Abstract
High telomerase activity in human breast cancer is associated with aggressive tumors resulting in decreased survival. Recent studies have shown that telomerase inhibitors may display anticancer properties in some human cancer cell lines. In the present study, we examined the effects of 4 reverse transcriptase inhibitors (RTIs), used for the treatment of HIV; Abacavir (AC), Lamivudine (LV), Stavudine (SV), and Tenofovir (TF) on proliferation, apoptosis, and migration in the normal human mammary epithelial cell line, hTERT-HME1, and the human breast cancer cell line, MCF-7. Cells were treated with AC, LV, SV, or TF alone or in combination with paclitaxel (PAC), a known drug used to treat breast cancer. Conduct of the thiazolyl blue tetrazolium bromide assay demonstrated that AC, SV, and TF had stronger cytotoxic effects on MCF-7 cells than in hTERT-HME1 cells. The combined treatment of RTIs and PAC caused high rates of cell death in MCF-7 and low rates of cell death in HTERT-HME1 by apoptosis. The percentages of apoptotic cells in the treatment of AC and SV in combination with PAC for 48 and 72 hours were higher than PAC. Significantly increased apoptosis and decreased migration levels were found in MCF-7 cells treated with AC and co-treatment of AC+PAC or SV+PAC than HME1 cells. These treatments can also prevent migration capacity more than PAC. Therefore, a combination strategy based on telomerase inhibitors such as AC or SV and anticancer drugs may be more effective in the treatment of certain breast cancers.
Introduction
Breast cancer is one of the most common cancers in women worldwide. Unfortunately, tumors often develop resistance to chemotherapy drugs used to treat these patients with breast cancer after surgery. For this reason, more effective therapies are needed. 1
Telomeric repeat amplification is a mechanism used by many cancer cells to induce limitless proliferation. High telomerase activity was detected in many breast cancers (95% of breast carcinomas), and its activity increases proportionally with tumor activity. 1,2 A significant association was also found between telomerase activity and tumor size, lymph node status, and metastasis. In addition, normal somatic cells treated with telomerase inhibitors are not adversely affected by treatment since most do not have telomerase activity. 2 The treatment of breast cancer cells with telomerase inhibitors can induce growth arrest, senescence, and death because most have very short telomeres. 2,3 The differences in telomerase activity in normal and tumor cells led to the hypothesis that the inhibition of telomerase can cause shorter telomeres which may be a suitable anticancer treatment. 2 -4
Reverse transcriptase inhibitors (RTIs) are predominantly used for the treatment of HIV infections. They are capable of blocking the activity of human telomerase reverse transcriptase catalytic subunit because of the RNA-dependent DNA polymerase activity of telomerase. 5,6 Previous studies have demonstrated that some RTIs can inhibit the reverse transcriptase activity in a number of human cancer cells, reducing the rate of cell growth and inducing differentiation. 7 Telomerase inhibition therapy may also be most effective when used in combination with chemotherapy, radiation, or other targeted therapeutics. 8 Therefore, RTIs may be a potential target for novel anticancer therapeutics.
Based on this evidence, the present study was designed to investigate the effects of 4 RTIs; Abacavir (AC), Lamivudine (LV), Stavudine (SV), and Tenofovir (TF) on the cell proliferation, apoptosis, and migration in human breast cancer cells in vitro. A review of the literature has demonstrated that data pertaining to the combined effects of RTIs and anticancer drugs in breast cancer cells are limited. These studies also included an investigation into potential combination therapy using paclitaxel (PAC), a known chemotherapy agent used to treat breast cancer.
Materials and Methods
Chemicals and Reagents
Abacavir, LV, SV, TF, PAC, thiazolyl blue tetrazolium bromide (MTT), ethidium bromide (EtBr), and acridine orange (AO) were purchased from Sigma. Eagle’s Minimum Essential Medium (EMEM) was obtained from ATCC. Fetal bovine serum (FBS), trypsin, and penicillin–streptomycin (PS; 10,000 U/mL) were purchased from Gibco. Mammary Epithelial Cell Growth Medium Bullet Kit (MEGM) was obtained from Lonza (cat. no. 3150).
A wide range of concentrations were selected to determine the effects of high and low concentrations of RTIs. After preparing 10-mM stock of each drug, the other concentrations (1, 0.1, 0.075, 0.05, 0.025, 0.01, 0.005, 0.0025, and 0.001 mM) were prepared by serial dilutions.
Cell Lines and Cell Culture
The human breast cancer cell line MCF-7 and hTERT immortalized human mammary epithelial cell line hTERT-HME1 were purchased from ATCC. MCF-7 cells were grown in EMEM supplemented with 10% FBS and 1% PS. hTERT-HME1 cells were cultured in MEGM supplemented with insulin, human epidermal growth factor, hydrocortisone, bovine pituitary extract, and antibiotic. All cultures were maintained at 37 °C in a 5% CO2 humidified atmosphere.
Cell Proliferation and Viability Assay
MCF-7 cells and hTERT-HME1 were seeded in a 96-well plate at the density of 1 × 104 cells/well. The cells were treated with different concentrations (0.001, 0.0025, 0.005, 0.01, 0.025, 0.05, 0.075, 0.1, 1, and 10 mM) of AC, LV, SV, and TF for 24, 48, 72, 96, and 120 hours with or without PAC (0.05 µM) for 24, 48, and 72 hours. The yellow tetrazolium salt (MTT) assay to measure viability was performed according to Mosmann 9 with some modifications. A 20 µL of MTT solution (5 mg/mL) was added to each well, and the plates were incubated at 37 °C for 4 hours to induce the reduction of MTT. After the media was removed, 100 µL of dimethyl sulfoxide was added to each well. The optical density in each well was evaluated by measurement of absorbance at 570 nm using a microplate reader (Biotek Reader). All experiments were performed in triplicate, and the results were expressed as percentage inhibition relative to control cells (considered to be 100%). The cell viability was calculated by using the following formula: Cell viability (%) = ([Sample absorbance average]/[Control absorbance average] × 100). Logarithmic regressions were performed to define 50% inhibitory concentrations (IC50; concentrations which inhibit the response by 50%).
Acridine Orange and EtBr Stain
Acridine orange/EtBr staining was performed to detect live, necrotic, and apoptotic cells concomitantly in MCF-7 and hTERT-HME1 cells. Cells were treated with AC, LV, SV, or TF (0.1, 1, and 10 mM) separately or together with PAC (0.05 µM) for 24, 48, and 72 hours following seeding in 96-well plates (1 × 104 cells/well). Cells were washed with PBS, 25 µL of trypsin was added into each well. After 5 minutes, an equal volume of growth medium with trypsin was added into each well suspension. A 2 µL of AO/EtBr stain (100 μg/mL AO and 100 μg/mL EtBr in PBS) was added to 50-µL treated and control cultured cell suspensions. The mixed suspensions were transferred to microscope slides. Cells were immediately examined using the fluorescent microscope (Leica DM2500) and digitally photographed. Apoptotic cells was examined and 100 cells were counted to calculate the percentage (Apoptosis % = Apoptotic cells/total cell count × 100%). Green fluorescence was indicative of the viable cells with normal nuclear chromatin, whereas the orange-red fluorescence indicated apoptotic cells, containing fragmented nuclear chromatin. 10
Cell Migration Assay
Wound-healing assay is a commonly used method for investigation of cell migration and was used to evaluate the effects of AC, LV, SV, or TF (0.1, 1, and 10 mM) with and without PAC (0.05 µM) on cell migration in the present study. MCF-7 cells were seeded in 24-well plates. Confluent cultures were scratch-wounded with a 200-µL sterile pipette tip. After washing with 1X phosphate-buffered saline to remove cellular debris, the cells were cultured in serum-free medium containing Mitomycin C (5 μg/mL) to inhibit cell proliferation. Wound images were photographed at marked regions (0 hour). The cells were treated with AC, LV, SV, and TF (0.1, 1, and 10 mM) with and without PAC (0.05 µM) for 6, 12, 24, 48, and 72 hours, and the same regions were photographed. The wound areas were analyzed using ImageJ software. The rate of migration of cells was calculated by the following formula: Migration rate (%) = (A0−At)/A0 × 100, where A0 is the area of the wound measured after scratching (0 hour) and At is the area of the wound at the end of the each treatment times (6, 12, 24, 48, and 72 hours).
Statistical Analysis
Thiazolyl blue tetrazolium bromide assay and AO/EtBr stain were repeated at least 3 times. All data were expressed as the mean ± SE. Statistical tests were performed using SPSS v. 14.0 (SPSS Inc). Data were analyzed using 1-way ANOVA followed by the Student t test. P < 0.05 was considered statistically significant when compared to control.
Results
Cell Proliferation and Viability
The IC50 values of RTIs for hTERT-HME1 and MCF-7 cells are illustrated in Table 1. Effects on cell viability of RTIs in hTERT-HME1 and MCF-7 cells are shown in Figures 1 and 2, reflective of the MTT assay. The results of MTT assay indicated that RTIs showed stronger inhibitory effects at the higher concentrations in hTERT-HME1 cells. 1 mM and/or 10 mM concentrations of AC, SV, and TF reduced the cell viability below 50% for all treatment times, whereas LV showed this reduction after 72-hour treatment. After 120-hour treatment, the cell viability values were below 50% at the 2 highest concentrations (1 and 10 mM) of AC, SV, and TF and the highest concentration of LV (10 mM) in hTERT-HME1 cells. The IC50 values were generally lower in MCF-7 cells than hTERT-HME1 cells expect for TF treatment.
IC50 Values of hTERT-HME1 and MCF-7 Cells Treated With RTIs.
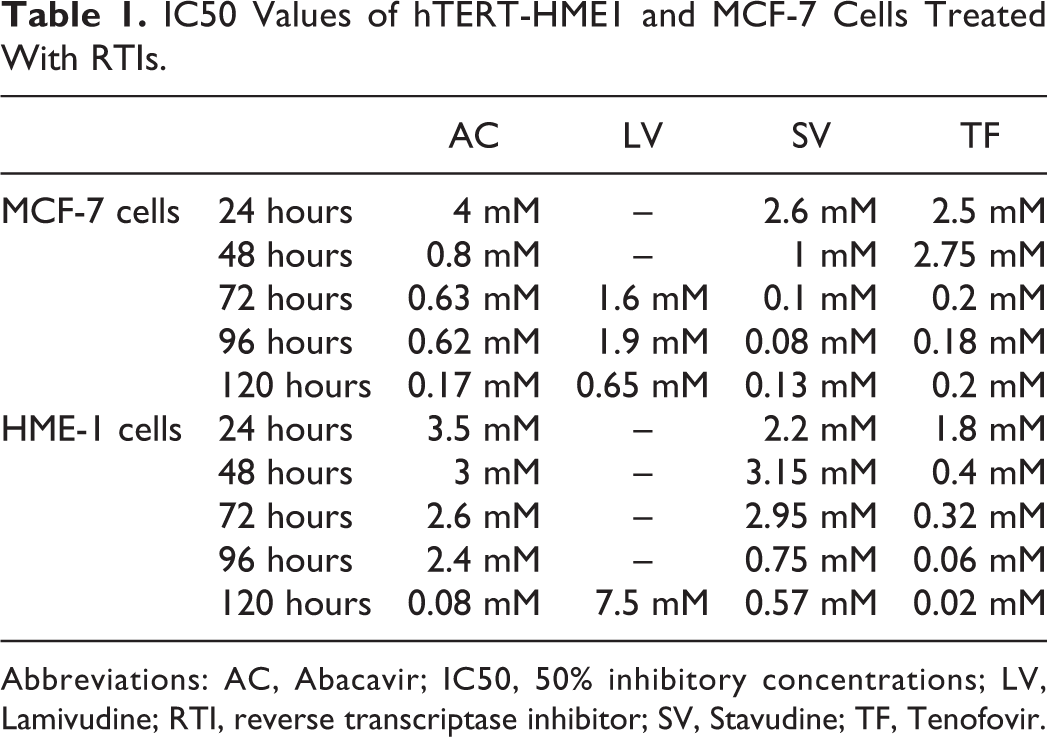
Abbreviations: AC, Abacavir; IC50, 50% inhibitory concentrations; LV, Lamivudine; RTI, reverse transcriptase inhibitor; SV, Stavudine; TF, Tenofovir.
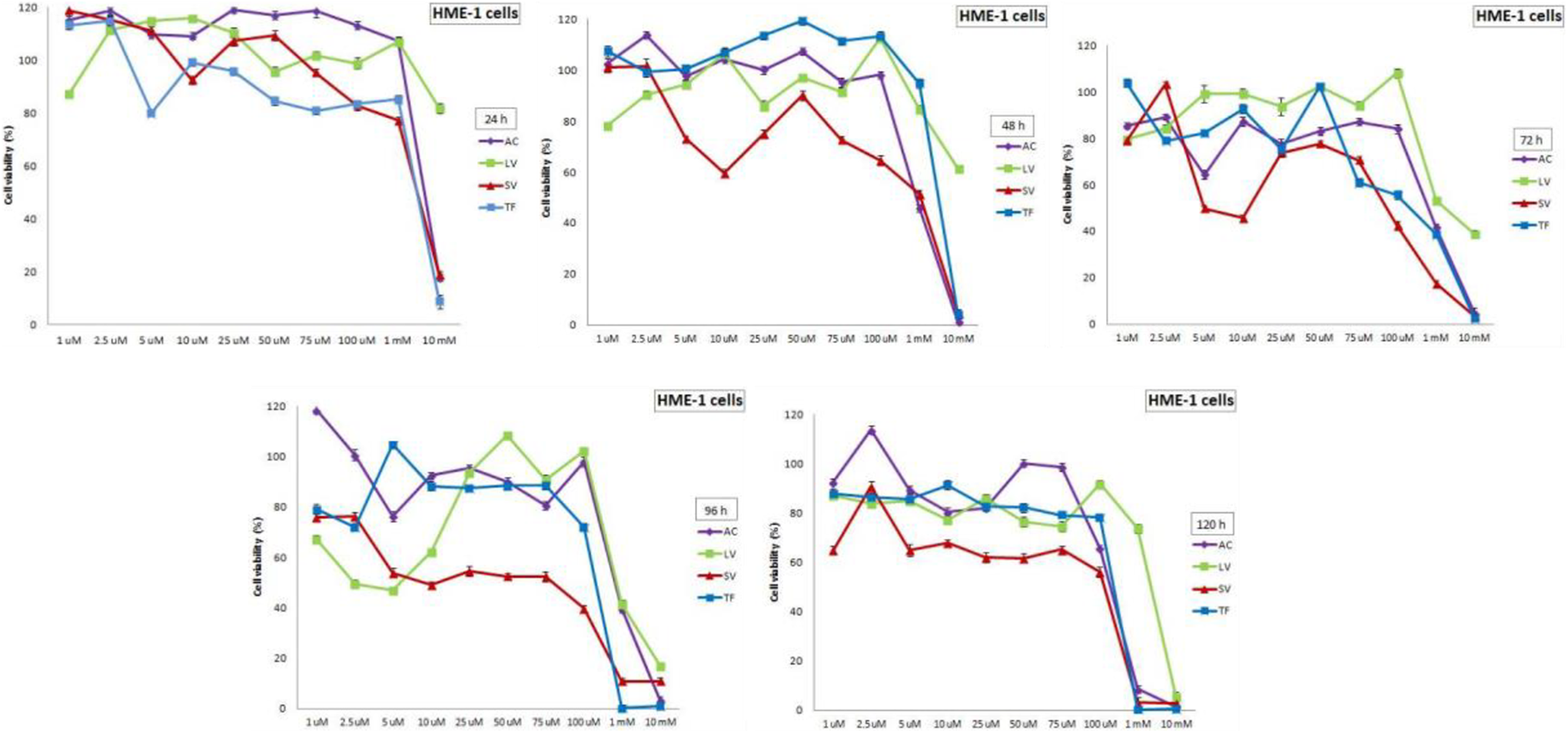
Effects of RTIs on cell viability of hTERT-HME1 cells after 24, 48, 72, 96, and 120 hours of treatments. RTIs showed stronger inhibitory effects at the higher concentrations in hTERT-HME1 cells. After 120-hour treatment, the cell viability values were below 50% at the 2 highest concentrations (1 and 10 mM) of AC, SV, and TF and the highest concentration of LV (10 mM). Data are expressed as mean ± SE for n = 3. AC indicates Abacavir; LV, Lamivudine; RTI, reverse transcriptase inhibitor; SV, Stavudine; TF, Tenofovir.
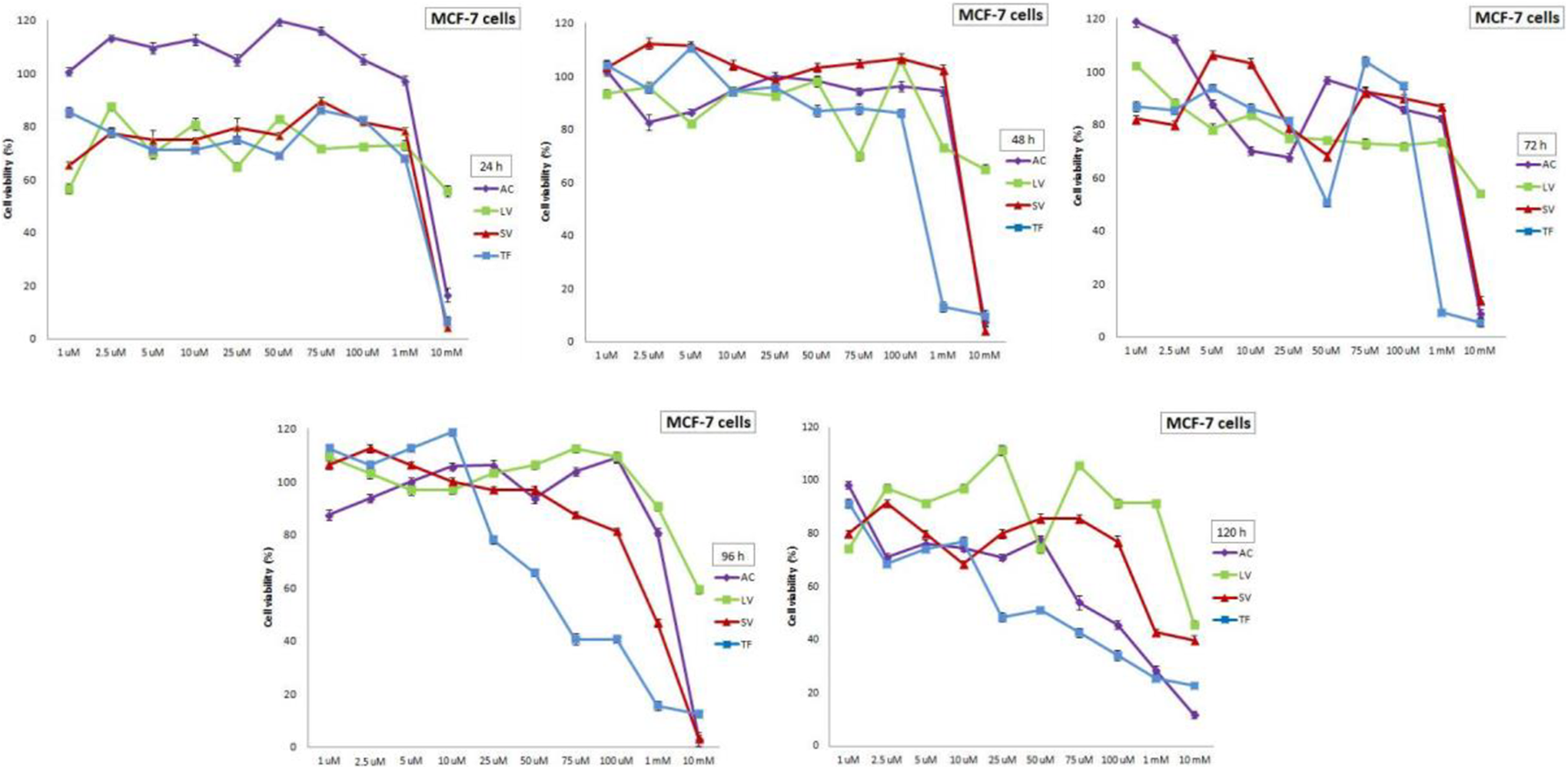
Effects of RTIs on cell viability of MCF-7 cells after 24, 48, 72, 96, and 120 hours of treatments. AC, SV, and TF showed stronger inhibitory effects at the higher concentrations in MCF-7 cells. After 120-hour treatment, the cell viability values were below 50% at the 3 highest concentrations (0.1, 1, and 10 mM) of AC, at the highest concentration (10 mM) of LV, at the 2 highest concentrations (1 and 10 mM) of SV, and the 4 highest concentrations (0.075, 0.1, 1, and 10 mM) of TF. Data are expressed as mean ± SE for n = 3. AC indicates Abacavir; LV, Lamivudine; RTI, reverse transcriptase inhibitor; SV, Stavudine; TF, Tenofovir.
As shown in Figure 2, the highest concentration (10 mM) of AC, SV, and TF reduced the cell viability greater than 50% at 24-hour treatment in MCF-7 cells. The cell viability below 50% was found only at the highest concentration (10 mM) for only 120-hour treatment in MCF-7 cells treated with LV. After 120-hour treatment, the cell viability values were below 50% at the 3 highest concentrations (0.1, 1, and 10 mM) of AC, at the highest concentration (10 mM) of LV, at the 2 highest concentrations (1 and 10 mM) of SV, and the 4 highest concentrations (0.075, 0.1, 1, and 10 mM) of TF in MCF-7 cells. These results indicated that AC, SV, and TF have stronger cytotoxic effects on MCF-7 cells than in hTERT-HME1 cells.
HTERT-HME1 and MCF-7 cells were also treated with both RTIs (0.1, 1, and 10 mM) and PAC (0.05 µM) to determine their effects when given together. The combined treatment decreased the cell viability of both HTERT-HME1 cells and MCF-7 (Figures 3 and 4). The highest cell viability values were 91.56%, 42.31%, and 16.01% in HTERT-HME1 cells and 41.35%, 32.11%, and 27.7% in MCF-7 cells 24, 48, and 72 hours, respectively.
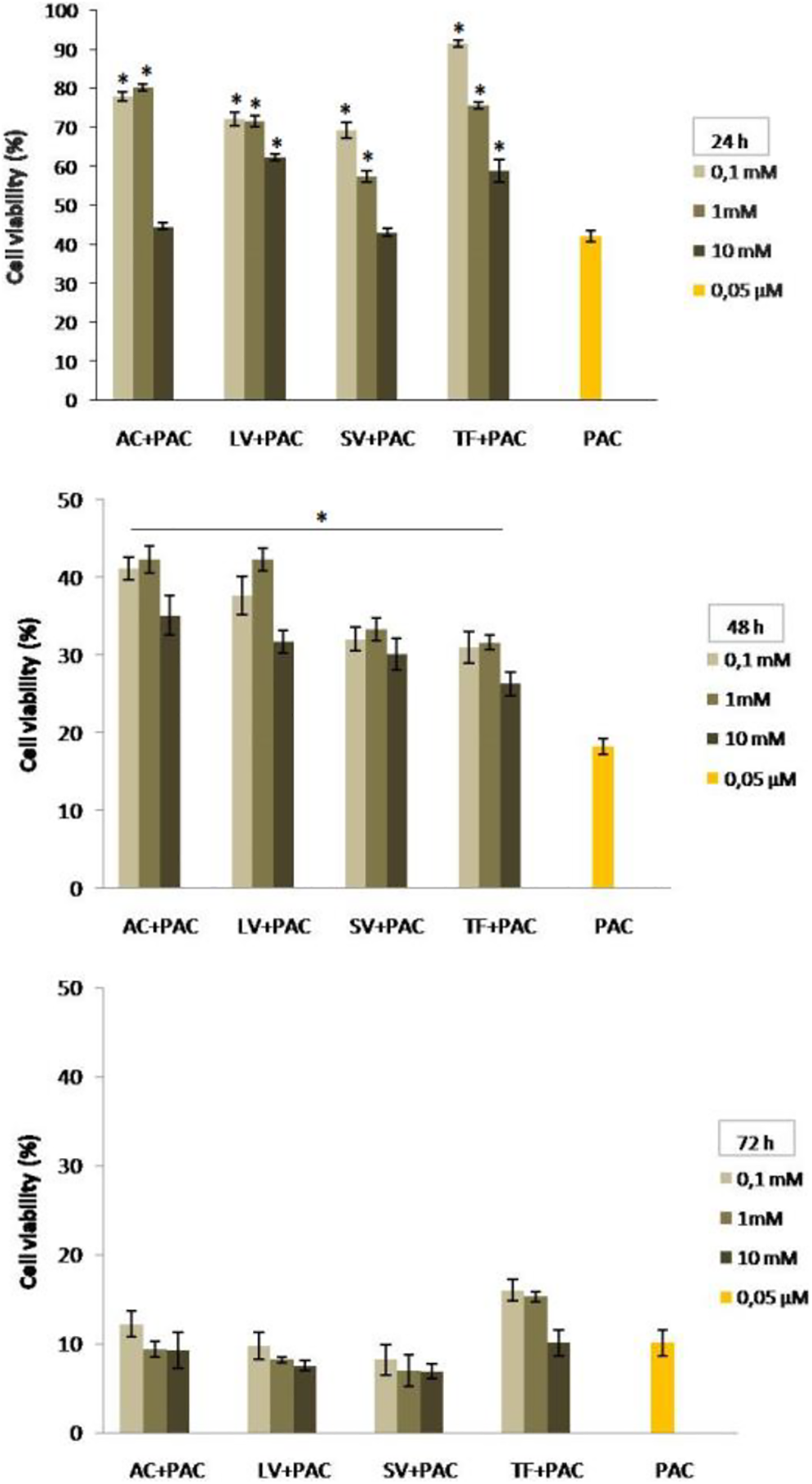
The combined effects of RTIs and PAC on cell viability of hTERT-HME1 cells after 24, 48, and 72 hours of treatments. Although the cell viability values in cultures treated with the combination of RTIs and PAC were significantly higher than PAC alone for 48 hours and 24 hours except for the highest concentration of AC and SV (P < 0.05), there were not found any significant increases for 72-hour treatments compared to PAC alone. Data are expressed as mean ± SE for n = 3. *significant against PAC at P < 0.05. AC indicates Abacavir; PAC, paclitaxel; RTI, reverse transcriptase inhibitor; SV, Stavudine.
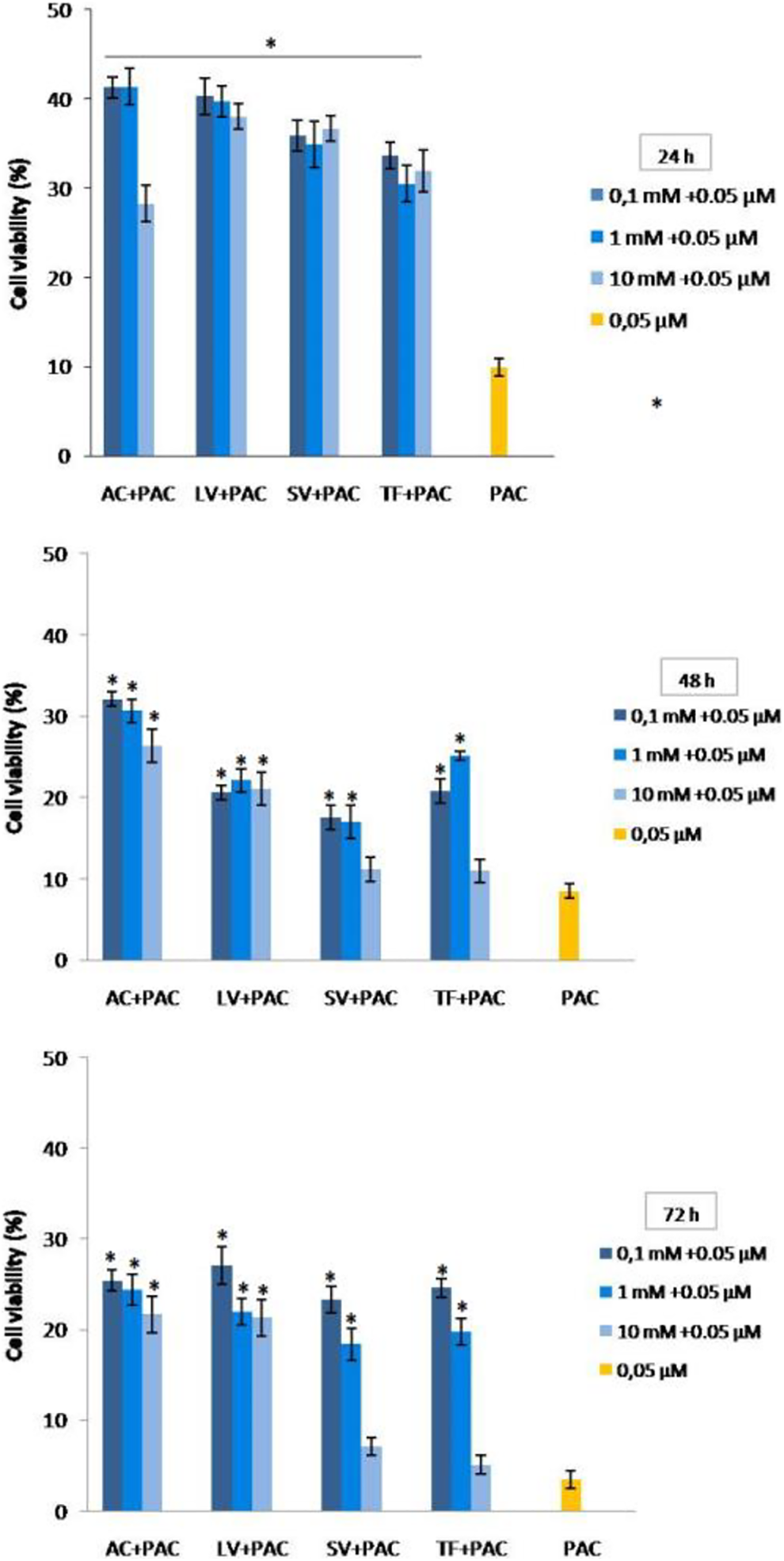
The combined effects of RTIs and PAC on cell viability of MCF-7 cells after 24, 48, and 72 hours of treatments. The cell viability values in cultures treated with the combination of RTIs and PAC were significantly higher than PAC alone in MCF-7 cells except for the highest concentration of TF and SV for 48 and 72 hours. Data are expressed as mean ± SE for n = 3. *significant against PAC at P < 0.05. PAC indicates paclitaxel; RTI, reverse transcriptase inhibitor; SV, Stavudine; TF, Tenofovir.
The lowest cell viability values were found in the combination of SV and PAC for 24 hours and 72 hours (43.15% and 6.91%) but in the combination of TF and PAC (26.25%) for 48 hours in HTERT-HME1 cells. Cell viability was also reduced for AC and PAC relative to PAC alone at 10 mM for 24 hours and at all concentrations at 72 hours. The percent survival values of HTERT-HME1 cells treated with PAC were 42.12, 18.20, and 10.10 for 24, 48, and 72 hours, respectively. All viability values in the combination of RTIs and PAC were higher than PAC in 24 and 48 hours, whereas the percent survival values of the combined treatment of LV+PAC and SV+PAC at all concentrations and AC+PAC at higher concentrations (0.1 and 1 mM) were lower than PAC in 72 hours in HTERT-HME1 cells. Although the cell viability values in cultures treated with the combination of RTIs and PAC were significantly higher than PAC alone for 48 hours and 24 hours with the exception of the highest concentration of AC and SV (P < 0.05), there were not any significant increases for 72-hour treatments compared to PAC alone. In other words, the cytotoxicity of the co-treatment of some of RTIs and PAC was greater than that of PAC for 72 hours in HTERT-HME1 cells (Figure 3).
The lowest cell viability values were found in the treatment of AC+PAC (28.25%) for 24 hours, TF+PAC (10.96% and 5.10%) for 48 and 72 hours. The percent survival values of MCF-7 cells treated with PAC were 9.88, 8.48, and 3.46 for 24, 48, and 72 hours, respectively. The cell viability values in cultures treated with the combination of RTIs and PAC were significantly higher than PAC alone in MCF-7 cells except for the highest concentration of TF and SV for 48 and 72 hours (P < 0.05; Figure 4).
Acridine Orange/EtBr Staining
A fluorescence micrograph of AO/EB-stained MCF-7 cells is illustrated in Figure 5. The viable cells with normal nuclear chromatin (green; Figure 5A), early apoptotic cells (green, yellow, or orange; Figure 5B), late apoptotic cells (orange or red) containing fragmented nuclear chromatin (orange-red; Figure 5C) and necrotic cells (red; Figure 5D) were detected after staining.
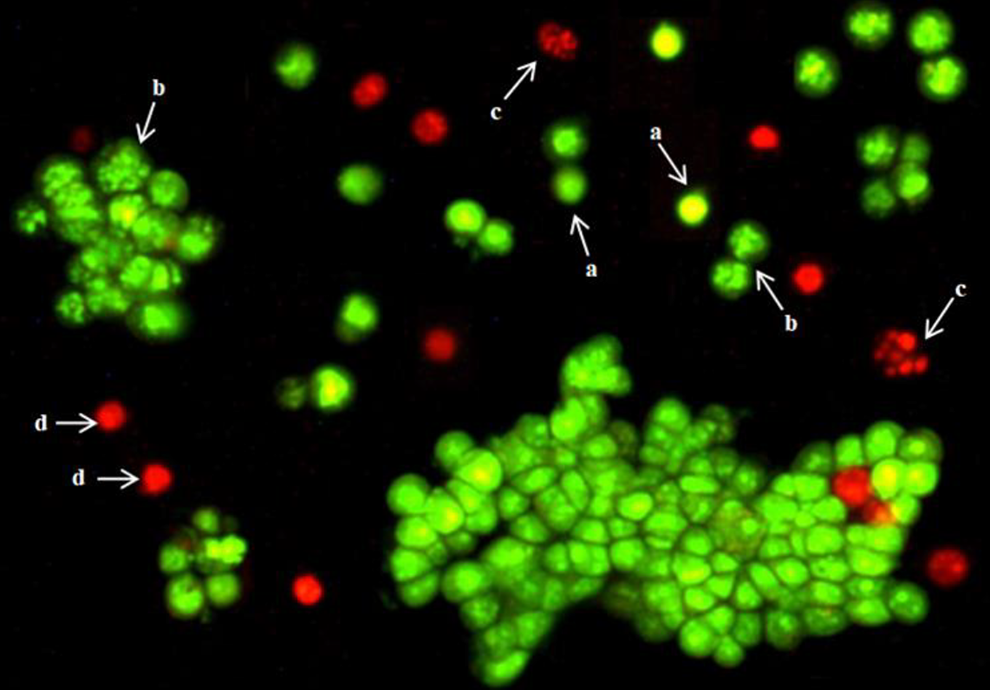
MCF-7 cells stained with AO/EB under a fluorescent microscope. a: Viable cells, b: early apoptotic cells, c: apoptotic cells, d: necrotic cells. AO indicates acridine orange.
Table 2 shows apoptosis rates of MCF-7 and HTERT-HME1 cells treated with RTIs and/or PAC. Paclitaxel treatments for 24, 48, and 72 hours significantly increased the percentage of apoptotic cells in both MCF-7 and HTERT-HME1 cells compared to their own control groups (P < 0.05). Although the values of apoptosis in MCF-7 cells treated with PAC were higher than the values in HME1 cells, only the rate of apoptosis of 24 hours treatment in MCF-7 cells was statistically significant compared to HME1 cells (P < 0.05). All treatments of RTIs alone and in combination with PAC significantly increased (P < 0.05) the percentage of apoptotic cells compared to control group, except LV treatments alone in MCF-7 cells for 24, 48, and 72 hours. Apoptotic rates in all LV treatments were very similar to the control. Although apoptosis rates at the highest concentrations of RTIs increased, they still were lower than PAC. However, the percentages of apoptotic cells in the treatment of AC and SV in combination with PAC for 48 and 72 hours were higher than PAC. Significant apoptosis values (P < 0.05) were also found in HTERT-HME1 cells treated with RTIs with or without PAC. However, the combined treatment of RTIs and PAC caused high rates of cell death in MCF-7 and low rates of cell death in HTERT-HME1 by apoptosis.
Apoptosis Rates of MCF-7 and HTERT-HME1 Cells Treated With RTIs and/or PAC.
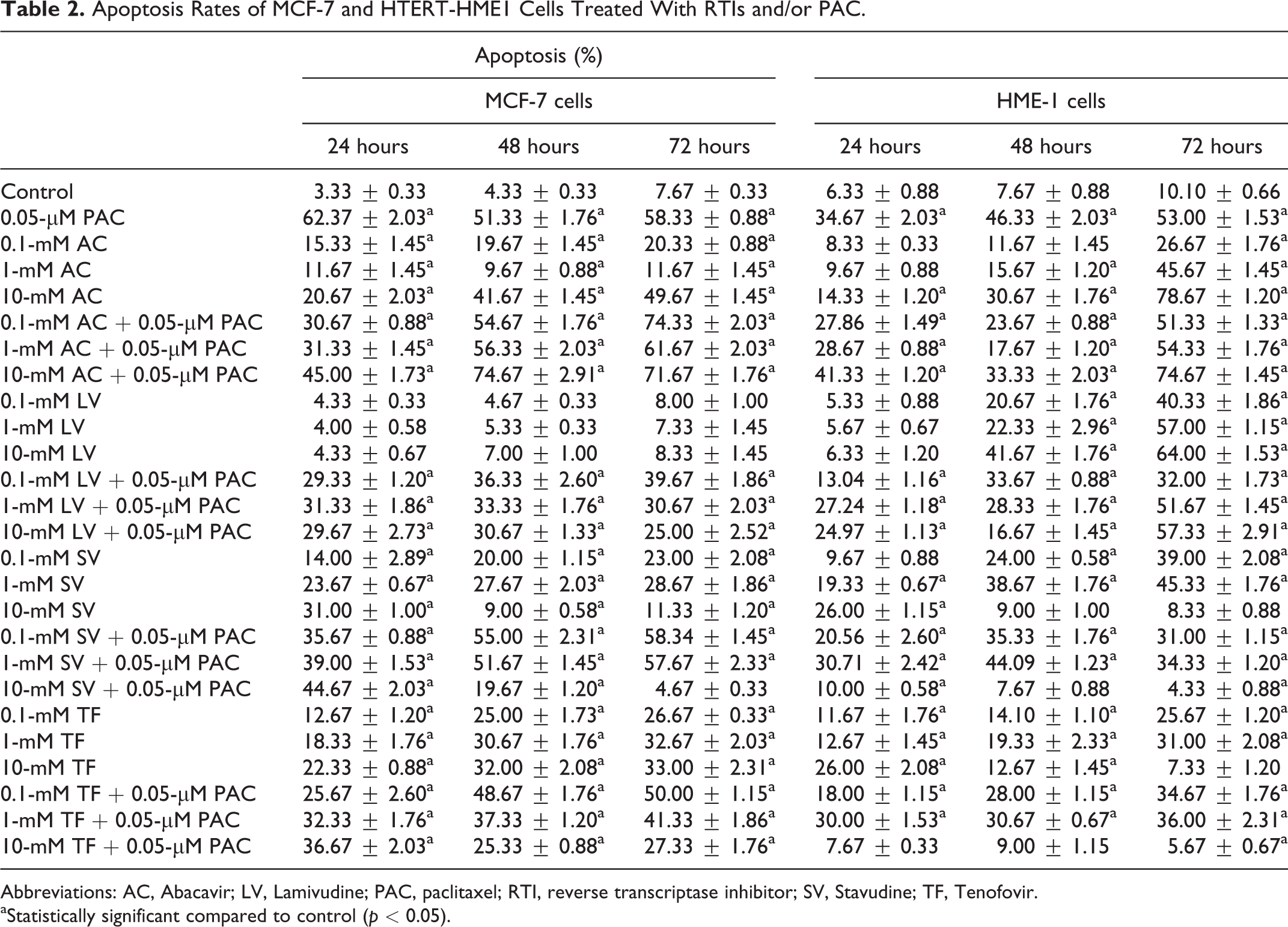
Abbreviations: AC, Abacavir; LV, Lamivudine; PAC, paclitaxel; RTI, reverse transcriptase inhibitor; SV, Stavudine; TF, Tenofovir.
aStatistically significant compared to control (p < 0.05).
Acridine orange/EtBr staining results showed that 10-mM AC for 48 hours, and 72 hours and 0.1-mM AC+PAC, 0.1-mM SV+PAC, and 10-mM SV+PAC for all treatment times significantly increased the apoptosis in MCF-7 cells than in HTERT-HME1 cells. The highest rate of apoptosis in MCF-7 cells treated with RTIs alone was 10-mM SV for 24 hours, whereas it was 10-mM AC for 48 and 72 hours. The concentration of 10-mM AC was the most effective in terms of impacting cancer cell apoptosis for all treatment times. In addition, when RTIs were given to MCF-7 cells in combination with PAC, apoptosis rates were increased compared to their treatments alone. When normal cells (HTERT-HME1) were compared with cancer cells (MCF-7), there were some significant increases in the level of apoptosis in normal cells treated with 1 and 10-mM AC and 0.1-mM SV for 72 hours and 1-mM SV and all LV concentrations for 48 and 72 hours (P < 0.05). These results indicate that LV may not an effective drug to induce apoptosis of cancer cells.
Necrosis rates in both cell lines were significantly higher in some treatment groups relative to controls (P < 0.05). Treatment of the cells with LV, SV, or TF alone and in combination with PAC showed stronger necrotic effects in HTERT-HME1 cells than MCF-7, especially in the highest concentration (Table 3).
Necrosis Rates of MCF-7 and HTERT-HME1 Cells Treated With RTIs and/or PAC.
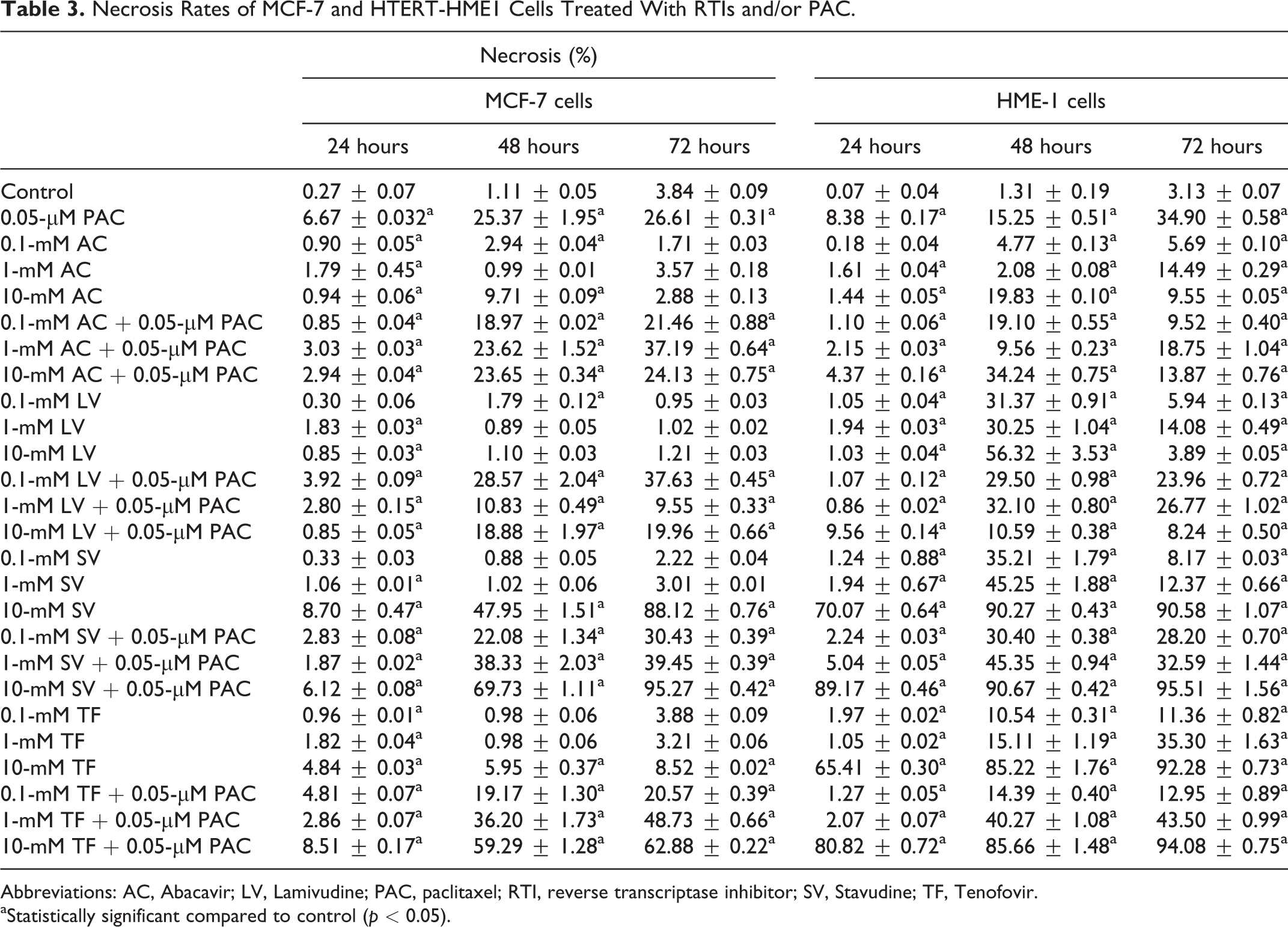
Abbreviations: AC, Abacavir; LV, Lamivudine; PAC, paclitaxel; RTI, reverse transcriptase inhibitor; SV, Stavudine; TF, Tenofovir.
aStatistically significant compared to control (p < 0.05).
Cell Migration Assay
The results of the wound healing assay are presented in Figure 6. In this assay, a large wound area shows that the tested agent can inhibit the migration of carcinoma cells. When cell proliferation was inhibited by mitomycin C, cell migration fell to 33% after 72 hours in the control group. Paclitaxel (0.05 µM) significantly decreased the cell migration of MCF-7 cells with 19.74, 16.61, 22.61, and 21.18% migration rates after 12, 24, 48, and 72 hours of treatments (P < 0.05). Treatment of cells with AC, LV, SV, or TF (0.1, 1, and 10 mM) with or without PAC altered the cell migration rates. However, these alterations were only found statistically significant in the cells treated with AC or SV when compared to the control group (P < 0.05). A 10-mM AC for 6, 12, and 48 hours and 0.1-mM AC+0.05-µM PAC for all treatment times significantly inhibited the migration rates compared to the control group (P < 0.05). Of total, 18.32%, 15.22%, 16.00%, 13.60%, and 13.19% of the wound areas remained unfilled in cells treated with 0.1-mM AC+0.05-µM PAC at 6, 12, 24, 48, and 72 hours, respectively. Significant migration rates were also found in cells co-treated with 0.1 and 10-mM SV with 0.05-µM PAC for 48 (16.06% and 18.85%, respectively) and 72 hours (18.19% and 18.07%, respectively) compared to the control group (P < 0.05). Ten-mM AC, 0.1-mM AC+PAC, 0.1-mM SV+PAC, and 10-mM SV+PAC treatments prevented the migration capacity of MCF-7 cells more than PAC. Our results showed that similar to the cytotoxicity experiment, 10-mM AC and co-treatment of AC (0.1 mM) or SV (0.1 mM and 10 mM) with PAC can significantly inhibit the migration ability of MCF-7 cells.
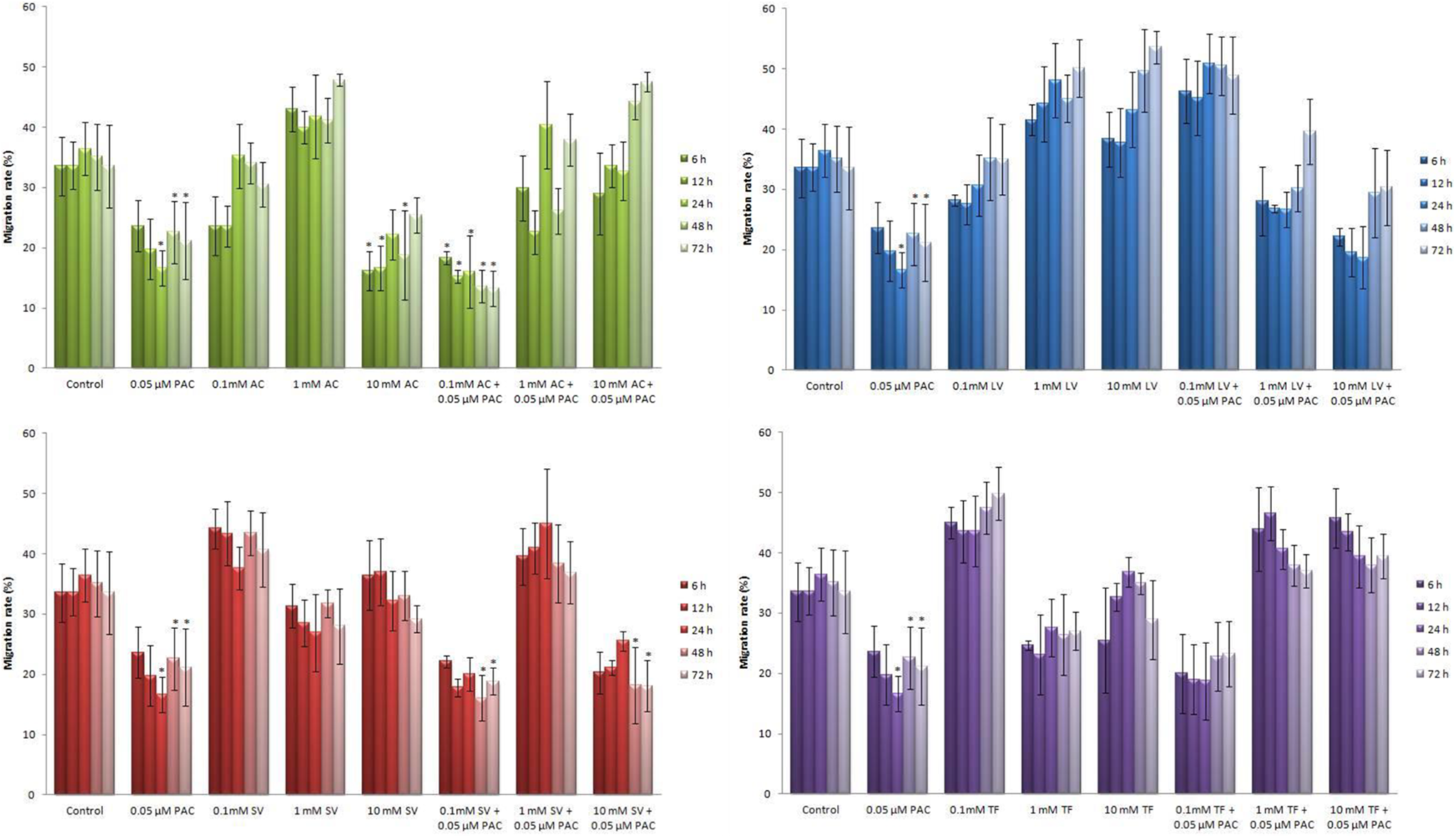
Migration rates of MCF-7 cells treated with RTIs and/or PAC. Although treatment of cells with AC, LV, SV, or TF (0.1, 1, and 10 mM) with or without PAC altered the cell migration rates, 10 mM AC, and co-treatment of AC (0.1 mM), or SV (0.1 mM and 10 mM) with PAC can significantly inhibit the migration ability of MCF-7 cells. Data are expressed as mean ± SE for n = 3. *significant against control at P < 0.05. AC indicates Abacavir; LV, Lamivudine; PAC, paclitaxel; RTI, reverse transcriptase inhibitor; SV, Stavudine; TF, Tenofovir.
Discussion
Several studies have shown that some RTIs, widely used in the treatment of HIV, may be a target in cancer therapy since they can inhibit the telomerase activity in human cancers. 7 Zidovudine inhibited the growth, colony formation in soft agar, and telomerase activity of breast cancer cell lines, MCF-7 and T47D, and Jurkat cells. It also inhibited the growth of normal breast cell line, MCF-1OF, although it required a much higher concentration. 11 Some RTIs such as azydothymidine (AZT), dideoxyinosine (ddI), and AZT-5’triphosphate (AZT-TP) reduced the proliferation with increasing concentrations in human MCAS ovarian mucinous cystadenocarcinoma cells and HEC-1 uterine endometrial carcinoma cells. The ddI and AZT-TP reduced telomerase activity and shortened the length of the telomere in HEC-1cells. The antitumor effects of cisplatin were enhanced in the presence of ddI in HEC-1 cells. Co-treatment of cisplatin and ddI caused a marked reduction in cell proliferation, the appearance of morphological changes, and senescent-like cells. 12 Zidovudine, nucleoside RTI, showed significant growth inhibitory effects in human liver carcinoma cell line HepG2. 13 Abacavir decreases cell proliferation and induces differentiation of human medulloblastoma cell lines Daoy and D283-MED through the downregulation of telomerase activity. 6 Abacavir significantly reduced cell growth, migration and invasion, and induced senescence and cell death in prostate cancer cells. Expression of some genes involving multiple cellular pathways was significantly altered after AC treatment. 7 It has been found that telomerase inhibitors target cancer stem cell populations in multiple myeloma, prostate, brain, breast, and pancreatic cancer. 8 Combination of emtricitabine (a cytidine analogue) and TF can induce DNA damage and cell cycle arrest in some human cancer cells such as SKOV3, OVCAR3, MCF7, and HeLa. They also enhanced the cytotoxic effect of doxorubicin. 14 Some genes associated with cell death, cell survival, cell cycle, cell growth, cell proliferation, replication, recombination, and repair of DNA differentially expressed in HepG2 cells treated with zidovudine compared to the immortalized human liver cell line THLE2. Therefore, HepG2 cells are more sensitive than THLE2 cells to the toxicity of zidovudine. 15 Some newly synthesized derivatives of zidovudine showed anticancer activities on MCF-7 cells. 16 Tenofovir and AC significantly inhibited the telomerase activity at therapeutic concentrations in peripheral blood mononuclear cells. 17 The results of previous studies support the results of our study because AC, LV, SV, and TF significantly reduced the cell viability and induced apoptosis in MCF-7. MCF-7 cells were also more sensitive than HTERT-HME1 cells to the effects of RTIs.
It has been stated that combining telomerase inhibitors with current therapies might be more effective in the treatment of breast cancer or prevent tumor recurrences. 2,18 Combining telomerase inhibition with common chemotherapeutic drugs may be more effective in killing tumor cells than telomerase inhibition alone in breast cancer cells. However, few studies have analyzed the effects of telomerase inhibition in combination with other anticancer drugs in breast cancer cells. 1 The breast cancer cell lines YCC-B2 and MCF-7 cells whose telomerase activity was inhibited were more sensitive than the controls to different anticancer drugs. The effects of combination approaches were often found cell line and drug type-specific. 1 The obtained results in the present study are an agreement with the above findings because when AC, LV, SV, or TF were given together with PAC, their cytotoxic and apoptotic effects were further enhanced. Based on all these results, it can be said that the combination of telomerase inhibition and anticancer drugs may be more effective in inducing cell death of breast cancer cells.
It has been stated that overexpression of hTERT may induce cell adhesion, migration, and transformation by altering the expression of some genes. 19 Co-treatment of morin (a flavonoid) with MST 312 (a telomerase inhibitor) inhibited the wound healing of MDA-MB 231 cells. 20 TMPyP4, a telomerase inhibitor, significantly inhibited the migration and adhesion of some breast cancer cells (MCF-7 and MDA-MB-231). 21 Similar to the results of these studies, treatment of AC and co-treatment of AC+PAC or SV+PAC significantly inhibited the migration of MCF-7 cells in our study.
Although chemotherapy is considered to be the most efficacious approach in cancer therapy, many anticancer drugs do not perform as expected due to the emergence of resistance. Termination of treatment commonly occurs due to indiscriminate damage to normal tissues and cancer cells. The development of novel anticancer drugs and strategies to improve treatment is critical. 22 Combination therapy is defined as 2 or more therapeutic agents were administered together, which is considered as an optimal approach to cancer therapy as multiple pathways may be targeted. Although, the monotherapy approach is still very common for many different types of cancer, it is generally considered less effective than using a combination. Traditional monotherapeutic techniques are nonselective and can destroy both healthy and cancerous cells. 23 The results from these studies demonstrate the utilization of combination therapy. We demonstrated that breast cancer cells were often more sensitive of RTIs except for TF because of lower IC50 values in MCF-7 cells compared to hTERT-HME1 cells. In regard to apoptosis, AC and SV increased the efficacy of PAC in MCF-7 cells as the rates were significantly higher than PAC alone for 48 and 72 hours of treatments. Conversely, combination therapy significantly reduced apoptosis rates in hTERT-HME1 cells in almost all treatments compared to PAC alone. These data indicate that AC and SV can increase the efficacy of PAC in breast cancer cells with the lowest toxic or side effects on normal breast cells. AC, 0.1-mM AC+PAC, 0.1-mM SV+PAC, and 10-mM SV+PAC treatments prevented the migration capacity of MCF-7 cells more than PAC. Our results indicated that similar to the cytotoxicity experiment, 10-mM AC and co-treatment of AC (0.1 mM) or SV (0.1 mM and 10 mM) with PAC can significantly inhibit the migration ability of MCF-7 cells. Furthermore, when AC was used alone or in combination with PAC, the migration of MCF-7 cells was inhibited. A similar inhibitory effect was also observed following SV+PAC treatment. These treatments significantly prevented the cell migration relative to PAC. Based on these data, the use of these drugs in combination with PAC in the treatment of breast cancer may be beneficial in terms of killing cancer cells at high rate and protect healthy cells.
The use of drugs for oncology applications is another therapeutic approach in cancer research, where current pharmaceutical agents primarily used for noncancerous diseases are applied for cancer treatment. This approach decreases the cost of bringing the drug to market as it has been approved. 23,24 Reverse transcriptase inhibitors are already used to treat HIV infection and are FDA-approved. The results of the present study described above show that they may example of old non-oncological drugs that can be repurposed as anticancer agents.
In conclusion, we confirmed that AC and SV which are already used for different diseases can be potentially used alone or in combination with chemotherapeutic agents in breast cancer because they may induce cytotoxicity and apoptosis and also inhibit migration and adhesion that can limit the aggressive potential of cancer cells. Therefore, a combination strategy based on telomerase inhibition and anticancer drugs may be more effective in the treatment of breast cancers.
Footnotes
Author Contributions
Z. Atlı Şekeroğlu contributed to conception and design; acquisition, analysis, and interpretation of data; drafted the manuscript; and critically revised the manuscript. V. Şekeroğlu contributed to acquisition, analysis, and interpretation of data; manuscript writing; and critically revised the manuscript. N. Küçük contributed to acquisition, analysis, and interpretation of data and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by the Scientific Research Funding of Ordu University (Turkey) (Project No: AR-1668).
