hyperoxic state that accompanied CS should also be considered.
To emphasize the role of placental tissue as a considerable source of elevated circulating MDA in PE, we measured the levels of MDA in PE and normal placentas after 1, 2 and 3 h of incubation at 37°C. The results revealed time-dependent increase in the production of MDA by placental tissue with PE compared with the normal placental tissue (data not shown). Additionally, our results demonstrated exaggerated response (% stimulation) of placental tissues with PE to the prooxidant effects of Fe2+ (0.5 mM) and ascorbate (0.1 mM) when compared with the corresponding normal tissue. Furthermore, the inhibition of lipoperoxide formation by α-tocopherol (100 µM) was significantly lower in placental tissue with PE than in normal placental tissue. This was attributed to higher rate of peroxidation and probably due to lower basal level of vitamin E in preeclamptic placentas.
Few studies reported the involvement of caspase-8 and -9 in PE. 1,2 Caspase-8 and -9 are mainly involved in the initiation of apoptosis. 13 Our study demonstrates higher activity of only caspase-9 in the preeclamptic than in the normal placentas. Additionally, ROC curve analysis revealed that both caspase-8 and -9 had similar sensitivity (88%) but the specificity of caspase-9 was much higher than that of caspase-8 (96% versus 36%). Since it was reported that the death receptor pathway of apoptosis was mediated by caspase-8, while the mitochondrial pathway of apoptosis was mediated by caspase-9, 29 our results demonstrate the involvement of both the caspases in the pathogenesis of PE, with the domination of mitochondrial caspase-9 signal transduction pathway.
The relationship between oxidative stress and apoptosis was explained previously. 2,12,30 In agreement with these reports, the current study revealed a positive correlation between the placental level of MDA and percentage of DNA fragmentation, which is a marker of the degradation (late) phase of apoptosis in PE. Meanwhile, the sensitivity and specificity of percentage DNA fragmentation and caspase-9 were much higher than those of placental MDA. This finding may suggest that apoptosis is more dominant than oxidative stress in the pathogenesis of PE.
In the current study, statistically significant differences were found upon comparing the groups of severe and mild PE with the normal control group. These differences included some of the investigated clinical data (SBP, DBP and gestational age) as well as some of the laboratory data (serum MDA, percentage DNA fragmentation, caspase-9 activity and serum level of uric acid). The results also demonstrated a decrease in the fetal birth weight in relation to the severity of PE.
In the context of PE, our study is one of the few reports that investigated the relationship between the activities of caspase-8 and -9, percentage of DNA fragmentation and MDA and the maternal risk factors and perinatal outcome. Pregnancy in older women was reported to be associated with many confounding factors that end with increased incidence of maternal and fetal complications. 31 Research increasingly suggests that changes in the levels of estrogen during aging may be a risk factor for many diseases. 32 Conflicting results were obtained regarding the relationship between maternal age and apoptosis. Yamada et al. 33 reported that placentas from older women overcome the age-related hypofunction by increasing the proliferative signals and reducing the apoptotic signals. Moreover, Smith et al. 34 reported the absence of any significant correlation between the maternal age and the incidence of apoptotic cells in normal placentas as demonstrated by microscopic examination.
Interestingly, our results revealed that the percentage of DNA fragmentation was negatively correlated with the maternal age in the preeclamptic as well as the control group. This may indicate that apoptosis decreases with the advance of the maternal age regardless of the presence or absence of PE. Meanwhile, the positive correlation between the activities of caspase-8 and -9 with the maternal age in the preeclamptic group and its absence in the normal control group may indicate the presence of unidentified factors in placentas of preeclamptic old women that abort the apoptotic cascade before reaching its late stage. So, further studies are recommended to elucidate the precise mechanism by which maternal aging affects the apoptotic and proliferative activity of placental tissue with PE.
Considering gestational age, our results demonstrated its decrease in the PE group compared with the control group. This confirms the previously reported correlation between PE and perinatal outcome, 35 which can be attributed to the enhanced apoptotic process in preeclamptic placental cells during late stages of pregnancy as suggested by Wang et al. 36 and loss of the normal placental protection that decrease the lipid peroxidation process with the advance of gestational age as reported by Takehara et al. 37 Our results support both mechanisms through (1) the negative (though statistically insignificant) correlation between the activity of both caspases (-8 and -9) and gestational age, (2) the positive correlation between placental MDA and gestational age in the PE group.
Apoptosis is a potentially important determinant of placental size and function, which is crucial for growth and development of the fetus throughout gestation. 38 Upon examining the relationship between apoptosis and placental and fetal growth, our results demonstrated a negative correlation between the activities of each of caspase-8 and -9, and both weights in the PE group. This result is consistent with the previously reported correlation between apoptosis and intrauterine fetal death in mice. 38 Meanwhile, the absence of the expected significant reciprocal relationship between fetal birth weight and placental level of MDA may indicate that the fetus was protected from the maternal oxidative stress but not from the apoptotic process.
Although sex difference and long-term perinatal outcome are not the topics studied in our study, it is worth mentioning that evidence suggests interplay of hormonal modulation and genetically determined apoptotic mechanisms, through which perinatal females may be afforded a level of protection against perinatal insult that is greater than that for perinatal males . 39 Clinical findings show that male infants are not only more vulnerable to perinatal insult but they also suffer more long-term cognitive deficits when compared with females with comparable injury. 39–41 This may be because the activation of the caspase-dependent apoptotic pathway in females may afford greater protection, potentially due to the actions of X-linked inhibitor of apoptosis (XIAP) within this pathway. 39,42,43
In conclusion, the elevations of MDA, caspase-9 activity and the percentage of DNA fragmentation in the placentas of women with PE implicate the involvement of lipid peroxidation and apoptosis in PE. The placenta represents a considerable source of the elevated circulating MDA in PE. The negative correlation of maternal age with the percentage of DNA fragmentation and its positive correlation with the activities of caspase-8 and -9 in placentas of women with PE support the need for further studies. Further studies are also recommended to test modulation of apoptosis by drugs inhibiting caspases and/or affecting the release of the mitochondrial cell death effectors in a model of preeclamptic placentas.
Footnotes
Conflict of interest
The authors declared no conflicts of interest.
Funding
The present work was partially supported by the financial assistance provided by Faculty of Medicine, Cairo University, Cairo, Egypt.
