Abstract
Hepatosteatosis is a complex disorder, in which insulin resistance and associated dyslipidemic and inflammatory conditions are fundamental. Dietary habit, especially regular consumption of fat and sugar-rich diet, is an important risk factor. Coconut and mustard oils (CO and MO) are medium-chain saturated and monounsaturated fats that are common dietary ingredients among the Indian populations. Present study analyzed the effect of prolonged consumption of the fresh and thermally oxidized forms of these oils on glucose tolerance and hepatosteatosis in male Wistar rats. Thermally oxidized CO (TCO) and MO (TMO) possessed higher amount of lipid peroxidation products and elevated p-anisidine values than their fresh forms. Dietary administration of TCO and TMO along with fructose altered glucose tolerance and increased hyperglycemia in rats. Dyslipidemia was evident by elevated levels of triglycerides and reduced high density lipoprotein cholesterol (HDLc) levels in fructose and edible oil-fed group (p < 0.05). Additionally, hepatic antioxidant status was diminished and oxidative stress markers were elevated in TCO- and TMO-fed rats. Substantiating these, hike in liver function marker enzyme activities were also observed in these animals. Supporting this, histological analysis revealed higher incidence of microvesicles and hepatocellular ballooning. Results thus suggest that consumption of thermally oxidized fats may cause hepatic damage.
Introduction
Hepatosteatosis is a frequent form of chronic hepatic disorder, which is characterized by the accumulation of triacylglycerol primarily in the liver. 1 Insulin resistance, impaired carbohydrate and lipid metabolisms, obesity and chronic inflammation are the hallmark of hepatosteatosis. 2 Other than genetic predispositions, several exogenous factors influence the development of this disease. Diet rich in fats and sugar are known to promote the lifestyle disorders including the development of hepatosteatosis. It has been also verified that reduced intake of dietary fat and lowered intestinal absorption of cholesterol have brought down the frequency of hepatosteatosis 3 indicating the role of dietary fats in the development of the disease. Fructose, which is known to upregulate the hepatic lipogenesis, is also identified as an independent risk factor. High fructose along with medium-chain saturated fats is widely being used as an experimental dietary model to induce insulin resistance. 4 –6 Increased incidences of hepatic disorders are reported worldwide, where lifestyle changes especially in the food habits and sedentary life are the main culprits. Consumption of fructose-sweetened beverages and high fat diet, especially fried fat, is thought to be a reason for increased hepatic disorders. 7,8 Recent report indicates that increased mortality rates are associated with high intake of sugar-sweetened beverages. 9 According to a study by Singh et al., 10 about 1.8 million people are died because of sugar-sweetened beverages consumption for the last 30 years.
Thermal oxidation such as deep frying, is known to produce harmful changes in fats and oils including increase in the peroxide value, free fatty acids content, lipid peroxidation products and formation of triacylglycerol polymers and dimers. 11 Thus, consumption of thermally oxidized oils has shown to augment oxidative and carbonyl stress thereby inducing preneoplastic lesions in the liver of rats. 12,13 Previous reports have indicated that thermally oxidized vegetable oils contain higher levels of cyclic fatty acid monomers (CFAMs), 14 which are known to inhibit biochemical reactions and enzyme activities. 15
Coconut oil (CO) and mustard oil (MO) are the generally consumed forms of edible oil in South India. They differ in fatty acid composition, where CO is rich in medium-chain saturated fatty acids reaching up to 90% of the total, with lauric acid as the predominant fatty acid (50%), whereas MO contains high amounts of monounsaturated fatty acids especially erucic acid. CO possesses several health benefits, which includes neuroprotection 16 and improvement of lipid metabolism in premenopausal women. 17 At the same time, reports indicate an increased incidence of cardiovascular diseases when saturated fat intake is high suggesting its possible health hazard. 18,19 Also, CO as a fatty acid source in high fructose diet induces hyperglycemia and oxidative stress in murines 20 and leads to hepatosteatosis-like symptoms. 21 On the other hand, MO has shown to possess antioxidant activity, 22 antimutagenic, 23 and chemopreventive effects. 24 However, studies have reported that dietary MO can induce hyperalgesia 25 and neurologic inflammation. 26 In addition, the MO has shown to enhance hepatic preneoplastic foci development in rats. 27 Consumption of MO increases the serum triglycerides (TG) with a reduction in mitochondrial cardiolipin content 28 and also known to induce cardiac fibrosis and vacuolation. 29
In view of all these, the present study aims to compare the effects of long-term consumption of the fresh and thermally oxidized coconut (TCO) and MOs along with a high fructose diet (60%) on the development and extent of hepatosteatosis in male Wistar rats.
Materials and methods
Animals
Male Wistar rats weighing approximately 200–230 g (8–10 weeks old) on an average were supplied by the Kerala Veterinary and Animal Sciences University. The animals were fed with a commercial diet from the Sai Durga feeds (Bangalore, Karnataka, India). The rats were randomly assigned into five groups of six each, on the basis of their weights using the spiral method, which resulted in almost equal total initial weights of groups. The animals were maintained at standard conditions and fed water ad libitum. The experimental procedure followed the established guidelines for the care and handling of laboratory animals by Institutional animal ethical committee and Committee for the Purpose of Control and Supervision of Experiments on Animals, Government of India (approval no. ACRC/IAEC/15/06-(2)).
Preparation of the thermally oxidized oil
Fresh CO and MO were purchased from local market. These oils were then divided into two equal portions: one portion was stored at 15°C to be used in the fresh oil-containing diets later on in the experiment, while the other portion was heated at 160°C for 48 h. 30 For heat treatment, these oils were put in 1-L glass beakers which were placed on a heating block set at the intended temperature. The thermally oxidized oils were stored at −20°C in order to prevent further oxidation during storage before being included in the respective diets. The extent of lipid peroxidation in the oil was estimated by thiobarbituric acid reactive substances (TBARS), conjugated dienes, and p-anisidine value. 31
Diets and experimental procedure
Experimental diets for inducing insulin resistance are based on the previous studies. 4,5,21 It composed of 60% fructose, 20% protein, and 10% nutrient and mineral mix. They consisted of either fresh or TCO/TMOs as fat source (10%). Thus, five experimental diets were prepared: (1) fresh CO-supplemented diet, (2) TCO-supplemented diet, (3) fresh MO-supplemented diet, (4) TMO-supplemented diet, and (5) reference diet fed (groundnut oil as fat source). Each of these experimental groups was maintained on one of the above diets for a 30-week period. All animals had free access to food and water during the experiment. At the end of experimental period, the animals were killed following an overnight fasting and blood was collected by heart puncture. Liver tissue was excised and a portion was fixed in 10% formalin for histological analysis and remaining tissue was kept under −80°C for further studies.
Estimation of glucose tolerance
The animals were fasted overnight before oral glucose tolerance test. Glucose tolerance was measured using the method of Narayanankutty et al. 21 Briefly, rats were orally administered with 2 g/kg glucose. Following this, blood glucose levels were measured using glucometer (One touch select, UK) at 0, 30, 60, 90, and 120 min intervals. The rate of glucose clearance and area under the curve were calculated.
Biochemical analysis
Various serum parameters such as total cholesterol (TC), high density lipoprotein cholesterol (HDLc), TG, alanine transaminase (ALT), aspartate transaminase (AST), alkaline phosphate (ALP), and blood glucose were determined using commercially available kits (Agappe diagnostics, Chennai, Tamil Nadu, India) as per the manufacturer’s instructions.
Analysis of oxidative stress in liver tissues of rats
Liver tissue was excised and washed in ice-cold saline. A small portion of each was fixed in 10% buffered formalin for histopathological studies. Liver homogenate was prepared (10% w/v) in 0.1 M Tris hydrochloric acid buffer (pH 7.4) and used for the estimation of lipid peroxidation 32 and protein carbonyls. 33
The homogenates were prepared by centrifuging at 10,000 r/min for 15 min at 4°C. The supernatant was collected and used for reduced glutathione (GSH), 34 superoxide dismutase (SOD), 35 catalase, 35 and glutathione reductase 36 assays.
Measurement of polyol pathway enzymes and ornithine decarboxylase
Polyol pathway enzymes, aldose reductase (AR) and sorbitol dehydrogenase (SDH), activities were measured by spectrophotometric procedures as described by Jang et al. 37 Ornithine decarboxylase activity was determined according to the methods of Ngo et al. 38
Histopathological analysis
A portion of the formalin-fixed liver tissue was embedded in wax following dehydration with series of alcohol. Serial sections of the tissues were then taken in a microtome at a thickness of 4 µm and stained with hematoxylin and eosin. Histopathological examination was carried out by a pathologist who was blind to the plan of this study. Scoring of hepatosteatosis was carried out according to standard method. 39
Statistical analysis
The values are represented as mean ± standard deviation. Statistical analysis of the data was done by one-way analysis of variance followed by Tukey–Kramer multiple comparisons test using Graph pad Instat software version 3.05.
Results
Physicochemical analysis of fresh oils and its thermally oxidized forms
The results indicated that levels of conjugated dienes were elevated during thermal oxidation of CO (Table 1). Upon heating, MO underwent severe oxidative modifications as indicated by a significant (p < 0.05) increase in the levels of both conjugated dienes and trienes (Table 1). p-Anisidine value, which is an indication of the aldehyde levels, also found to be high in the TCO than its fresh counterpart. However, these changes were much higher in MO. In addition, lipid peroxidation products, as measured by TBARS, were also elevated in the TCO as well as TMO (p < 0.001). Aldehyde content of TMO was also found to be higher than its fresh oil (p < 0.001).
Physicochemical properties of fresh and thermally oxidized edible oils.a
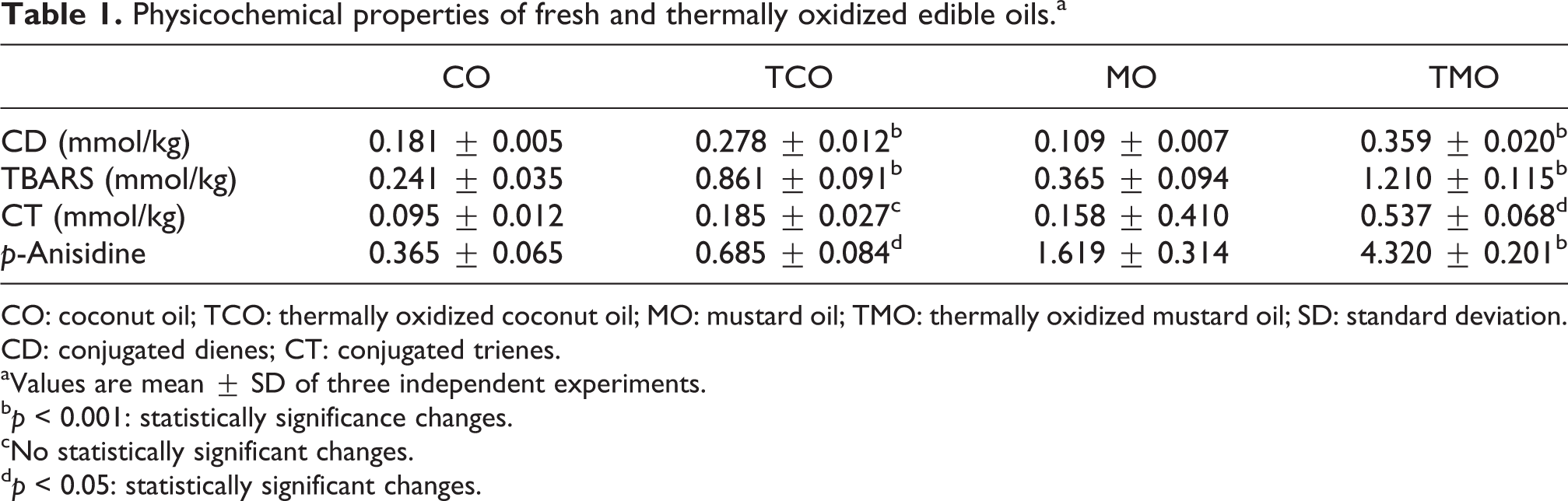
CO: coconut oil; TCO: thermally oxidized coconut oil; MO: mustard oil; TMO: thermally oxidized mustard oil; SD: standard deviation. CD: conjugated dienes; CT: conjugated trienes.
aValues are mean ± SD of three independent experiments.
b p < 0.001: statistically significance changes.
cNo statistically significant changes.
d p < 0.05: statistically significant changes.
Effect of diet-containing high fructose and either of CO, MO, or their thermally oxidized products on blood glucose levels and glucose tolerance
The animals fed with diet-containing high fructose and different edible oils had a gradual increase in the blood glucose level in comparison with the reference group of rats. Reference group of animals had an initial blood glucose level of 81.12 ± 12.46 mg/dL, which gradually increased to 103.69 ± 5.72 mg/dL over a period of 30 weeks (net increase of 21.18 ± 3.21 mg/dL). In the same experimental period, the changes in blood glucose level of CO-fed animals were from initial 88.53 ± 6.52 mg/dL to a final 210.3 ± 7.68 mg/dL (net increase of 114.01 ± 6.45 mg/dL). However, the increase in TCO-fed group was more profound with a net increase of 145.13 ± 10.24 mg/dL in blood glucose (initial 88.72 ± 6.69 and final 225.90 ± 14.0 mg/dL). In MO group of rats, from an initial level of 89.07 ± 8.68, the blood glucose was raised to 211.43 ± 12.80 mg/dL (net increase of 122.36 mg/dL). In the TMO-fed animals, blood glucose level had an increase from 89.62 ± 7.08 to 233.98 ± 8.47 mg/dL (net increase of 144.36 mg/dL).
Before killing, oral glucose tolerance was assessed in these rats to evaluate the level of insulin resistance. All animals were given an oral gavage of 2 g/kg glucose after an overnight fasting and blood glucose level was estimated at different time intervals (before glucose administration, 30, 60, 90, and 120 min after glucose administration). In reference group of animals, from an initial level 100.12 ± 1.24 mg/dL, blood glucose was elevated to 204.69 ± 2.54 mg/dL within 30 min (Figure 1(b)). The level returned to normal within 120 min (100.23 ± 3.41 mg/dL), with a glucose clearance rate of 52.23 ± 2.39 mg/dL/h. In the case of CO-fed animals, the blood glucose was elevated to 329.24 ± 12.67 from the initial level 205.26 ± 6.18 mg/dL. Even after 120 min, the levels failed to return to their basal glucose levels (251.07 ± 8.59 mg/dL). Blood glucose level in TCO group was also found to be elevated within 30 min of glucose administration (2 g/kg) to 335.57 ± 5.61 mg/dL, which was returned to a level of 272.97 ± 8.41 mg/dL at 120 min time. TMO diet-fed animals had reduced glucose tolerance (31.3 ± 5.23) than the MO-fed group (39.9 ± 4.84 mg/dL/h; Figure 1(b)).
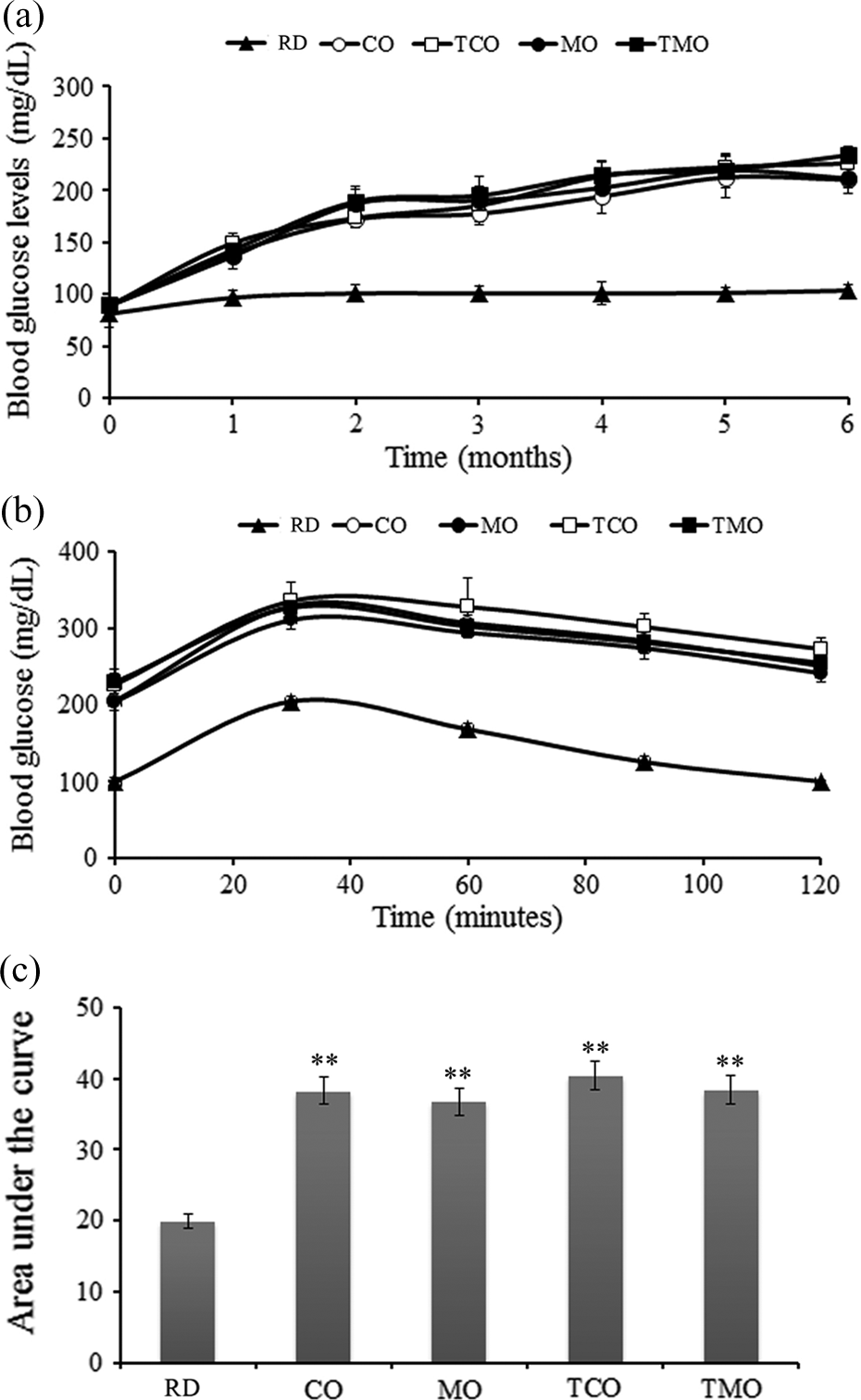
(a) Variation in blood glucose levels in RD, CO, MO, and their fried products (TCO and TMO) containing diet-fed groups from initial to 6th month. (b) Oral glucose tolerance levels at the end of experimental period in RD-, CO-, TCO-, MO-, and TMO-containing-diet-fed animals administered with glucose (2 g/kg) at different time intervals. (c) Area under the curve calculated for different experimental groups. Values are mean ± SD of six animals in each treatment group. **p < 0.01. RD: reference diet; CO: coconut oil; MO: mustard oil; TCO: thermally oxidized coconut oil; TMO: thermally oxidized mustard oil; SD: standard deviation.
Effects on serum and hepatic lipid profile
In reference diet-fed animals, the serum TC was found to be 61.73 ± 6.83 mg/dL; whereas the CO- and TCO-containing-diet-fed animals had higher levels of TC (94.61 ± 5.98 and 95.9 ± 5.82 mg/dL). Similar observations were made in the case of MO- and TMO-containing-diet-fed animals (94.9 ± 3.61 and 99.37 ± 4.84 mg/dL). HDLc levels of reference diet-fed and CO-fed groups were more or less the same (28.06 ± 3.94 and 28.17 ± 6.05 mg/dL) In TCO-, MO-, and TMO-containing diet-fed groups, the HDLc levels reduced nonsignificantly without much difference among the individual groups (26.91 ± 5.82 26.04 ± 3.10 and 24.55 ± 2.67 mg/dL), respectively (Figure 2(a)). TGs in reference diet-fed rats were 84.46 ± 16.92 mg/dL. An increase in the TG level was observed in the CO (165.74 ± 11.4 mg/dL), TCO (192.51 ± 13.1 mg/dL), MO (165.57 ± 11.55 mg/dL), and TMO (199.97 ± 13.07 mg/dL) containing diet-fed groups (Figure 2(a)).
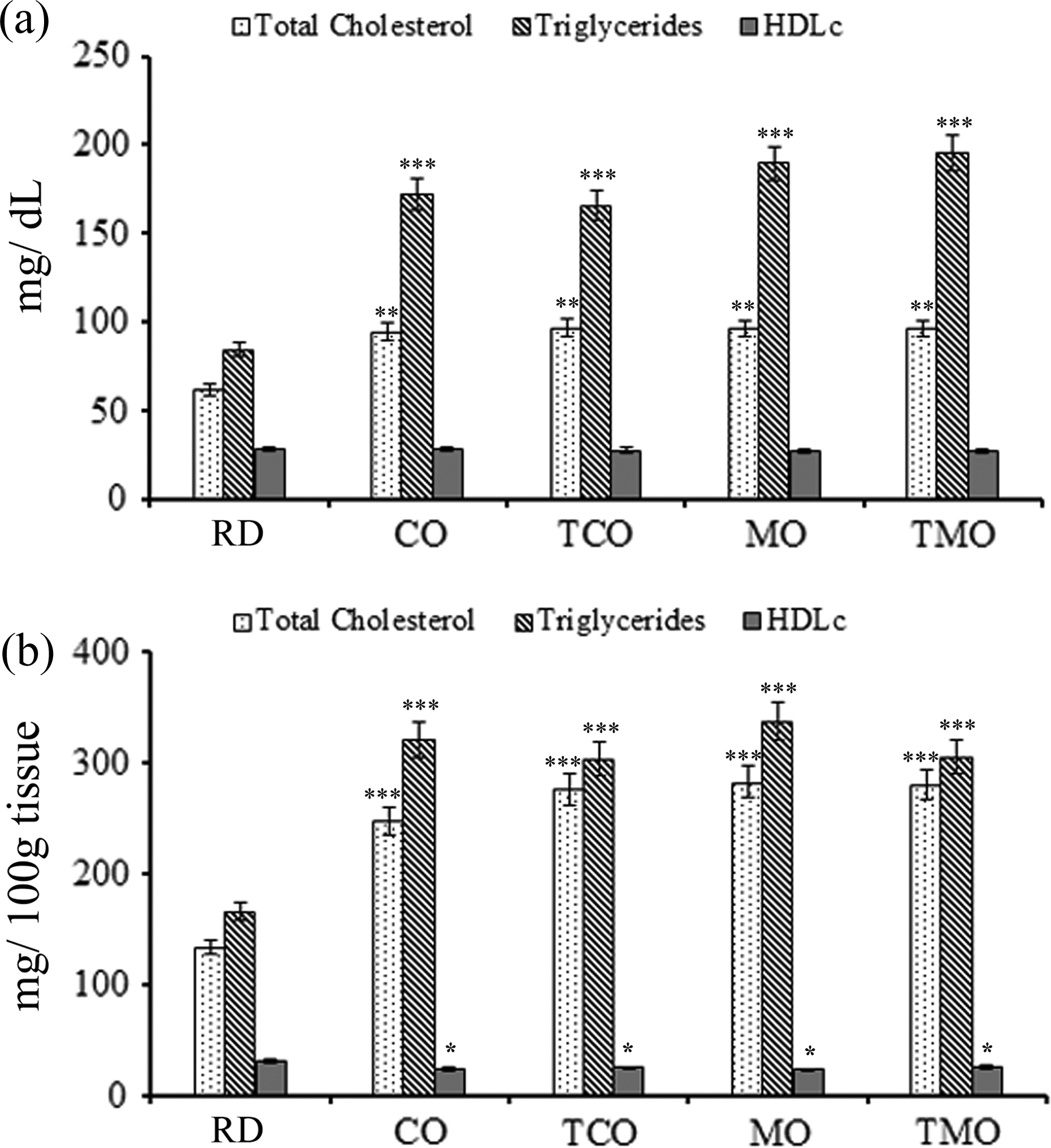
Serum (a) and liver (b) lipid profile of experimental animals fed with RD, CO, MO and their thermally oxidized products (TCO and TMO) fed groups at the end of 30 weeks. Values are mean ± SD of six animals in each treatment group. *p < 0.05; **p < 0.01; and ***p < 0.001. RD: reference diet; CO: coconut oil; MO: mustard oil; TCO: thermally oxidized coconut oil; TMO: thermally oxidized mustard oil; SD: standard deviation.
TC of the liver tissue was significantly elevated in CO-, TCO-, MO-, and TMO-fed animals (Figure 2(b)) compared to reference diet-fed group. The TC levels in the reference diet-fed rats were 128.86 ± 3.34 mg/100 g of tissue. Among the oil-fed animals, the CO-fed animals had the lower TC levels (247.3 ± 19.1 mg/100 g tissue). Animals fed with a diet-containing TCO were shown to have elevated liver cholesterol levels (282.04 ± 6.86 mg/100 g of tissue). However, difference between the TC levels of TMO (279.63 ± 12.79 mg/100 g of tissue) and MO-fed group (275.07 ± 12.16 mg/100 g of tissue) animals was not significant.
Hepatic triglyceride level in reference diet-fed animals was 164.89 ± 15.63 mg/100 g of tissue. In CO and TCO groups, an increased level of 320.4 ± 23.1 and 336.2 ± 21.9 mg/100 g of tissue was observed. The TG levels were more or less similar in the MO and TMO-fed animals (302.77 ± 14.06 and 303.97 ± 20.12 mg/100 g of tissue). HDLc levels showed a reduction from 30.32 ± 1.62 mg/g in reference diet-fed rats to 23.2 ± 3.0 and 22.6 ± 2.3 mg/100 g of tissue in CO- and TCO-fed rats. It was estimated to be 24.55 ± 1.95 and 25.25 ± 2.30 mg/100 g of tissue in MO- and TMO-fed animals.
Effect on hepatic oxidative stress markers
As shown in Table 2, in comparison with reference diet-fed animals, CO-, TCO-, MO-, and TMO-fed animals had reduced levels of GSH (p < 0.05). Among these, animals fed with fresh oils, CO-fed rats was shown to have higher GSH levels than MO-fed animals. Feeding with fried oils (TCO and TMO) was shown to have similar GSH levels however with significant reduction from their respective fresh counterparts (p < 0.01).
Antioxidant levels in the hepatic tissue of experimental animals fed with RD, CO, MO, and their thermally oxidized forms (TCO and TMO).a
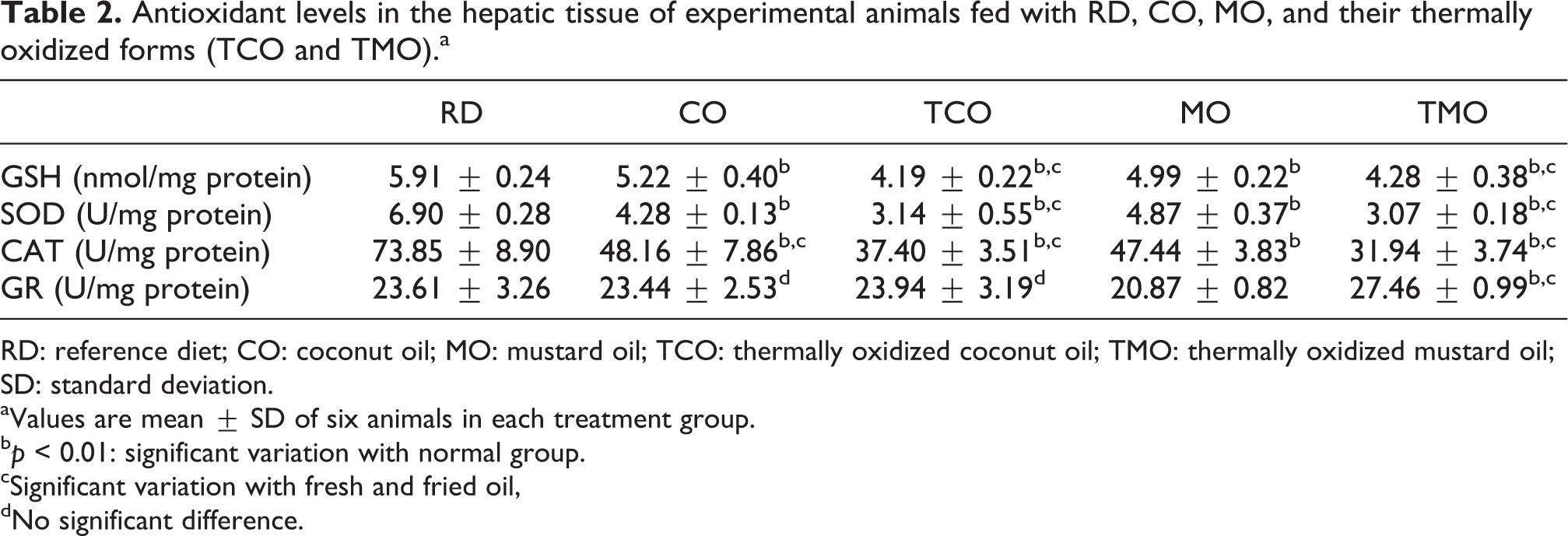
RD: reference diet; CO: coconut oil; MO: mustard oil; TCO: thermally oxidized coconut oil; TMO: thermally oxidized mustard oil; SD: standard deviation.
aValues are mean ± SD of six animals in each treatment group.
b p < 0.01: significant variation with normal group.
cSignificant variation with fresh and fried oil,
dNo significant difference.
SOD activity was also decreased in animals fed with fried oil (p < 0.001). On contrary to the pattern seen in GSH levels, MO-fed animals showed a higher SOD activity than the CO-fed animals. However, the difference between the fried oil-fed animals was not statistically significant. The activity of catalase, which is known to involve in the detoxification of peroxide radicals, was found to be reduced in all the experimental groups than those of the reference diet-fed rats (p < 0.01), of which TCO and TMO had the least levels of catalase activity. There was no significant difference observed among the animals of CO and MO-fed animals in terms of catalase activity. In contrast with other antioxidant enzyme activity levels, GR, which takes part in the conversion of oxidized GSH to its reduced form, remained unchanged in all the experimental groups except TMO-fed rats. The values were found to be 23.61 ± 3.26, 23.44 ± 2.53, 23.94 ± 3.19, 20.87 ± 0.82, and 27.46 ± 0.9 U/mg protein in reference, CO, TCO, MO, and TMO groups, respectively.
As shown in Table 3, a significant increase in carbonyl stress as indicated by serum protein carbonyls adducts (p < 0.001) was observed in CO-, MO-, TCO-, and TMO-containing-diet-fed groups. However, serum TBARS did not show increase in CO and MO groups with that of reference diet-fed groups, where as in TCO and TMO groups, significant (p < 0.01) increase in TBARS was observed. Hepatic TBARS (p < 0.001) and protein carbonyl adducts (p < 0.05) were also got elevated in the CO, MO, TCO, and TMO groups.
Hepatic and serum oxidative and carbonyl stress in rats fed with RD, fresh oils (CO and MO), and their fried forms (TCO and TMO).a
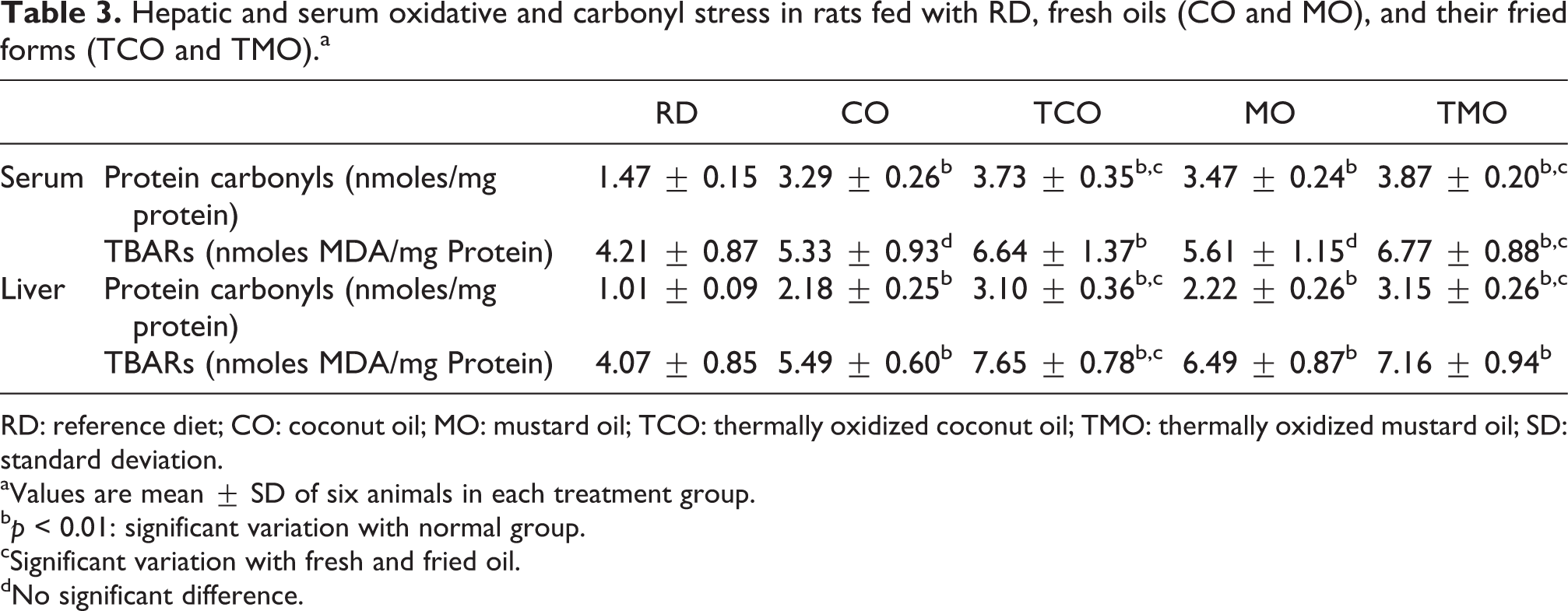
RD: reference diet; CO: coconut oil; MO: mustard oil; TCO: thermally oxidized coconut oil; TMO: thermally oxidized mustard oil; SD: standard deviation.
aValues are mean ± SD of six animals in each treatment group.
b p < 0.01: significant variation with normal group.
cSignificant variation with fresh and fried oil.
dNo significant difference.
Effect on polyol pathway enzymes and ornithine decarboxylase
In the reference diet-fed rats, the AR activity was 1.42 ± 0.41 U/mg protein (Figure 3). In the CO- and TCO-containing-diet-fed rats, the respective activity was increased to 2.82 ± 0.46 and 3.59 ± 0.47 U/mg protein. In MO- and TMO-containing-diet-fed rats, there was a marginal decrease in the activity (3.12 ± 0.49 and 3.69 ± 0.66 U/mg protein, respectively).
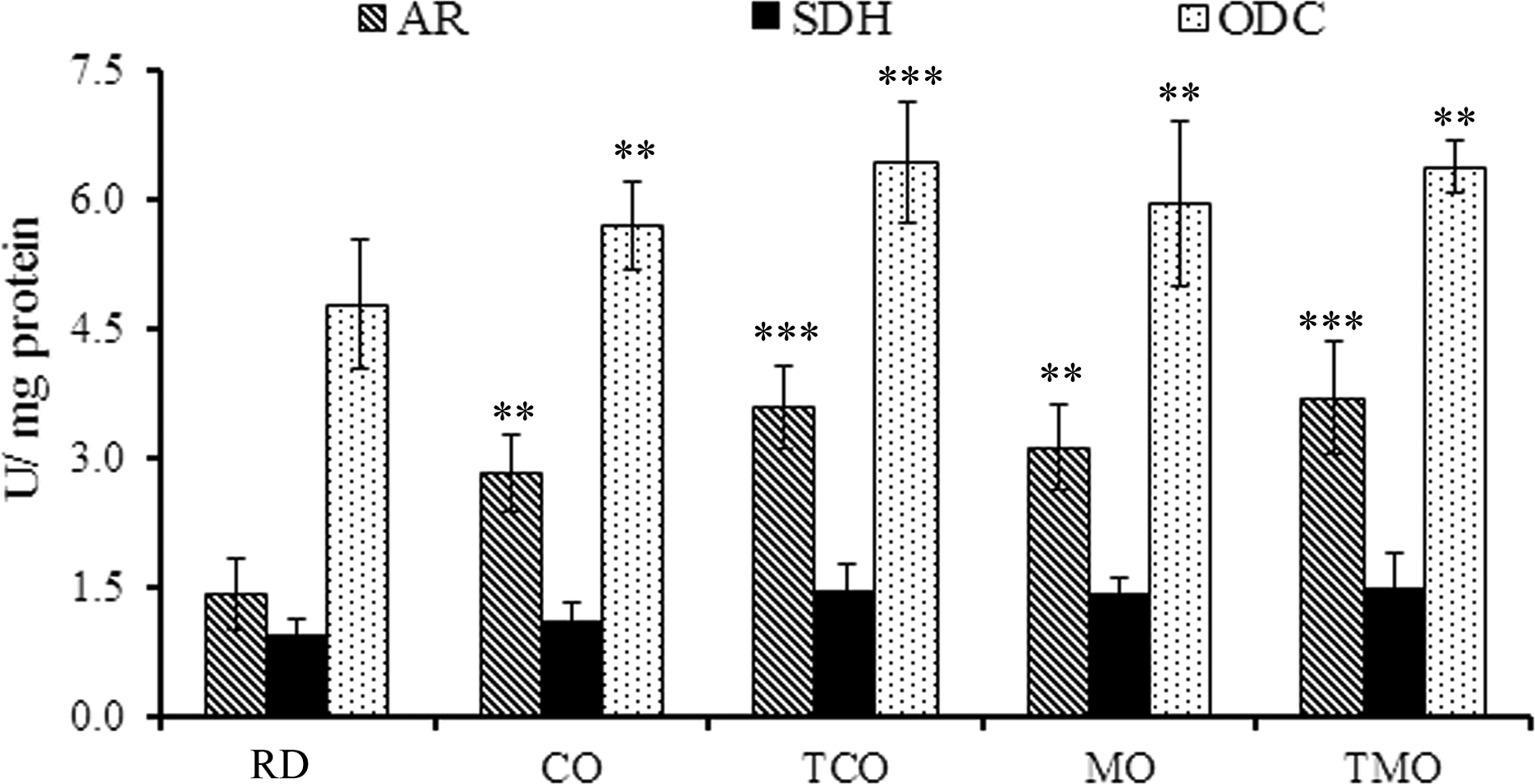
Activities of AR, SDH, and ODC enzymes in the hepatic tissue of rats fed with the experimental diets. Values are mean ± SD of six animals in each treatment group. **p < 0.01; ***p < 0.001. AR: aldose reductase; SDH: sorbitol dehydrogenase; ODC: ornithine decarboxylase; SD: standard deviation.
SDH activity was 0.94 ± 0.18 U/mg protein in the reference diet-fed animals. In the CO- and TCO-containing-diet-fed rats, levels were increased to 1.11 ± 0.21 and 1.46 ± 0.29 U/mg protein. However, the variation between MO and TMO was not statistically significant. The SDH activities in these animals were 1.42 ± 0.18 and 1.49 ± 0.41 U/mg protein, respectively.
Ornithine decarboxylase activity was found to be 4.78 ± 0.76 U/mg protein in reference diet-fed rats (Figure 4), which was slightly increased in CO-containing-diet-fed rats (5.69 ± 0.52 U/mg protein). In TCO-fed rats, the level was further raised to 6.42 ± 0.71 U/mg protein. Whereas in the MO- and TMO-fed rats, the levels were 5.95 ± 0.95 and 6.37 ± 0.29 U/mg protein.
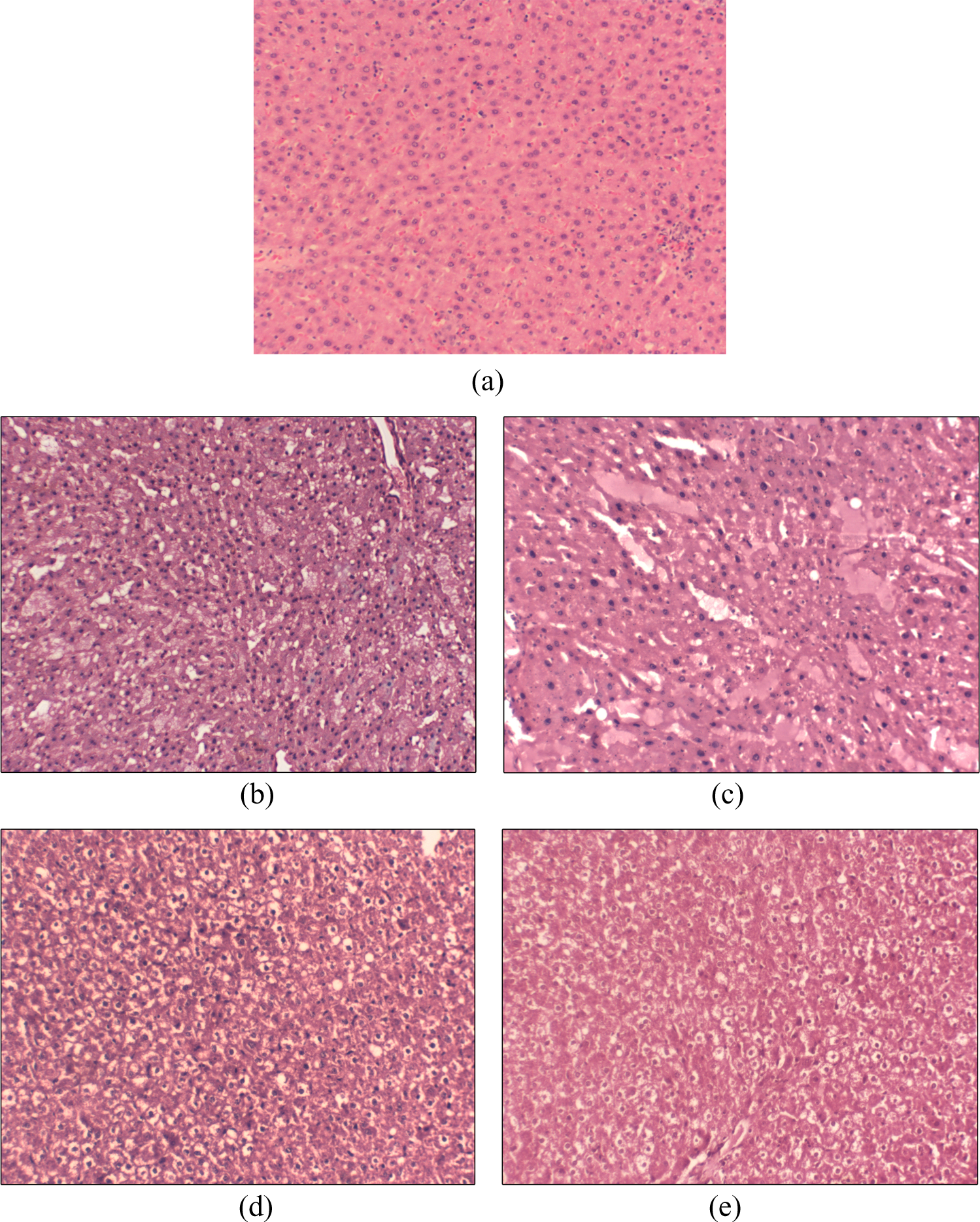
Histological evaluation of hematoxylin–eosin-stained liver tissues of reference diet-fed rats (a), CO (b), MO (c), thermally oxidized CO (d), and thermally oxidized MO (e) fed animals. Photographs were taken under ×200 magnification in microscope (Magnus, New Delhi, India). CO: coconut oil; MO: mustard oil.
Liver function parameters
The liver function marker enzyme analysis showed significant change with respect to ALT and AST activity, where threefold increase in the AST and twofold increase in ALT levels were observed in the fried oil-fed animals (Table 4). AST levels were shown to be higher in the thermally oxidized oil-fed groups (TCO and TMO) than their fresh oil-fed ones (p < 0.01). Similarly, all these groups had significantly higher levels of AST activity than the reference group (p < 0.001). The ALT activity of reference diet-fed animals was 32.63 ± 2.8 U/L. It was found to be significantly increased (p < 0.05) in TCO-fed animals than CO-containing-diet-fed animals (Table 4). Similar results were observed in the case of MO- and TMO-fed animals, where a higher hepatic marker enzyme activity (p < 0.001) was reported in the fried oil-fed animals. Alkaline phosphatase activity showed only a slight increase (p < 0.05) in the fresh oil-fed animals. No significant variation was observed among the CO- and TCO-fed groups (Table 4), whereas the difference in ALP activity between MO and TMO was statistically significant (p < 0.05).
Liver function parameters (IU/L) of the experimental animals fed with RD and high fructose diet-containing CO, MO and their fried forms (TCO and TMO).a
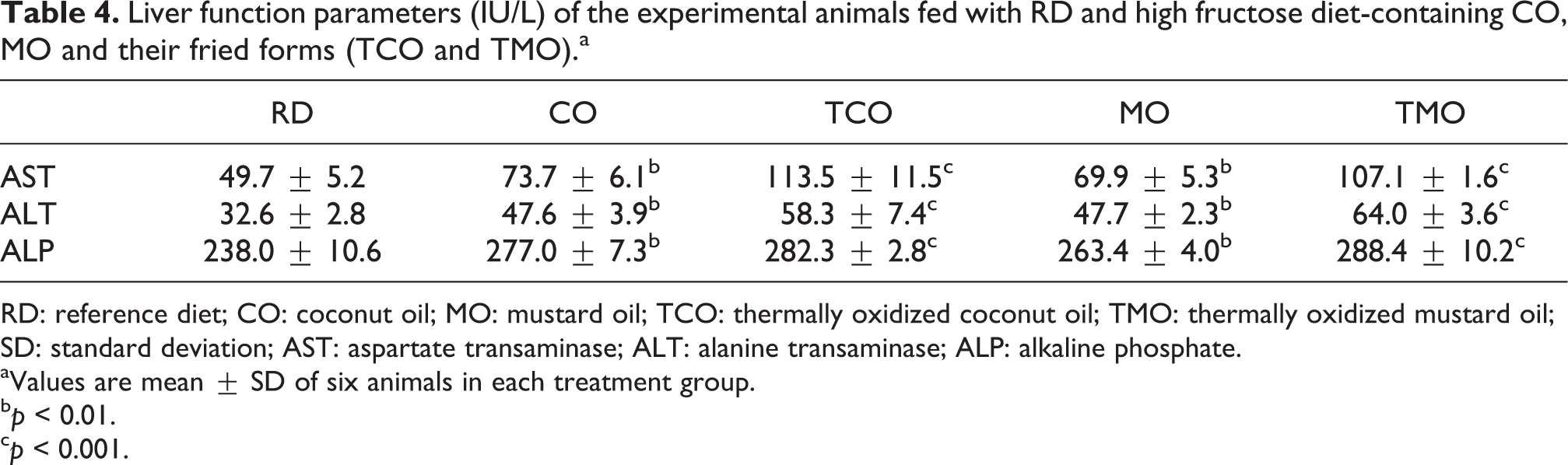
RD: reference diet; CO: coconut oil; MO: mustard oil; TCO: thermally oxidized coconut oil; TMO: thermally oxidized mustard oil; SD: standard deviation; AST: aspartate transaminase; ALT: alanine transaminase; ALP: alkaline phosphate.
aValues are mean ± SD of six animals in each treatment group.
b p < 0.01.
c p < 0.001.
Histopathological analysis
Histopathological analysis of the liver tissues of animals fed with reference diet (Figure 4(a)) and those fed on high fructose and CO (Figure 4(b)), MO (Figure 4(c)), TCO (Figure 4(d)), and TMO (Figure 4(e)) containing diet-fed animal are shown in Figure 4. The reference diet-fed animals showed normal hepatic architecture with normal central venous system, the portal triads, sinusoidal spaces, and Kupffer cells, which was graded as ‘0’. CO- and MO-containing-diet-fed groups showed signs of low intensity hepatosteatosis, correlating the grading between 0 and 1. However, TCO- and TMO-containing-diet-fed animals showed progressed hepatosteatosis, which was evident from the increased incidence of microvesicles, hepatocellular ballooning, glycogenated nuclei as well as lipogranulomas with a grading of 1–2 (Table 5). Incidence of lipogranuloma was noted in TCO-fed rats.
Hepatosteatosis stages in rats fed on RD and diet-containing CO, MO, and fried forms of both (TCO and TMO).a
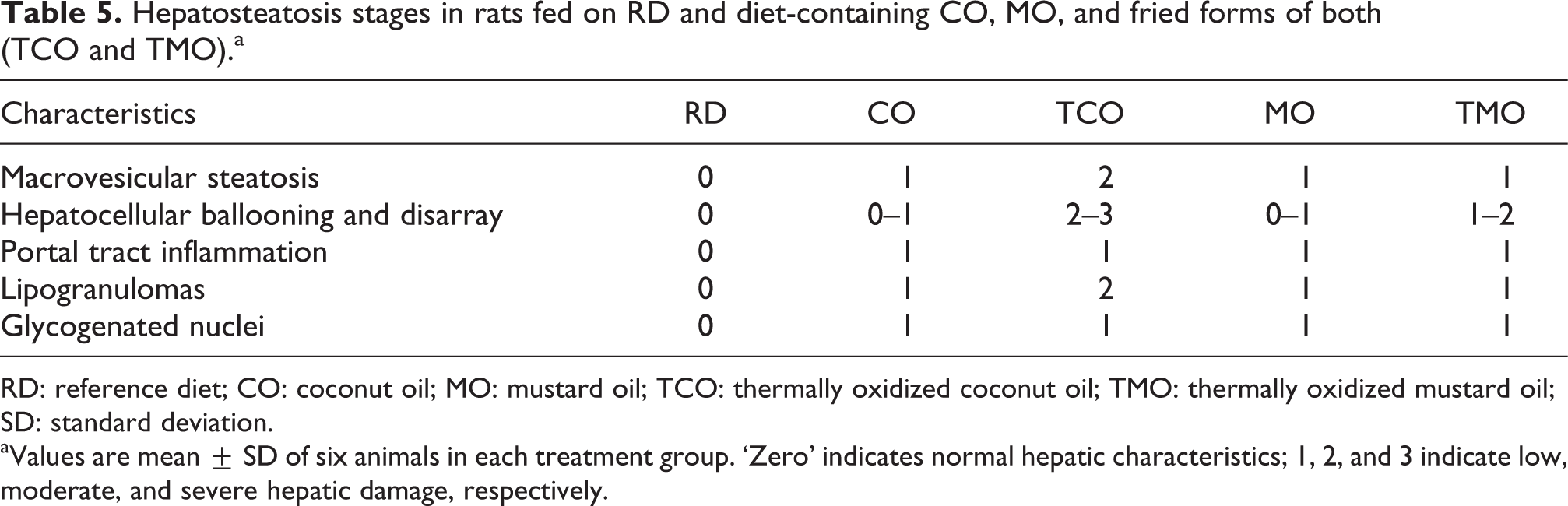
RD: reference diet; CO: coconut oil; MO: mustard oil; TCO: thermally oxidized coconut oil; TMO: thermally oxidized mustard oil; SD: standard deviation.
aValues are mean ± SD of six animals in each treatment group. ‘Zero’ indicates normal hepatic characteristics; 1, 2, and 3 indicate low, moderate, and severe hepatic damage, respectively.
Discussion
Natural edible oils are rich sources of medium- and long-chain saturated or unsaturated fatty acids. Heating of these oils, especially those containing unsaturated fatty acids, generates fatty acid decomposition products mostly carbonyl molecules. These fatty acid–derived molecules are known to induce toxic and inflammatory signaling in the body. 40 In addition to these, studies reported the formation of triacylglycerol polymers and dimers 11 along with toxic CFAMs in deep fried vegetable oils. 15 In the present study, deep frying of CO and MO documented increased peroxide and aldehyde molecules. Compared to medium-chain saturated fatty acid-rich CO, unsaturated fatty acid-containing MO has increased levels of these peroxidation products. This could be due to the presence of higher erucic acid content in MO (45%). Copra oil also contains minor amounts of monounsaturated and polyunsaturated fatty acids (5–9% of oleic and linoleic acids) accounting for the increased lipid peroxidation products, which is observed during its thermal oxidation. Further, prolonged consumption of high amount of fructose together with fresh and thermally oxidized edible oils shows symptoms of advanced changes related to hepatosteatosis and hepatotoxicity.
Hepatosteatosis is a severe metabolic disorder of liver, where insulin resistance, atheromatous dyslipidemia, and oxidative stress are contributing to excessive lipotoxicity and associated hepatic damages. 41 In this study, compared to reference diet-fed animals, rats fed with fructose and fresh or thermally oxidized oil-containing diet show increased levels of blood glucose and reduced glucose tolerance. This is a clear indication of the progressive insulin resistance developed in these animals. Associated with this, an increase in TG and reduction in HDLc levels are seen in fresh or thermally oxidized oil-containing-diet-fed animals. According to Song et al., 42 insulin resistance is manifested by dyslipidemia, where increased levels of TG and low HDL are evident. TC and low-density lipoprotein levels are also found to be higher among these groups. Therefore, it is assumed that insulin resistance and associated dyslipidemia might have contributed to the progressive hepatosteatosis, which is observed in the experimental diet-fed animals.
Hypertriglyceridemia-associated lipotoxicity is characterized by the vacuolation of hepatocytes and disruption of their normal functioning, 43 which is the major contributor of hepatic damages. Here, in this study, animals fed with a diet containing fructose and the edible oils (fresh and fried) shown to have higher triglyceride levels in comparison with reference diet-fed animals. It is already reported that high fructose intake increases triglyceride level and hepatic lipogenesis. 44 In addition, lipid oxidation products formed during the deep frying of these oils for instance CFAM are known to inhibit the hepatic lipase activity. 15 This may have contributed to the observed increase in the TG in the experimental animals. On the contrary, studies have reported that a diet rich in medium-chain saturated and monounsaturated fatty acids reduces the triglyceride levels in body by enhancing the enzymatic activity of lipases. 45,46
Higher levels of protein carbonyls and lipid peroxidative changes are noted in TCO and TMO groups, which is in line with the studies by Adam et al., 47 where increased lipid peroxidation is noted in fried palm oil-fed rats. Previous reports have indicated that thermally oxidized vegetable oils contain higher levels of CFAM. 14 Here, in this study, TCO and TMO are found to contain more lipid hydroperoxides and aldehydes than their fresh counterparts. The oxidative and inflammatory insults by these oxidation molecules may be responsible for the observed increase in lipid peroxidation products in the serum and liver tissue of fried oil-fed animals. Accumulation of these lipid oxidation products is often recognized as a risk factor for many of the liver diseases including nonalcoholic fatty liver. 48,49 Providing further insight into this, reduced GSH level is found diminished in TCO- and TMO-fed rats. GSH is actively involved in the elimination of toxic aldehydes; the reduction of GSH thus might have contributed to the observed increase in lipid peroxidation products. Interestingly, hyperglycemia-associated activation of polyol pathway enzymes, particularly AR, is observed in the TCO- and TMO-fed rats. Aldehyde reductase, which is a key enzyme involved in Polyol pathway, has significant role in maintaining GSH level. It converts excess blood glucose to sorbitol, which disturbs oxidative balance by exhausting reduced form of nicotinamide adenine dinucleotide phosphate (NADPH). 50 On the other hand, this depletion in NADPH affects GSH regeneration, as it is necessary for the restoration of GSH from glutathione disulfide by the action of GR. 50 Thus it could be possible that enhanced polyolyl pathway enzyme activities account for the observed reduction in GSH levels. Further, the diminished activity of hepatic antioxidant enzymes, comprising of SOD and catalase, is observed in the TCO and TMO groups, which is in line with the previous studies. 51,52 Reduction in the antioxidant enzyme activity is also thought to be involved in the progression of nonalcoholic fatty liver disease. These observations are in corroboration with the increased incidence of microvesicles, hepatocellular ballooning, and lipogranulomas in the TCO- or TMO-fed rats. These results thus indicate a comparatively higher degree of hepatic damage in the animals fed with high fructose and thermally oxidized edible oil. Considering these, results of the present study might support the fact that long-term consumption of sugar-sweetened beverages with fried fats increases metabolic diseases.
Conclusion
Overall, the present study revealed that heated monounsaturated and medium-chain saturated fats equally contribute to the development of hepatic disorders by disrupting the antioxidant defense. These finding are thus suggestive of the role of oil fried foods in the causation of lifestyle-associated diseases.
Footnotes
Acknowledgments
First author is thankful to the Council of Scientific and Industrial Research, India, for the financial support. JJM acknowledges the receipt of DST-Inspire fellowship. SEM and SPI are recipients of fellowship from University Grants Commission-Faculty Development Program and Kerala State Council for Science Technology and Environment (letter no. 01/FSHP MAIN/2015/KSCSTE). Authors also acknowledge Dr Gopinathan, Pathologist, Jeeva laboratory and Pathcentre, Thrissur, for histological analysis.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Council of Scientific and Industrial Research, India, in the form of Junior Research Fellowship (09/869 (0012)/2012-EMR-I).
