Abstract
Studies have revealed that impairment of the pregnant body weight reduces the fetal body weight and causes minor changes in skeletal development. The aim of the present study was to assess the effects of maternal feed restriction during pregnancy in offspring immune system development. Pregnant Wistar rats were distributed into 5 groups: 1 control in which dams received food ad libitum and 4 experimental groups in which dams were fed restricted amounts of rodent ration (16, 12, 9, or 6 g/rat/day) from the 6th to 17th gestation day. Teratogenicity was assessed using classical teratological evaluation and developmental immunotoxicology protocols. Maternal body weight gain, fetus weight, and placenta weight were reduced for feed-restricted females from the groups fed 12, 9, and 6 g/rat/day (p < 0.05). No pup mortality was observed immediately after cesarean sections among the groups, and no visceral or skeletal malformations were detected. An immunoteratological study revealed an increase in the relative weight of the thymus and an increase in the phorbol myristate-acetate solution-induced hydrogen peroxide release by inflammatory cells in 21-day-old pups. Alterations in the delayed-type hypersensitivity response and the humoral immune response against sheep red blood cells were observed in pups from feed-restricted mothers. Feed restriction in Wistar rats during organogenesis did not promote structural malformations but resulted in offspring with lower birth weights and promoted significant changes in the immune responses of the rat pups.
Introduction
In recent decades, the regulatory guidelines for reproductive toxicity testing of agricultural and industrial chemicals have been extensively revised with the addition of many new reproductive functions and offspring development end points. 1,2 One of the end points most often in question is whether decreased maternal food consumption causes decreased fetal body weight. 3 Although the demonstration of some degree of maternal toxicity is required in regulatory developmental and reproductive toxicology assessments, 4 marked maternal signs of toxicity may be a significant confounding factor in the study design and data interpretation. 5
Developmental toxicity results from a direct effect on the embryo/fetus, an indirect effect through toxicity of the agent to the mother, or a combination of direct and indirect effects. 6 Thus, maternal physiological conditions (e.g. maternal nutritional status) can adversely affect the developing embryo/fetus. 7
Khera 8 demonstrated that impairment of pregnant body weight resulting from maternal toxicity caused embryo lethality or fetal malformations. In contrast, other studies on the effects of maternal body weight loss on fetus development revealed that a reduction in fetal body weight and/or delays in skeletal ossification alone were generally not associated with malformations in rats 9 –11 or rabbits. 12 –14
The observation of malformations and/or anomalies alone does not reflect the xenobiotic risk of teratogenesis. Thus, developmental toxicology should consider the functionality of the body systems in addition to weight and morphology. 6 Indeed, the developing endocrine, 15 central nervous, 16 and immune systems 17 –20 are generally more sensitive to toxicant insults in utero compared to other age groups. Considering this theory, we can assume that feed restriction could also prejudice the development of the immune system. As a result, we hypothesized that impairment of their functions would be reflected only in the postnatal periods.
The nature of changes in the immune system may be subtle; consequently, most routine assessment end points do not detect these changes, especially those associated with developmental toxicity. 21 Therefore, developmental immunotoxicology protocols can be a potent tool to determine the effects of maternal dietary restrictions on the offspring as a complement to the classical teratological protocols. Thus, the objective of the present study was to determine whether restriction of maternal food intake during pregnancy caused disturbances in normal immune system development in rat offspring.
Materials and methods
The Bioethics Committee of the School of Veterinary Medicine and Animal Science from the University of São Paulo, Brazil (process no. 1403/2008) approved the animal care and experimental procedures.
Animals and housing
One hundred twenty pregnant 70-day-old Wistar rats that were inbred in the Research Center for Veterinary Toxicology, Department of Pathology, School of Veterinary Medicine and Zootechny were used. Gestational day (GD) 0 was defined as the day when spermatozoids were detected in the vaginal lavage. The rats were housed individually in polycarbonate cages with contact bedding of heat-treated hardwood chips in a 12-h light/12-h dark cycle room. The rats were allowed to acclimate for at least 7 days prior to the initiation of feed restrictions.
All animals were observed twice daily for alterations in behavior or demeanor.
Clinical observations were performed every other day to evaluate soft feces, alterations in behavior or demeanor and mortality as proposed by Fleeman et al. 11 The dams were assigned to 5 groups of 10 (experiment 1) or 11–14 (experiment 2) rats based on their body weights on GD 0 to ensure homogeneity within the groups of rats under food restriction.
Feed restriction regimen
The feed-restriction experiment was based on a study by Fleeman et al. 11 with subtle modifications. Briefly, 4 groups of dams were fed restricted amounts of NUVILAB NUVITAL-CR1® rodent ration (16, 12, 9, or 6 g/rat/day). The rations were provided each morning from GD 6–17 and were equivalent to approximately 85%, 60%, 45%, and 30% of the typical amount of food consumed by the pregnant rats of the control group, respectively; the control group females were allowed ad libitum access to feed during organogenesis (GD 6–17). Before GD 6 and after GD 17, females from all groups were allowed ad libitum access to feed. Water was provided ad libitum to all dams during the course of the study. The experiments were conducted as a classical teratogenic study (experiment 1) and two immunological evaluations (experiment 2) conducted with 21-day-old weaned offspring (trial A) and 70-day-old adults (trial B; Figure 1).

Overview of experimental design and study schedule. Solid lines: ad libitum feeding; dashed lines: feed restriction period; GD: gestation day; PND: postnatal day.
Maternal study
The females were observed for mortality and clinical signs daily during the study. Food consumption and body weight gain (BWG) were measured every 3 days from GD 6 to the cesarean section (GD 21). In experiment 2, the behavior of the mothers, BWG and food consumption were recorded from parturition to the end of lactation (postnatal day or PND 21).
Experiment 1: Classical teratological protocols
After the feed-restriction regimen, the dams were anesthetized on GD 21 with intraperitoneal (ip) injections of xylazine and ketamine (5 and 50 mg/kg, respectively) to prepare for the cesarean sections. The number of corpora lutea, implantations, and resorptions were recorded. The fetuses were removed from the uterus, weighed, and sexed. The viability was evaluated immediately after birth and the numbers of live and dead fetuses were recorded. The placental weight was also measured. After this procedure, alternate fetuses were randomly selected for either skeletal examination (2–3 fetuses/litter for a total of 25 fetuses) according to the technique of Staples and Schnell 22 or visceral examination by the Wilson method 23 using serial sectioning (2–3 fetuses/litter for a total of 25 fetuses). The fetal observations were recorded in accordance with the internationally developed terminology. 24
Experiment 2: Effect of maternal feed restriction on the immune systems of weaned pups (trial A) and adults (trial B)
The dams gave birth naturally, and the pups from each litter were weighed immediately after birth (PND 0). To avoid the interference of sex hormones, only male pups were used in the immunotoxic study. On PND 3, the litters were culled to obtain the maximum number of males from each litter, leaving each mother with 8 pups. Six to ten male rats/group were used in each assay for the postnatal study with heterogenic hosts. The BWG of the male pups from trials A and B was recorded weekly until weaning and PND 70, respectively.
Hemogram, lymphoid organ evaluation, and bone marrow cellularity
On PND 21 and PND 70, male rats from the 5 groups of dams were anesthetized intraperitoneally with xylazine and ketamine (5 and 50 mg/kg, respectively) to collect blood from the caudal vena cava. After euthanasia by cervical dislocation as preconized by the American Veterinary Medical Association, 25 the lymphoid organs (thymus and spleen), and bone marrow from the left femur were harvested to investigate the immunopathological effects of feed restriction during gestation. Blood collection was performed with ethylenediaminetetraacetic acid to evaluate hematological parameters using automated Horiba® ABX equipment.
For the analysis of lymphoid organs, the thymus and spleen were removed from the euthanized rats and weighed. Bone marrow cell suspensions were obtained by flushing the marrow cavity of the left femur of each rat with ice-cold Roswell Park Memorial Institute (RPMI) 1640 medium using a sterile syringe with a 26-gauge needle. Bone marrow cell viability was assessed using a trypan blue dye exclusion test, and cell numbers were determined with a hemocytometer.
Innate immune function: Macrophage activity
Seven days before euthanasia (PND 14 (trial A) and PND 63 (trial B)), the pups were injected intraperitoneally with a 3% thioglycolate solution to elicit macrophage migration to the cavity. At the end of the experimental period, the activity of the peritoneal macrophages was evaluated using the protocols previously described in Rabinovitch and Destefano. 26
Briefly, macrophages were obtained by lavage of the peritoneal cavity with 10 mL of cold phosphate-buffered saline (PBS). The cells were centrifuged and resuspended in RPMI 1640 medium and adjusted to a density of 2 × 106 cells/mL. Duplicate 200 µL aliquots of the cell suspension from each animal were placed in 6-well tissue culture plates. To assess the phagocytic activity, 1.0 mg of opsonized zymosan A solution (5.0 mg/mL) was added to each well and the cells were incubated for 60 min at 37°C. Then, the wells were rinsed with PBS to remove uningested zymosan and any nonadherent cells. Thereafter, 200 cells/well were analyzed, and the macrophage phagocytic index (PI%) was calculated as follows: PI% = number of phagocytic cells (PI) × 100 µ. A total of 200 adherent cells were counted. The PI was the number of cells that ingested ≥ one particle. Each well was examined twice, and the mean of the four values obtained from the duplicate counts from two wells/rat was used for the statistical analysis.
The spontaneous and phorbol myristate-acetate (PMA) solution-induced hydrogen peroxide (H2O2) release by macrophages was measured according to the method of Russo et al. 27 The H2O2 concentration was calculated from the absorbance measurements as described by Pick and Mizel. 28 Additionally, the concentrations of nitric oxide (NO) in the supernatants of macrophages seeded with lipopolysaccharide (100 ng/mL) for 24 h were measured with the Griess reagent. 29 Briefly, the Griess reagent was prepared by mixing an equal volume of 1% sulfanilamide in 5% phosphoric acid and 0.1% naphthylethylenediamide for immediate use. The Griess reagent (100 µL) was mixed with an equal volume of the cell supernatant. After incubation at room temperature for 10 min, the micromolar value was measured using a microplate reader (PowerWave HT, BioTek®, Winooski, Vermont, USA) at 540 nm. The NO measurements were performed in triplicate.
Humoral immune function
The humoral immune response was evaluated as previously described by Sharma et al. 30 with modifications. On PND 14 and PND 63, pups from trial A and trial B, respectively, were immunized by ip injection of 2.0 × 10 9 sheep red blood cells (SRBC) in 0.9% saline. Seven days later, blood samples were harvested from the euthanized rats, allowed to clot in a vertical tube at room temperature for 45 min, and then centrifuged at 4000 r/min for 10 min. The serum was collected, and the anti-SRBC antibody titer was estimated as follows: 25 µL of twofold diluted serum in 0.9% saline was challenged with 25 µL of a 1% v/v SRBC suspension in microtiter plates. The plates were incubated at 37°C for 1 h and observed for hemagglutination. The highest dilution that caused hemagglutination was used as the antibody titer. The antibody titers were expressed in a graded manner with the minimum dilution (1/2) ranked as one, and the mean ranks of different groups were compared for statistical analysis.
Cellular immune function: Delayed-type hypersensitivity
Delayed-type hypersensitivity (DTH) was assessed as reported by Henningsen et al. 31 Seven days before the end of each experimental period (PND 14 and 63), the rats were injected subcutaneously in the caudal tail fold with 0.1 mL of bovine serum albumin (BSA; 1.0 mg/mL; BSA-fraction V; Sigma, St Louis, Missouri, USA) emulsified in Freund’s complete adjuvant (Sigma) at a 1:1 ratio. The immunological challenge was performed 7 days later. Heat-aggregated BSA (hBSA) was prepared by heating BSA (10 mg/mL) in saline to 75°C for 1 h. After removing the excess saline, the hBSA was injected intradermally into the left hind paw. Prior to and 24 h after the hBSA injection, the footpad was measured with a caliper (Mytutoyo number 7309). Swelling was calculated by subtracting the data obtained after the hBSA injection from the data obtained prior to challenge.
Statistical analysis
The maternal BWG, maternal food consumption, fetal and placental weights, percent preimplantation loss (100 × ((no. of corpora lutea − no. of implantations)/no. of corpora lutea)), percent postimplantation loss (100 × ((no. of implantations − no. of viable fetuses)/no. of implantations)), fetal sex ratio, and immune evaluation data were analyzed with the SAS statistical software package (Version 9.2; SAS Institute, Cary, North Carolina, USA) using one-way analysis of variance followed by Dunnett’s test for multiple comparisons. Fisher’s exact test was used for contingency analyses of visceral and skeletal observations. The data were expressed as the mean ± standard error of the mean, and the differences were considered statistically significant at p < 0.05.
Results
Experiment 1
The feed-restricted females showed apparent increase of activity that returned to normal behavior one day after returning the dams to ad libitum food consumption. Maternal BWGs on GD 7–18 in the 4 feed-restricted groups were significantly lower (p < 0.05) compared to the control group and were dependent on the amount of feed restriction; dams from the 9 and 6 g/rat/day feed-restricted groups lost weight during this period (Table 1). However, maternal BWGs during GD 19–21 (after feed restriction) in all restricted groups were higher (p < 0.05) than in females with ad libitum access to food during the entire gestation period. In terms of total dam BWG (GD 0–21), mothers from the 12, 9, and 6 g/rat/day feed-restricted groups weighed significantly less (p < 0.05) than the control mothers.
Reproductive performance, maternal measurements, and fetal viability of dams with feed restrictions from gestational day 6 to day 17 (mean ± SEM).

BWG: body weight gain; BW: body weight; GD = gestation day; SEM: standard error of the mean.
Different letters (a, b, c, d and e) within the same line showed significant (p < 0.05) differences among the groups.
1Number of observations.
Table 1 shows that feed restriction did not induce any changes in the numbers of corpora lutea, preimplantation or postimplantation losses, viable fetuses, sex ratios, and fetal lengths. No dams underwent total litter resorption. However, significant differences in the placental and fetal weights were observed among fetuses from mothers fed 12, 9, and 6 g/rat/day compared with the controls.
No fetal external (data not shown), visceral, or skeletal malformations associated with the feed restrictions were detected (Tables 2 and 3).
Skeletal observations.

Visceral observations.

aSignificantly different from control group at p < 0.05.
Experiment 2 (trials A and B)
Table 4 shows food consumption for both the gestation and lactation periods in dams administered the feed restriction regimen during pregnancy. The total BWG of these dams and offspring are presented. A significant dose–response reduction in feed intake was observed in all restricted groups of dams during pregnancy (p < 0.05). No significant effect was observed for the dams with the 15% feed restriction (16 g feed offered). However, a significant reduction in the BWG of the dams from the other groups under the feed restriction regimen occurred in a dose-dependent fashion (p < 0.05).
Total body weight gain and feed intake during pregnancy and lactation of dams with feed restrictions from gestational day 6 to 17, and the body weight and body weight gain of their offspring at birth, at weaning (21 days) and at adulthood (70 days).a

BWG: body weight gain; BW: body weight; GD = gestation day; SEM: standard error of the mean.
aNumber of observations. Data are expressed as mean ± SEM. Different letters within the same line showed significant (p < 0.05) differences among the groups.
A significant increase in the BWG of restricted mothers during lactation also occurred in a dose-dependent fashion, primarily for the dams that were provided 9 and 6 g of feed (p < 0.05).
A reduction was observed in the body weight at birth of litters from dams fed the 12, 9, and 6 g/rat/day diets (p < 0.05). The lowest BWG was observed in litters from dams fed the 9 and 6 g/rat/day diets throughout both trial periods (21 and 70 days of life; Table 4).
The hemograms of pups evaluated at 21 and 70 days showed no differences among the groups (Table 5). Regarding the lymphoid organ analysis of the pups (relative weights and bone marrow cellularity), an increase was found in the thymus relative weight at 21 days in all feed-restricted groups compared with the control group (p < 0.05; Table 6).
Hematological parameters of male offspring rats 21 and 70 days old from dams with feed restrictions.

SEM: standard error of the mean.
aNumber of male pups evaluated. Data are expressed as mean ± SEM.
Evaluation of lymphoid organs and bone marrow cellularity of male offspring rats 21 and 70 days old from dams with feed restrictions (mean ± SEM).
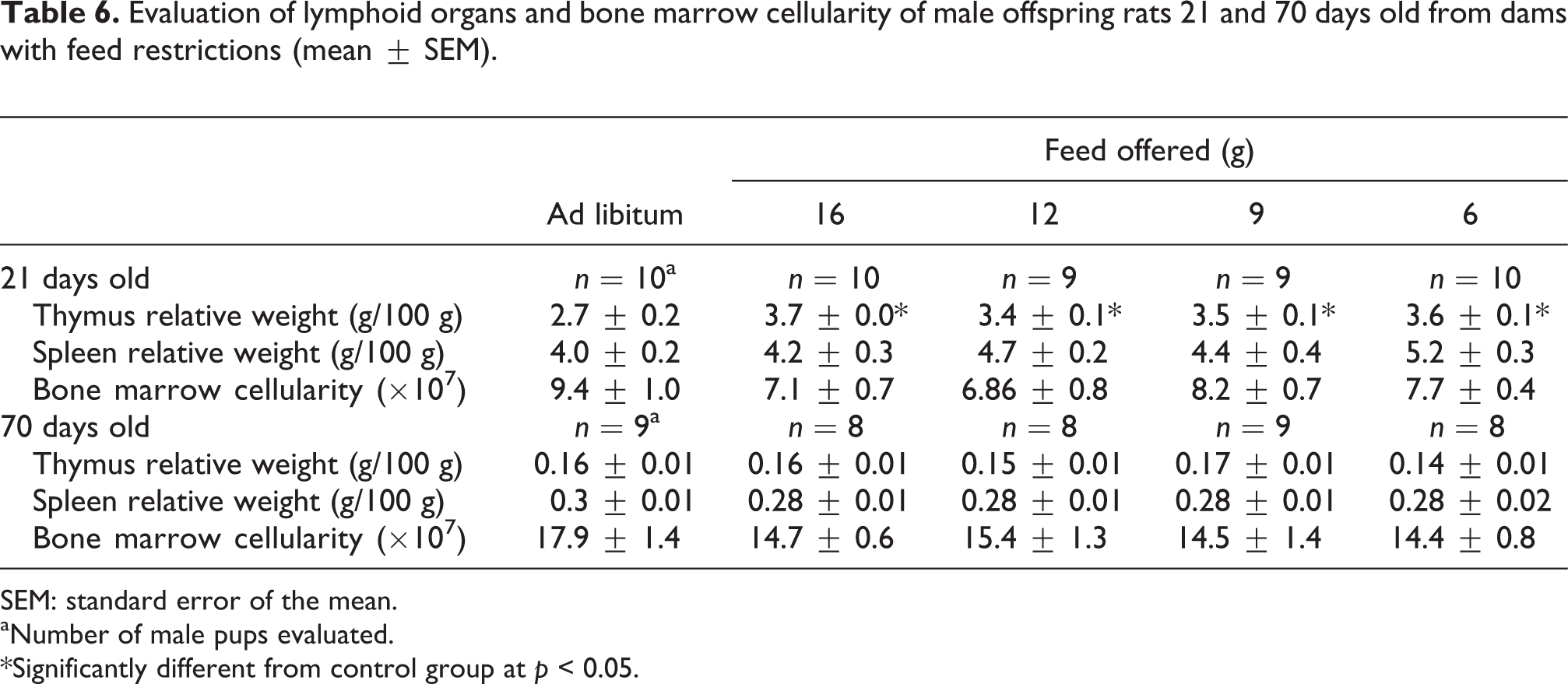
SEM: standard error of the mean.
aNumber of male pups evaluated.
*Significantly different from control group at p < 0.05.
Table 7 shows the peritoneal macrophage activities of 21- and 70-day-old pups. An increase in the PMA-induced H2O2release by inflammatory cells occurred in the 21-day-old pups from all dams with feed restrictions (p < 0.05).
Peritoneal macrophage activities of male offspring rats 21 and 70 days old from dams with feed restrictions (mean ± SEM).
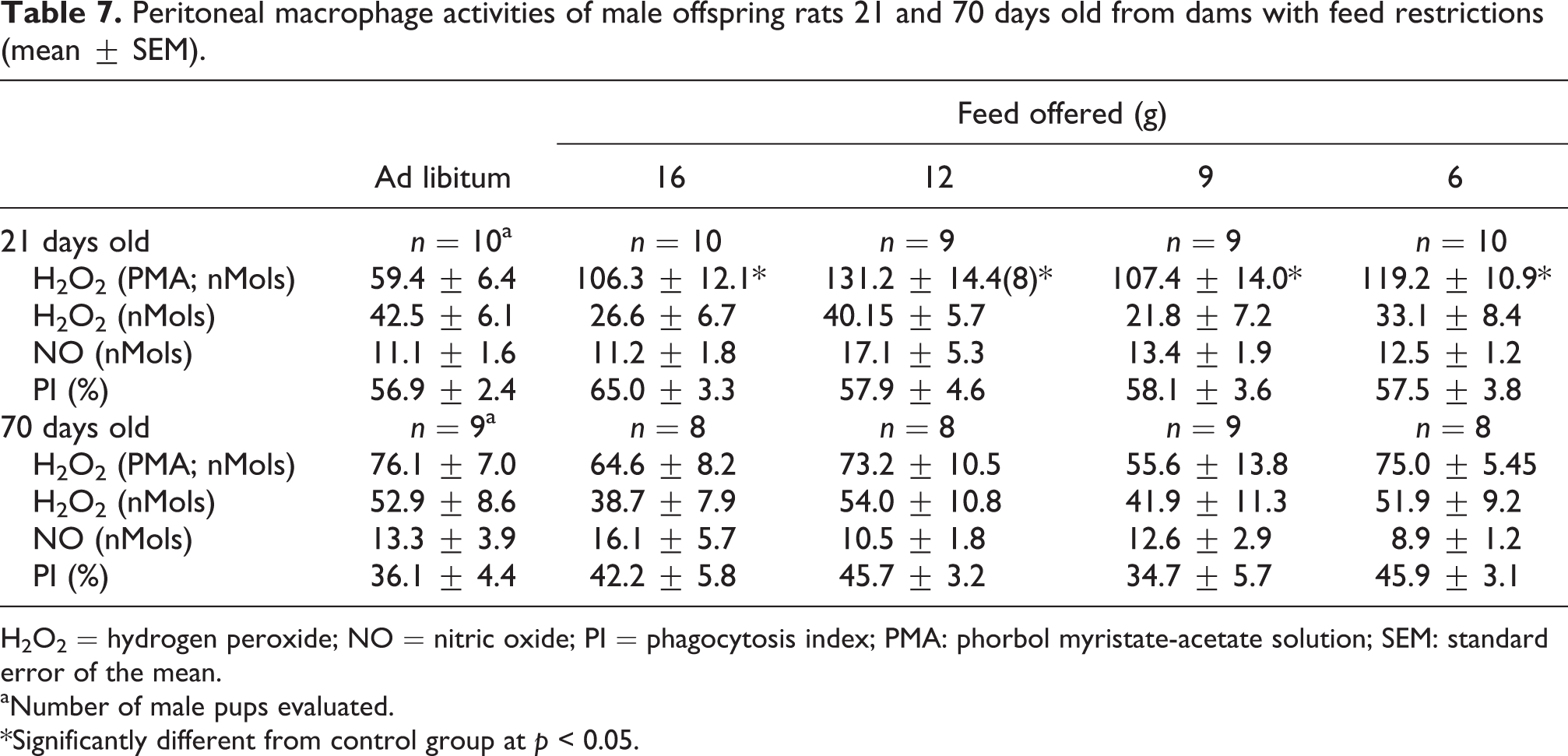
H2O2 = hydrogen peroxide; NO = nitric oxide; PI = phagocytosis index; PMA: phorbol myristate-acetate solution; SEM: standard error of the mean.
aNumber of male pups evaluated.
*Significantly different from control group at p < 0.05.
In the acquired immune function evaluations, the 21-day-old pups from the experimental groups showed an increased hypersensitivity response to BSA (12, 9, and 6 g feed diets; p < 0.05). Conversely, pups from restricted mothers revealed an increase in the DTH response (9 and 6 g feed diets, p < 0.05) and the serum titer against SRBC at adulthood compared with their control groups (p < 0.05), as shown in Table 8.
Experiment 2. Immune functions of male offspring rats 21 and 70 days old from dams with feed restrictions (mean ± SEM).

SRBC = sheep red blood cell; DTH = delayed type hypersensitivity; SEM: standard error of the mean.
aNumber of male pups evaluated.
*Significantly different from control group at p < 0.05.
Discussion
Developmental toxicity bioassays are largely standardized throughout the world. The protocols require at least three dose levels of the study agent and two species, with rats the most preferred animal species. 3,32 However, some debate existed concerning the interstrain response differences in toxicological tests. 33 –35 Sprague–Dawley (SD) and Wistar Hannover (WH) rats generally have similar parameter in reproductive toxicology. 36 –38 Thus, we expected to obtain similar findings using the WH strain using the protocol proposed by Fleeman et al., 11 who used SD rats to evaluate embryo-fetal development in feed-restricted mothers. Indeed, we found a similar profile in maternal measurements and reproductive performance data compared with the Fleeman study. We detected reductions in fetal body weight and minor changes in skeletal development, but no external, visceral, or skeletal malformations associated with any of the levels of maternal feed restriction. Therefore, the WH rats were assumed to be a suitable strain to reproduce the protocol of feed restriction during organogenesis proposed by Fleeman et al. 11 Interestingly, the comparison of control SD and WH rats showed that both the feed consumption and body weight of the WH dams were lower than those of the SD dams. Okamura et al. 39 and Noritake et al. 38 reported similar results. Campion et al. 37 proposed that the requirement for less test material was one of the primary reasons for the increased use of WH rats as an animal model in toxicology.
Curiously, although the lactating dams from the feed-restricted groups did not increase their total food intake, the dams from the 9 and 6 g/rat/day feed-restricted groups showed a significant increase in BWG during this period. Hence, studies are planned to determine the physiological mechanism(s) underlying this effect. We are also performing additional studies to clarify whether there is a decrease in milk yield, and if so whether this decrease is accompanied by a decline in milk quality. This latter evaluation will be relevant when interpreting the BWG of pups, because a decrease in this parameter was observed in pups from mothers treated with 9 and 6 g/rat/day of feed in both periods assessed (birth to 21 days and 21–70 days). One theory to explain this result is poor milk quality/quantity from feed-restricted lactating dams.
Increases in the numbers of pups/litter and the frequency of their suckling behavior increases milk production, which leads to an increase in the food intake of the dam 40 –42 and a safe, gradual decrease in the maternal body weight in different animal species, 43,44 including women who are nursing. 45 However, because the number of pups in the present study was approximately equal among the groups and the food supply was ad libitum for all dams during the lactation period, a second hypothesis is required to explain both results. Thus, it is also feasible to theorize that the increase in the BWG of feed-restricted mothers during the lactation period and the decrease in the BWG of pups could be associated with deficits in pup suckling behavior as a consequence of the feed restriction in utero.
There is great concern among researchers about whether the existing protocols used to determine potential exposure risks of numerous chemical substances during fetal development reflect real conditions. Claudio et al. 46 noted limitations in the protocols used to assess developmental toxicity. One of the most important limitations of these protocols was the lack of examination of the offspring during the postnatal period, which could lead to mistakes in the assessment of the developmental toxic effects of the substances under examination. In a study of pregnant rats performed in this laboratory, alterations were evident after weaning pups from mothers treated with cyanide during gestation. In contrast, when all of the parameters analyzed in reproductive toxicology were considered (i.e. skeletal morphology and visceral analysis of the fetuses), no significant differences were observed between the control and experimental animals. 47 Therefore, in this study the possible developmental effects of feed-restriction on the mothers were identified using not only the conventional protocols but also immunotoxic parameters during later stages of the postnatal development period.
The relationship between the development of the immune system and the maternal nutritional status during pregnancy has been studied for at least four decades. In another feed restriction study, Carney et al. 48 found that female rats that were feed-restricted during pregnancy and lactation had offspring with thymus impairment. However, this result was contradictory with the present study because our data revealed that weaned pups from feed-restricted pregnant rats had increased relative thymic weights.
The thymus is a primary lymphoid organ that is the site of the differentiation, proliferation, and maturation of T-lymphocytes into immune competent cells. In rats, the development of the thymus has its organogenesis on approximately GD 13, which is when the thymocytes migrate to and begin to colonize the tissues. 49,50 The process is characterized by a rapid growth in size due to the proliferation and differentiation of thymocytes. The thymus reaches its maximum size early in the postnatal period, when it begins to shrink as a primary feature of age-related thymic involution. 51,52 Lymphoid organs are vulnerable to malnutrition. Deficiencies in micronutrient support, such as with zinc, 53,54 vitamins, 55,56 and amino acids, 57 results in precocious thymus involution and atrophy, as reported by Carney et al. 48 In the present study, we evaluated the thymus status of pups after weaning. It is important to note that the lactating females had free access to food until the end of the experimental period. Thus, in relation to the results obtained here, there was not an increase in the relative weight of the thymus of pups with nutrient restriction in utero, but a natural thymus involution of those offspring from mothers with a balanced diet. Therefore, pups of mothers without feed restriction reached a natural process of thymic involution, whereas pups from mothers with feed restrictions during gestation were still in the process of thymus colonization, indicating a lag period in thymus development. Corroborating this hypothesis was the observation that no significant differences in thymus relative weights were found in the adults.
Studies on perinatal protein-energy malnutrition in children of pregnant women with food restrictions revealed that nutrient deficits induced profound immunodeficiencies in all branches of the immune system: cell-mediated immunity, humoral immunity, and innate immunity. 58 However, contrary to expectations, we found an increased DTH response in offspring from mothers of all feed-restricted groups when we evaluated the competence of the cellular immune function of the offspring for both periods (PND 21 and 70) . Moreover, all pups from feed-restricted mothers at adulthood had augmented antibody anti-SRBC titers when the humoral immune response was assessed.
Environmental factors can influence fetal development in a variety of ways that result in direct malformations due to DNA lesions or even epigenetic modifications that induce heritable changes. Among these environmental factors, we highlighted those related to in utero undernourishment that led to adaptive changes in the fetal phenotype with permanent modifications. 59 Several studies have attempted to elucidate the influence of these nutritional epigenetic effects in different organs, including adipose tissue. Jousse et al. 60 studied the effects of perinatal maternal undernutrition on DNA methylation and expression of the leptin gene and found that removal of methyl groups from 5′–C–phosphate–G–3′ sites in the promoter region of the leptin gene caused a permanent modification in the dynamics of leptin gene expression, particularly in response to a meal when leptin expression was more strongly induced in the offspring from undernourished dams than in control offspring.
Studies investigating leptin and its interaction with the immune system reported that this hormone had an immunomodulatory effect on both the innate and adaptive immune responses, with direct effects on macrophage up-regulating functions, phagocytosis, proinflammatory cytokine secretion, 61 –63 and B-cell activity, including an increase in immunoglobulin G isotype production in the process of the T-cell switch towards Th1 64 and constraints on regulatory T-cell proliferation and suppressive activity. 65 Thus, additional studies must be conducted to better address and concatenate the effects of maternal feed restrictions and offspring immunomodulation with epigenesis and leptin blood levels. It is possible that the observed improvement in the acquired immune responses was due to an in utero undernutrition that led to an epigenetic effect on the promoter region of the leptin gene. Thus, the increased levels of this hormone/cytokine after meals could interfere in the immune response, as shown here.
In conclusion, this study corroborated the assumption that SD and WH rats produced similar results in teratological studies. In the same manner described by Fleeman et al., 11 we concluded that feed restriction during organogenesis only produced a reduction in fetal body weight and minor changes in skeletal development. However, using the postnatal evaluation it was possible to demonstrate the immunomodulatory effects of feed restriction during gestation, reinforcing the necessity for complementary evaluation of the neonate after weaning, which could improve the teratogenicity testing procedures used today.
Footnotes
Acknowledgments
We thank Paulo Cesar Fabricio Raspantini, Leonila Ester Reinert Raspantini, Estevão Belloni, Marco Antonio Faustino dos Santos, and Adilson Baladore for their valuable assistance with the study and for the animal care.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Conselho Nacional de Desenvolvimento Científico e Tecnológico - CNPq.
