Abstract
Sensorineural hearing loss, ataxia, pyramidal signs, and vestibular deficits characterize superficial siderosis of the central nervous system. This study investigated changes in vestibular function, free radical formation, and phosphorylated cJun expression in the vestibular end organs after middle ear treatment with a ferric chloride (FeCl3) solution. A single injection of 70% FeCl3 solution into the unilateral middle ear cavity caused static vestibular symptoms, such as spontaneous nystagmus and head tilt. Asymmetric expression of c-Fos protein was observed in the bilateral vestibular nuclei and prepositus hypoglossal nuclei within 6 h after injection. Histopathologic examinations revealed partial hair cell loss, degeneration of the supporting stroma, and terminal deoxynucleotidyl transferase dUTP nick end labeling-positive cells in the neuroepithelial layer of the crista ampullaris in FeCl3-treated animals. 5-(And-6)-chloromethyl-2′,7′-dichlorodihydrofluorescein diacetate, acetyl ester and diaminofluorescein–2 diacetate fluorescence and immunoreactivity for nitrotyrosine increased markedly in the sensory neuroepithelial layer and nerve bundles of the crista ampullaris after 2 h. Strong immunoreactivity for phospho-cJun and cJun was observed in the type I hair cells of the crista ampullaris 120 h after injection. Thus, a single short-term treatment with a high concentration of FeCl3 in the unilateral middle ear cavity can induce activation of intracellular signals for cJun protein and oxidative stress through the formation of reactive oxygen species and nitric oxide in vestibular sensory receptors, resulting in vestibular dysfunction. These results suggest that activation of intracellular signals for cJun protein and oxidative stress may be a key component of the pathogenesis of vestibular deficits in patients with superficial siderosis.
Keywords
Introduction
Oxidative stress is the cellular consequence of overproduction of free radicals, such as reactive oxygen species (ROS) and reactive nitrogen species (RNS), which are by-products of oxidative metabolic processes. Excessive ROS/RNS results in oxidative damage to lipids, proteins, and DNA, which then leads to dysregulation of mitochondrial functions and gene expression, finally causing either apoptosis or necrosis. Oxidative stress is thus an important intracellular mechanism underlying the development or progression of various diseases in humans. 1,2
It has been demonstrated that oxidative stress is involved in dysfunction of the inner ear, such as ischemia- or noise-induced hearing loss, ototoxicity, presbycusis, and even vestibular neuronitis. 3 –6 Excessive ROS/RNS and inducible nitric oxide synthase are produced in vestibular sensory cells after exposure to ototoxic aminoglycosides, lipopolysaccharides, or glutamate. 7 –10 Some studies of the direct effects of an oxidant on vestibular functions or vestibular hair cells have been reported. Five daily applications of hydrogen peroxide (H2O2) into the middle ear fail to elicit vestibular evoked potentials in response to linear acceleration stimuli in less than 50% of the animals tested. 11 Previous in vitro studies have demonstrated that direct application of H2O2 may affect the morphology of cochlear outer and inner hair cells in the guinea pig. Direct application of H2O2 also reduces cell viability, concomitant with morphologic changes, such as swelling, bleb formation, and neck shortening in isolated vestibular hair cells. 12,13
Because ferrous (Fe2+) and ferric (Fe3+) ions produce hydroxyl radicals in the cell via the Fenton and Haber−Weis reactions, 14 an iron solution is frequently used to induce oxidative stress and cell death in many experimental studies. 15,16 Direct application of ferric chloride (FeCl3) solution to vessels causes thrombus formation through endothelial injury, leading to local ischemic injury. 17 Binding of aminoglycosides to iron is believed to potentiate oxidative stress in sensory hair cells of the inner ear. 18
Superficial siderosis of the central nervous system is caused by repeated or continued bleeding into the subarachnoid space, resulting in the deposition of iron from hemosiderin onto the surface of the brain, which is characterized by sensorineural hearing loss, ataxia, pyramidal signs, and vestibular deficits. Clinical symptoms of superficial siderosis are caused by gliosis, neuronal loss, and demyelination resulting from the deposition of hemosiderin. 19 –22 However, the pathogenesis of vestibular deficits due to superficial siderosis is not clear. A few studies have described that the possible factors involved in vestibular deficits include atrophy of the vestibular nerve, loss of vestibular hair cells, and decreased peripheral blood flow in the inner ear. 22 –24
Therefore, the present study investigated the effect of middle ear application of FeCl3 solution on vestibular functions and oxidative stress in the inner ear by observing production of free radicals and expression of nitrotyrosine (NT). NT is formed by peroxynitrite-mediated nitration of protein tyrosine residues and is a biomarker of nitrosensitive stress in various pathological conditions. 25 We also assessed the expression of cJun and phosphorylated cJun (p-cJun), which participate in cell death 26 in the vestibular end organs of rats.
Methods
Animals and middle ear treatment with FeCl3 solution
Sprague−Dawley male rats weighing 250−350 g were used and were randomly divided into two groups: the FeCl3-treated group and the sham group. All animals were anesthetized with 4% isoflurane and fixed on a surgical table. Under surgical microscopic examination, the left middle ear cavity was opened by rupturing the tympanic membrane using a surgical forceps through the external auditory meatus. The stapes and incus were removed to expose the oval window. Sterile cotton saturated with about 50 µl of 70% FeCl3 dissolved in normal saline was applied to the oval window. After 10 min, the cotton was removed and the animals were allowed to recover from anesthesia. In some animals (n = 6), a 7% FeCl3 solution was applied to the middle ear to determine the dose effect on static vestibular symptoms. As a sham operation, the same surgical procedures were performed as in FeCl3-treated animals, but sterile cotton saturated with normal saline only was applied to the oval window. All animal protocols and procedures described were performed in accordance with the rules and regulations of the Animal Research Committee of the Wonkwang University.
Evaluation of spontaneous nystagmus
Recovery of spontaneous nystagmus (SN), a static vestibular symptom caused by imbalance of bilateral vestibular functions, was monitored over time after middle ear treatment with FeCl3 solution. Eye movements were recorded using a video camera with a zoom lens (Samsung, Korea) at 1, 2, 6, 12, and 24 h after treatment with FeCl3. The frequency of SN per 10 s was counted three times by replaying the video tape.
Hematoxylin–eosin staining
Five days after surgery, histological examination was performed to observe structural changes to the vestibular end organs in FeCL3-treated animals (n = 3) and sham animals (n = 3). After cardiac perfusion with 4% paraformaldehyde, the temporal bones were removed from the skull and kept in a decalcification solution (Sigma-Aldrich, St Louis, Missouri, USA) for 5 days. The tissues were then rinsed through a graded alcohol series, washed with xylene, and embedded in paraffin for sectioning. Sections (7 µm thick) were mounted on subbed slides, heated briefly, dried overnight, and stained with hematoxylin and eosin. Histological changes of the vestibular end organs were observed by light microscopy (Olympus, Tokyo, Japan).
Immunohistochemistry
Animals were killed at 0 (n = 6), 2 (n = 6), 6 (n = 6), or 24 h (n = 6) after middle ear treatment with 70% FeCl3 solution to determine c-Fos expression in the vestibular nuclei. Experimental animals were deeply anesthetized with urethane (10 mg/kg), transcardially perfused, fixed in 4% paraformaldehyde dissolved in 0.1 M phosphate buffer (PB), and decapitated. The brains were then removed, postfixed, rinsed in PB, and immersed in 30% sucrose solution for 1−2 days at 4°C. The tissue was sectioned at 40 µm on a freezing microtome (Leica, Wetzlar, Germany), incubated for 30 min with 6% H2O2, rinsed twice for 10 min each with 0.1 M phosphate-buffered saline (PBS) containing 5% dry milk, and incubated with 0.8% Triton X-100 dissolved in 0.1 M PBS containing 0.5% bovine serum albumin (PBS + BSA). After a brief wash, tissue was incubated overnight at room temperature with an antibody against polyclonal rabbit c-Fos (1:1000; Cell Signaling Technology, Danvers, Massachusetts, USA) in PBS + BSA. The following day, the tissue was rinsed with PBS + BSA and incubated with a biotinylated secondary antibody (goat anti-rabbit; DAKO, Glostrup, Denmark) using the ABC Elite Kit (Vector Lab, Burlingame, California, USA). The neurons expressing c-Fos protein were visualized by incubating tissue with 0.05% diaminobenzidine (DAB) hydrochloric acid and 0.003% H2O2. After the DAB reaction, the tissue was rinsed with 0.1 M PB, mounted on gel-coated slides, air-dried, dehydrated, xylene cleared, coverslipped with Permount (Fisher Scientific, Hampton, New Hampshire, USA), and analyzed by bright-field microscopy (Olympus).
Immunofluorescent staining
Animals were killed at 0 (n = 3), 24 (n = 3), or 120 h (n = 3) after middle ear treatment with 70% FeCl3 solution to determine the expression of cJun and p-cJun in the vestibular end organs. After perfusion fixation, vestibular end organs were isolated from the temporal bone under a surgical microscope. The isolated sample was embedded in a 1.5% gelatin solution and fixed again for 3 h. The gelatin specimen was cryoprotected in a 30% sucrose solution overnight at 4°C. Forty-micrometer sections were cut using a freezing microtome, collected on PBS. The free-floating gelatin sections containing vestibular specimens were blocked with PBS containing 0.1% Triton X + 0.5% BSA. Sections were incubated overnight at room temperature with an antibody against polyclonal rabbit cJun (1:1000), or cJun phosphorylated at serine-73 (p-cJun; 1:1000; Cell Signaling Technology), or NT (Chemicon, Billerica, Massachusetts, USA). The following day, tissues were rinsed with PBS + BSA and incubated with Alexa Fluor 594-conjugated secondary antibody (Cell Signaling Technology) for 30 min. Sections were rinsed with 0.1 M PB, mounted on gel-coated slides, air-dried, coverslipped, and analyzed by confocal microscopy (Fluoview FV-1000; Olympus). For double-immunofluorescent staining of p-cJun and parvalbumin proteins, free-floating gelatin sections stained with p-cJun antibody were washed with PBS containing 0.1% Triton X for 30 min. Thereafter, the sections were incubated with primary antibody against parvalbumin (1:500; Chemicon) overnight at room temperature. The next day, sections were incubated with Alexa Fluor 488-conjugated secondary antibody (Cell Signaling Technology) for 30 min and mounted on gel-coated slides. Immunofluorescent staining for phalloidin, which binds specifically to F-actin, was adopted to visualize the cilia of vestibular hair cells. Free-floating gelatin sections were washed with PBS containing 0.1% Triton X for 30 min and then incubated with Alexa Fluor 594-conjugated phalloidin (Cell Signaling Technology) for 30 min. After a brief wash, sections were mounted on a slide.
Terminal deoxynucleotidyl transferase dUTP nick end labeling staining
Immunohistochemical evidence for DNA strand breaks was obtained with the terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) assay using the In Situ Cell Death Detection Kit (Roche Diagnostics, Basel, Switzerland) as per the manufacturer’s instructions. After incubation with DAB solution, sections were rinsed with 0.1 M PB, mounted on gel-coated slides, air-dried, coverslipped, and analyzed by light microscopy (Olympus).
Detection of ROS and nitric oxide
Three hours after middle ear treatment with 70% FeCl3 solution, animals (n = 3) were deeply anesthetized with urethane and immediately decapitated. Procedures for the detection of ROS and nitric oxide (NO) in the vestibular end organs were as previously described, 8 with some modifications. The temporal bones were quickly removed; washed with artificial perilymph (AP) containing 130 mM sodium chloride, 5.4 mM potassium chloride, and 1.25 mM calcium chloride:2H2O; buffered to pH 7.2 with 5.0 mM 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid; adjusted to 300 mOsm with glucose; and equilibrated with 95% O2:5% CO2. A small opening at the cochlear promontory was made to facilitate penetration of the solution into the inner ear structure. The temporal bone specimens were loaded with 10 mM diaminofluorescein–2 diacetate (DAF-2DA; Cell Signaling Technology; for detecting NO) or with 10 mM 5-(and-6)-chloromethyl-2′,7′-dichlorodihydrofluorescein diacetate, acetyl ester (CM-H2DCFDA; for detecting ROS) in AP for 1 h at 37°C in the dark. After fluorescent dye loading, the specimens were washed with AP three times for 5 min each and quickly fixed in 4% paraformaldehyde for 2 h at room temperature. Vestibular end organs were isolated from the temporal bone under a surgical microscope. The isolated preparations were embedded in a 1.5% gelatin solution, cut to 40 µm thickness using a microtome, and mounted on slides. Fluorescent signals produced by DAF-2DA or CM-H2DCFDA in vestibular end organs were analyzed using confocal microscopy (Fluoview FV-1000; Olympus)
Quantitative analysis
Dark brown-colored immunoreactive neurons expressing c-Fos protein in the medial vestibular nucleus (MVN) were captured, digitized with a digital camera (Olympus DP-70), mounted under a microscope (X-14; Olympus), and images were obtained. After uniform adjustment of the brightness and contrast in each image file, the number of immunoreactive neurons was quantified using image analysis software (Image Pro Plus, Media Cybermetics, Silver Spring, USA). Only cells that had significant levels of DAB reaction product above that of tissue background levels were counted, and cells that were lightly immunolabeled were not counted. In each animal, at least three counts per nucleus were performed, and the mean number of immunoreactive neurons was calculated.
Statistical analysis
All data obtained from quantitative data were represented as mean ± SD. Differences between groups were evaluated using the nonparametric Mann−Whitney U test. The values of p below 0.05 were considered statistically significant.
Results
Vestibular symptoms
All animals receiving single treatment with 70% FeCl3 solution into the middle ear showed SN with fast eye movement toward the side contralateral to the affected ear. SN appeared within 1 h and the frequency of SN peaked at 29.4 ± 3.9/10 s at 2 h, after which it decreased gradually over time, and finally disappeared at 24 h after 70% FeCl3 injection. Additionally, SN was also observed at a low frequency and with rapid disappearance in all animals receiving 7% FeCl3, as compared to those receiving 70% FeCl3 (Figure 1). Treatment with 70% FeCl3 induced a mild to moderate head tilt toward the ipsilateral side to the affected ear, but there was no circling movement of the body during experimental period (data not shown). Sham animals did not show any deficits in eye movement and posture.

Static vestibular symptoms after middle ear treatment with FeCl3. (a) Photographs showing ocular eye deviation and head tilt at 2 h after FeCl3 treatment. (b) Temporal changes in SN after middle ear injection of FeCl3. The number of animals in each group was 6. Values are mean ± standard deviation. **p < 0.01: significant difference between 70% FeCl3 and 7% FeCl3. SN: spontaneous nystagmus; FeCl3: ferric chloride.
Expression of c-Fos in neurons of the MVN
A few neurons in the MVN (11.6 mm from the bregma, AP 6.4 mm, ML 1.5 mm in stereotaxic atlas) expressed c-Fos 2 h after the sham operation. Compared with sham animals, the expression of c-Fos protein in the MVN was asymmetric at 2 h after middle ear treatment with 70% FeCl3; the number of c-Fos protein-expressing neurons was significantly higher in the MVN ipsilateral (ipsi-MVN) than in the MVN contralateral (contra-MVN) to the affected ear (p < 0.01). The contralateral prepositus hypoglossal nucleus (contra-PrH; PrH: 11.6 mm from the bregma, AP 6.8 mm, ML 0.5 mm in stereotaxic atlas) showed significant expression of c-Fos with ipsi-PrH at 2 h after 70% FeCl3 treatment. The increased expression of c-Fos protein in the ipsi-MVN and contra-PrH continued until 6 h after 70% FeCl3 treatment and disappeared by 24 h after 70% FeCl3 treatment. In contrast, treatment with FeCl3 resulted in negligible expression of c-Fos in the contra-MVN and ipsi-PrH for the 24 h of observation (Figure 2).

Expression of c-Fos protein in the MVN after middle ear injection of 70% FeCl3. (a) Expression of c-Fos immunoreactive neurons in the MVN and PrH. (b) Line histograph depicting temporal changes in the number of c-Fos immunoreactive neurons in the MVN after treatment with 70% FeCl3. ISPI: ipsilateral to the side of the affected ear; CONT: contralateral to the side of affected ear; 0 h: immediately after injection of FeCl3. The number of animals for each time period was 6. Values are mean ± standard deviation. *p < 0.05; **p < 0.01: significant difference between IPSI and CONT. Scale bar: 500 µm. MVN: medial vestibular nucleus; PrH: nucleus prepositus hypoglossi; FeCl3: ferric chloride.
Histopathological findings
Histopathological examinations using hematoxylin–eosin and TUNEL stainings were performed 120 h after middle ear treatment. Light microscopic findings showed a partial loss of hair cells and degeneration of the supporting stroma, causing atrophy of neuroepithelial layer in the ampulla of FeCl3-treated animals. A large number of inflammatory cells had infiltrated into the endolymphatic space and the stroma. Apoptosis was monitored using TUNEL staining. Compared with sham animals, TUNEL-stained cells with a dark brown color were scattered through the neuroepithelial layer and stroma in the ampulla of FeCl3-treated animals, indicating induction of apoptosis in the hair cells by FeCl3. Immunofluorescent staining for phalloidin was performed to visualize the cilia of vestibular hair cells and revealed many intact cilia in the vestibular end organs at 120 h after middle ear treatment with 70% FeCl3, suggesting partial loss of hair cells (Figure 3).
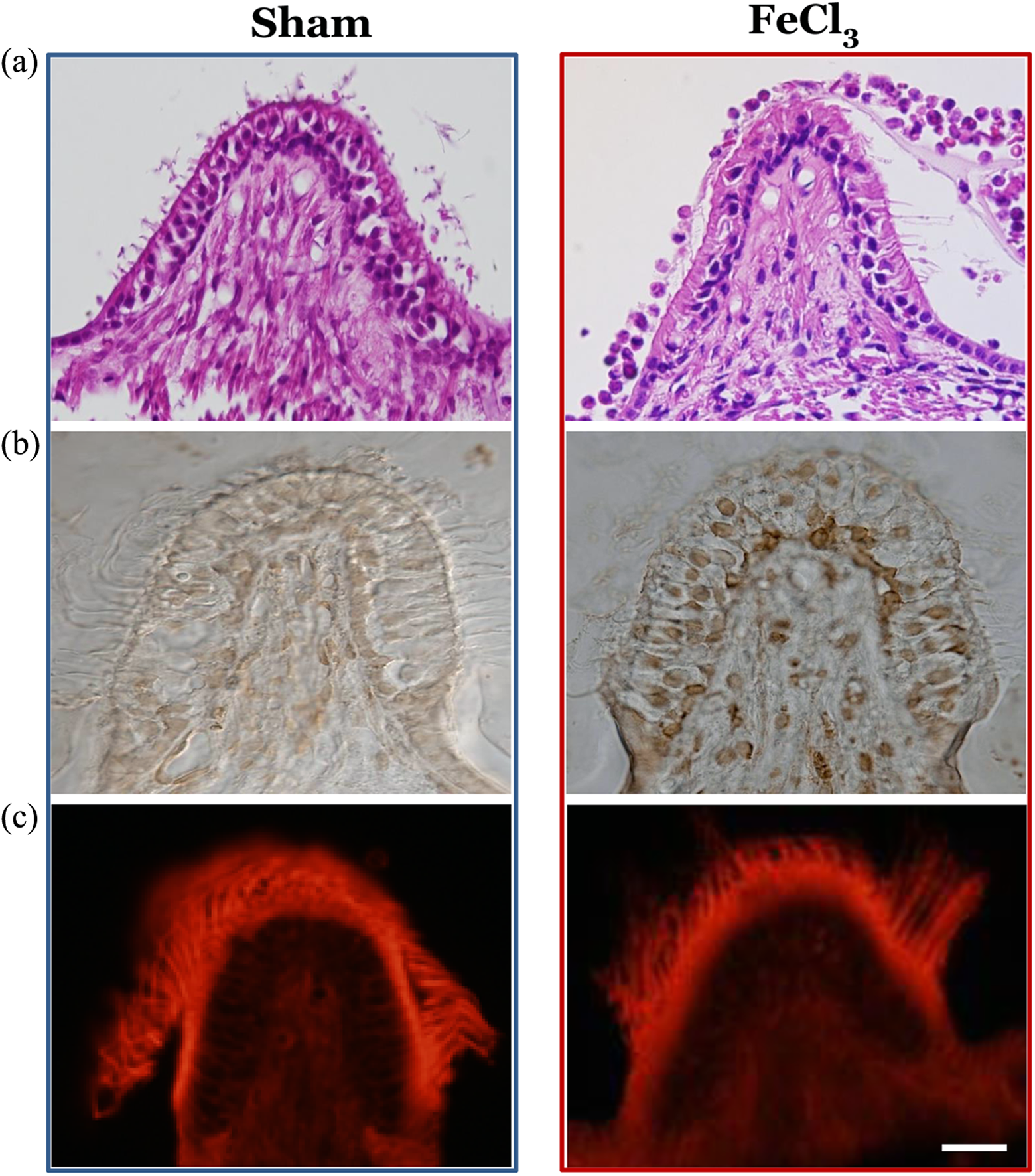
Histopathological changes in the vestibular sensory epithelium at 120 h after middle ear injection of 70% FeCl3. (a) Hematoxylin–eosin staining showing partial loss of hair cells and infiltration of inflammatory cells into the endolymphatic space after FeCl3 treatment. (b) TUNEL staining showing the presence of some apoptotic cells in the neuroepithelial layer. (c) Phalloidin staining showing partial loss of the cilia of hair cells after FeCl3 treatment. Magnification: ×400; scale bar: 10 µm. FeCl3: ferric chloride; TUNEL: terminal deoxynucleotidyl transferase dUTP nick end labeling.
Production of ROS and NO in the vestibular end organs
Production of NO and ROS was determined by the observation of green fluorescence after incubation of the vestibular end organs with DAF-2DA or CM-H2DCFDA, respectively. A relatively low intensity of CM-H2DCFDA and DAF-2DA signals was seen in the neuroepithelium and nerve bundles of the crista ampullaris of sham animals. In contrast, the intensity of CM-H2DCFDA fluorescence increased markedly in the sensory neuroepithelial layer and nerve bundles of the crista ampullaris at 2 h after middle ear treatment with 70% FeCl3. Moreover, green fluorescence signals for DAF-2DA were more diffuse yet stronger than those of CM-H2DCFDA throughout the crista ampullaris. Some type I hair cells generated a very strong DAF-2DA signal in response to middle ear treatment with 70% FeCl3 (Figure 4).

Expression of ROS and NO in the crista ampullaris at 2 h after middle ear injection of 70% FeCl3. (a) CM-H2DCFDA staining for detecting ROS. (b) DAF-2DA staining for detecting NO. Magnification: ×400; scale bar: 10 µm. ROS: reactive oxygen species; NO: nitric oxide; CM-H2DCFDA: 5-(and-6)-chloromethyl-2′,7′ -dichlorodihydrofluorescein diacetate, acetyl ester; DAF-2DA: diaminofluorescein–2 diacetate; FeCl3: ferric chloride.
Immunoreactivity for NT in vestibular end organs
To determine the level of NO-related RNS in the hair cells, we immunohistochemically detected the changes of immunoreactivity for NT, a biomarker of the NO-related peroxynitrite (ONOO–) reaction with protein tyrosine residues. 27 The immunoreactivity for NT was faint in the crista ampullaris of sham animals but was increased mainly in the neuroepithelial layer 2 h after middle ear treatment with 70% FeCl3. The increased immunoreactivity for NT also appeared in the nerve bundles of the stroma (Figure 5).

Expression of n-nitrotyrosine in the crista ampullaris at 2 h after middle ear injection of 70% FeCl3: (a) 2 h after sham operation and (b) 2 h after middle ear injection of 70% FeCl3. Magnification: ×400; scale bar: 10 µm. FeCl3: ferric chloride.
Expression of cJun and p-cJun in vestibular end organs
We determined the expression of cJun and p-cJun, which are known as intracellular molecules related to cell death in vestibular end organs, at 24 and 120 h after middle ear treatment with 70% FeCl3. Confocal microscopy showed very low immunoreactivity for cJun and p-cJun in type I hair cells and in primary nerve fibers throughout the stroma in the crista ampullaris immediately after middle ear treatment with 70% FeCl3. The stereocilia and supporting cells were not immunoreactive. There was a slight increase in immunoreactivity for p-cJun and c-Jun in the crista ampullaris at 24 h after 70% FeCl3 treatment. In contrast, the crista ampullaris showed strong immunoreactivity for p-cJun and cJun at 120 h after FeCl3 treatment. The shape of the neuroepithelial cells showing distinct expression of p-cJun and cJun proteins was flask shaped, suggesting that they are type I hair cells. In the hair cells, expression of p-cJun and cJun was mainly localized in the neck and outer cell membrane of the cell, rather than in the inner cytoplasm or nucleus (Figure 6). In order to confirm whether expression of p-cJun and cJun was specific to type I hair cells, expression of p-cJun and parvalbumin, a marker of type I hair cells, was determined. Most hair cells immunoreactive for parvalbumin also labeled for p-cJun protein in FeCl3-treated animals, indicating colocalization of parvalbumin and p-cJun in the type I hair cells (Figure 7).

Confocal microscopy illustrating expression of cJun and p-cJun in the crista ampullaris after middle ear injection of 70% FeCl3. The translational level and phosphorylation of cJun were increased simultaneously by FeCl3-induced oxidative stress to the vestibular end organs. 0 h: immediately after middle ear injection of 70% FeCl3. Magnification: ×400; scale bar: 10 µm. p-cJun: phosphorylated cJun; FeCl3: ferric chloride.

Confocal microscopy illustrating double labeling of parvalbumin (green) and p-cJun (red) in the crista ampullaris at 0 h and 120 h after middle ear injection of 70% FeCl3. (a) Parvalbumin, (b) p-cJun, and (c) parvalbumin + p-cJun. Magnification: ×400; scale bar: 10 µm. p-cJun: phosphorylated cJun; FeCl3: ferric chloride.
Discussion
Unilateral loss of vestibular afferent signals due to dysfunction of sensory hair cells in the inner ear disrupts the balance of neuronal activity between bilateral vestibular nuclei in the brain stem. This asymmetric neuronal activity in the central vestibular system is a major cause for the appearance of vestibular symptoms. 28,29 The present study observed a rapid onset of static vestibular symptoms, such as SN and roll head tilt, as early as 1 h after middle ear treatment with 70% FeCl3. SN, with a fast eye component toward the contralateral side to the affected ear, peaked at 2 h and disappeared at 24 h after FeCl3 administration. These data were comparable with the results obtained with other chemicals. For instance, sodium arsanilate produces static vestibular symptoms 6 h after injection into the middle ear, and SN toward the intact ear peaked at 12 h after this treatment. 30 A middle ear injection of α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid 31 or streptomycin 32 induces SN toward the intact ear, which peaks at 9 h after the treatment. After intratympanic instillation of 4% lidocaine, which achieves a transient blockade of the vestibular nerve, SN appeared within 15 min and peaked after about 20 min. 33 The frequency of SN observed in the present study showed a dose dependence but was lower than that observed in surgically or chemically labyrinthectomized animals. 30,34
Compared to the onset time of vestibular symptoms caused by other ototoxic chemicals or excitatory neurotransmitters, application of a high concentration of an oxidant, such as FeCl3 solution, results in a more rapid dysfunction of vestibular sensory end organs, which gives rise to asymmetric neuronal activity in the vestibular nuclei. The rapid action of FeCl3 solution on the vestibular end organs is similar to an earlier finding that the cochlear action potential (CAP) threshold sensitivity in response to sound stimuli is disrupted within 10 min of direct infusion of H2O2 into the scala tympani. 35 This disruption of the CAP threshold sensitivity by H2O2 reflects impairment of afferent synaptic transmission, due to dysfunction of the inner hair cells or the type I spiral ganglion cells. The early appearance of vestibular symptoms in the present study can in part be attributed to disturbance of afferent synaptic activity by the FeCl3 solution.
c-Fos is rapidly expressed in neurons in response to various stimuli and is thus a marker of neuronal activation for neural circuits in the brain. 36 Several lines of experimental evidence indicate that spatiotemporal changes in the expression of c-Fos in the vestibular nuclei correlate strongly and positively with recovery of static symptoms during vestibular compensation. 37 –39 In this study, the increased expression of c-Fos protein in the ipsi-MVN and contra-PrH was seen at 2 and 6 h after middle ear treatment with 70% FeCl3. The expression of c-Fos protein had disappeared in these nuclei by 24 h after treatment. On the other hand, there was no significant expression of c-Fos protein in the contra-MVN and ipsi-PrH after 70% FeCl3 treatment
Surgical unilateral labyrinthectomy (UL) induces higher expression of c-Fos protein in the contra-MVN than in the ipsi-MVN at 2 h after surgery. The subsequent asymmetry in expression of c-Fos is continued due to increased expression in the ipsi-MVN by at least 24 h after surgical UL. 34 In chemical UL, induced by sodium arsanilate, asymmetric expression of c-Fos, due to higher labeling of c-Fos in the contra-MVN than in the ipsi-MVN, has also been noted by 6 h after treatment. 30 Therefore, the spatiotemporal patterns of c-Fos protein expression in the MVN differ markedly between this study and earlier surgical or chemical UL studies. Considering that c-Fos is a metabolic marker for cellular depolarization in neural circuits, 36 FeCl3 treatment in the middle ear may cause different levels of vestibular end organ damage, or different levels of neuronal excitation in the central vestibular neural circuits, which also involve different processes for the recovery from static vestibular symptoms as compared with surgical or chemical UL.
In patients with superficial siderosis, vestibular deficits are functionally demonstrated by hyporeflexia of caloric tests and diminished or absent vestibular evoked potential 22 and are histopathologically demonstrated by degeneration of the neuroepithelium of the vestibular end organs, loss of hair cells, and vestibular nerve atrophy. 24 In the present study, middle ear treatment with 70% FeCl3 caused loss of hair cells, atrophy of the neuroepithelial layer in the ampulla, and apoptosis of the hair cells, which are consistent with the results obtained from patients with superficial siderosis. 24
In the presence of Fe2+ and Fe3+ ions, intracellular H2O2 and O2 − can undergo the Fenton reaction, which increases the levels of hydroxyl radicals. The hydroxyl radical is highly reactive and is thought to be the ROS most responsible for oxidative damage. 14,40 Zorov et al. 41,42 described ROS-induced ROS release as a positive feedback mechanism for enhanced ROS production, leading to potentially significant mitochondrial and cellular injury. When a FeCl3 solution was injected into the rat isocortex, superoxide radicals were increased in a cylinder of tissue obtained at the injection site 15 min after completion of the injection. 15 Direct application of FeCl3 to the adventitial surface in the mouse testicular artery induced the formation of thrombi, leading to complete occlusion by 12 min after endothelial injury and significant ROS production within 5 min after the treatment. 17 In the present study, the intensity of CM-H2DCFDA and DAF-2DA fluorescence had increased markedly in the sensory neuroepithelial layer and nerve bundles of the crista ampullaris, indicating a significant production of ROS and NO, 2 h after middle ear treatment with 70% FeCl3. The immunoreactivity for NT was increased mainly in the neuroepithelial layer at 2 h after middle ear treatment with 70% FeCl3. The present study thus confirmed that in vivo application of 70% FeCl3 can increase the production of ROS and NO, which elicits nitrosive stress.
The isolated vestibular and cochlear outer hair cells show rapid morphologic changes, such as swelling, bleb formation, and neck shortening, in a concentration-dependent manner within 30 min after H2O2 loading. 12,13 Hydroxyl radicals generated by Fe2+ or H2O2 inhibit the postsynaptic excitatory evoked potentials through inhibition of presynaptic Ca2+ entry in hippocampal slices. 43,44 A low concentration of Fe3+ blocks the activity of ion channels and affects intracellular Ca2+ levels via stimulation of the activity of the Na+–Ca2+ exchanger. 45,46 As a result, in addition to morphological deformation through direct oxidative toxicity, middle ear treatment with 70% FeCl3 solution in the present study may have affected multiple cellular processes involved in ion homeostasis or neural synaptic transmission in the vestibular end organs.
The cJun N-terminal region contains cJun N-terminal kinase (JNK) binding and phosphorylation sites. cJun can be activated by JNK, a subfamily of the MAPKs, through phosphorylation, which may occur in response to physiological or pathological stimulation. 47 In the case of the JNK1–3, phosphorylation at both Ser73 and Ser63 is critical for JNK-mediated effects. There is a close relationship between the increase in p-cJun at Ser73 and cell death. 48,49 Functional studies employing in vivo strategies, using gene deletion, targeted expression of dominant-negative isoforms, and pharmacological inhibitors, suggest a bipotential role for cJun in mediating neurodegeneration and cell death as well as in plasticity and repair. 26,50
Direct administration of H2O2 onto cultured cells induces the expression of immediate early genes, such as c-fos and cjun. 51,52 Additionally, direct application of H2O2 to the cultured cells also induces phosphorylation of JNK and cJun. 53,54 However, there have been no experimental studies reporting an increase in cJun expression by direct application of FeCl3.
Although the expression of cJun and p-cJun by direct application of H2O2 or FeCl3 in the inner ear has not been reported to date, there is considerable evidence that activation of cJun by JNKs is involved in the death of vestibulocochlear hair cells or sensory neurons in the cochlear spiral ganglion by ototoxic agents. An in vitro experiment using cochlear explants from neonatal rats showed that exposure to neomycin results in phosphorylation of both JNK and cJun. 55 After loss of cochlear hair cells caused by kanamycin, all cochlear spiral ganglion neurons undergoing apoptosis exhibited an increase in the expression of p-cJun during the cell death period. 56 In chick utricles, JNK activation lies upstream of the release of cytochrome c from the mitochondria into the cytoplasm, which in turn is upstream of the activation of caspase-9. 57,58
In explants of the organ of Corti of P3 mice, D-JNK-1, a peptide inhibitor of JNK that blocks JNK-mediated phosphorylation of cJun, prevented more than 90% of the hair cell death caused by exposure to neomycin. 59 Indirect inhibition of JNK activation by CEP-11004 also inhibits aminoglycoside-induced hair cell death in chick utricles 58 and neonatal rat explants of the organ of Corti. 60
In the present study, there was a significant increase in immunoreactivity for cJun as well as p-cJun in type I hair cells and the primary nerve fibers throughout the stroma in the crista ampullaris at 120 h after FeCl3 treatment. In hair cells, expression of p-cJun and cJun was mainly localized to the outer cell membrane rather than to the inner cytoplasm or nucleus. To the best of our knowledge, this study for the first time showed that FeCl3-induced oxidative stress stimulates a cellular signaling pathway involving translation of cJun protein in an in vivo experiment. Considering the role of the JNK pathway in the hair cells undergoing ototoxic reactions, the strong expression of p-cJun in this study may suggest that a signaling pathway involving activated JNK participates in cell death of hair cells or in the stroma of the crista ampullaris after middle ear treatment with 70% FeCl3. The histopathological investigations in the present study confirmed that partial hair cell loss by cellular events related to apoptosis after middle ear treatment with 70% FeCl3. However, this requires further experiments to evaluate the effects of JNK inhibitors on hair cell death or injury after middle ear treatment with 70% FeCl3.
In cultured utricles, addition of neomycin enhances expression of p-cJun in vestibular hair cells, with p-cJun protein localized to the nuclei. 58,61 Ruan et al. 62 reported that increased immunoreactivity for p-cJun was noted throughout the cytoplasm of the surviving hair cells at 5 and 13 days after noise exposure. The differences in localization and temporal changes of p-cJun protein expression in the hair cells between the earlier studies and the present study can possibly be attributed to different experimental conditions or animal species or different types of injury.
Type I sensory hair cells of the vestibular epithelium are completely ensheathed by an afferent nerve ending, that is, the vestibular nerve calyx. The nerve calyx is therefore very close to the cell membrane of type I hair cells. 63 The resolution of the confocal imaging technique used in the present study did not allow definition of the calyx from the cell membrane of hair cells, and thus, we could not rule out expression of p-cJun and cJun in the calyx of type I hair cells after FeCl3 treatment. Further research should be undertaken to elucidate the exact localization of p-cJun and cJun in the hair cells.
Conclusion
A single short-term treatment with a high concentration FeCl3 into the middle ear is sufficient to result in activation of cJun and oxidative stress through the formation of ROS and NO in the vestibular sensory receptors, resulting in vestibular dysfunction. Therefore, activation of cJun and oxidative stress may be key components of the pathogenesis of vestibular deficits in patients with superficial siderosis. Moreover, a single short-term unilateral treatment with a high concentration FeCl3 into the middle ear may be a useful experimental model for research of inner ear abnormalities caused by oxidative stress.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
