Abstract
Developmental immunotoxicity (DIT) occurs when exposure to environmental risk factors prior to adulthood, including chemical, biological, physical, or physiological factors, alters immune system development. DIT may elicit suppression, hyperactivation, or misregulation of immune responses and therefore may present clinically as decreased resistance to pathogens, allergic and autoimmune diseases, and inflammatory diseases. When evaluating DIT in an animal model, specific endpoints are assessed, which can reveal the potential for a risk factor to alter immune system development. However, linking DIT evaluation in an animal model with clinical realities observed in human populations requires that DIT testing regimens evaluate critical windows in immune system development. In addition, pathways leading to DIT may not be apparent without the stressors that induce aberrant and detectable responses. This review contains brief descriptions of recently published work that addresses disease patterns associated with DIT and solutions for altering such patterns of disease. We also comment on gaps between DIT testing in animal models and the clinical manifestation of immune-based diseases in children that can be filled by a better understanding of critical windows in immune system development and DIT testing that includes multiple functional assays.
The immune system is complex, with evolving disease-surveillance functions that protect the body from birth until adulthood. Given recent increases in the incidence of childhood immune-based diseases such as recurrent otitis media, asthma, allergies, and type 1 diabetes, understanding the role of environmental stressors and vulnerable periods of immunological maturation is of increasing concern (Dietert and DeWitt 2010). Compromised immunity that leads to increased susceptibility to infection, for instance, has been demonstrated in rodents exposed to polychlorinated biphenyls (PCBs) during early stages of immunological development (Dietert and DeWitt 2010). This is supported in epidemiology studies, indicating positive correlations between PCB exposure and rates of recurrent otitis media and recurrent respiratory infections in children (Dietert and DeWitt 2010).
Research investigating developmental immunotoxicity (DIT) and the role of environmental stressors in childhood disease is becoming a global priority, as evidenced by several recent workshops and symposiums (Burns-Naas et al. 2008). This is supported by the increasing awareness that the developing immune system is typically more sensitive to toxicant exposure than is the adult immune system (Luebke et al. 2006). Further evidence of this priority concern to the global community is consideration of inclusion of DIT testing in regulatory developmental and reproductive toxicology (DART) protocols along with developmental neurotoxicity testing (Burns-Naas et al. 2008).
Immune Dysfunction and Early-Life Health Risks
In response to the increasing prevalence of pediatric immune-based diseases, Dietert and Zelikoff (2008, 2009, 2010) developed a system for identifying patterns of immune-based diseases associated with immune dysfunction and early-life health risks. The focus of the first review (Dietert and Zelikoff 2008) was on avoidance of specific environmental risk factors that affect the developing immune system, culminating in asthma and allergic diseases, including allergic rhinitis, atopic dermatitis, and food hypersensitivities. Dietert and Zelikoff (2009) expanded this approach with the development of a matrix for health risks associated with pediatric immune dysfunction, leading to correlations of primary immune-based diseases with comorbid diseases that developed later in life. For example, patients diagnosed with the primary immune-based disease inflammatory bowel disease had an increased risk of developing another immune-based disease, such as asthma or multiple sclerosis, later in life. Furthermore, Dietert and Zelikoff (2010) suggested that later-life immune-based diseases that are comorbid with a primary immune-based disease can be prevented if the primary disease is treated as an immune disease rather than as a disease in the presenting system. In doing so, they identified four main patterns of pediatric immune-related diseases associated with prototypical “entry-way” diseases: allergic, autoimmune, inflammation, and infection related. The authors hypothesized that if the entry-way disease was treated as an immune-based disease early in life, the risk of developing a comorbid immune-based disease later in life would decrease (Figure 1).
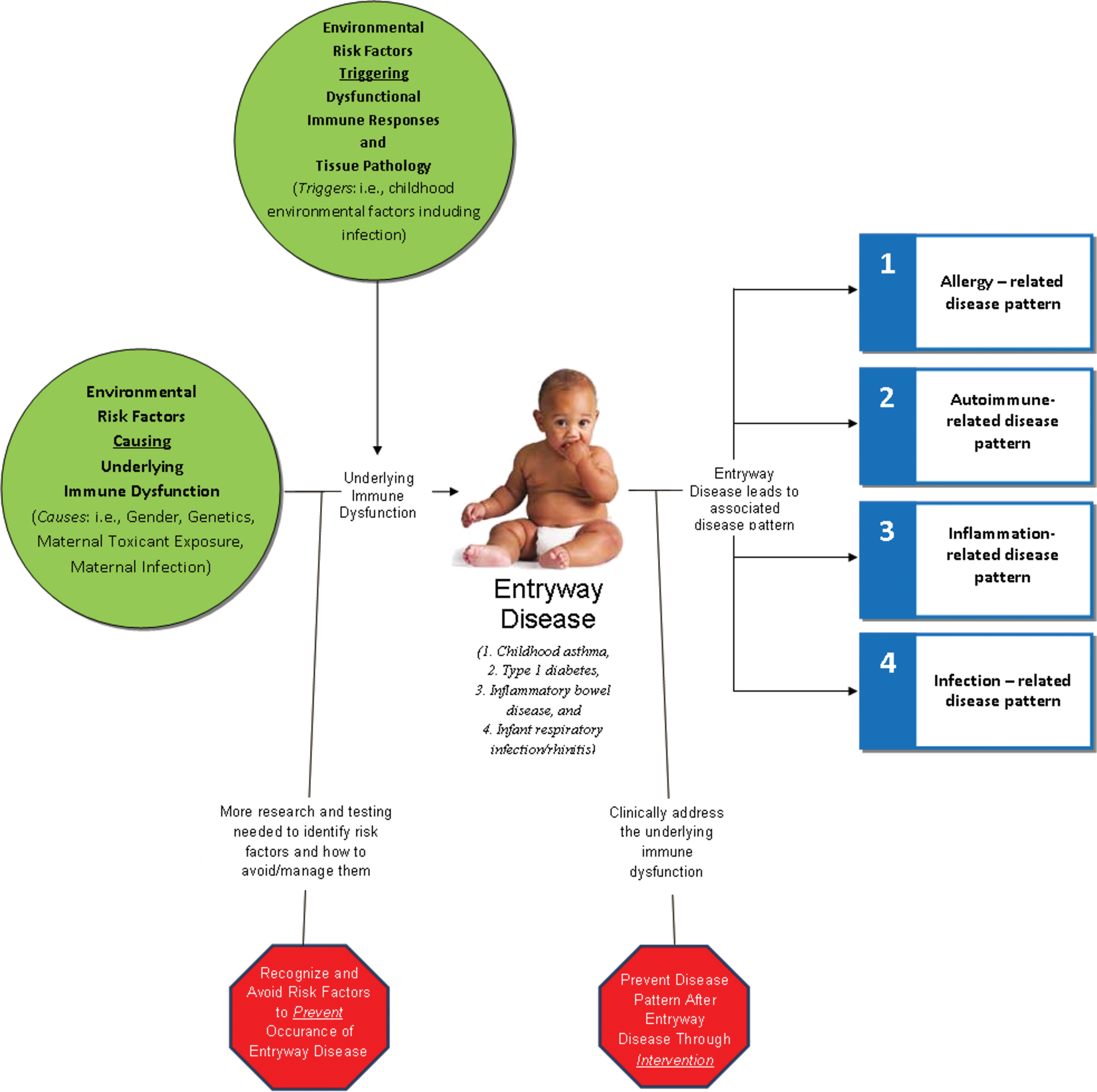
Generalized schematic of entry-way diseases and disease pattern concept proposed by Dietert and Zelikoff (2010). This figure demonstrates the overlapping relationships between causative agents for underlying immune dysfunction with regard to later-life triggers (Dietert 2009) that lead to entry-way diseases spanning allergy-, autoimmune-, inflammatory-, and infectious-related dysfunctions.
In a subsequent review article, Dietert et al. (2010) took the proposals elucidated by Dietert and Zelikoff (2008, 2009, 2010) one step further and suggested that breaking immune dysfunction–based patterns of disease requires improved identification of environmental risk factors as well as appropriate treatment of the early-life immune-based disease. The authors highlighted metabolic syndrome as an immune-based disorder induced by environmental risk factors. Inflammatory dysfunction is a hallmark of metabolic syndrome and linked diseases, including heart disease, stroke, and type 2 diabetes. Therefore, these diseases may be managed by treating the underlying, early-life immune dysfunction. Dietert et al. (2010) suggested that there are two significant opportunities during the lifetime of a potentially afflicted individual to reduce pattern-associated health risks: avoiding environmental risk factors associated with immune dysfunction and managing the entry-way disease as an immune dysfunction–based disease to reduce the risk of associated diseases. The authors articulated that to accomplish the latter, therapeutic approaches must focus on the health risks within a pattern rather than on just treating the clinical symptoms (Figure 1). In addition, Dietert et al. (2010) asserted that to accomplish the former, DIT testing should be a required component of safety testing for drugs and chemicals and that evaluated endpoints should be directly relevant to immune dysfunction–based diseases.
Critical Windows of Immunological Maturation
Just like every other system in the body, development of the immune system is a regulated process that involves spatial and temporal interactions that culminate in specific structures and functions. When the process of immune system development is disturbed, it is possible to identify critical windows, or periods of vulnerability, for cells, tissues, and processes, including differentiation, selection, and seeding of cells to the periphery. Five major maturational events occur in immune system development: (1) initiation of hematopoiesis, (2) stem cell migration and cell expansion, (3) colonization of bone marrow and thymus, (4) maturation to immunocompetence, and (5) establishment of immune memory (Figure 2; Dietert et al. 2000). If a disturbance occurs during a particular maturational event, altered morphology or dysfunction may be detectable in the offspring once they have attained immunocompetence. For example, in a study of the effects of cyclosporin, an immunosuppressant used to prevent organ transplant rejection, Barrow et al. (2006) observed DIT in pups exposed postnatally but not prenatally. In 10-week-old pups exposed to cyclosporin from postnatal day 4 through 28, splenic hyperplasia of the periarterial lymphatic sheaths and mantle zone and suppression of T-cell–dependent antibody responses (TDARs) were observed. No such changes were observed in pups exposed to cyclosporin during gestation. This example illustrates that, in some instances, changes to both organ structure and function are detectable. However, in other instances, detectable changes in organ structure do not accompany changes in function (Peden-Adams et al. 2007, 2008; Keil et al. 2004, 2009; Allen et al. 2003) and suggest that for certain toxicants or when exposure occurs during specific critical windows of immune maturation, changes in organ structure may not correlate with immune dysfunction. Therefore, an approach that combines both structural (histopathology, lymphoid organ weights, immunophenotyping) and functional (TDAR, cytotoxic T-lymphocyte activity, natural killer [NK] cell activity, etc.) endpoints may be more predictive of DIT (Weinstock et al. 2010). For example, Watanabe, Shimizu, Sawamura, Hino, Konno, and Kurokawa (2010) reported that after perinatal exposure to a flame retardant, decabrominated diphenyl ether (DBDE), the exposed offspring developed a functional disorder of primary immunity in response to a respiratory syncytial virus (RSV). However, when 5-week-old mice were fed DBDE for 4 weeks, pulmonary viral titers to RSV were similar to what was observed in untreated mice (Watanabe, Shimizu, Sawamura, Hino, Konno, Hirose, et al. 2010). These studies illustrate the differential sensitivity of immune system development associated with exposure to DBDE during the perinatal colonization of bone marrow, thymus, and maturation to immunocompetence. In contrast, when DBDE exposure occurred during establishment of immune memory, immune functions were not affected.
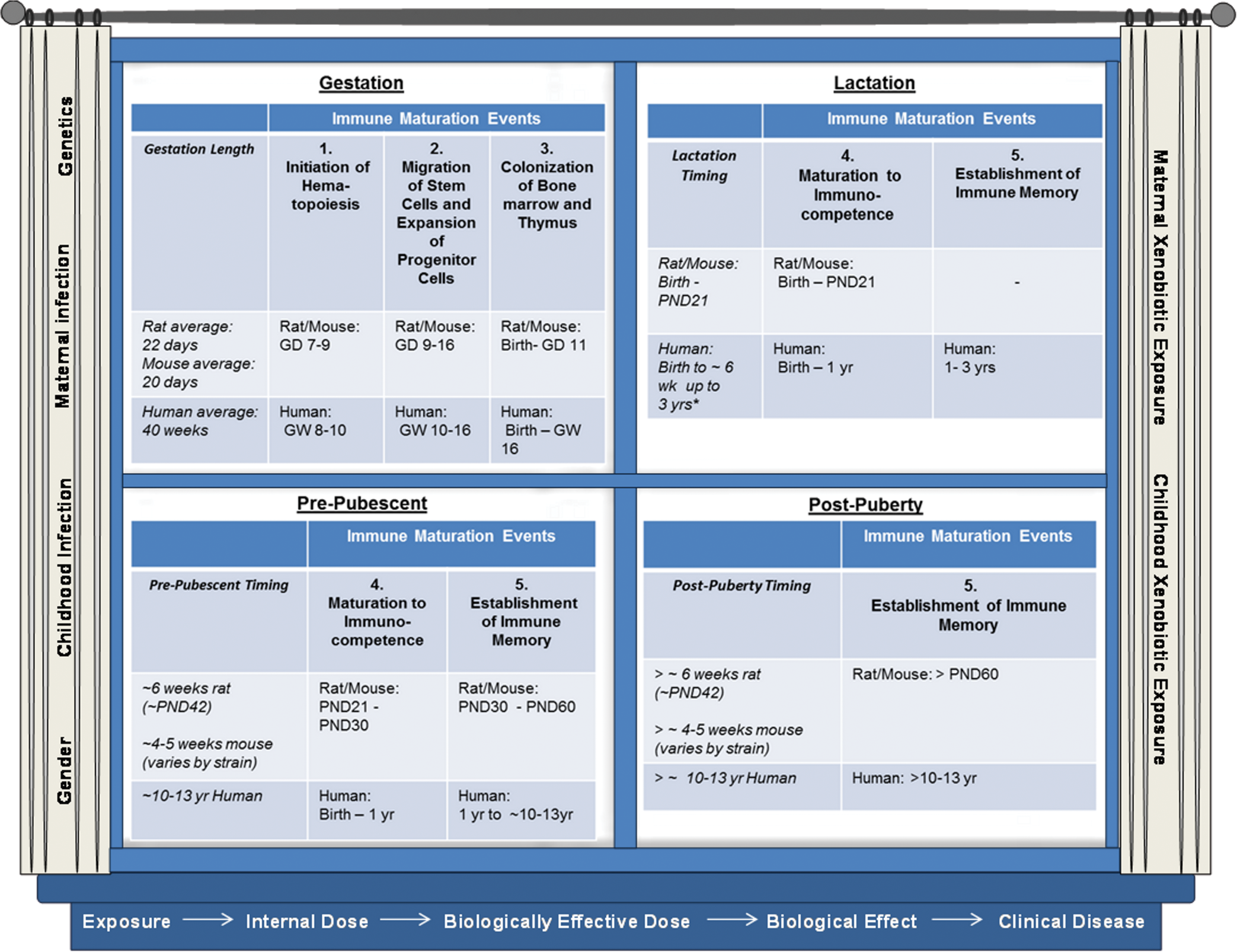
Critical windows of immune system development in relation to environmental risk factors and the exposure–disease continuum. Typical critical windows of immune development are shown in the window “panes” in relation to the corresponding immune maturation timing in rats, mice, and humans. Environmental triggers and biomarkers of susceptibility are shown on the “curtains.” The critical windows, maturation events, markers of susceptibility, and environmental triggers all relate to each step of the exposure disease continuum listed on the “window sill,” such that exposure during various critical windows with varied differences in susceptibility will modulate the outcome of DIT. This timing will vary from child to child, as some mothers may not breast-feed and some may breast-feed anywhere from 6 weeks to 3 years. Therefore, lactation exposure in humans may either be nonexistent or last for varied times, with the typical breast-feeding time generally ranging from 6 weeks to 1 year (Dietert et al. 2000; Dietert and Holsapple 2007). GW = gestation week; GD = gestation day; PND = postnatal day.
Lead (Pb) is an example of a well-studied immunotoxicant that induces different immunotoxicological effects when given at different life stages. One of its classic effects is inducement of a shift in the T helper (TH) cell balance, where Pb shifts immune responses away from TH1-dependent responses toward TH2-dependent responses. This ultimately affects host responses to antigens as TH2-dependent responses are involved in humoral immunity and immunoglobulin production, whereas TH1-dependent responses promote cell-mediated immunity. With regard to DIT, Dietert et al. (2004) illustrated that the period of T-cell hypersusceptibility to Pb corresponds to the perinatal period, which includes seeding of the thymus with bone marrow–derived precursors and subsequent thymocyte maturation. This hypersusceptibility was demonstrated in a study by Bunn et al. (2001): rats given Pb late in gestation experienced the TH1/TH2 shift, whereas those given Pb early in gestation experienced a different DIT profile that precluded the TH1/TH2 shift. Pb is also known to affect macrophages and B cells as well as T cells, and the severity of the effects vary with the timing of exposure during development. Additional studies with Pb and other immunotoxicants suggest that there are at least seven specific T-cell maturational events that represent hypersusceptibilities for DIT. However, depending on the toxicant being evaluated and/or the endpoint being assessed, additional hypersensitivities may exist. This is particularly true when resident immune populations in nonlymphoid tissues are included in consideration. For example, Dietert and Dietert (2008) suggested that nine DIT vulnerabilities of immune maturation warrant special consideration when it comes to potential developmental vulnerabilities of autism spectrum disorders. These DIT vulnerabilities include both those that are systemic, such as seeding of the thymus with thymocytes, and those that are specific to immune maturational events within the nervous system, such as appearance and expansion of astrocyte populations (Dietert and Dietert 2008). DIT evaluation would, therefore, potentially capture not only systemic immune responses but also responses that include immune populations within nonlymphoid tissues.
Need for DIT
Although recommended for inclusion in DART protocols (Burns-Naas et al. 2008), under current U.S. regulatory requirements for evaluating the safety of exogenous agents, DIT testing is not routinely performed and is generally initiated only by evidence of immunotoxicity from adult exposure studies (Dietert and DeWitt 2010). As the developing immune system is generally regarded as being more sensitive than is the adult immune system, immunotoxicity testing in adults would not adequately account for potential impacts to the developing immune system (Luebke et al. 2006). However, with regard to nondrug agents, adult immunotoxicity testing is required only under the Federal Insecticide, Fungicide, and Rodenticide Act for pesticides but not under the Toxic Substances Control Act (TSCA) for other types of chemicals. Typically, under the TSCA, initiation of immunotoxicity testing requires changes in immune organ mass or cellularity, which are not always sensitive immune endpoints. Various studies have shown that effects in the immunotoxicity tests chosen for assessment by the U.S. Environmental Protection Agency (EPA) within the Immunotoxicity Test Guidelines (OPPTS 870.7800) can occur in the absence of immune organ mass or cellularity changes (Peden-Adams et al. 2007, 2008; Keil et al. 2004, 2009; Allen et al. 2003). Leubke et al. (2006) indicated that for diethylstilbestrol, diazepam, lead, 2,3,7,8-tetrachlorodibenzo-p-dioxin, and tributyltin oxide, the developing immune system was found to be at greater risk of modulation than the adult system because effects were observed at lower doses, were persistent, or both. However, the increased sensitivity of the developing immune system relative to the adult immune system is not yet comprehensively characterized, as our understanding of the processes of the developing immune system is still the focus of intense investigation (Holsapple et al. 2004). If DIT testing is routinely performed before an agent undergoes adult immunotoxicity testing, it is likely that positive DIT results would be predictive of adult immunotoxicity and be sufficient for protection of both adult and developing immune systems. In the review of the five environmental chemicals by Luebke et al. (2006), all five chemicals induced immunotoxicity in developing organisms at lower doses and/or persisted for a longer period of time than in adult organisms. Therefore, if data from developmentally exposed animals were used to establish safe exposure guidelines, the adult immune system would likely be sufficiently protected at doses that induce DIT.
Even as the knowledge of vulnerable time periods in immune system development is expanded, the need to perform DIT testing using the assays already selected for immunotoxicity testing under EPA Immunotoxicity Test Guidelines (OPPTS 870.8700) in adult rodent models is not precluded. Inclusion of DIT testing in DART protocols would be advantageous because (1) the same animals could be used without incurring additional costs, (2) it would be less time-consuming than conducting a new study specifically to assess DIT, (3) it would provide information to better determine the need for immunotoxicity testing adult models, and (4) it has the added benefit of having evaluated the most sensitive age group for potential immunotoxicity.
Two of the problems with establishing guidelines for DIT testing, as discussed in a companion article by Holsapple et al. (submitted), are that the timing/duration of exposure and the most appropriate endpoint(s) to evaluate have not yet been established. But given increased prevalence of immune dysfunction–based diseases such as childhood asthma and type 1 diabetes, there is ample pressure to initiate relevant and timely DIT testing of chemicals and drugs (Dietert 2011a, 2011b). Dietert and Holsapple (2007) proposed a DIT exposure scenario that has been carried forward by others calling for DIT testing. In their DIT exposure scenario, rodent offspring would be exposed transplacentally, lactationally, and directly from weaning through young adulthood. Immune parameters would be assessed once the offspring reached adult-level immunocompetence at approximately 6 weeks of age. This approach was developed by a diverse group of scientists and reflects the most appropriate exposure scenario for assessing DIT in animal models. An additional advantage to this approach is that if an agent induces DIT, follow-up studies can, for example, expose offspring during lactation, but not gestation, to determine if the period of heightened susceptibility is during lactation.
Also included in the approach developed by Dietert and Holsapple (2007) is a suite of immune assays recommended for determining DIT in the young adult offspring. These include TDAR, delayed-type hypersensitivity (DTH), NK cell or cytotoxic T lymphocyte (CTL) assays, histopathology, and immunophenotyping. These assays combine functional (TDAR, DTH, NK, and CTL) assays and observational (histopathology and immunophenotyping) assessments and represent tests that have a relatively high potential for predicting immunotoxicity risk. Notably, TDAR, NK cell activity, and immunophenotyping are considered three of the most predictive measures of immunotoxocity (Luster et al. 1988, 1992) and are the three tests chosen by the EPA for their Immunotoxicity Test Guidelines (OPPTS 870.8700) in adult rodents. Identifying DIT is also more effective when a multifunctional analysis evaluates different immune response dimensions (Dietert 2008). This approach is also amenable to follow-up studies to determine cells, tissues, and processes that are DIT targets. For example, Keil et al. (2008) exposed B6C3F1 mice to perfluorooctane sulfonate (PFOS) from gestational days 1 through 17 and evaluated the suite of recommended assays (Dietert and Holsapple 2007) in the offspring. Suppression of TDAR following gestational exposure coupled with studies in adult rodents led to additional experiments to determine the pathway(s) involved in TDAR suppression (Peden-Adams et al. 2008, submitted; Fair et al. 2011), which will ultimately help to establish a mechanism by which PFOS affects immune system development and safety guidelines for protection of human health. Had Keil et al. (2008) assessed only morphology or innate immunity, the ability of PFOS to suppress TDAR would have been overlooked. Therefore, when evaluating an agent for potential DIT, both the exposure scenario and the endpoints to be assessed must factor into the experimental design.
Although there is some overlap between established immunotoxicity assays and DIT testing, little has been done to examine host resistance assays that are applicable in DIT testing. This area of testing is critical for examining the overlapping functional capacity of the immune system and may facilitate risk assessment should quantitative relationships of the individual immune parameters such as TDAR, lymphocytic subpopulations, or NK cell activity be established. Keil et al. (1999, 2001) established quantitative relationships between immune parameters and host resistance with traditional immunotoxicity testing, and this approach may serve as a template for establishing similar relationships in DIT. Furthermore, this approach could assist in distinguishing causes versus triggers of immune dysfunction associated with developmental exposure.
Causes versus Triggers
Finally, identifying environmental risk factors associated with immune dysfunction requires separating causes of immune dysfunction from triggers of disease. The distinction between causes of immune dysfunction and triggers of diseases was thoroughly discussed in a review by Dietert (2009). Briefly, an environmental risk factor that is a cause of immune dysfunction is one that profoundly alters the status of the immune system following exposure during key periods of immune maturation (Dietert 2009). Typically, these environmental risk factors are experienced during gestational development, but exposure to some factors may occur postnatally, provided that they alter the course of immune system development (Figure 1). Lead, methylmercury, TCDD, and environmental tobacco smoke are all examples of environmental risk factors that cause immune dysfunction. Alternatively, triggers of disease are risk factors that may cause immune dysfunction, but when exposure occurs, they most likely uncover or facilitate the existence of an underlying immune dysfunction (Dietert 2009). Infectious agents often serve as triggers of immune dysfunction. Dietert (2009) proposed that the causative agent imprints the basis for the dysfunctional response onto the developing immune system and then the trigger serves as the critical factor that leads to pediatric disease.
Does acknowledging that separate causes and triggers of immune dysfunction exist discount the previous discussion on DIT testing? Would all DIT testing have to include tests of host resistance to see if the infectious agent triggers the underlying immune dysfunction induced by the causative agent? If DIT testing includes multifunctional analyses that evaluate different immune responses, then in essence, the challenge given to elicit the immune response can serve as the trigger. For example, when evaluating TDAR for DIT, offspring are exposed to the purported causative agent during a proscribed period of development and then given an antigen challenge to elicit the response. An appropriate antigen challenge can serve as the trigger that signals the underlying immune dysfunction. The evaluation of several different immune responses in a DIT test is critical; measuring several functional endpoints therefore provides several opportunities to challenge different arms of the immune response to essentially trigger the immune dysfunction if it exists.
Conclusions
Important in DIT testing is the role of exposure during critical windows of susceptibility and the understanding of risk factors including causes and triggers of immunotoxicity. Exposure during different times of development can result in differing effects on the immune system, as discussed earlier with the example of lead exposure (Dietert and DeWitt 2010). The concept of effects related to critical developmental windows is integral to the EPA Framework for Assessing Health Risks of Environmental Exposures to Children (Brown et al. 2008). However, comparatively few compounds have been extensively studied for DIT (Burns-Naas et al. 2008; Dietert and DeWitt 2010), but in light of the possible role of immune alterations in various increasing childhood diseases, it is important to assess DIT to determine the potential risk to children’s health. Critical to this is the need for more DIT testing in the safety assessment of chemicals and drugs. Moreover, preventing patterns of immune-based disease relies on four research-supported actions: (1) ensuring that DIT testing protocols have maximal relevance for priority human disease, (2) expanding the knowledge base of critical windows in immune system development, (3) identifying environmental risk factors that may disrupt critical windows of early life, and (4) linking DIT outcomes to triggers that will uncover underlying immune dysfunction. We are at a point where we can continue to strengthen the communication pipeline between clinicians, regulatory scientists, and research scientists to develop a DIT testing scheme that is relevant for human diseases of highest priority, meets regulatory requirements, and expands our knowledge of critical windows of immune system development.
Footnotes
The authors declared no potential conflicts of interests with respect to the authorship and/or publication of this article. The authors received no financial support for the research and/or authorship of this article.
