Abstract
Acquired immunodeficiency syndrome (AIDS) is a worldwide disease characterized by impairments of immune function. AIDS can be associated with oxidative stress (OS) that can be linked to selenium (Se) deficiency. Se is fundamental for the synthesis of selenoproteins, such as glutathione peroxidase and thioredoxin reductase. These enzymes catalyze the decomposition of reactive oxygen species and contribute to maintain equilibrium in cell redox status. Literature data indicate that organoselenium compounds, such as ebselen and diphenyl diselenide, have antioxidant properties in vitro and in vivo models associated with OS. Nevertheless, selenocompounds can also react and oxidize thiols groups, inducing toxicity in mammals. Here, we tested the potential cytotoxic and genotoxic properties of six analogs of the prototypal anti-HIV drug azidothymidine (AZT) containing Se (5′-Se-(phenyl)zidovudine; 5′-Se-(1,3,5-trimethylphenyl)zidovudine; 5′-Se-(1-naphtyl)zidovudine; 5′-Se-(4-chlorophenyl)zidovudine) (C4); 5′-Se-(4-methylphenyl)zidovudine (C5); and 5′-(4-methylbenzoselenoate)zidovudine). C5 increased the rate of dithiothreitol oxidation (thiol oxidase activity) and C2-C4 and C6 (at 100 µM) increased DNA damage index (DI) in human leukocytes. Moreover, C5 (200 µM) decreased human leukocyte viability to about 50%. Taken together, these results indicated the low in vitro toxicity in human leukocytes of some Se-containing analogs of AZT.
Introduction
Acquired immunodeficiency syndrome (AIDS) is a worldwide known disease characterized by progressive depletion of the CD4C helper/inducer subset of T lymphocytes, which leads to severe impairment of immune function. 1,2 In addition, AIDS can be associated with depletion of antioxidant system and an increase in the oxidative stress (OS), 3 –7 which is defined as an imbalance of oxidants and antioxidants in favor of the oxidants. 8 –12
Another factor that can contribute to the progression of OS is a decrease in total body content of selenium (Se) in HIV patients. 13 –15 Se is involved in the synthesis of important organic molecules, including the rare amino acid selenocysteine. 16 –18 This amino acid is present in selenoproteins and selenoenzymes, such as glutathione peroxidase (GPx) and thioredoxin reductase (TrxR), which are involved in the maintenance of equilibrium between the production and decomposition of reactive oxygen species (ROS) in the body. 19 –21
In the last four decades, the synthesis and the search for new organoselenium compounds with potential antioxidant properties have been increased (for comprehensive review, see literatures 22 –26 ). Ebselen and diphenyl diselenide, two organoselenium compounds that have been extensively studied in the literature, 22 –26 present antioxidant effects in different in vitro and in vivo models associated with OS. The mechanisms involved in their antioxidant activity are related to their transformation to selenol intermediates (see Online Supplementary content 1) either via their direct reduction by endogenous reduced thiols (normally glutathione, pathway 2 in Online Supplementary content 1) or via a catalyzed reaction involving TrxR (pathway 1 in Online Supplementary content 1). 27 –29 The selenol intermediates can then decompose potentially toxic peroxides. However, selenocompounds can react and oxidize a variety of thiols groups without a concomitant degradation of peroxides 30,31 and can induce toxicity in mammals. 22,32
Zhan et al. 33 described the synthesis of several 1,2,3-selenadiazole thioacetanilides derivatives and tested them against in vitro HIV-1 activity by the inhibition of the virus-induced cytopathic effect in the human T lymphocyte (MT-4). Some of these compounds possess HIV-1 inhibitory activity, nevertheless, were also cytotoxic. De Souza et al. 34 show the synthesis and some in vitro biological and biochemical activities of news chalcogenozidovudine derivatives containing Se. In this work, the authors show that some of the Se molecules could exhibit antioxidant and antitumoral activity but the toxicological effects of these new molecules are unknown.
In this context, we have investigated the in vitro toxicological effects (cytotoxicity and genotoxicity) of six organoselenium compounds, which are analogs of 1-[(2 R,4 S,5 S)-4-azido-5-(hydroxymethyl)oxolan-2-yl]-5-methypyrimidine-2,4-dione (AZT). This study is important because new AZT analogs can be of pharmacological significance for AIDS treatment.
Methods
Chemicals
Dimethyl sulfoxide (DMSO), dithiothreitol (DTT), and 5,5′-dithiobis-(2-nitrobenzoic acid) (Ellman’s reagent or DTNB) were obtained from Sigma-Aldrich (St Louis, Missouri, USA). All other chemicals were of analytical grade and obtained from standard commercial suppliers.
Compounds
Compounds 1–6 (Figure 1) were prepared according to de Souza et al.
34
The purity of compounds 1–5 and also zidovudine were higher than 95% (the purity data were present in the article cited above). However, only compound 5′-(4-methylbenzoselenoate) zidovudine (C6) have a purity higher than 90% (supplemental content 2).
Compound 1: 5′-Se-(phenyl)zidovudine; Compound 2: 5′-Se-(1,3,5-trimethylphenyl) zidovudine; Compound 3: 5′-Se-(1-naphtyl)zidovudine; Compound 4: 5′-Se-(4-chlorophenyl)zidovudine; Compound 5: 5′-Se-(4-methylphenyl)zidovudine; Compound 6: 5′-(4-methylbenzoselenoate)zidovudine; and Zidovudine (azidothymidine or AZT).
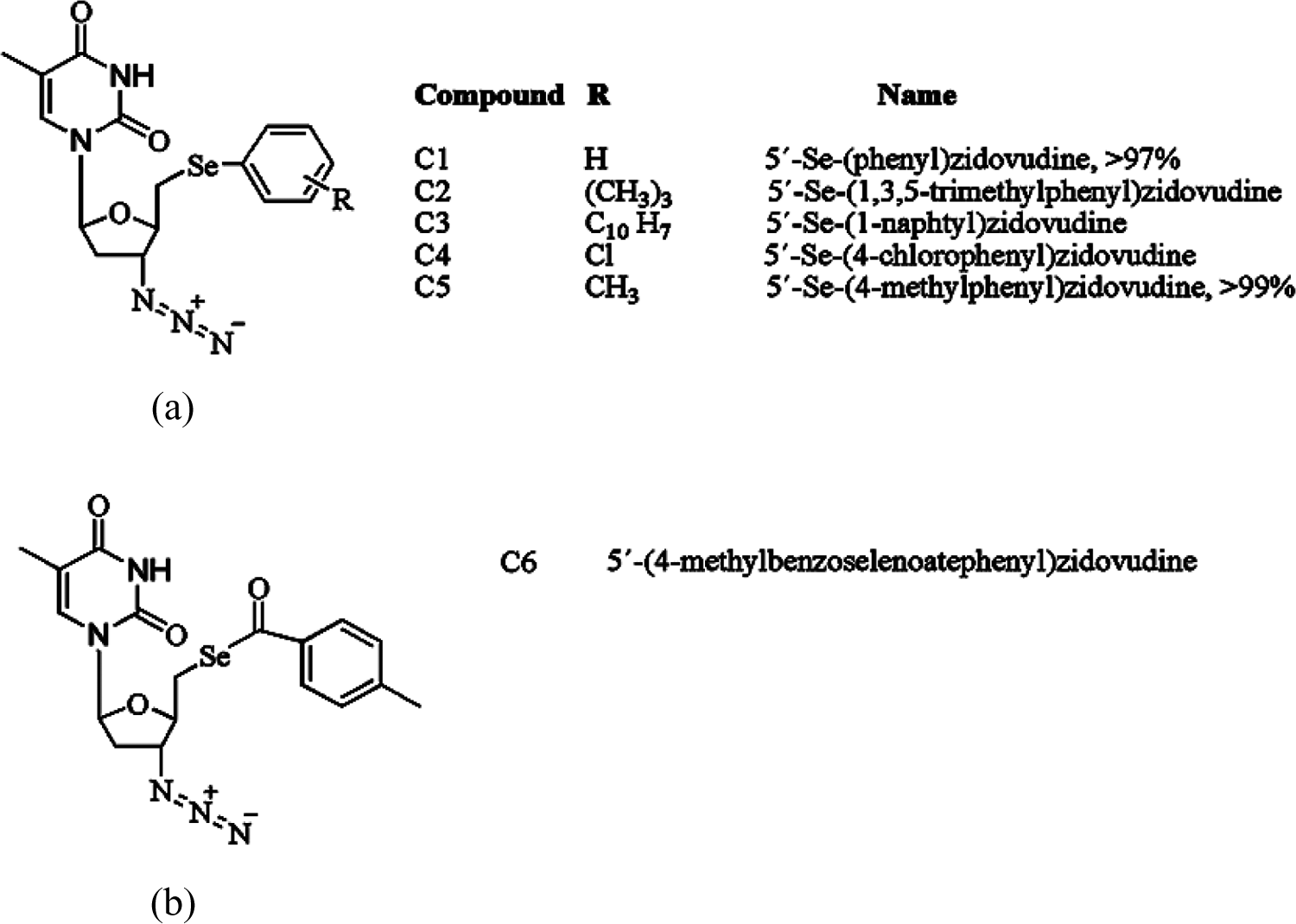
(a) Chemical structure of compounds 1–5 showing that the difference between each compound was the radical group bound to the aromatic ring. (b) Chemical structure of compound 6.
Blood samples
Heparinized venous blood was obtained from healthy volunteer donors from the Hospital of Federal University of Santa Maria (UFSM), Santa Maria, RS, Brazil (age 30 ± 12). The protocol of study was reviewed and approved by the appropriate institutional review board from Guidelines of the Committee of UFSM (0089.0.243.000-07).
Leukocyte isolation
Peripheral blood leukocytes were isolated and adjusted to 2 × 106 leukocytes/mL with hank’s balanced salt solution (HBSS)/heparin, according Santos et al. 35
Comet assay
Comet assay was performed according to Santos et al. 35 Leukocytes were incubated for 3 h, at 37°C, with AZT or AZT analogs (100 µM) or DMSO (vehicle; final concentration 0.05%). One hundred randomly selected cells per sample were scored visually by three independent individuals, which were blind to the treatments. Cell damage was scored according to tail intensity into five classes (0 for undamaged and 4 for highly damaged cells). Damage index (DI) was calculated using the formula: (n1 × 1 + 2n × 2 + 3 × n3 + 4n × 4), in arbitrary units. Methyl methanesulfonate (10 µM) was used as positive control in the comet assay. The concentration was selected in the basis of previous study from our laboratory with Se compounds. 30,35 –37 The concentration used here is expected to be in the high toxic range.
Cell viability assay
The percentage of viable cells was measured according Santos et al. 35 Leukocytes were incubated for 3 h, at 37°C, with DMSO (vehicle; final concentration 0.05%) or AZT or AZT analogs (5, 25, 50, 75, 100, and 200 µM). A solution of hydrogen peroxide (2 mM) plus sodium azide (1 mM) was used as positive control for cell viability. An aliquot of samples was added in a medium containing HBSS and 0.04% of trypan blue. Cell viability was calculated as the number of living cells divided by the total number of cells multiplied by 100. The concentrations used were expected to cover the pharmacological and the toxicological concentrations of the compounds. 36
Thiol oxidase activity assay
Oxidation of 1 mM DTT was determined in a medium containing 30 mM Tris-hydrochloric acid, pH 7.4, in the absence or in the presence of AZT or Se-containing analogs (100 µM). We have also tested the oxidation of thiols with 10 µM of Se-AZT analogs, but they did not increase the rate of DTT oxidation (data not shown). This was the maximal concentration selected to avoid any interference in the color reaction (i.e. the compound after dilution for color development was lower than 5 µM and this is not expected to oxidize 2-nitro-5-thiobenzoate or TNB back to DTNB). 38 The oxidation of –SH groups of DTT as function of time was quantified using the Ellman’s reagent as described in Rocha et al. 39
Statistical analysis
Statistical analysis was performed either by one-way analysis of variance (ANOVA), followed by Duncan’s multiple range test when appropriate. DTT oxidation (Figure 2 and Table 3) were analyzed by two-way ANOVA (eight compounds (DMSO, AZT, and six selenocompounds) × five times of sampling) with time treated as repeated measures. Statistical analysis revealed a significant interaction between compounds and time (p < 0.05). All values were presented as mean ± standard error of mean (SEM) and the differences were considered significant when p < 0.05.
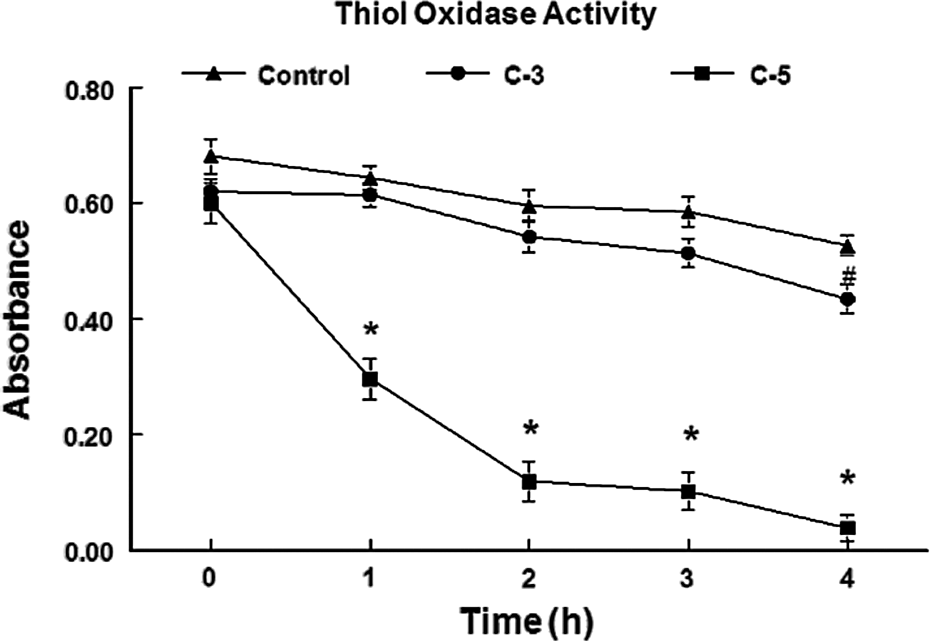
Thiol oxidase activity of compounds 3 and 5 during 4 h at 100 µM. Data are expressed as mean of DTT absorbance ± SEM. #A significant difference (p < 0.05) from control. *Significant difference (p < 0.01) from control. SEM: standard error of mean; DTT: dithiothreitol.
Results
DNA damage in blood leucocytes
Compounds 3, 4, and 6 caused a significantly decrease in the frequency of undamaged cells (all compounds were tested at a final concentration of 100 µM). Compounds 2, 3, 4, and 6 increased the total DNA DI and the frequency of cells in the level 4 of damage, when compared with control group (p < 0.01; Table 1). Compound 6 also increased the frequency of cells in the level 1 of DNA damage (p < 0.05; Table 1).
DNA damage is expressed as DNA damage levels of percentage of cells and DNA DI as a.u.a
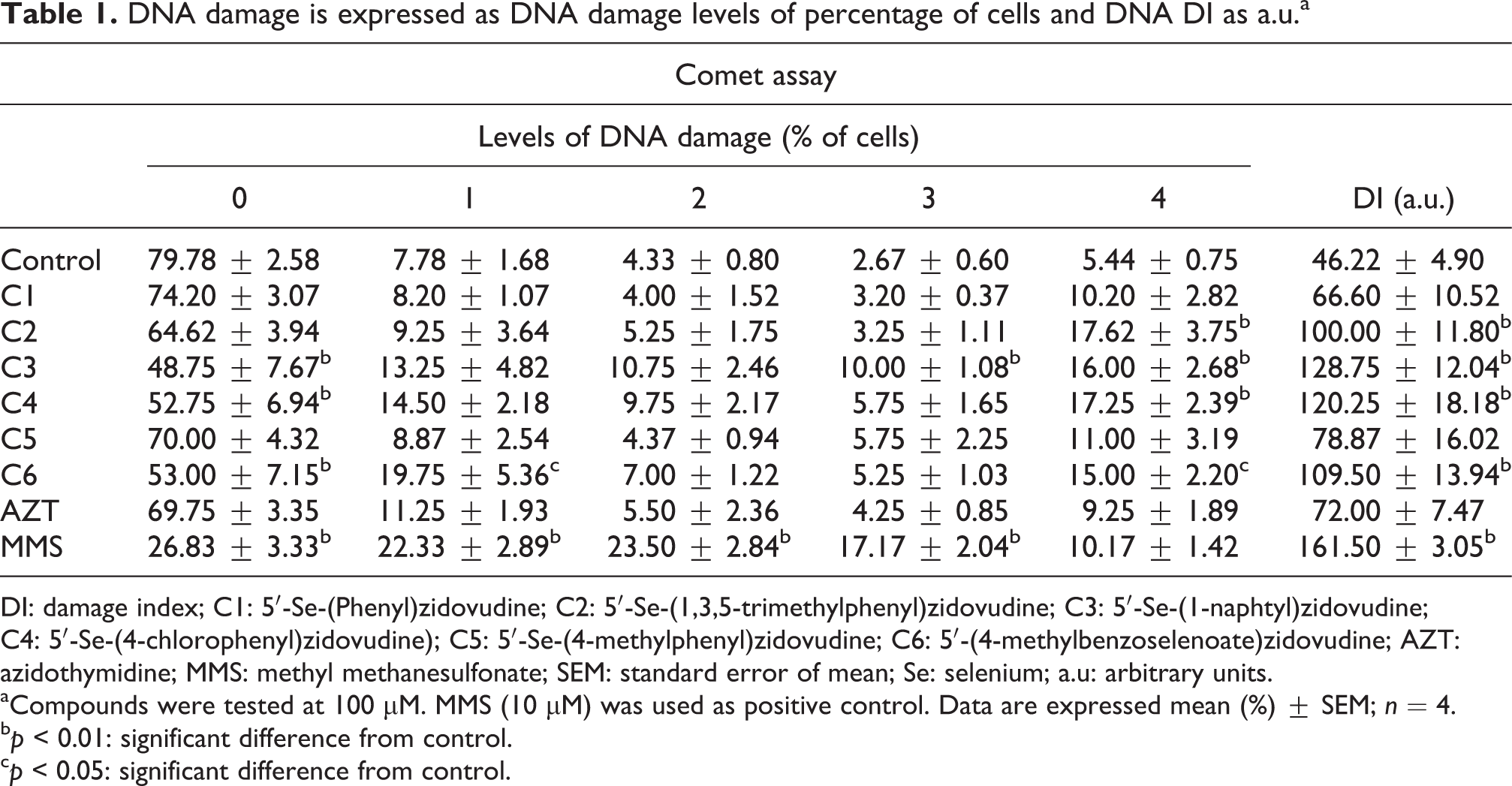
DI: damage index; C1: 5′-Se-(Phenyl)zidovudine; C2: 5′-Se-(1,3,5-trimethylphenyl)zidovudine; C3: 5′-Se-(1-naphtyl)zidovudine; C4: 5′-Se-(4-chlorophenyl)zidovudine); C5: 5′-Se-(4-methylphenyl)zidovudine; C6: 5′-(4-methylbenzoselenoate)zidovudine; AZT: azidothymidine; MMS: methyl methanesulfonate; SEM: standard error of mean; Se: selenium; a.u: arbitrary units.
aCompounds were tested at 100 µM. MMS (10 µM) was used as positive control. Data are expressed mean (%) ± SEM; n = 4.
b p < 0.01: significant difference from control.
c p < 0.05: significant difference from control.
Cell viability assay
Human leukocytes viability was decreased by compounds C1–C5 and AZT (p < 0.01; Table 2). However, compound 5 was more toxic to leukocytes than the other analogs; which caused a decrease in cell viability even when tested at 5 µM. In contrast, compound 6 did not change the viability of leukocytes (Table 2).
Cell viability is expressed in percentage of viable cells.a
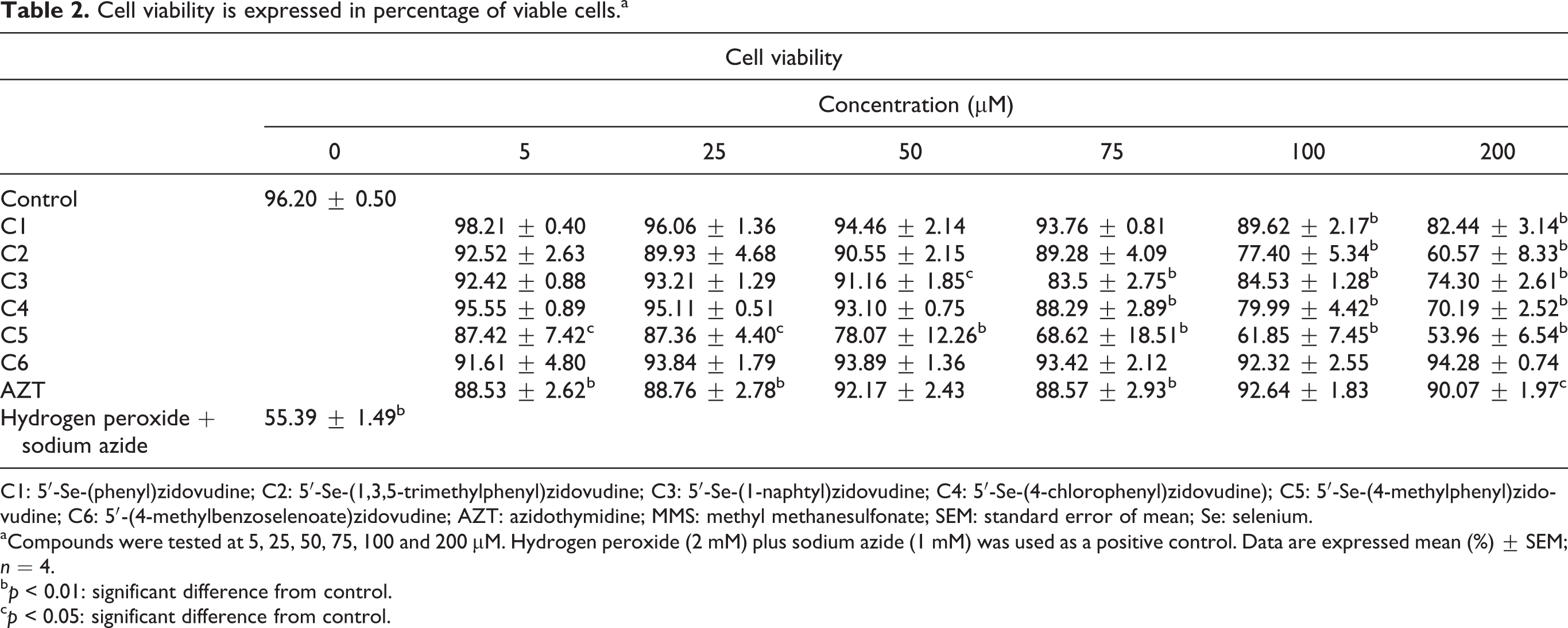
C1: 5′-Se-(phenyl)zidovudine; C2: 5′-Se-(1,3,5-trimethylphenyl)zidovudine; C3: 5′-Se-(1-naphtyl)zidovudine; C4: 5′-Se-(4-chlorophenyl)zidovudine); C5: 5′-Se-(4-methylphenyl)zidovudine; C6: 5′-(4-methylbenzoselenoate)zidovudine; AZT: azidothymidine; MMS: methyl methanesulfonate; SEM: standard error of mean; Se: selenium.
aCompounds were tested at 5, 25, 50, 75, 100 and 200 µM. Hydrogen peroxide (2 mM) plus sodium azide (1 mM) was used as a positive control. Data are expressed mean (%) ± SEM; n = 4.
b p < 0.01: significant difference from control.
c p < 0.05: significant difference from control.
Thiol oxidase activity of seleno-AZT analogs
From the tested compounds, C5 exhibited a high thiol oxidase activity; whereas compound 3 had a small but statistically significant thiol oxidase activity after 4 h of reaction (p < 0.01; Table 3 and Figure 2).
Thiol oxidase activity of selenium AZT analogs.a
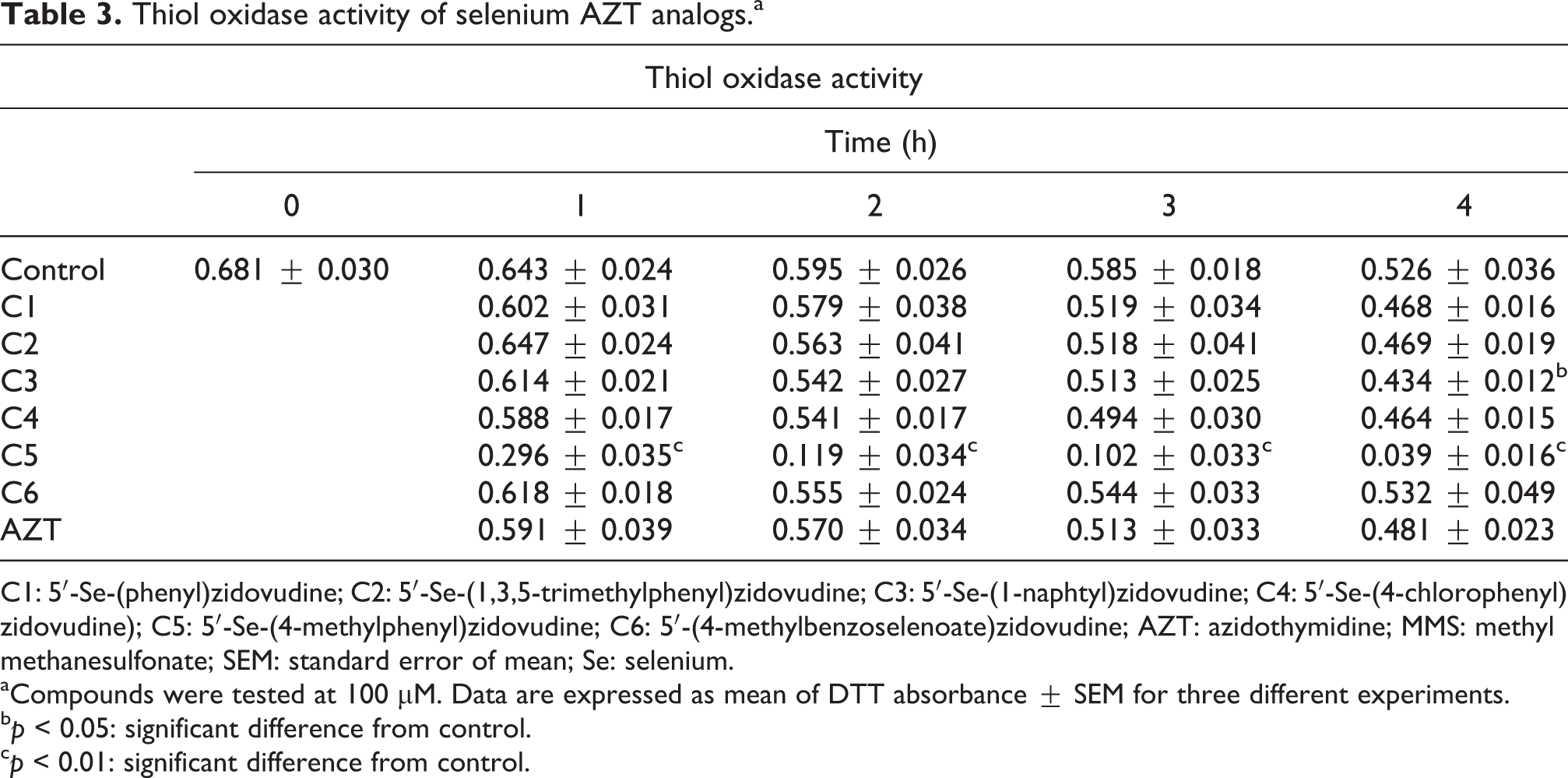
C1: 5′-Se-(phenyl)zidovudine; C2: 5′-Se-(1,3,5-trimethylphenyl)zidovudine; C3: 5′-Se-(1-naphtyl)zidovudine; C4: 5′-Se-(4-chlorophenyl)zidovudine); C5: 5′-Se-(4-methylphenyl)zidovudine; C6: 5′-(4-methylbenzoselenoate)zidovudine; AZT: azidothymidine; MMS: methyl methanesulfonate; SEM: standard error of mean; Se: selenium.
aCompounds were tested at 100 µM. Data are expressed as mean of DTT absorbance ± SEM for three different experiments.
b p < 0.05: significant difference from control.
c p < 0.01: significant difference from control.
Discussion
AIDS infection can be associated with OS. 3 –7 AZT is one of the most common drugs used in the treatment of AIDS (40); however, AZT can present a prooxidant activity. Accordingly, AZT has been reported to increase ROS and MDA formation in mouse cardiac mitochondria, macrophages, and endothelial cells. 40 –43
De Souza et al. 34 tested compounds 1–5 against antioxidant activities. They show that Se-containing AZT analogs are weak inhibitors of Fe (II)-induced thiobarbituric acid reactive substances formation in rat’s brain homogenate. Also, only compounds 4 and 5 displayed relatively good GPx-like activity at higher concentration.
But, under in vitro aerobic conditions, compound 5 (C5) had a clear and apparent thiol oxidase activity. As suggestion, the activating or deactivating groups bound to Se-containing ring may not significantly contribute in thiol or specifically (in this case) DTT oxidation. The thiol oxidase was determined in the presence of molecular oxygen. The presence of molecular oxygen is important in determining the thiol oxidase activity of Se compounds. Thus, extrapolating to in vivo conditions, we can expect that C5 be able to oxidize thiol groups found in cell.
Of particular toxicological significance, compounds 2, 3, 4, and 6 had weak in vitro genotoxic properties in human leukocytes, when tested at a high concentration (100 µM). Compounds 1 to 5 also caused a significant decrease in leukocytes viability, when they were tested at high concentrations (100 and 200 µM). Furthermore, compound 5 decreased cell viability even when tested at a relatively low concentration (5 and 25 µM).
When tested during 48 h against human bladder carcinoma cells (5637), the growth inhibition rate (half maximal inhibitory concentration) of compound 1 was 78.85 µM. 34 Also, at 100 µM, this molecule increased the number of apoptotic cells and apoptosis-associated DNA damage in this carcinoma cells. It is difficult to correlate different assays but we observed that at 100 µM, C1 shows only a weak genotoxicity and decrease in cell viability in human leukocytes. This may indicate a selective toxicity toward tumor cells, when compared with normal blood cells.
Some compounds, for instance, compound 6, exhibited only a modest genotoxic effect and had no toxic additional activity. In contrast, compound 5 had cytotoxic activity and had no genotoxic property. It is interesting to note that different organochalcogens compounds can exhibit genotoxicity to human and rodent cells, depending on the concentration and on the chemical structure. 30,35 –37,44
Here, we have observed that at relatively high concentrations, compounds 2, 3, 4, and 6 increased DNA damage. Literature has indicated that AZT can cause DNA breaks, which can be repaired in different cell types. 45 Here, AZT did not cause a significant increase in DNA damage, indicating that Se analogs of AZT (C2, C3, C4, and C6) were more toxic than the prototype compound AZT and C1 and C5. A question to be answer is how much the damage caused by these compounds could be restored by related DNA repair enzymes. 45
However, as a rule, Se-containing-AZT analogs were less toxic than ebselen and diselenide in the comet assay and viability test. 36 Consequently, it would be interesting to test the in vivo toxicity of AZT analogs in animal models.
One important aspect that was not investigated here is the use and application of this interesting class of AZT analogs as possible HIV pharmacological agents. Whether it can inhibit reverse transcriptase enzymes against HIV is yet to be explored. Recently, Sancineto et al. 46 describes the synthesis and anti-HIV activity of a series of organoselenium compounds derived from 2,2′-dithiobisbenzamides. Some 2,2′-diselenobisbenzamides molecules had anti-HIV activity at low concentration (half maximal effective concentration < 14 µM; concentration of compound required to achieve 50% protection) but also induced cytotoxicity between 115 and 170 µM (CC50: concentration of compound that reduces the viability of mock-infected cells by 50%) in MT-4 cells.
Furthermore, the monoselenide analogs of AZT are expected to give limited protective effects against the prooxidant situation in AIDS. Logically, the presence of Se atom can give to the AZT molecule some advantages over the parent compound (AZT). However, as mentioned above, the detail and mechanistic studies are needed.
Conclusion
The development and the pharmacological use of organoselenium compounds can be considered an additional tool in the combat of diseases associated with OS and inflammation. 47 Here, we observed two AZT analogs containing Se (C1 and C6) have low toxic activity at 100 µM. At high concentration, compounds 2 and 4 caused DNA damage and were cytotoxic; compounds 3 and 5 exhibited a thiol oxidase activity and cytotoxicity. These results may indicate potential pathways via which they can be toxic to mammal cells. Consequently, the potential inhibitory effects of these compounds against HIV reverse transcriptase can be highly informative and can indicate whether or not the toxicity of these compounds should be investigated in rodents.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We are thankful for the financial support from FAPERGS/PRONEX, INCT for Excitotoxicty and Neuroprotection, CNPq, CAPES, and UFSM.
Supplemental material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
