Abstract
Methyl tert-butyl ether (MTBE), a widely used gasoline additive and a ubiquitous environmental pollutant in many countries and regions, can cause various kinds of toxic effects on human health. However, the molecular mechanism underlying its toxic effects remains elusive. The present study aimed to explore the cytotoxicity, DNA damage and oxidative damage effects of MTBE on human bronchial epithelial cells (16HBE) and the possible role of DNA polymerase β (pol-β) in this process. RNA interference (RNAi) was used to obtain pol-β gene knocked-down cells (pol-β−). CCK-8 assay was adopted to analyze the cell viability. Alkaline single-cell gel electrophoresis (SCGE) was performed to detect the DNA damage effects of MTBE. The enzyme activity of GSH-Px, SOD, CAT and the level of MDA were assessed. The data indicated that when treated with MTBE at the concentration exceeding 50 μmol/L and for the time exceeding 24 h, the pol-β− exhibited significantly decreased cell viability and increased DNA damage effects, as compared to the control (P < 0.05). Furthermore, there was significant difference in the levels of GSH-pX, SOD, CAT and MDA between the pol-β− and the control (P < 0.05). Our investigation suggests that MTBE can cause obvious cytotoxicity, DNA damage and oxidative damage effects on 16HBE cells. DNA polymerase β may be involved in protecting 16HBE cells from the toxic effects induced by MTBE exposure. These findings provide a novel insight into the molecular mechanism underlying the toxic effects of MTBE on human cells.
Keywords
Introduction
In 1970s, the U.S. Congress passed the Clean Air Act (CAA) to prohibit the use of lead alkyls in gasoline, and, for the first time, methyl tert-butyl ether (MTBE) was used in gasoline instead of lead alkyls to improve oxygen content and octane number, so as to reduce the hazardous exhaust of motor vehicles. Since then, MTBE has been widely used in gasoline around the world.1,2 Due to the characteristics of strong volatility, solubility and low biodegradation, each step of producing, transporting and use of MTBE may be an important source of environmental MTBE.1,3,4 MTBE can migrate rapidly through the atmosphere, soil and water environment and become a ubiquitous environmental pollutant.5,6 Most of people are exposed to MTBE through air and drinking water.7–9 Especially, the occupational workers, who exposed to relatively high concentrations of MTBE, may face greater potentials of health risks.10–12 Our previous investigation in gas stations in South China suggested that the average concentration of MTBE in occupational air could be 388.38 μg/m3 (4.388 μmol/L). 13 In consumers’ pulmonary alveolar, the concentration of MTBE could be 108.7 μmol/L. 14
It has been reported that MTBE could enter the human body through respiratory and digestive systems and distributed to various organs and systems with blood circulation.10,15,16 In both animals and human, liver is the main metabolic organ of MTBE.17,18 There are three major cytochrome P450 (CYP) enzymes involved in the metabolism of MTBE.19,21 In animals, CYP2B1 and CYP2E1 play the most important role, while in human body CYP2A6 and CYP2E1 play the dominant role.17,19 The main metabolites of MTBE are tert butyl alcohol (TBA) and formaldehyde, and, both of them have been proved to be carcinogens.20,21
Previous study found that MTBE could induce obvious DNA damage in human cells. 22 Animal tests also found that MTBE exposure could induce DNA damage 23 , DNA adducts.24,25 and increase the potentials of cancer risks26,27; It was suggested that oxidative stress is one of the important mechanism underlying the toxic effects of MTBE exposure.28,29 Actually, the International Agency for Research on Cancer (IARC) has listed MTBE as a “possible human carcinogen”. 30
DNA pol-β, the core enzyme in base excision repair (BER) system in mammalian, is the smallest (39 kDa) eukaryotic DNA polymerase, was isolated from a variety of mammalian cells in 1971. 30 It was found that when the quantity or quality of DNA pol-β changes, the function of DNA damage repair will be abnormal, which may result in the acute or chronic death or malignant transformation of cells when exposed to environmental chemicals.31–33 However, the possible role of DNA pol-β in the toxic effects induced by MTBE in human cells remains unknown.
This study was designed to investigate the cytotoxicity, DNA damage and oxidative damage effects of MTBE on human bronchial epithelial cells (16HBE) and the possible role of DNA polymerase β (pol-β) in this process.
Materials and methods
Chemicals and reagents
All of the chemical materials and reagents adopted in our investigation were purchased from commercial suppliers and used following the manufactures’ protocol: Dulbecco’s Modified Eagle Medium (DMEM), G418 and lipofectamine 2000 were purchased from ThermoScientific (USA). 0.25% EDTA-Trypsin (TE), phosphate buffer saline (PBS), fetal bovine serum (FBS), extraction buffer, phenylmethanesulfonyl fluoride (PMSF) and penicillin-streptomycin were obtained from the Solarbio (China). MTBE (99.8% purity) and ethidium bromide (EB) were acquired from Sigma-Aldrich (USA). Anti-DNA polymerase beta antibody and horseradish peroxidase-conjugated goat anti-rabbit IgG were bought from Abcam (UK). Bicinchoninic acid (BCA) protein assay kit was obtained from the beyotime biotechnology (China). Cell Counting Kit-8 was bought from Dojindo (Japan). pSIREN-RetroQ vector was purchased from BD Company (USA). pEGFP-C1 vector was bought from Clontech (USA). Restriction enzymes (BamH1, EcoRI, Bgl II) were bought from TaKaRa (USA); DNA synthesis, sequencing, dsRNA were commercially prepared by Sangon (China). Glutathione peroxidase (GSH-Px), malondialdehyde (MDA), superoxide dismutase (SOD), and catalase (CAT) detection kits were purchased from Jiancheng Bioengineering Institute (China).
Cell culture
16HBE cells (PCS-300-010) used in this study were obtained from American Type Culture Collection (ATCC). The cells were cultured in DMEM containing 10% FBS, penicillin (100 U/mL)-streptomycin (100 μg/mL), at 37°C, with 5% CO2. The cell culture medium was renewed on time every 2 days. Once the confluence reached about 85%, the cells were trypsinized with 0.25% TE, and then evenly distributed to the other two culture plates. All cells utilized were in the exponential growth phase.
RNA interference on DNA pol-β of 16HBE cells
DNA pol-β knocked-down cells (pol-β−) were obtained by using RNA interference (RNAi). Briefly, according to the cDNA sequence of pol-β gene published in Genbank, dsRNA used in RNA interference was designed with dsRNA oligonucleotide designer. DNA recombination technology was used to construct the pEGFP-C1-pU6-dsRNA recombinant. Then recombinant of pEGFP-C1-pU6-dsRNA was transfected to 16HBE cells by using Lipofectamine 2000, and empty vector of pEGFP-C1 was transfected to 16HBE cells acting as control. Finally, western blot analysis was used to assess the efficiency of pol-β interference. The detailed steps refer to the data we reported previously. 34
Western blot analysis on pol-β
Briefly, 5 × 106 cells of each group were treated with 1 mL cold RIPA lysis containing 1 mM PMSF for 10 min on ice. Gathered the lysate and transferred them to a micro-centrifuge tube, respectively. Each sample was centrifuged at 14,000 g for 15 min. BCA protein assay kit was used to measure the concentration of total protein. 60 µg protein samples were subjected to 10% SDS-PAGE gels and transferred to PVDF membranes (0.45 μm, Millipore). The membranes were incubated with TBST containing 5% non-fat milk for 4 h at room temperature (RT) and subsequently probed with primary antibody including rabbit anti-DNA polymerase β (1:1000 dilution) and anti-β-actin (1:5000 dilution) overnight at 4°C. The membranes were further incubated with secondary antibody for 2 h at RT. The protein levels were normalized with the level of β-actin. Enhanced chemiluminescence was performed for immunodetection analysis and the gray values were quantified with software Image J (NIH, MD, USA).
Cell treatment with MTBE
Our previous investigation in gas stations in South China suggested that the average concentration of MTBE in the occupational air could be 388.38 μg/m3 (4.388 μmol/L). 13 In consumers’ pulmonary alveolar, the concentration of MTBE could be 108.7 μmol/L. 14 Accordingly, we selected the range of 5–100 μmol/L (5, 50, 100 μmol/L) MTBE as the experimental concentrations, which were close to the real environmental burden. Briefly, each group of cells were seeded into the culture plates at a density of 1 × 106 cells/mL and directly exposed to MTBE at different concentrations for 12, 24, 36 h, respectively. Then the cells were harvested for further analysis.
Cell viability analysis
Cell viability of each group of cells was assessed using CCK-8 assay following the manufacturers’ instruction. Briefly, different groups of cells were incubated in 96-well plates at a density of 3 × 104 cells per well. After 24 h, the cells were exposed to MTBE at the environmental relevant concentrations (5, 50, 100 μmol/L) for 12, 24, 36 h, respectively. After the culture medium was renewed, 10 µL CCK-8 solution was added into each culture well, which was then incubated at 37°C for another 3 h. The OD values of all samples were detected with an automated microplate reader (ThermoScientific, USA) at 450 nm wavelength. The experiment was repeated for three times.
Cell growth analysis
The cell growth curves were estimated by using CCK-8 assay. Briefly, 2 × 103 cells per well of each group were seeded in 96-well culture plates and incubated with 100 µL of cell culture medium. Cells were allowed to grow in medium for 7 days and cell proliferation was detected at a designated time every day. For analysis, each well was added 10 µL of CCK-8 working solution and incubated for 3 h under standard cell culture conditions, then the OD values of each well were measured by an automated microplate reader (ThermoScientific, USA) at 450 nm wavelength.
Alkaline single-cell gel electrophoresis analysis
DNA damage of each group of cells was assessed by using alkaline single-cell gel electrophoresis (SCGE). Briefly, each group of cells were trypsinized with 1 mL 0.25% TE, centrifuged at 300 g for 5 min, and then suspended in PBS at a density of 1 × 106 cells/mL. 35 µL of cell suspension was mixed with 100 µL of 0.6% low melting agarose (LMA) at 37°C, and applied to a microscope slide on which 0.8% normal molten agarose (NMA) was pre-coated, the prepared slides were cold at 4°C for 20 min. Cells were lysed in cell lysis solution at 4°C for 4 h. After that, the microscope slides were immersed in cold alkali electrophoresis solution for 20 min to unwind DNA and then put in a fixed voltage (20 V/300 mA) electrophoresis tank to perform electrophoresis for 20 min. After electrophoresis, the microscope slides were put in the neutralization solution for 10 min to perform neutralization, stained with 50 mL ethidium bromide (EB) for 10 min in darkroom, and detected immediately. Comet images were analyzed under a fluorescence microscope (NIS-Elements BR 3.1) at ×100 magnification with Comet Assay Software Project (CASP). Comet tail lengths were automated calculated. Fifty nuclei comet images of each specimen were scored.
Biochemical measurement
Based on the manufacturer’s protocol, the enzyme activity of GSH-Px, SOD, CAT and the levels of MDA in 16HBE cells treated with various concentrations of MTBE for different time were analyzed by using the detection kits. Bradford protein assay was used to determine the protein levels. The enzyme activity of GSH-Px, SOD and CAT were separately calculated according to standard curves and expressed as U/mg protein. The MDA levels was expressed as nmol/mg protein. All samples were conducted independently in triplicate.35,36
Data analysis
Statistical analysis was performed with SPSS20.0 (IBM SPSS, USA). One-way analysis of variance (ANOVA) and the Students’ t-test were used to analyze the inter-group difference of quantitative data. All of the quantitative data was presented as mean ± standard deviation. The cutoff level of statistical significance was set at α = 0.05.
Results
Morphological images and cell growth curves of different groups of cells
Morphological images of different groups of cells, including 16HBE, pol-β− and 16HBE cells transfected with the empty vector of pEGFP-C1 (16HBE-NC), were observed under an inverted optical microscope. The result showed that at the early stage of cell transfection (24 h), pol-β− and 16HBE-NC cells were shrunk and floated to some extent, and there was no other significant difference in the morphology among different groups of cells (Figure 1(a)). CCK-8 assay showed that there was no significant difference in cell growth among different groups of cells [Figure 1(b)].

(a) Morphological images of different groups of cells (a: 1: 16HBE; 2. pol-β−; 3.16HBE-NC); (b) cell growth curves of different groups of cells.
The efficiency of RNAi on DNA pol-β expression
In order to assess the efficiency of RNAi on DNA pol-β− cells, western blot analysis was conducted to detect the expression level of DNA pol-β protein in 3 groups of cells (16HBE; pol-β−; 16HBE-NC.). Western blot gray scale results showed that the expression level of DNA pol-β protein in pol-β− was approximately 16.22% of the control group of 16HBE. While there was no significant difference in gray values between the groups of 16HBE and 16HBE-NC (Figure 2).
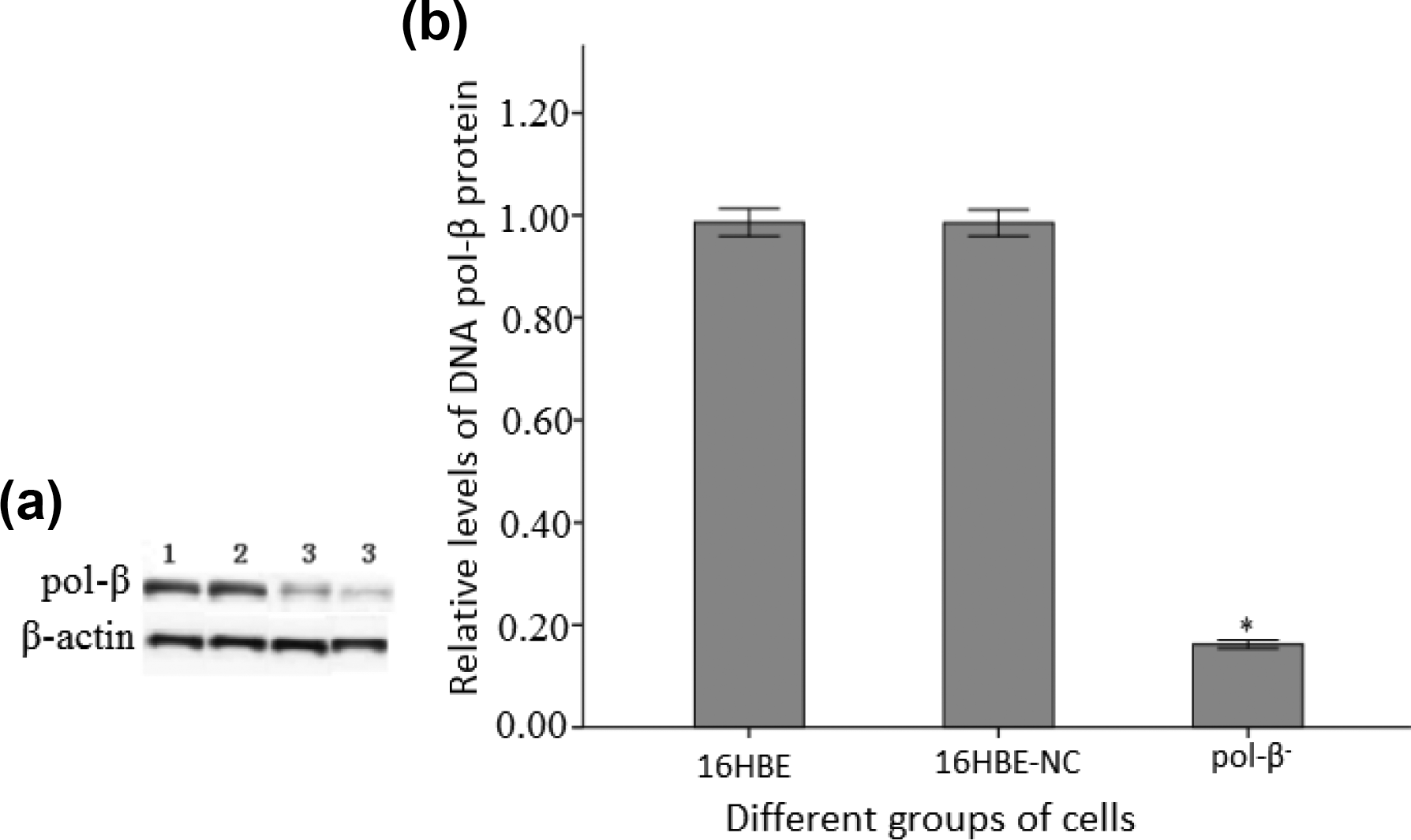
Western blot analysis on pol-β protein: (a) Western blot image (1:16HBE; 2: 16HBE -NC; 3: pol-β−). (b) Relative level of different groups of cells.
Decreased viability of pol-β− cells treated with MTBE
CCK-8 assay was used to assess the viability of different groups of cells. The result showed that when treated with MTBE at the environmental relevant concentration of 5, 50, 100 μmol/L for 12, 24, 36 h, respectively, the pol-β− exhibited significantly decreased viability in approximate dose- and time-dependent manner, as compared with the control group of 16HBE (P < 0.05). While there was no significant change in cell viability between the 16HBE and 16HBE-NC (P > 0.05) (Figure 3(a) to (c)).

Cell viability of different groups of cells (* shows the significant difference in the viability between the group of pol-β− and 16HBE; P < 0.05; n = 3).
pol-β− exhibited increased DNA damage effect induced by MTBE
SCGE assay was conducted to detect the DNA damage effects induced by MTBE in different groups of cells. The result showed that when treated with MTBE at the environmental relevant concentrations of 5, 50, 100 μmol/L for 12, 24, 36 h, respectively, the cells in each group showed obvious DNA damage effect in approximate dose- and time-dependent manner. The average comet tail lengths of pol-β− group was significantly longer than that of the 16HBE group (P < 0.05). While there was no significant difference in DNA damage effects between the groups of 16HBE and 16HBE-NC under the same experimental conditions (P > 0.05) (Figure 4(a) to (c)).
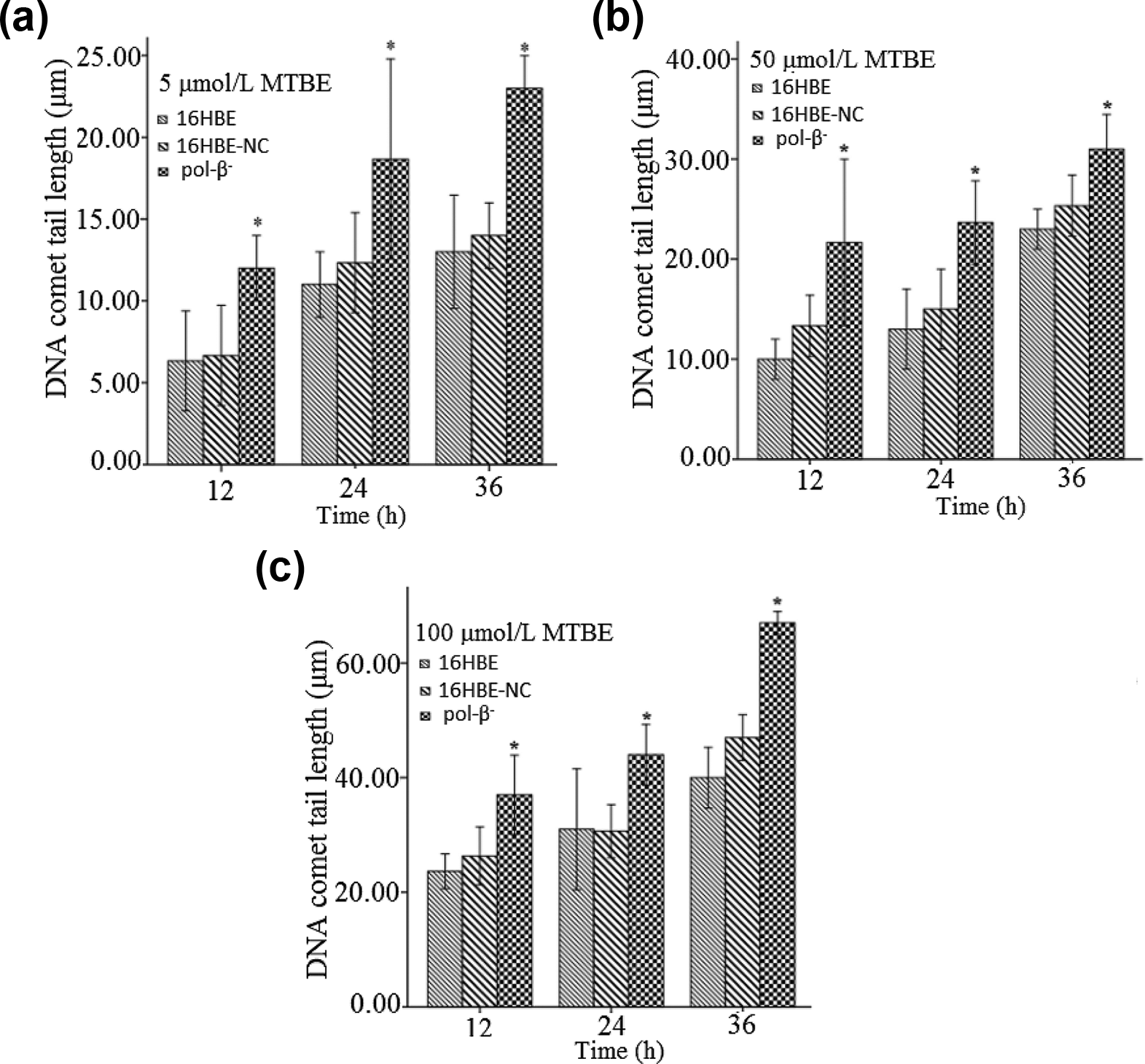
DNA damage effects induced by MTBE in different groups of cells (* shows the significant difference in comet tail lengths between the groups of pol-β− and 16HBE; P < 0.05; n = 3).
DNA pol-β gene knocked-down triggered oxidative stress in 16HBE cells treated with MTBE
When different groups of cells were treated with MTBE at different concentrations and for different time, their oxidative stress exhibited various degrees of change. The results indicated that when the concentration of MTBE exceeded 50 μmol/L and the exposure time exceeded 24 h, the enzyme activity of antioxidant molecules including GSH-pX, SOD and CAT in pol-β− cells was significantly lower than that of 16HBE cells (P < 0.05). On the contrary, the level of oxidant molecule MDA in pol-β− cells was significantly higher than that of 16HBE cells (P < 0.05). That is to say, DNA pol-β gene knocked-down triggered the oxidative stress in 16HBE cells treated with MTBE (Table 1).
Oxidative stress of different groups of cells treated with MTBE.

a: 16HBE; b: 16HBE-NC; c: pol-β−; * significant difference of P < 0.05 when compared to the group of 16HBE under the same condition).
Discussion
It’s well known that under the impact of a variety of internal and external environmental toxicants, genomic DNA molecules in organisms will inevitably suffer from various kinds of damage. Simultaneously, organisms can maintain the stability of their own genome through a variety of pathways and exhibit a certain degree of tolerance to toxicity of toxicants. For example, cells have the ability to protect genomic DNA against damage via DNA pol-β-dependent short-patch (1 nt) base excision repair (SP-BER) and long-patch (2–10 nt) base excision repair (LP-BER), which are predominant pathways involved in the process of DNA repair with high efficiency.
DNA pol-β, a single polypeptide chain composed of 335 amino acid residues, has two special domains and is the most important component in BER process. For instance, during the process of SP-BER, DNA pol-β plays a dual role, one is DNA polymerase activity, the other is 5′-deoxyribose phosphate (dRP) lyase activity. Its amino-terminal domain connected with the main protein globule, acts as a lyase, can remove dRP produced in BER process. The larger other domain, depending on the template of DNA chain, acting as a nucleotidyl transferase, can bind to dsDNA, the correct nucleoside triphosphate and catalytic metals, so as to achieve its normal functions. Studies have found that when the quantity or quality of DNA pol-β changes, DNA damage repair function will be abnormal, thus resulting in acute or chronic death or malignant transformation of cells under the attack of environmental chemicals.31,32 Whether this kind of phenomenon also occurs in the cytotoxicity and DNA damage effects induced by MTBE exposure in human cells is unknown.
In order to investigate the possible role of DNA pol-β on the toxic effects induced by MTBE exposure in human cells. We used RNA interference technology to obtain the DNA pol-β gene knocked-down cells as we reported previously. 34 The expression level of DNA pol-β gene was reduced to about 16.22% of normal 16HBE cells, which meant that the RNA interference on DNA pol-β gene is success. Simultaneously, there was no significant change in the morphological and cell growth characteristics of DNA pol-β gene knocked-down cells, which meant that the down regulation of DNA pol-β gene expression itself had no obvious effect on the biological characteristics of cells.
When treated with MTBE at the environmental relevant concentrations of 5, 50, 100 μmol/L for 12, 24, 36 h, respectively, 16HBE cells exhibited no significant morphological changes and no typical cytotoxic effects. The morphology and growth characteristics of the cells could be maintained at normal status under the environmental relevant concentrations. These results indicated that 16HBE cells have the ability to tolerate relatively low levels of the environmental relevant concentrations of MTBE. While SCGE analysis revealed that obvious DNA damage effect was induced by MTBE exposure under the same conditions. This is consistent with the previously reported materials22,23 Our results suggested that the risk of genotoxicity existed in exposure to low concentrations of MTBE, which should be paid enough attention.
It worth noting that, when DNA pol-β gene knocked-down cells were treated with MTBE at the environmental relevant concentrations of 5, 50, 100 μmol/L for 12, 24, 36 h, respectively, they exhibited significantly decreased cell viability and increased DNA damage effects in approximate dose- and time-dependent manners, as compared with the control. While there was no significant change in cell viability and DNA damage effects in the two controls. Our data suggested that DNA pol-β gene was involved in the cytotoxicity and DNA damage effects induced by MTBE exposure. Furthermore, the data showed that when treated with MTBE at the concentrations exceeding 50 μmol/L and for the time exceeding 24 h, DNA pol-β gene knocked-down cells exhibited increased oxidative stress also in approximate dose- and time-dependent manners, as compared with the controls. It means that oxidative stress pathway may be in an important mechanism underlying the effects of DNA pol-β on the toxicity of MTBE. These findings provided a novel insight into the molecular mechanism of the toxicity of MTBE. Herein, we conclude that the congenital genetic defect of DNA pol-β gene may be a contraindication of occupationally exposure to MTBE.
Conclusion
Taken together, this study indicates that MTBE can cause obvious cytotoxicity, DNA damage and oxidative damage effects on 16HBE cells. DNA polymerase β may be involved in protecting 16HBE cells from the toxic effects of MTBE. These findings provide a novel insight into the toxic mechanism of MTBE on human cells.
Footnotes
Authors’ note
Zhonghan He, Hongyi Xian, Meilin Tang, Ying Chen and Zhenwei Lian authors contributed equally to this paper.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Natural Science Foundation of China (21677066); Science and Technology Project of Guangzhou City (201803030027); Science and Technology Project of Shenzhen City (JCYJ20170306162947321).
