Abstract
The testis is sensitive to cadmium, but studies investigating cadmium-induced testicular injury have not yet clearly revealed the underlying mechanisms. This study aimed to investigate the injurious effects of cadmium on rat testes and the role that autophagy plays in this process. Wistar rats were randomly divided into four groups and intraperitoneally injected with 0.2 (low), 0.4 (middle), and 0.8 mg/kg·body weight (high) cadmium chloride for 5 weeks, while the control rats were injected with equal volume of saline. Rats exposed to cadmium appeared inactive and had reduced body weights and increased testicular organ coefficients at the end of treatment compared with control rats. Atomic absorption results showed that cadmium levels increased with increased cadmium exposure. Hematoxylin and eosin staining of testicular sections showed seminiferous tubular atrophy, decreased pipe diameter, spermatogonial stem cells falling off the inner lining, and reduced germ cell layers of disorderly arrangements in cadmium-treated rats. Immunohistochemical and western blot results both showed that levels of the autophagy-related proteins Beclin1 and microtubule-associated protein 1 light chain 3B (LC3B) increased with increased cadmium exposure. We also found that LC3B-II and calcium-sensing receptor (CSR) levels in cadmium-exposed rats significantly increased. By immunofluorescence, we found that the percentage of cells that expressed the CSR was significantly higher in LC3B-positive than LC3B-negative cells. Together, our results showed that cadmium accumulates in the testes causing testicular injury, which may be related to increased autophagy levels. Furthermore, calcium disorders associated with the CSR may reveal a potential way to activate autophagy.
Introduction
Cadmium is a commonly used chemical in agricultural and industrial operations and is released into the environment causing atmosphere, soil, and water pollution. Due to rapid industrial and agricultural development, 1,2 cadmium pollution has become a widespread issue, as exposure is a risk for both workers in these certain occupations and the general population, for example, from smoking or rice consumption.
Cadmium can accumulate in the body for up to 40 years, so it can harm human health over an extended period of time. 3 –5 Studies have found that low-dose cadmium exposure may increase thyroid hormone levels and prostate cancer mortality 5 , influence female reproductive health and embryos development. 6,7 Many studies have established that reproductive organs are highly sensitive to cadmium, 8 –11 which can influence levels of reproductive hormone, reduce sperm quality, increase sperm cell apoptosis, and even lead to male infertility.
However, studies of the mechanism underlying cadmium-induced testes damage are limited and lack consensus. 12,13 Some researchers have proposed that the apoptosis pathway and oxidative stress account for the pathological mechanisms, while others argue that it may involve effects on metabolism. 14,15 Other studies have found that autophagy can be used as a sensitive indicator of renal injury caused by low-dose cadmium exposure, which can activate autophagic cell death in the liver. We hypothesized that cadmium may damage the reproductive system through the same pathway. Currently, studies of the mechanism whereby autophagy is induced by cadmium-mediated toxicity in the testis are insufficient, and as this cadmium-sensitive organ is essential for human reproduction, it is of great importance to better characterize this response and to develop more effective ways to treat cadmium-mediated testes injury.
Autophagy 16,17 is a collection of processes that delivers a portion of the cytoplasm to lysosomes for degradation, eliminating of damaged organelles and misfolded proteins to maintain cellular homeostasis. 18,19 Excessive autophagy can damage intestinal epithelial cells, and it is possible that autophagic cell death can cause physiologically relevant injury. 20,21 Beclin1 can promote autophagy, and microtubule-associated protein 1 light chain 3B (LC3B) is a protein marker of autophagic activity. Together, the expression of Beclin1 and LC3B can reflect the autophagic state of a cell. 22 –24 Previous studies have suggested that calcium ions are involved in protein metabolism, and it was subsequently reported that disrupting homeostasis could induce autophagy and a calcium-sensing receptor (CSR) regulates intracellular calcium levels, thus these reports suggest a link between CSR and autophagy. This study explored the role of autophagy in mediating the toxic effects of cadmium in the testes by analyzing Beclin1, LC3B, and CSR expression.
Materials and methods
Experimental animals
A cohort of 40 male Wistar rats purchased from the Laboratory Animal Center of Jilin University (Changchun, China) with weights ranging from 220 g to 260 g were used in this study and were provided food and water ad libitum in cages with an indoor temperature of 21 ± 2°C, a relative humidity of 30–35%, and12-h light/12-h dark cycles. Rats were randomly divided into four groups (10 each): control, low, middle, and high (0.0 (vehicle), 0.2, 0.4, and 0.8 mg/kg·body weight, respectively) cadmium treatments. All operations were carried out in strict accordance with the care and use of laboratory animal guidelines established by the National Research Council and were supported and approved by the Animal Ethics Committee of Jilin University.
Establishing a rat model of cadmium-induced testis injury
Wistar rats received daily intraperitoneal injections of cadmium chloride in saline solution at the concentrations of 0.2, 0.4, or 0.8 mg/kg·body weight in the experimental groups, and saline of equal volume was injected into control rats. All rats were treated 5 days/week for 5 weeks.
Animal-based testing
Rats were anesthetized by intraperitoneal injection of 10% chloral hydrate, and bilateral testes were quickly removed after cervical dislocation. Testes were immediately weighed and the organ index was calculated using the following formula: visceral index = visceral weight/body weight × 100.
Detection of testes-accumulated cadmium
Fresh testicular tissue (0.8 g) was weighed, and then 8 ml concentrated nitric acid and 2 ml perchloric acid were added, mixed, and allowed to react overnight in a fume hood. The next day, the mixture was heated on a hot plate until the liquid became colorless and transparent, and then distilled water was added to a total volume of 5 ml and mixed well. Finally, an atom absorption spectrophotometer (AA-6300; Shimadzu, Inc., Kyoto, Japan) was used to measure cadmium concentration in the samples. Cadmium contents accumulated in the testes are expressed as cadmium content (μg)/tissue weight (g).
Histological evaluation
Testicular paraffin sections were stained with hematoxylin and eosin (H&E). 25 Histopathological changes in the testes were observed using a light microscope (Olympus, Tokyo, Japan).
Immunohistochemical analysis of Beclin1, LC3B, and CSR
Paraffin slices were dewaxed and hydrated following standard protocols, incubated at 98°C in citron acid solution for 5 min for antigen retrieval, reacted with oxidation enzyme blocking agent at room temperature for 12 min, washed three times with phosphate buffer saline (PBS), closed with non-immune animal serum for 30 min, and finally incubated with anti-Beclin1 antibody (1:150; ProteinTech, Chicago, Illinois, USA), anti-LC3B antibody (1:200; ProteinTech), and anti-CSR antibody (1:200; ProteinTech) at 4°C overnight. The following day, slides were washed in PBS three times, incubated with biotinylated secondary antibodies for 30 min at room temperature, washed with PBS three times, reacted with streptomycin avidin peroxidase for 30 min, washed with PBS three times, stained with 3,3’-diaminobenzidine (MaiXin, Fuzhou, China) and then with hematoxylin for 2 min, incubated with weak ammonia for a few seconds until the slices turned blue, dehydrated and brightened using standard protocols, and finally fixed using rhamsan gum. The integrated optical density and tissue area were analyzed using Image Pro Plus 6.0 (Media Cybernetics Inc., Rockville, Maryland, USA); the mean optical density was used to assess the expressions of autophagy-related proteins in the testes from the different groups. The mean optical density was calculated by the following formula: mean optical density = integrated optical density/tissue area.
Double immunofluorescence labeling of testes to detect LC3B vesicles and CSR
Paraffin slices were dewaxed with xylene and hydrated using an alcohol gradient and then incubated in 98°C citrate solution for antigen retrieval for 5 min. Slides were incubated with 10% donkey serum at room temperature for 30 min, washed with PBS three times, incubated with anti-LC3B (1:50; ProteinTech) and anti-CSR (1:50; ProteinTech) antibodies at 4°C overnight, and then washed with PBS three times. A mixture of fluorescent-conjugated secondary antibodies, including AlexaFluor 568 donkey anti-mouse (1:100; Life Technologies, Carlsbad, California, USA) and AlexaFluor 488 donkey anti-rabbit (1:100 dilution; Life Technologies) antibodies, were incubated with the slides at room temperature for 1 h, following the slides were washed with PBS three times, stained with 4’,6-diamidino-2-phenylindole (Sigma, St Louis, Missouri, USA) for 5 min, and sealed with 5% glycerol. Images were captured by laser confocal microscopy (Olympus).
Western blotting method to detect Beclin1 and LC3B expression
Total testicular proteins were extracted, and 15% separation glue and 5% concentrated glue were prepared. A 30-μg protein sample was pulsed into each sample well, and samples were subjected to 80 V electrophoresis and 100 mA transfer to film for 2 h (Bio-Rad, Hercules, California, USA). Skim milk was dissolved in Tris-buffered saline with tween-20 (TBST) to block the films for 2 h, and films were then incubated with anti-LC3B, anti-Beclin1, and anti-β-actin antibodies (1:2000; ProteinTech) at 4°C overnight, washed at room temperature with TBST, incubated with horseradish peroxidase-conjugated goat anti-rabbit/mouse antibodies (1:2000; ProteinTech) at room temperature for 45 min, and then washed three times as described above. Enhanced chemiluminescence (ECL) glow liquid was added in the darkroom for exposure and the images were fixed. Quantity One software (Bio-Rad) was used to analyze grayscale values. Mean of values of repeated measurements were divided by the reference β-actin value, which represented the quantitative gray value of the target proteins.
Statistical analyses
The software package SPSS version 20.0 (SPSS Inc., Chicago, Illinois, USA) was used for data analysis; all data are expressed as mean ± standard deviation (SD). Variance was analyzed by single-factor analysis followed by Fisher’s least significant difference test. The threshold for statistical significance was set at p < 0.05.
Experimental results
General status and the body weight of rats
Body weights of rats in all four groups increased over the course of the experiment. However, after 3 weeks of treatment, weights of middle and high-dose groups were statistical lower than that of the control group. After 4 weeks of cadmium treatment, the rat cadmium toxicity model was established; we observed that rat weights in all the cadmium exposure four groups were decreased a statistically significant amount compared with the control group. However, testicular weights were not significantly different. The organ indexes of the cadmium exposure groups improved compared with the control group, although only the difference between the high-dose and control groups was significant (Figure 1).

Body and testis weights and the organ index of testes. (a) Body weight of rats from each group at the end of the fifth week. (b) Bilateral testes weights obtained after the rats were sacrifice. (c) Organ index of rat testes from each group after sacrifice. The data are given as mean ± SD. *p < 0.05: compared with the control group.
Testicular cadmium content
Cadmium accumulated in the testicular tissue of rats in the cadmium exposure groups and was scarcely detectable in the control group. A comparison of the mean of each group revealed that the differences were significant (p < 0.05; Figure 2).

Cadmium accumulation in rat testes. The data are given as mean ± SD. *p < 0.05: compared with the control group; # p < 0.05: compared with the low-dose group; and & P < 0.05: compared with the middle dose group.
Histopathological observations based on H&E staining
We observed different degrees of testes injury in the cadmium-treated groups, which frequently presented as seminiferous tubule atrophy, endothelial cell swelling, germ cell shedding from the endothelial wall, irregular arrangements of spermatogenic cells, and reduced layers of mature cells. As shown in Figure 3, H&E staining of control group rat testicular tissues revealed normal, seminiferous tubule morphology with regular, inner cells that were closely connected to the endomembrane and with all levels of spermatogenic cells clearly visible and regular. The low-dose group exhibited limited pathological changes, including tube atrophy, seminiferous tubules shrinkage, reduced layers, relatively inflated and loose cells, slightly disordered cell arrangement, and fewer sperm in the tube cavity. In the middle dose group, seminiferous tubules were obviously atrophied, cells within the basement membrane had obviously been shed, cell size varied, and cell forms and arrangements were disordered. Finally, the seminiferous tubules in high-dose group exhibited severe shrinkage, few cell layers remained, and cells with disordered nuclei in the tube and cellular debris were visible in these samples.

Histopathological assessment of H&E staining and immunohistochemical detection of the autophagy-related proteins in rat testes. (a) H&E staining and immunohistochemical detection of the autophagy-related proteins Beclin1, LC3B, and CSR in rat testes. Brown indicates the target protein and blue indicates the nuclei. (b) Quantitative results of Beclin1, LC3B, and CSR expression in rat testes. The data are expressed as mean optical density and given as mean ± SD. *p < 0.05: compared with the control group; # p < 0.05: compared with the low-dose group; and & p < 0.05: compared with the middle dose group. H&E: hematoxylin and eosin; LC3B: light chain 3B; CSR: calcium-sensing receptor.
Immunohistochemical analysis of LC3B, Beclin1, and CSR protein expression
Immunohistochemical analyses of the expression of the autophagy-related proteins Beclin1 and LC3B in the different groups of rats revealed various degrees of expression, and the levels of Beclin1 and LC3B expression in the high-dose cadmium exposure group exhibited a wider range and greater intensity; differences among groups were statistically significant (Figure 3). The expression of CSR in rat testes treated with cadmium was increased significantly compared with control group (Figure 3).
Immunofluorescence-based assessment of LC3B and CSR
The percentage of LC3B-positive cells (a marker of autophagy) was greater in the cadmium-exposed groups than that in the control group and was correlated with increased cadmium exposure. Furthermore, CSR expression was significantly higher in LC3B-positive cells than LC3B-negative cells (Figure 4).

Immunofluorescent assessment of CSR, LC3B, and CSR expression in rat testes. (a) Immunofluorescence images of CSR, LC3B, and CSR expression in rat testes. (b) The rate of LC3B and CSR positive cells. (c) CSR positive rate in cells. Green indicates LC3B, red indicates CSR, and blue indicates DAPI (bar = 50 μm). The data are expressed as the percentage of cells stained positive for the target proteins and given as mean ± SD. *p < 0.05: compared with the control group; # p < 0.05: compared with the low-dose group; and & p < 0.05: compared with the middle dose group. CSR: calcium-sensing receptor; LC3B: light chain 3B; DAPI: 4’,6-diamidino-2-phenylindole.
Western blot analysis of Beclin1 and LC3B expression
We detected significant differences in the expression of the autophagy-related proteins Beclin1 and LC3B in rats testes from the middle- and high-dose cadmium-treatment groups compared with the control group (p < 0.05). Beclin1 and LC3B expression increased with increased cadmium exposure levels (Figure 5). LC3B-II, the active form of LC3B, also showed the same tendency.
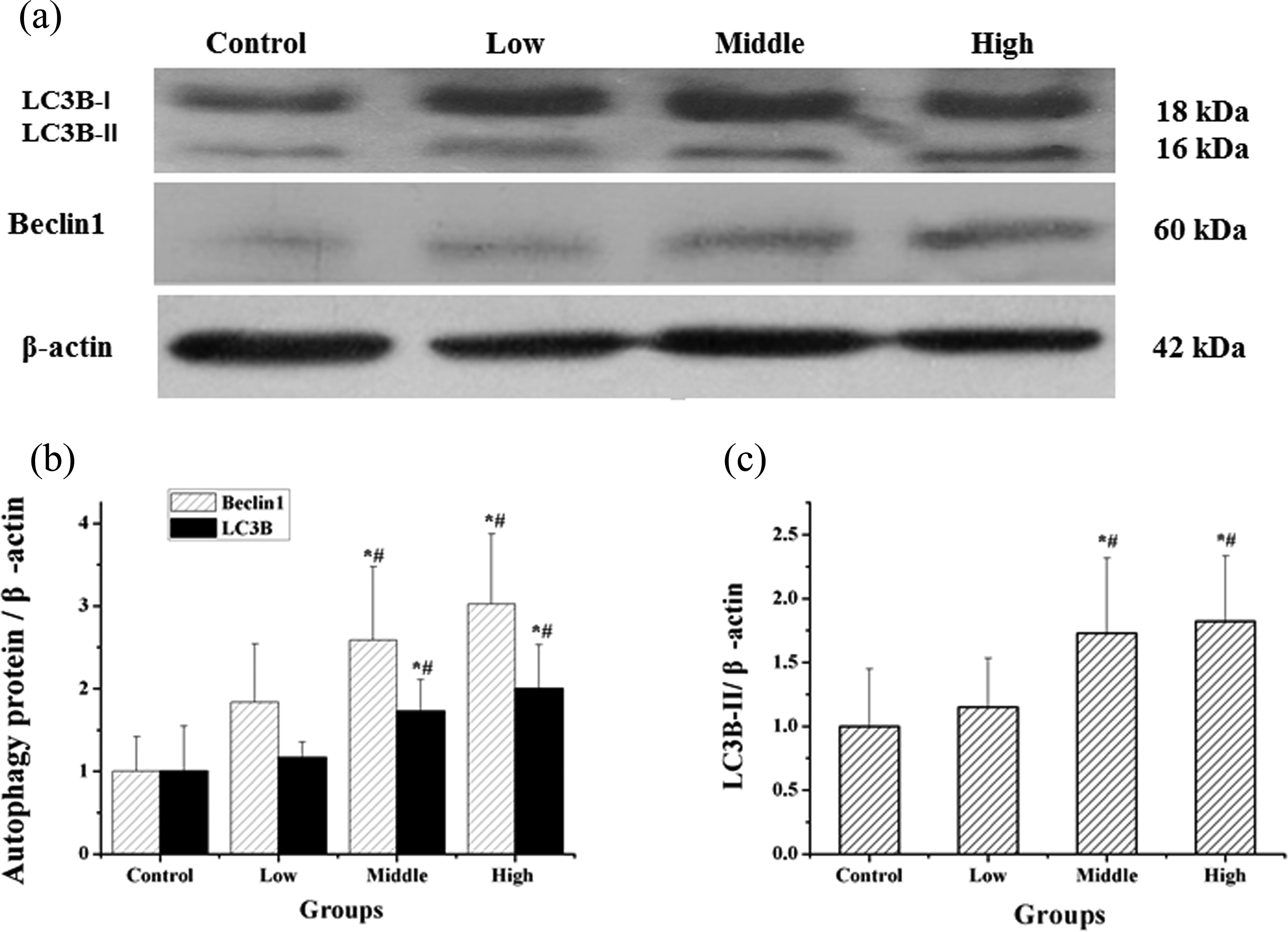
Levels of Beclin1 and LC3B protein expression in the testes. The results presented as protein expression versus the control group. (a) Western blot analysis of Beclin1 and LC3B expression in rat testes. (b) Quantitative results of Beclin1 and LC3B expression levels. (c) Quantitative results of LC3B-II expression levels. The bar diagram shows the band intensities of the target proteins and the data are given as mean ± SD. *p < 0.05: compared with the control group and # p < 0.05: compared with the low-dose group. Beclin1: Bcl2-interacting protein; LC3B: light chain 3B.
Discussion
Cadmium accumulation in the liver and kidney has been carefully studied; however, the testis is another key organ affected by cadmium, and because of its critical role in male reproduction, the deleterious effects of cadmium exposure are of great clinical importance. Previously, researchers have found that exposure to different levels of cadmium can cause testicular injury in rats. Our present findings showed that compared with control group rats, the cadmium-exposed rats exhibited a poor general status, including darker disheveled fur, appearing timid and more easily startled, and eating and drinking less. Testicular cadmium content in the cadmium-exposed groups was significantly higher than that in the control group (p < 0.05), and the content was found to be highly dose dependent, which confirmed that cadmium can enter and accumulate in the testes. H&E staining of rat testes in the cadmium-exposed groups showed an abnormal morphology compared with the control group. The reduction of spermatogenic cells and mature spermatozoa reflected the diminution of spermatogenic capability, which undoubtedly affected reproductive health. 26,27 These experimental findings were consistent with previous studies.
Recently 28,29 , several studies have reported that autophagy is one of the underlying mechanisms of cadmium-induced hepatic and renal damage; however, its effect in cadmium-induced testicular injury has not been carefully examined. 30,31 Autophagy can reduce the secretion of reactive oxygen and apoptosis, which can reduce the magnitude of cadmium-induced damage. However, the protective effects of autophagy on the body are somewhat limited because, when the overall dosage and time exceed the safety threshold, 30 cadmium can cause irreversible cell damage by inducing autophagic death. 31 Cadmium can damage cells by inhibiting intercellular communication via gap junctions, and autophagy can exacerbate this effect, resulting in aggravated damage. 32 Others have also found that cadmium can increase inflammatory responses in the testes. 6 When rats were exposed to cadmium, they could not synthesize functional metallothionein which normally acts to reduce inflammatory responses. 33 Therefore, increasing levels of inflammatory cytokines caused by cadmium absorption result in an increased number of cells undergoing autophagy. 34 Furthermore, some have considered cadmium exposure alone insufficient to cause cell injury and proposed that autophagic cells were highly sensitive to cadmium, resulting in exacerbated autophagy-induced organ injury in response to cadmium. In this study, immunohistochemical and western blot analyses suggested that the expression of the autophagy proteins Beclin1, LC3B, and its autophagy active form LC3B-II in the testes increased along with the exposure dose, and there were significant differences between the exposure groups and the control group. These aforementioned findings suggested that autophagy might contribute to cadmium-induced testicular injury.
Autophagy 35,36 can be controlled by various molecules, including the CSR, which was found to be activated prior to injury. 37 Wang et al. suggested that cadmium-induced autophagy could be mediated by a calcium signaling pathway in vitro; therefore, we hypothesizes that CSR activation might be an important trigger for autophagic cell death that resulted in testicular damage. 37 In this study, immunohistochemical analyses revealed that CSR protein expression was markedly higher in the testes of cadmium-exposed rats.
Previous study 38 demonstrated that cadmium could activate the endoplasmic reticulum, and that calcium within the endoplasmic reticulum was released inducing an iron disorder, which in turn triggered autophagic cell death. LC3B-II is the active form of LC3B and represents the autophagy. Our findings revealed that the protein CSR was highly expressed in the cadmium-treated groups, and our dual-signal fluorescence results indicated that CSR expression co-localized with markers of autophagy and the percentage of autophagy-positive cells was significantly higher than the percentage of autophagy-negative cells. These immunofluorescence data also showed that CSR were co-expressed with active LC3B-II. Together, our findings suggest that the CSR might be related to autophagy.
In summary, autophagy represents one of the mechanisms that contribute to cadmium-induced testicular injury, and the CSR might regulate these autophagy-induced damages. Autophagy-related proteins may represent important targets for the treatment of cadmium-induced testicular injury. This study focused on the adverse effects of cadmium on the male reproductive organ from the perspective of autophagy, which was scarcely reported and our in vivo results suggest that cadmium-induced testicular injury is associated with autophagy. Future experiments will be needed to confirm these results in human patients.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article. This study was funded by the Foundation of Science and Technology Development Program of Jilin Provincial Science and Technology Department, PR China (grant number: 20160101230JC), the Scientific Research Foundation of Health and Family Planning Commission of Jilin Province, PR China (grant number: 2014Z046), and Science & Technology Program during the 12th Five-Year Plan Period of the Education Department of Jilin Province, PR China (grant number: 2015533).
