Abstract
The present research task is aimed to evaluate the role of exogenous α-lipoic acid (ALA) (100 mg/kg body weight) as hepatoprotective and potent antioxidant in amelioration of copper nanoparticle (CNP)-induced hepatotoxicity. Forty male rats were randomly assigned into four equal groups: group I (control), group II received CNPs, group III received CNPs + ALA, and finally group IV received ALA for 2 months. At the end of the experimental period, the rats were decapitated, and blood and liver tissue samples were collected for measurement of liver function tests, antioxidant status, lipid peroxidation (LPO), copper content, expression of some apoptotic genes, and histopathological analysis. CNPs induced marked hepatic damages as evident by severe alteration in hepatic biomarkers. This was accompanied by a significant elevation in hepatic LPO and induced nitric oxide, copper content, and expression level of apoptotic genes (C-myc and C-jun). In contrast, marked depletion for antioxidant parameters was detected. These findings were confirmed with severe pathological alterations. Coadministration of ALA as a powerful antioxidant attenuates the hepatotoxic effects of CNPs through improvement of liver parameters, oxidative status, genetic changes, and preservation of liver integrity through histopathological analysis. These results suggest that consumed ALA could be used as an applicable hepatoprotective agent against oxidative damage mediated by nanoparticles intoxication.
Introduction
Nanotechnology is a new promising field with great potential applications in domestic, industrial, and biomedical products. 1 In recent years, the use of nanoparticles (NPs) in human activities has been increased. Therefore, studies of the biological effects of different NPs, especially their effects on human and animal organs, need special attention. Published literatures showed that nanosize materials have relatively greater toxicity rather than large size materials, because the smaller size NPs are highly reactive and cause oxidative stress in humans and animals. 2 NPs can easily pass through the cell membrane and even through blood–brain barrier and blood–testes barrier. 3 CNPs are widely synthesized and used as a metal catalyst, heat transfer fluids in machine tools, semiconductors, and even in antibacterial medications. 4 It is mentioned that CNPs are dispersed in animal causing particles structural changes. 5 In the study by Lei et al. 6 and Ibrahim et al., 7 hepatotoxic effect of CNPs via different routes and doses has been reported. 6,7 Most work to date has suggested that reactive oxygen species (ROS) overproduction and consequent oxidative stress are frequently observed with NP toxicity. 8 Liver is the main detoxifying organ for many toxic substances and drugs that contribute to oxidative stress. 9 The increased ROS generation makes mitochondrial membranes highly susceptible to oxidative damage. ROS plays a critical role in the development of endothelial injury and hepatic fibrosis. 9 α-Lipoic acid (ALA) has been identified as a natural antioxidant and reduced the risk of diseases associated with oxidative stress. Its antioxidant properties give it the ability to scavenge ROS. 10 In addition, ALA had the ability to regenerate endogenous antioxidants, including vitamin C, vitamin E, and intracellular glutathione (GSH). 11 ALA was considered as a potential therapeutic agent for heavy metal toxicity. 12 Also, ALA was used as a therapeutic or protective agent in a wide variety of conditions related to liver disease. 9,13 Health problems caused by toxic environmental compounds are becoming a global challenge. Increasing urbanization and ensuing industrialization may contribute to continuous human exposure to many toxic environmental pollutants. Based on the antioxidant activity of ALA, we designed the present study to investigate the potential hepatoprotective role of ALA against the metabolic disorder, oxidative stress, and genotoxicity induced by CNPs.
Materials and methods
Animals
Forty male albino rats, weighing 150–170 g, were obtained from the breeding unit of the Department of Forensic Medicine and Toxicology, Faculty of Veterinary Medicine, Cairo University. The animals were housed in plastic cages and fed a standard laboratory diet and water ad libitum. The animals were exposed to 12-h light/12-h dark cycle and temperature (25°C ± 2°C). Rats were acclimatized for 1 week prior to the start of the experiment. The experiment was carried out in accordance with the guide of the care and use of laboratory animals published by the material institute of health and approved by the animal experiment local ethics committee at the Cairo University.
Chemicals
CNPs (20–30 nm) at 99.5% purity, spherical in shape, and mineral in nature were purchased from Sigma Aldrich (St. Louis, Missouri, USA). A stock suspension of CNPs was prepared by dispersing CNP powder in deionized water followed by vigorous vortexing and sonication. Prior to each use, the stock solutions were sonicated for approximately 20 s to ensure proper particle suspension in solution. According to Chen et al., 14 LD50 for CNP-induced gravely toxicological effects and heavy injuries for liver tissue is 413 mg/kg body weight (bw). The selected dose of CNPs (40 mg/kg bw, 1/10 LD50) used in the present study was used based on a previous study in which 1/10 LD50 induced biochemical alteration in rat without morbidity. 14 ALA was obtained from EVA Pharma (Egypt). The selected dose for ALA (100 mg/kg bw) was reported to be hepatoprotective for rats. 15
Experimental design
After acclimatization, rats were randomly divided into four different equal groups. All the treatment was performed via oral gavage for 5 days per week for 2 months. Group I acts as control received distilled water only. Group II received CNPs (40 mg/kg bw) only. Group III orally received CNPs (40 mg/kg bw) + ALA (100 mg/kg bw). Group IV was given ALA (100 mg/kg bw) only.
Rats were treated for 5 days per week for 2 months. Throughout the experimental period, no signs of toxicity in the animals treated with CNPs were detected. At the end of the experiment, the animals were fasted overnight, anesthetized, and killed by cervical dislocation. Blood and liver samples were collected for further estimations.
Biochemical analysis
The blood samples were collected in plain tubes that were centrifuged at 10,000 r/min for 10 min and the serum was stored at −20°C for subsequent serum alanine aminotransferase (ALT), aspartate transaminase (AST), total protein (TP), and total bilirubin (TB) concentration measurement according to the instructions provided by the manufacturer of the kits. The liver tissue was homogenized in ice-cold 0.1 M phosphate buffer saline (pH 7.4) using a Teflon tissue homogenizer. The crude tissue homogenate was centrifuged at 14,000 r/min for 15 min at 4°C and used for the measurement of malondialdehyde (MDA), 16 superoxide dismutase (SOD) activity, 17 catalase (CAT) activity, 18 reduced GSH concentration, 19 and TP concentration according to the method described by Bradford. 20
Estimation of copper content in the liver tissue
Liver tissue was digested with nitric acid solution using a microwave. The concentration of copper in digested fluid was analyzed using an atomic absorption spectrophotometer according to the method described by Zheng et al. 21
Quantitative reverse transcription polymerase chain reaction analysis
Approximately 100 mg of liver tissue was used for total RNA extraction using Qiagen Rneasy Mini Kit following the manufacturer’s protocol. The RNA yields and purity were determined using a spectrophotometer (Thermo Scientific, Waltham, Massachusetts, USA). Ten micrograms of transfer RNA was treated with DNase I for 20 min at 37°C. The complementary DNA (cDNA) synthesis was carried out using reverse transcriptase (Invitrogen, Carlsbad, California, USA) and oligo-dT following the manufacturer’s protocol. After initial heat denaturation of 1 µg of total RNA (65°C for 5 min), the reaction mixtures (20 µl) were incubated for 60 min at 42°C. Real-time polymerase chain reaction (PCR) was done in (color for research Laboratory, Qiagen, Egypt). cDNA was added to a SYBR Green qPCR Master Mix (Qiagen) containing 30 pg/ml of each primer with the following sequence: C-jun forward 5′-AGA CGC GTG CCT ACG GCT ACA GTA A-3′, reverse 5′-CGA CGT GAG AAG GTC CGA GTT CTT G-3′; C-myc forward 5′-TAC CCT CTC AAC GAC AGC AG-3′, reverse 5′-TCT TGA CAT TCT CCT CGG TG-3′; and inducible nitric oxide synthase (iNOS) forward 5′-GGAGCGAGTTGTGGATTG-3′, reverse 5′- CCAGGAAGTAGGTGAGGG-3′. The cDNA was amplified by 40 cycles of denaturation at 95°C for 15 s, annealing at 60°C for 20 s, and extension at 72°C for 45 s. During the first cycle, the 95°C step was extended to 1 min. The size of all amplicons was confirmed by 2% agarose gel electrophoresis stained with SYBR Safe DNA gel stain (Invitrogen). The GAPDH gene was amplified in the same reaction to serve as the reference gene. Each measurement was repeated three times, and the values were used to calculate the gene/GAPDH ratio, with a value of 1.0 used as the control (calibrator). The normalized expression ratio was calculated using the method described by Livak and Schmittgen. 22
Histopathological examinations
Liver sections of different groups were fixed in 10% neutral buffered formalin for 72 h, washed, dehydrated, and embedded in paraffin sections; 5-µm thick sections were stained with hematoxylin and eosin for histopathological examination.
Immunohistochemical analysis
Expression of iNOS in the liver tissue of normal and treated rats was demonstrated in immunohistochemically stained sections using rabbit polyclonal antibody diluted at 1:100 (Lab Vision Corporation, Fremont, California, USA). The immune-reactive cells were visualized using 3,3-diaminobenzidine tetrachloride (Sigma Chemical Co.). The positive immune-reactive cells exhibited brown cytoplasm. The positive iNOS immune-reactive cells were counted in five random high microscopic field (40×).
Terminal deoxy nucleotidyl transferase-mediated dUTP nick-end labeling assay
Terminal deoxy nucleotidyl transferase-mediated dUTP nick-end labeling (TUNEL) assay was performed according to the method described by Hong et al. The TUNEL positive apoptotic hepatocytes appeared shrunken with condensation and or fragmentation of chromatin. The apoptotic hepatocytes were counted in five high microscopic field (40×).
Statistical analysis
The different analytical determinations in the biological samples were carried out in duplicate and results are expressed as mean ± SEM. Data for multiple variable comparisons were analyzed using SPSS version 16 package for Windows through one-way analysis of variance test followed by Tukey’s HSD test for multiple comparisons. The difference was considered significant when p < 0.05.
Results
Blood biomarker parameters
Copper nanoparticles (CNPs) supplementation at doses of 40 mg/kg bw produced significant elevation in liver tissue biomarkers (AST, ALT, and TB) and significant reduction in TP concentration compared to the control group, p > 0.05 (Table 1). The oral coadministration of ALA showed significant reduction in ALT by 14.3%, AST by 38.09%, and TB by 56.7% in comparison to group II. In contrast, significant increase in TP concentration was detected in group III by 30.98%. Insignificant change was detected between control and group IV.
The influence of ALA on serum biochemical parameters of CNP-intoxicated rats.a
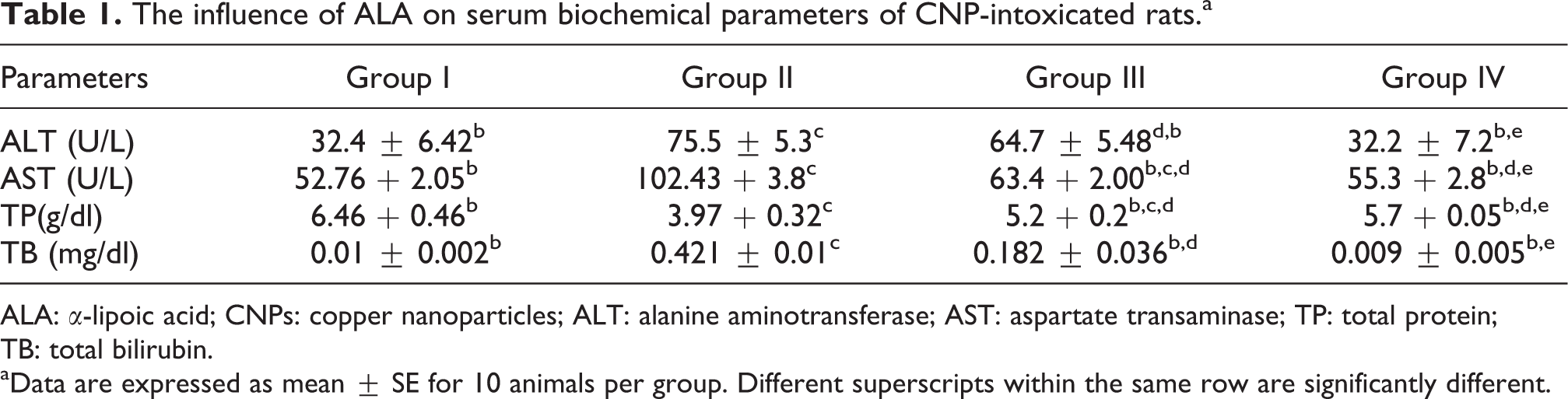
ALA: α-lipoic acid; CNPs: copper nanoparticles; ALT: alanine aminotransferase; AST: aspartate transaminase; TP: total protein; TB: total bilirubin.
aData are expressed as mean ± SE for 10 animals per group. Different superscripts within the same row are significantly different.
Oxidative stress parameters
Considering the key roles of ROS in the development of CNPs hepatotoxicity, it was necessary to investigate the changes of the hepatic antioxidant system, which are displayed in Table 2. The measurement of MDA is commonly used to monitor lipid peroxidation (LPO) and oxidative stress. Administration of CNPs to experimental rats showed a significant (p < 0.05) elevation in MDA level (Table 2) compared to group I. In the same consequence, CNPs caused a highly significant reduction (p < 0.05) in GSH concentration, CAT, and SOD enzymes activity. ALA administration to group III caused significant reduction in the elevated MDA by 48.9% and maintained its normal values (Table 2). Also, ALA caused significant increase in GSH concentration by 52.65% and significant elevation for CAT by 49.2% and SOD by 23.3% enzyme activities in comparison to group II. Both groups I and IV showed no significant differences in all measuring oxidative stress parameters (Table 2).
Influence of ALA on the oxidative stress parameters in liver of CNP-intoxicated rats.a
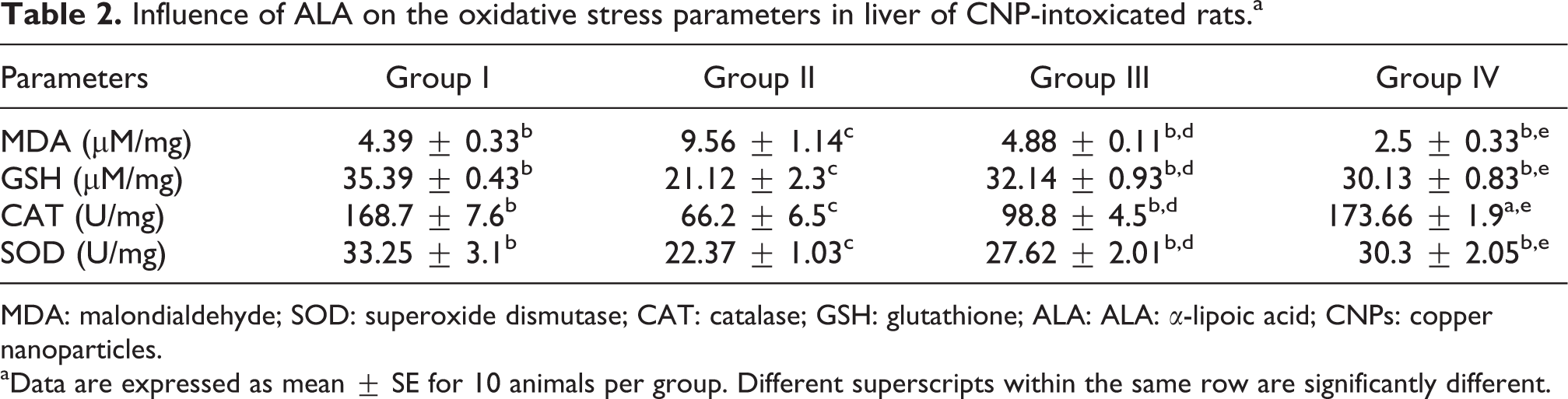
MDA: malondialdehyde; SOD: superoxide dismutase; CAT: catalase; GSH: glutathione; ALA: ALA: α-lipoic acid; CNPs: copper nanoparticles.
aData are expressed as mean ± SE for 10 animals per group. Different superscripts within the same row are significantly different.
iNOS expression
Nitric oxide (NO) is the oxidative metabolite of L-arginine. There are two constitutive NOS: endothelial NOS (eNOS) and iNOS responsible for NO synthesis. Under physiological conditions, only eNOS is active in the liver, whereas iNOS is activated in pathological conditions. In the present study, we detected significant overexpression of iNOS in group II treated by CNPs to 5.90 ± 0.58 fold (Figure 1(a)). This overexpression enhanced the NO overproduction and leads to nitrosative stress, which plays an important role in different liver pathologies. Oral administration of ALA attenuates the iNOS overexpression and downregulates by 84.06%. Insignificant change in iNOS expression level was detected between groups I and IV.
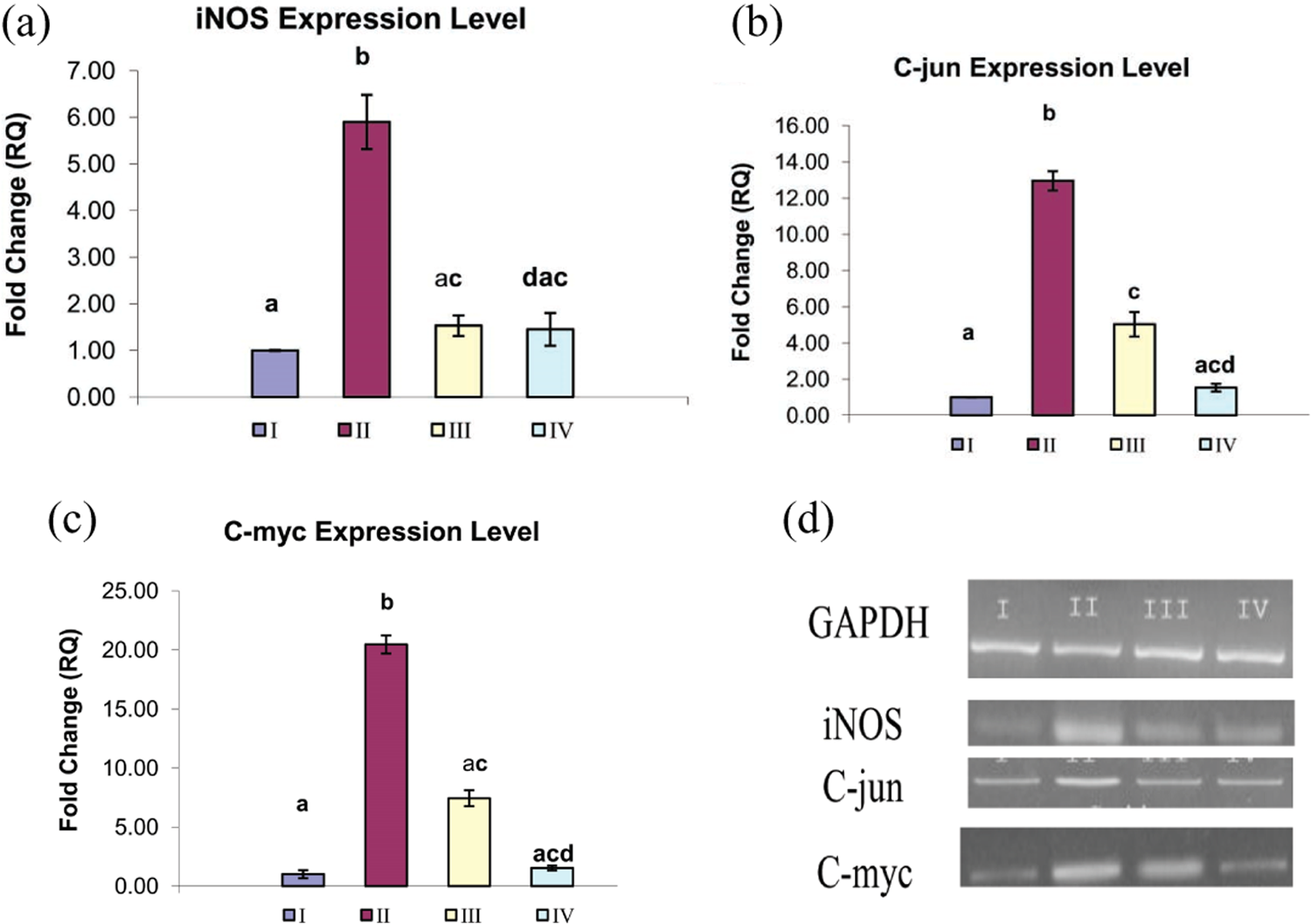
Influence of the ALA administration on iNOS, C-jun, and C-myc expression level in hepatic tissue exposed to CNPs. Fold change of mRNA expression of iNOS (a), C-jun (b), and C-myc (c) in rat liver of different experimental groups (I = group I, II = group II, III = group III, and IV = group IV) using qPCR. Data are represented as mean ± SEM. Different superscripts are significantly different. (d) The results obtained by real-time PCR analysis were confirmed by agarose gel electrophoresis analysis of the PCR product compared to GAPDH. ALA: α-lipoic acid; iNOS: inducible nitric oxide synthase; CNPs: copper nanoparticles; qPCR: quantitative polymerase chain reaction.
C-jun and C-myc expression
Apoptosis/programmed cell death is a specialized form of cell death by which the cell undergoes intentional suicide mediated by specific receptors and signal transduction. In the present investigation, qPCR was used to analyze the messenger RNA (mRNA) expression level of apoptotic genes C-jun and C-myc in hepatic tissue exposed to CNPs. The mRNA expression level of apoptotic gene C-jun was significantly upregulated to 12.95 ± 0.53 fold in group II treated by CNPs as compared to group I (Figure 1(b)). Administration of ALA to group III significantly reduced the overexpression by 61.15%. The C-myc gene is considered as one of the existent apoptotic genes in hepatic tissue. The mRNA expression of C-myc was significantly upregulated to 20.46 ± 0.76 in group II (Figure 1(c)) compared to controls. Oral administration of ALA for group III attenuated this overexpression and downregulated by 63.63%.
Copper content in liver tissue
Human health risk from heavy metal accumulation has been a subject of growing concern in recent years. In our present study, after the period of repeated oral administration of CNPs to group II, liver tissue displays marked retention of copper judging by an over increase in its concentration (from 2.22 to 4.98 ppm/g) in comparison with the control group (Figure 2). By contrast, ALA supplementation reduced copper accumulation by 40.36% compared to group II. Insignificant change was detected between control and group IV.
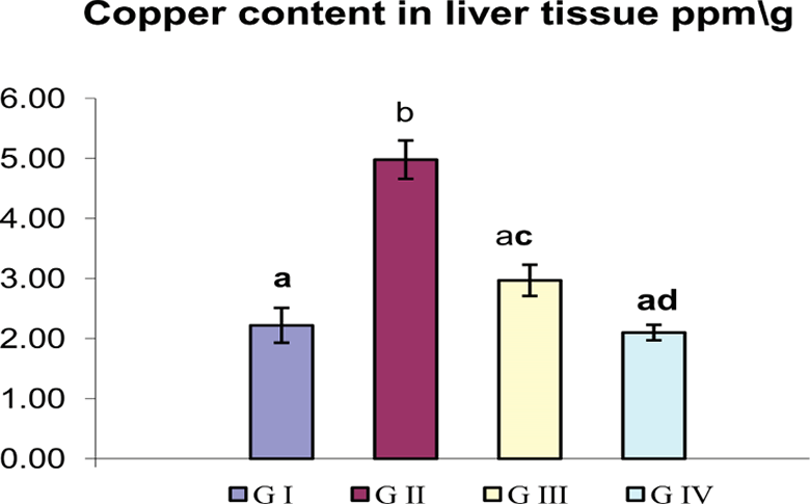
Influence of oral administration of ALA on copper accumulation in the liver of CNP-intoxicated rats. Data are represented as mean ± SEM. Different superscripts are significantly different. ALA: α-lipoic acid; CNPs: copper nanoparticles.
Histopathological analysis
Liver pathology serves as an important tool for identifying and characterizing liver injury. Liver of control rats showed normal hepatic parenchyma with preserved lobular architecture (Figure 3(a)), whereas those of CNP-treated group showed severe histopathological alterations characterized by dilatation of central vein with disruption of lobular architecture and disorganization of hepatic cords as well as severe necrobiotic changes ranged from vacuolar degeneration of hepatocytes to massive hepatic cell necrosis associated with increased number of apoptotic cells (Figure 3(b)). Multifocal mononuclear cell aggregation associated with sinusoidal dilatation (Figure 3(c)) was also observed. Portal area revealed mild inflammatory cell infiltration, oval cell proliferation, and massive necrosis of the periportal hepatocytes (Figure 3(d)). The liver of the lipoic acid + CNPs group showed regression of the histopathological lesions compared to CPs-treated one manifested by granular degeneration of hepatocytes with decreased number of necrotic and apoptotic hepatocytes, which was restricted to single cells (Figure 3(e)). No obvious histopathological changes were demonstrated in the liver of group treated with lipoic acid alone and the liver appeared normal (Figure 3(f)) greatly similar to the control group.
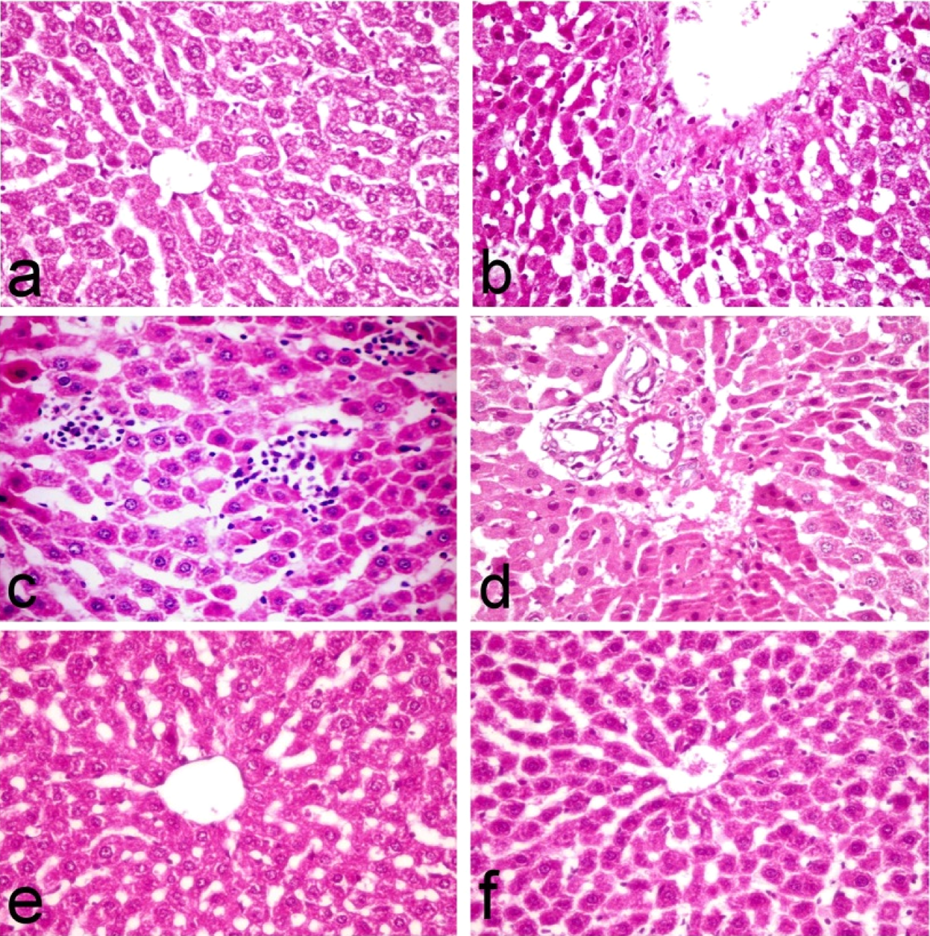
Liver tissue of control rats (a) showing normal hepatic parenchyma with preserved lobular architecture, nanocopper-treated rats showing (b) necrobiotic changes ranged from vacuolar degeneration of hepatocytes to massive hepatic cell necrosis associated with increased number of apoptotic cells, (c) multifocal mononuclear cell aggregation associated with sinusoidal dilatation, (d) massive necrosis of the periportal hepatocytes, nanocopper-treated and lipoic acid–treated rats showing (e) single cell necrosis and lipoic acid treated group showing (f) normal liver parenchyma (H&E, ×40). H&E: heamatoxylin and eosin.
Immunohistochemistry
Table 3 illustrates the results of immunohistochemical estimation of iNOS immune-reactive cells in the liver of normal control and treated rats. A significant increase of iNOS immune-stained cells was recorded in CNP-treated group (138.50 ± 22.75) (Figure 4(b)) compared to the control one (1.00 ± 0.40) (Figure 4(a)). On the other hand, a significant reduction of iNOS immune-reactive cells was recorded in lipoic acid and CNP-treated group (22.75 ± 4.32) (Figure 4(c)). Lipoic acid–treated group revealed nonsignificant difference (3.00 ± 1.6) (Figure 4(d)) compared to the normal control one.
TUNEL: terminal deoxy nucleotidyl transferase-mediated dUTP nick-end labeling; iNOS: inducible nitric oxide synthase.
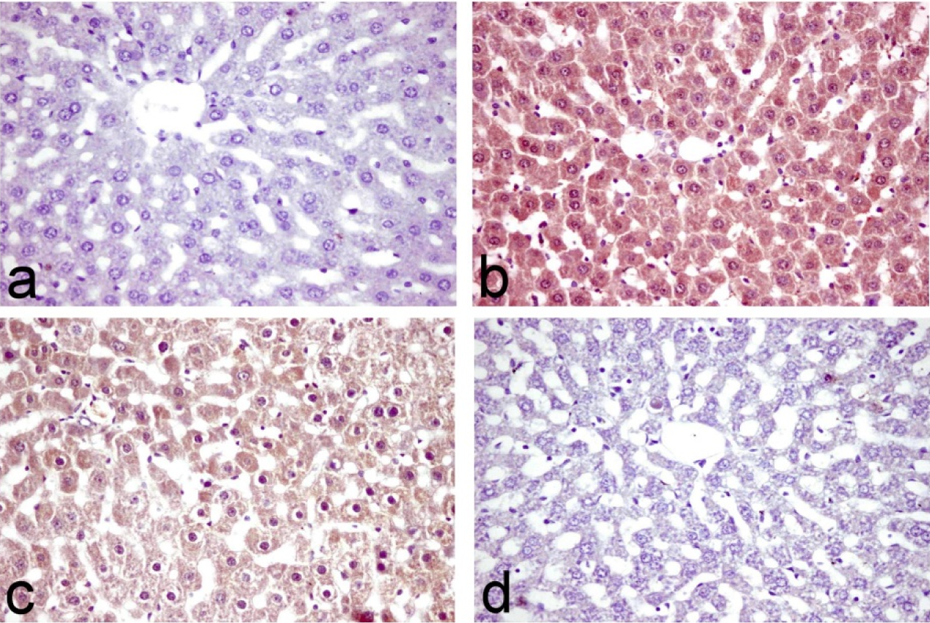
Liver tissue of (a) control rats showing no immune-reactive cells, (b) nano copper-treated rats showing a significant increase of iNOS immune-stained cells, (c) nanocopper-treated and lipoic acid–treated rats showing reduction of iNOS immune-reactive cells, and (d) lipoic acid–treated group showing single immune-reactive cell (immunohistochemical staining for iNOS, ×40). iNOS: inducible nitric oxide synthase.
TUNEL assay
Table 3 demonstrates the results of TUNEL assay for the estimation of apoptotic hepatocytes in the liver of normal control and treated rats. The liver of control rats showed individual positive TUNEL apoptotic cells (1.00 ± 0.54) (Figure 5(a)). A significant increase of apoptotic hepatocytes was recorded in nanocopper-treated group (14.00 ± 2.60) (Figure 5(b)). These positive apoptotic hepatocytes were greatly reduced in lipoic acid and CNP-treated group (5.40 ± 0.50) (Figure 5(c)). Sparse apoptotic hepatocytes were observed in lipoic acid-treated group (0.80 ± 0.58) (Figure 5(d)).
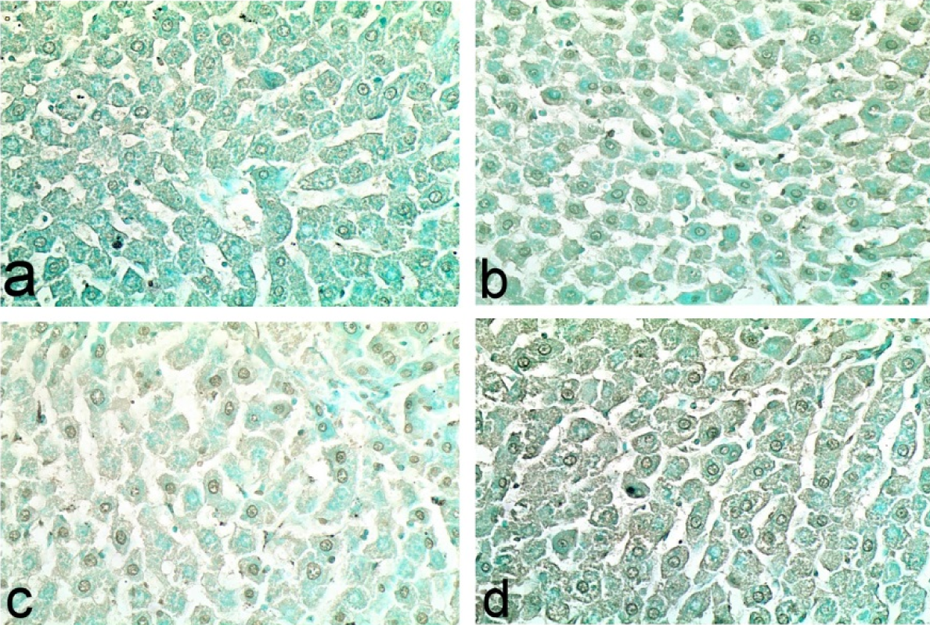
Liver tissue of (a) control rats showing, individual positive apoptotic cells (b) nanocopper-treated rats showing significant increase of apoptotic hepatocytes, (c) nanocopper-treated and lipoic acid–treated rats showing reduced number of positive apoptotic hepatocytes, and (d) lipoic acid–treated group showing sparse apoptotic hepatocytes (TUNEL assay, ×40). TUNEL: terminal deoxy nucleotidyl transferase-mediated dUTP nick-end labeling.
Discussion
Several studies had reported the toxicity of CNPs was associated with hepatic dysfunction. 5 –7 Liver tissue is a target organ and primary site for detoxification and therefore prone to various disorders as a consequence of exposure to the toxins of extrinsic and intrinsic sources. The magnitude of hepatic damage is usually assessed by measuring the level of released cytosolic transaminases in circulation. The present study revealed that rats received CNPs had marked elevation in serum ALT and AST enzymes activity and the concentration of TB compared to control (Table 1). In contrast, significant reduction in TP concentration was detected. Our results were closely correlated with those mentioned by Doudi and Setork, 5 Lei et al., 6 and Ibrahim et al. 7 Alterations in cytosolic transaminases were liver specific and used as a tool for measuring the cell viability and cell membrane permeability. 23 Whereas increases in both TB level is measures of overall liver function. An elevation in transaminase in conjunction with a rise in bilirubin level to more than double is considered as a potent marker for hepatotoxicity. 23 As a result of hepatic injury, alteration in the membrane permeability causes the enzymes release into the circulation. 24 Coadministration of ALA to the rats received CNPs (40 mg/kg bw) + ALA (100 mg/kg bw) orally significantly altered the liver biomarkers. Similar findings were reported about the protective influence of ALA against hepatotoxicity. 25,26 Exposure to several chemicals and environmental contaminants had been reported to increase the oxidative stress by disturbing the prooxidant/antioxidant balance. 27 A statistically significant elevation in MDA level as well as significant reduction in GSH concentration and SOD and CAT activities in group II of our present study indicate the oxidative damage produced by CNPs (Table 2). Similar results were previously reported by Lei et al., 6 Zhang et al., 25 and Morsy et al. 27 A variety of NPs including metal-induced ROS is one of the principal mechanisms of cytotoxicity. 8 ROS induction by NPs is considered the primary cause of nanotoxicity and had been attributed to the presence of pro-oxidant functional groups on their reactive surface and the NP–cell interactions. 8 In our study, the administration of ALA to group III significantly decreased the concentration of MDA. ALA also induced improvement in the level of GSH and activities of liver function of enzymes (Table 2). ALA appeared to elevate the GSH levels by improving the cellular availability of cysteine through conversion of cystine into cysteine. 28 ALA is considered as an inducer to antioxidant gene expression. 29 In the same consequence, ALA has the ability to scavenge free radicals and increase antioxidant enzyme activates. 28 Besides acting as a potent antioxidant, ALA either increases or maintains levels of other antioxidants such as vitamin E and vitamin C. 28 Several studies reported the protective effect of ALA against hepatic oxidative stress induced by toxic agents. 30,31 NO is a highly reactive oxidant produced in the liver in response to inflammatory mediators. NOS is an enzyme that catalyzes the formation of NO and iNOS is one of three isozymes of NOS. 32 The significant elevation of iNOS mRNA expression levels detected in our present study (Figure 1(a)) may be attributed to the inflammatory reaction occurred after CNPs exposure. This result was in agreement with previous reports that detected the overproduction of NO with NPs toxicity. 33 NPs deposition in tissues causes induction of inflammatory cells, the source for reactive nitrogen species (RNS) overproduction. 34,35 ALA was found to have an inhibitory effect on iNOS induced by different insults. 36,37 ALA can inhibit the synthesis and activity of iNOS and also scavenge RNS. 38 Others suggested that ALA improved the carbohydrate metabolism, and this is responsible for the inhibitory action of ALA on NO production in hepatocytes. 39 The anti-inflammatory effect of ALA has been demonstrated by several researchers. 40,41 Due to their special chemical characteristics, ROS/RNS can induce LPO, DNA damage, and oxidize all macromolecules in biological membranes and tissues, resulting in severe cellular injury. 42 Overgeneration of ROS is considered to be an upstream event for the initiation of NP-induced apoptosis. 43 Our findings suggested that CNPs significantly induced apoptosis as evidenced by overexpression of both apoptotic genes C-myc and C-jun (Figure 1(b) and (c)). Immediate early genes such as C-jun and C-myc are considered as important genes in the control of cellular proliferation and differentiation based on their early transcriptional activation in normal cells exposed to mutagenic substances. 44 Several studies have identified the oxidative stress as a common pathway for NP-induced cellular damage by targeting the mitochondrial apoptotic pathway. 6,7 Over ROS production in NPs toxicity can result in damage to mitochondrial membrane phospholipids inducing membrane depolarization. 8 Moreover, NPs are able to induce mitochondrial damage through the direct interaction with undissolved particles following endocytotic uptake, 45 pro-apoptotic proteins 46 confirmed by TUNEL assay. Also, NPs had been reported to influence intracellular calcium concentrations that induce expression of immediate early genes responsible for apoptosis. 8 All those could be the main causes for C-jun and C-myc overexpression detected in group II in our present study (Figure 1(b) and (c)). The ability of CNPs to induce apoptosis through the alteration of the expression of apoptotic genes was proven previously. 7,47,48 ALA downregulates the expression levels of both C-myc and C-jun (Figure 1(b) and (c)). Several researchers report the antiapoptotic effects of ALA. 49,50 ALA exerts inhibitory effect on cell proliferation via its effects on epidermal growth factor receptors. 51 Besides that, ALA induced restoration of the optimal level of Bax:Bcl-2 ratio (1:2), the key factor in pushing the cells toward survival. 52 Also, ALA reduced the amount of the hydroxyl radical and also scavenges the peroxide and superoxide radicals, the main factors induced cell death. 53 All those could be the main speculation that explain the antiapoptotic effect of ALA detected in our study (Figure 1). The sizes of NPs as well as their surface characteristics are the key parameters that can alter the biodistribution in tissues. The primary site of copper accumulation has consistently been demonstrated to be the liver tissue. 54 Exposure of male rats to CNPs induces a significant increase in copper concentration in liver compared to control (Figure 2). The greater accumulation of CNPs in the liver may be attributed to the fenestrated, discontinuous endothelia that allow the passage of NPs up to 100 nm from the blood into the liver parenchyma as well as the phagocytosis for undissolved particles. 54,55 Administration of ALA to group III produced significant reduction in liver copper concentration. Gomes and Negrato 56 mentioned that ALA was considered as chelator compounds because they are able to chelate divalent transient metals ions. This metal chelating property of ALA may be responsible for the observed reduction of copper connection detected in our study. The characteristic histopathological lesions in the liver of CNPs-treated group (Figure 3) could be attributed to the cyto- and genotoxic effect of CNPs correlated with higher copper accumulation in the liver tissue. 45 Evidence from this study and previous work revealed that ALA was considered as an ideal antioxidant because it was a naturally existing, low-molecular-weight compound with very powerful antioxidant properties in both aqueous and lipid domains, which makes it effective against a broader range of free radicals. The protective role of ALA against hepatotoxicty and oxidative stress is well documented in several reports. 57,58 In conclusion, the present results demonstrate that the oral administration of CNPs causes oxidative damage and apoptosis in hepatocytes. ALA as an antioxidant agent reduced these degenerative changes through preserving the activities antioxidant parameters, reduction of LPO, NO production, and inhibiting the development of apoptosis.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
