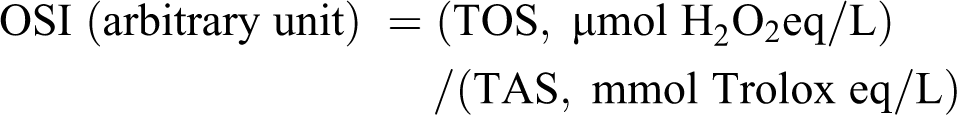Abstract
Background:
The widespread use of synthetic cannabinoids (SCs) among youth has become an important public health problem. Several life-threatening side effects of SC have been reported, including cardiovascular, gastrointestinal, neurological, renal, metabolic, ophthalmologic, and pulmonary effects, besides skin toxicity and hepatotoxicity.
Methods:
Given that high levels of SC can lead to oxidative stress, DNA damage, and inflammation, it has been aimed in this study to investigate the effects of SC in aspects of primary DNA damage, plasma total oxidant status (TOS)/total antioxidant status (TAS), thiol–disulfide homeostasis, myeloperoxidase (MPO) level, and cytokine levels (interleukin 1 beta (IL-1β), interleukin 6 (IL-6), and tumor necrosis factor-alpha (TNF-α)) of 40 SC users (SCUs) in Turkey.
Results:
Mean plasma TOS levels were significantly higher in the SCUs group than in the healthy group (HG). Similarly, mononuclear leukocyte DNA damage, plasma TOS, MPO activity, disulfide, oxidative stress index levels, IL-1β, IL-6, and TNF-α levels were significantly higher in the SCU group than in the HG, whereas plasma TAS, total, and native thiol levels were significantly lower in the SCU group than in the HG.
Conclusion:
It is concluded that SC can cause increase in oxidative stress and in inflammatory processes in addition to its potential for DNA damage. Additional studies with larger sample sizes and longer durations should be held to understand more specific outcomes of SC use.
Introduction
Since ancient years cannabis (marijuana) has been widely used for its pharmacologic effects as in the treatment of various diseases, or for its psychoactive properties in recreational use, synthetic cannabinoids (SCs), which were initially created for medicinal purposes, eventually became a common drug of abuse and the most widely used illegal drugs in the world. 1 The widespread use of SC among youth has become an important public health problem. 2
SC is sold under various names such as “spice,” “bonsai,” “K2,” “dream,” and “JWH-018.” Several life-threatening side effects of SC have been reported including cardiovascular effects, gastrointestinal, neurological, renal, metabolic, ophthalmologic, pulmonary, skin toxicity, and hepatotoxicity. 3 –5
Obtained from the plant Cannabis sativa or Cannabis indica, there are more than 500 chemical compounds and 100 cannabinoids, also named phytocannabinoids. 6,7 Due to their herbal aspect, SC is misused as “natural products.” 4 The most effective compounds in SC are tetrahydrocannabinol (THC), cannabidiol, cannabinol, cannabigerol, tetrahydrocannabivarin, cannabichromene, tetrahydrocannabivarin acid, delta-8 THC, and cannabidivarin. 8,9
There are a few studies focusing on the toxicity of cannabinoids describing cellular alterations caused by THC. 10 Given that high levels of SCs can lead to oxidative stress, 11,12 genotoxicity, 13,14 and inflammation, 15 the effects of the molecule at the blood concentrations of bonsai users in Turkey have been investigated in the aspects of total oxidant status (TOS)/total antioxidant status (TAS), thiol–disulfide homeostasis, myeloperoxidase (MPO) level, primary DNA damage (comet assay), and cytokine levels.
Studies are showing that an important underlying cause of cannabinoid toxicity can be oxidative stress, and this can be attributed to the effect of THC, which may induce oxidative stress (in vitro and in vivo). 11,12,16
Oxidative stress emerges when the prooxidant–antioxidant balance gets disturbed due to the accumulation of reactive oxygen species (ROS). Minor changes in the redox balance are compensated with immediate cellular environmental adaptations, which are explained with the concept that ROS at homeostatic level plays a central role in cellular processes. 17
On the other hand, severe accumulation of ROS due to disruption in the redox balance can cause severe deficiencies by interrupting the regular functions of the cell irreparably, thus, affecting the system by the whole. 18 For example, stroke is an important toxic effect among cannabinoid users, which is stated in several studies, 19 –21 and the generation of ROS is one of the known mechanisms of stroke in human. 22 It is also emphasized that the pathological effects of ROS accumulation such as diabetes, 23 liver necrosis, 24 and cancer 25 are mechanistically due to oxidized macromolecules, which can be sorted as lipid peroxidation, protein modifications, and DNA oxidation.
The antioxidant defense system consists of two major groups: antioxidant enzymes such as SOD, CAT, GR, and GPx and the nonenzyme antioxidant molecules such as GSH, vitamin E, and vitamin C. 26,27
The thiol–disulfide homeostasis takes place in critical cellular functions such as antioxidant protection, apoptosis, detoxification, and cellular signaling. 28 It has been seen that abnormal thiol–disulfide homeostasis can be involved in the underlying mechanisms of many disorders. 29 –31 Considering that the determination of thiol–disulfide homeostasis provides useful information about critical biochemical processes, it has been measured in the present study.
In the present study, genotoxicity was examined by the comet assay, which indicates DNA damage in single cells, which is based on the migration of DNA using electrophoresis cells. 32,33 According to the findings of recent investigations, the toxic effects of drugs may be due to the DNA-damaging properties. 13,14 Koller et al. report that damage of the genetic material can be an important cause of SC-induced toxicity. 14,34,35
Along with genotoxicity and oxidative stress 13 induction of inflammation can play a significant role in the occurrence of various diseases including cancer. 36 –38 Therefore, in the present study, the cytokine blood levels of the drug users were measured to see the effects of SC on inflammatory reactions.
Materials and method
The study was conducted in accordance with the Basic & Clinical Pharmacology & Toxicology Policy for experimental and clinical studies. 39 The study was planned as an observational, case–-control study to investigate the roles of DNA damage, systemic oxidative stress, thiol–disulfide homeostasis, and inflammation in SC addicts. Ethical approval for the study was obtained and was conducted with the principles of the Declaration of Helsinki. Written informed consent was obtained from each participant.
From February 2019 to August 2019, this prospective case–control study included 40 adult users (sex: 40 male; age: 18–45) who were diagnosed with SC addiction in the University of Health Sciences Fatih Sultan Mehmet Education and Research Hospital. All selected users were untreated individuals without using any other drugs but SC at least 2 years. For the healthy control group, 40 participants (sex: male; age: 18–45) were included in the study group with age and sex matching with the user group. This study has been approved by Bezmialem Vakif University Ethics Committee with the decision number 16/17.
Blood sample collection
After confirmation, blood samples were taken from the antecubital vein of each user, collected into heparinized tubes. Blood tubes were transferred to the biochemistry laboratory and were analyzed without delay. For peripheral mononuclear leukocyte separation, 1-mL heparinized blood was layered over 1-mL Histopaque 1077 (Sigma-Aldrich, St. Louis, Missouri, USA) and centrifuged for 25 min at 700 × g and 25°C. The interface band containing mononuclear leukocytes was washed with phosphate-buffered saline (PBS) and then collected by 15-min centrifugation at 500 × g. The resulting pellets were resuspended in PBS. Membrane integrity was assessed by Trypan Blue exclusion method. The remaining blood samples were centrifuged at 3000 × g for 10 min in order to collect the plasma. The separated plasma was frozen immediately at –80°C until further analysis of plasma interleukin 1 beta (IL-1β), interleukin 6 (IL-6) and tumor necrosis factor-alpha (TNF-α), MPO, total thiol (TT) and native thiol (NT) and TOS and TAS were measured. Oxidative stress index (OSI) and thiol–disulfide level were calculated by a mathematical equation.
Mononuclear leukocyte isolation for DNA damage analyses
The comet assay was conducted as described by Singh et al. 32 with slight modifications: 10 mL of fresh mononuclear leukocyte (MNL) cell suspension (about 20,000 cells) was mixed with 80 µL of 0.65% low melting point agarose in PBS at 37°C. Afterward, 80 µL of the mixture was plated onto a 1% normal melting point agarose-coated-slide and covered with a coverslip. The slides were kept at 4°C for 5 min to solidify the agarose. After the gel became solid, the slides were incubated with fresh cold (4°C) lysis buffer (2.5 M sodium chloride, 100 mM Na2Ethylenediaminetetraacetic (EDTA), 10 mM Tris-HCl, pH = 10–10.5) for at least 1 h: 1% Triton X-100 and 10% dimethyl sulfoxide (DMSO) were added just ahead of using the buffer. After that, the slides were immersed at 4°C in a freshly made alkaline buffer (0.3 mol/L sodium hydroxide and 1 mmol/L Na2EDTA, pH = 13) in the electrophoresis tank. After a 20-min DNA unwinding period, the electrophoresis was performed at 4°C for 30 min at 0.7 V/cm and 300 mA.
After the electrophoresis process, the slides were washed at 4°C for 5 min with neutralization buffer (0.4 M Tris, pH = 7.5) and dehydrated with ethanol. The slides were stained with ethidium bromide (2 μg/mL in distilled H2O; 70 μL/slide) covered with a coverslip and analyzed with a fluorescence microscope (Leica DM 1000, Solms, Germany) with a rhodamine filter (an excitation wavelength of 546 nm and a barrier of 580 nm). All experiments were done under minimal light. A computerized image analysis system (Comet Assay IV; Perceptive Instruments, Bury St Edmunds, UK) was used for the evaluation of the degree of DNA damage levels. As the primary criterion for DNA damage, the percentage of DNA in the tail intensity (tail intensity %) was used. 40 All steps were performed three times.
Determination of TOS
Plasma TOS was measured with a novel automated analyzing method developed by Erel. 41 Oxidants oxidize the ferrous ions of o-dianisidine compounds to ferric ions. The oxidation leads to the formation of ferric ions that are seen as colorful compounds in acidic environments using xylenol orange; the density of the color is proportional to the oxidant level. In this study, hydrogen peroxide (H2O2) was used for calibration of the assay. The results are expressed as micromoles of H2O2 equivalent per liter.
Determination of TAS
Plasma TAS was measured with an automated colorimetric assay 42 based on the quantitative measurement of OH radicals. The (Fe+2 + o-dianisidine) compound leads to Fenton-type reactions with H2O2 to form OH radicals which react with the o-dianisidine molecules creating yellow-brown dianisidine radicals. Antioxidants stop the color formation by inhibiting oxidant reactions. The experiments were performed by spectrophotometry at 240 nm with an automated analyzer (Thermo Scientific Varioskan Flash Multimode Reader, Waltham, Massachusetts, USA). Trolox, a water-soluble analog of vitamin E, was used as the calibrator. This assay shows the antioxidative capability of the sample against potent free radical reactions triggered by the OH radical. The results are given as millimoles of Trolox equivalent per liter millimole.
OSI
OSI is an indicative rate of oxidative stress. It is showed as a percentage of the ratio of plasma TOS to TAS which is calculated with the following formula
Activity of MPO
Plasma MPO activity, which is one of the main oxidative enzymes, was determined by the method of Bradley et al. 43 Based on the kinetic measurement of the formation rate of the yellowish-orange product of the oxidation of o-dianisidine with MPO in the presence of H2O2, at 460 nm, the MPO activity was given as units per liter of plasma.
Determination of thiol/disulfide homeostasis
Plasma TDH was measured by a method previously explained. 42 An automated clinical chemistry analyzer (Thermo Varioskan Multireader) was used in this study. Disulfide bonds are reduced to free functional thiol groups with sodium borohydride. Unused reductant sodium borohydride was used and removed with formaldehyde to avoid the reduction of 5,5′-dithiobis-(2-nitrobenzoic) acid. The thiol groups (reduced and NT) were detected after the reaction with 5,5′-dithiobis-(2-nitrobenzoic) acid. Fifty percent of the difference between the TTs and NTs provides the dynamic disulfide quantity. After the determination of –SH and –SH+–S–S–, –S–S– and –S–S–/–SH %, –S–S/–SH+– S–S%, and –SH/–SH+–S–S–% were calculated.
Cytokine levels
Plasma IL-1β, IL-6, and TNF-α levels were measured with an enzyme-linked immunosorbent assay kits for human, according to protocols provided by manufacturers (Elabscience, Houston, Texas, USA). All analyses were done with a microplate reader (Varioskan Flash; Thermo), and the results were recorded.
Statistical analysis
All data analyses were conducted using IBM SPSS version 22.0 software (IBM Corporation, Armonk, New York, USA). The numerical data were expressed as mean ± standard deviation, whereas categorical variables were expressed as numbers of users (n). Intergroup comparisons (controls vs. users) were made using χ 2 and Mann–Whitney U tests as appropriate. Student’s t-test was used to compare parametric variables between the user and control groups. Bivariate correlation analyses were done by Spearman’s test.
Results
Among the 40 drug users included in our research, the mean age was 31.23 ± 5.82 years (age range: 18–42 years), and all subjects are males. Among the 40 healthy volunteers, the mean age was 31.18 ± 5.82 years (age range: 19–45 years), and all volunteers are males. There was no significant difference between the synthetic cannabinoid users (SCUs) and healthy groups (HGs) in terms of age (p = 0.973). Mean plasma TOS levels were significantly higher in the SCU group than in the HG. Similarly, lymphocyte DNA damage (Figure 1); plasma TOS, MPO activity, disulfide, and OSI levels; and IL-1β, IL-6, and TNF-α levels were significantly higher in the SCU group than in the HG, whereas plasma TAS, total, and NT levels were significantly lower in the SCU group than in the HG.

DNA damage (SCU no: 1 and healthy control no: 1). SCU: synthetic cannabinoid user.
Table 1 presents the comparisons of the SCU and HG in terms of lymphocyte DNA damage, plasma oxidative stress parameters (TAS, TOS, OSI), thiol–disulfide homeostasis parameters (TT and NT, disulfide), and inflammatory cytokines levels (IL-1β, IL-6, and TNF-α).
DNA damage, oxidative stress, and inflammation parameters of 40 SCUs and 40 healthy controls.a

HG: healthy group; SCU: synthetic cannabinoid user; TOS: total oxidant status; TAS: total antioxidant status; OSI: oxidative stress index; MPO: myeloperoxidase; IL-1β: interleukin 1 beta; IL-6: interleukin 6; TNF-α: tumor necrosis factor-alpha; TT: total thiol; NT: native thiol; SD: standard deviation.
a Data are presented as mean ± SD and number of volunteers.
DNA damage was positively correlated with TOS (r = 0.734, p = 0.001), OSI (r = 0.834, p = 0.001), MPO (r = 0.828, p = 0.001), disulfide (r = 0.381, p = 0.001), IL-1β (r = 0.765, p = 0.001), IL-6 (r = 0.899, p = 0.001), and TNF-α (r = 0.878, p = 0.001) and negatively correlated with TAS (r = −0.819, p = 0.001), TT (r = −0.760, p = 0.001), and NT (r = −0.845, p = 0.001).
There was a strong positive correlation between addict duration and OSI (r = 0.887, p < 0.001) and between addict duration and TOS (r = 0.530, p < 0.001). There was a just negative correlation between addict duration and TAS (r = −0.672, p < 0.001). There was no significant correlation between the duration of addiction inflammation and DNA damage.
Discussion
As an important problem, SCs addiction becomes more prominent nowadays. A detailed literature survey showed us that there is not enough study on human subjects evaluating oxidative stress and the inflammatory effect of SC with different oxidative parameters, and enzyme profiles are inadequately examined. Especially, MPO and thiol/disulfide levels in human subjects have not been investigated by any researcher. However, there are many studies investigating oxidative parameters of SC on cell lines and in vivo. 34,44 –46 Researchers investigating the in vitro effects of SC have one opinion in common which is that cell viability reduces upon administration of SC on various cell lines.
Jacobsson et al. have suggested that activation of vallinoid and cannabinoid receptors has been associated with SC exposure leading to cellular toxicity. 47 Increased serum levels of ROS has been seen in users who are addicted to SC. Comparison of the measured TOS levels with the control group showed a statistical difference in the present study. Similar to our study, Bayazit et al. found that cannabis addicts had higher TOS and OSI levels compared to control but they found no difference in TAS. 48
Oxidative stress emerges when the prooxidant–antioxidant balance gets disturbed due to the accumulation of ROS. Thiol–disulfide homeostasis is a novel biomarker for oxidative stress. 31 Existing oxidative stress and inflammation causes a shift toward disulfide formation and, at the same time, NT and TT levels increase as a response. 49 NT and TT are able to increase the antioxidant capacity. To the best of our knowledge, there were no studies in the literature which are investigating the effects of SC usage on thiol–disulfide homeostasis. According to our finding, a statistical significance was seen between the groups. In our result, we detected increased ROS levels in serum of SCUs. Another research group showed an in vitro study that inflammation and oxidative stress are key mediators of neurotoxicity caused by SC. 50
Mitochondria are undeniably important sources of ROS. When there is excessive leakage of free radicals from mitochondria, the consequences can be seen in mitochondrial functions. Wolff et al. 12 demonstrated in vivo that THC impairs the mitochondrial respiratory chain and causes uncoupling which ultimately increases the production of free radicals.
The human body has different proteins and enzymes to cope with oxidative stress. Homeostasis is achieved via different pathways in biological systems. As a quick response to oxidative stress, MPO levels increase. MPO is one of the important inflammatory and oxidative enzymes which has antimicrobial properties using H2O2 to produce hypochlorous acid. 51 In our work, a statistical significance has been achieved in the MPO enzyme levels which is present in serum indicating inflammation and oxidative stress. On the other hand, there is also a study indicating that cannabidiol inhibited MPO activity in neutrophils suggesting cannabidiol as a potential neuroprotective and anti-inflammatory agent. 52 Nonetheless, this result might seem controversial to our results but this may be due to their experimental model which was conducted in mice and the exposure time was rather shorter, whereas our study is investigating the outcome of chronic exposure in human. On the other hand, the amount of the substance and difference of the constituents may also be related with the opposite outcomes.
The prooxidative/antioxidative properties of SC is a controversial topic. There are studies determining antioxidant effects of SC. Most of these effects are seen by small doses administered and differ by the treatment duration (chronic exposure most probably leads to oxidative stress, as we found in our study). But yet, it has been stated by Archie and Cucullo 53 that oxidative stress emerged from smoking SC can be due to the combustion rather than the SC itself.
Koller et al. 34 stated in their work that some of the most common molecules of SC has genotoxic properties. In their study, genotoxic effects of SC on lymphocytes were investigated with the comet assay. Similar to their study, we examined genotoxicity with the comet assay and a statistical significance has been found between HG and SCU. Bileck et al. also tested the impact of SC and found micronuclei formation which indicates chromosomal aberrations and comet formation. They also gave a reasonable explanation which is that a downregulation in the protein expression of DNA repair proteins in human lymphocytes may be the cause of the detected DNA damage. 15
The same group reported in their study that pro- and anti-inflammatory cytokines expression has been raised with SC application. In their study, IL-1β has showed an upregulation. On the other hand, a significant increase in IL-6 levels was observed by some other scientists. 54 –56 Inflammatory cytokines were found increased in cell lines and animal models; however, endocannabinoids application did not increase cytotoxicity. 57,58 Contrary to this finding, exogenous cannabinoids triggered the anti-inflammatory cytokine levels as well as the immune cells such as T and B cells. 56 However, the exact mechanism which initiated this increase of cytokine levels is not known. Our results of IL-1β and IL-6 levels were consistent with the literature. A significance has been acquired in the case of IL-6 levels of HG and SCU. TNF-α is another important inflammatory cytokine found elevated after cannabinoid usage in the literature. 56 There was also a statistical significance between the groups HG and SCU. As our results and literature correlated, SC usage has inflammatory effects on human cells leading to inflammation; thus, a harmful cascade could be initiated.
However, our study has some limitations as well, such as low number of participants, lack of the exact structural knowledge of the SC (JWH, AM, WIN, and CP derivatives) used as well as the SC serum levels.
Conclusions
The results obtained from this study pointed out that SC causes both increase in oxidative stress and inflammatory processes. SC usage is a priori cause to genotoxicity. Either short-term (other than therapeutic purposes) or long-term exposure is likely to be harmful to the human body, and the negative outcomes of substance abuse adversely affect the society as a whole. Programs or initiatives to quit these highly addictive substances should be supported by each individual and institution. Additional studies with larger sample sizes and longer durations should be held to understand more specific effects of SC use. We hope this study will put light upon the path of other studies and scientists.
Footnotes
Authors’ note
EMG and MYB are co-lead authors.
Acknowledgments
The authors would like to thank Bezmialem Vakif University and Fatih Sultan Mehmet Training Hospital for the given opportunity.
Author contributions
All authors contributed to and have approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by researchers and approved by Bezmialem Vakif University Clinical Research Ethics Committee.