Abstract
The growing use of carbon nanotubes (CNTs) emphasizes the importance of its potential toxic effects on the human health. Previous studies proved that CNTs caused oxidative stress and decreased cell viability. On the other hand, reactive oxygen species (ROS) and oxidative stress impaired β-cell functions and reduced the insulin secretion. However, there is not any study on the effects of CNTs on islets and β-cells. Therefore, the present study aimed to evaluate the effects of single-walled CNTs (SWCNTs) on oxidative stress in islets in addition to the protective effects of naringin (NRG) as an antioxidant . We examined the effects of SWCNTs and naringin on islets by 3,4 3-(4,5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide (MTT) assay; measurement of insulin secretion, ROS, and malondialdehyde (MDA); activities of superoxide dismutase (SOD), catalase (CAT), and glutathione (GSH) peroxidase (GSH-Px); and content of GSH and mitochondrial membrane potential (MMP). The MTT assay demonstrated that decreased viability of islets cells was dose-dependent with exposure to SWCNTs. Further studies revealed that SWCNTs decreased insulin secretion and MMP, induced the formation of ROS, increased the level of MDA, and decreased the activities of SOD, GSH-Px, and CAT and content of GSH. Furthermore, the pretreatment of islets with naringin significantly reverted back these changes. These findings revealed that SWCNTs might induce the oxidative stress to pancreatic islets, causing the occurrence of diabetes, and the protective effects of naringin that was mediated by augmentation of the antioxidant defense system of islets. Our research indicated the necessity for further in vivo and in vitro researches on the effects of SWCNTs and naringin on diabetes.
Introduction
Nanoscale substances are defined as materials with at least a main dimension of fewer than 100 nm. These substances can be approximately engineered from any chemical material. The new properties of these compounds can have enormous potential in new uses compared to their original forms. However, nanoscale materials have multiple functional uses; but the studies on its effects on humans and environment have a long way ahead. 1 Toxic effects of nanoparticles on the human and environmental health are the subjects of growing concern as their manufacture is anticipated to increase in the future; hence, the study on the toxic effects and mechanisms should be given importance to allow reputable experts to produce harmless products. 2 –4 Between the different forms of nanoparticles, there is a special attention to carbon nanotubes (CNTs) and their exceptional mechanical, electrical, magnetic, and hollow nanostructure properties. 5 –7 CNTs are the tube-shaped molecules with micrometer sizes and diameters of 0.4–2 nm for single-walled CNTs (SWCNTs) and 2–100 nm for multi-walled CNTs (MWCNTs). SWCNTs are more widely used than MWCNTs especially in medical applications such as in the diagnosis of diabetes. 8 Various kinds of increasing nanotechnology applications for CNTs will certainly result in the increased potential for both human and environmental contacts to this nanomaterial. Therefore, it is necessary to examine the toxicity and biocompatibility of CNTs. However, there are only a few studies on the biocompatibility and cytotoxicity of CNTs, and the obtained results are usually different. For instance, Shvedova et al. 9 indicated that the dermal contact to CNTs may lead to dermal toxicity because of enhanced oxidative stress, defeat of cell viability, and morphological alterations. Jia et al. 10 exposed alveolar macrophages, which were isolated from guinea pigs, to SWCNTs for 6 h and reported that the SWCNTs produced a more toxic response than MWCNTs. Cui et al. found that SWCNTs can inhibit the proliferation of human embryo kidney cells (HEK293) by inducing cell apoptosis and diminishing cellular adhesive ability. 11 On the contrary, other researchers established that the CNTs displayed cytotoxicity without the production of apoptosis or apoptosis without cytokine release and inflammation. 12 –14
Diabetes mellitus (DM), which affects numerous organs in the body, is one of the most widespread metabolic disorders in the world. In vivo examination indicated that excessive production of reactive oxygen species (ROS) occurred in diabetes. Oxidative stress is the end result of an imbalance among antioxidants and free radicals which are involved in cellular impairment. 15 For instance, cumulative ROS in pancreatic β-cells decreases the insulin gene expression and insulin release and also injures the islets. 16 Since the β-cells have little level of antioxidant protection enzymes such as superoxide dismutase (SOD) and catalase (CAT), these cells enormously have excessive sensitivity to free radical-induced impairment. 17 One of the damaging effects of ROS is lipid peroxidation which may result in β-cells death and loss of insulin secretion through apoptosis progression. 18,19 Therefore, it seems that the contact of pancreatic islets to antioxidant agents is important in the treatment of diabetes.
It is usually accepted that the apoptosis of β-cells and damage of islets, which are produced by increasing ROS, are among the main factors for induced DM. On the other hand, previous studies proved that the CNTs cause oxidative stress and decrease the cell viability. However, there is not any study on the effects of SWCNTs on islets and β-cells. Therefore, the present study aimed to evaluate the effects of SWCNTs on the viability of β-cells and oxidative stress in islets as well as the effects of naringin as an antioxidant for improving all of these defects. We examined the effects of SWCNTs and naringin on islets according to the 3,4 3-(4,5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide (MTT) assay; measurement of ROS and malondialdehyde (MDA); activities of SOD, CAT, glutathione (GSH) peroxidase (GSH-Px), and GSH; and mitochondrial membrane potential (MMP) content.
Naringin (4′,5,7-trihydroxyflavanone 7-rhamnoglucoside, NRG) is a main and active flavone glycoside in citruses and grape. 20 NRG is known for its physiological and pharmacological properties including the antihypercholesterolemic, 21 –23 cardioprotection, 24,25 anti-inflammatory, 21,26 –28 antioxidant, 29 –31 and antiapoptotic effects. 32 –34 In addition, most studies found that the NRG could normalize plasma glucose, insulin, glycosylated hemoglobin, and some important enzymes of carbohydrate metabolism in diabetic animals. They also reported that naringin treatment stimulates insulin secretion from remnant pancreatic β-cells and reduces blood glucose levels in diabetic animals. Furthermore, some studies discovered that the naringin treatment could have valuable effects on pancreatic β-cells by counterbalancing the oxidative stress and inhibited lipid peroxidation and increased antioxidant status in diabetic rats. Naringin was thus selected as an antioxidant to scavenge excessive free radicals which were produced by SWCNTs and reduced oxidative stress by virtue of its antioxidant effect.
Materials and methods
Materials
The SWCNTs (length: 50–300 nm and diameter: 1–2 nm) were purchased from US Research Nanomaterials, Inc (Houston, TX 77084, USA). Naringin, 4-(2-Hydroxyethyl)-1-piperazineethanesulfonic acid) (HEPES), mannitol, ethylene glycol tetraacetic acid, bovine serum albumin (BSA), 2,7-dichlorofluorescein diacetate (DCFH-DA), MTT, thiobarbituric acid, trichloroacetic acid (TCA), 1,1,3,3-tetramethoxypropane, reduced GSH, oxidized GSH, and Coomassie Brilliant Blue powder were purchased from Sigma-Aldrich (St Louis, Missouri, USA), and sucrose 5,5′-dithiobis(2-nitrobenzoic acid) (DTNB), dimethyl sulfoxide (DMSO), sodium chloride (NaCl), potassium chloride (KCl), calcium chloride (CaCl2), magnesium chloride (MgCl2), and sodium bicarbonate (NaHCO3) were obtained from Merck company (Darmstadt, Germany). GSH-Px (Cat. No. S0058) and GSH assay kit (Cat. No. S0052) were purchased from Beyotime Institute of Biotechnology (Jiangsu, China).
Dispersion of nanomaterials
The CNTs were dispersed in distilled water. Then, for decrease agglomeration, the stock suspensions (600 μg/mL) were mixed by vortex for 20 s and sonicated for 20 s by a sonicator probe (Ultra sonic processor VCX-750 W, Vibra-Cell™). The stock suspensions were then diluted with distilled water and sonicated as before to prepare test concentrate.
Animals
Eighty-four male NMRI mice (25–30 g) were obtained from the animal house of Ahvaz Jundishapur University of Medical Sciences (Ahvaz, Iran). They were housed in cages (22 ± 2°C, under a standard 12 h light: 12 h dark cycle) and allowed ad libitum feed access. All experimental procedures were done according to standards for animal care, established by the ethical committee of Ahvaz Jundishapur University of Medical Sciences (IR.AJUMS.REC.1396.294).
Isolation of mice pancreatic islets
Pancreatic islets were isolated from overnight-fasted male NMRI mice by Lacy and Kostianovsky modified collagenase digestion method.
35
In brief, after cervical dislocation, abdomen of animals was opened. The common bile duct was occluded at distal end close to the duodenum and 5 mL of Hank’s Balanced Salt Solution (HBSS; 115 mmol/L NaCl, 10 mmol/L NaHCO3, 5 mmol/L KCl, 1.1 mmol/L MgCl2, 1.2 mmol/L NaH2PO4, 2.5 mmol/L CaCl2, 25 mmol/L HEPES, and 5 mmol/L
Dose optimization for SWCNTs nanoparticles
Before starting the experiments, the isolated islets were cultured overnight in RPMI-1640 medium, in 5% CO2 at 37°C. Selected doses are based on previous studies by Davoren et al. 6 , Cui et al. 11 , and Yehia et al. 14 , and in order to select the best dose, optimization of dose was done by pretreating the islets with various concentrations of SWCNTs nanoparticles (40, 80, 160, and 320μg) for 24 h to reach effective doses. The islets were divided into six groups of 10 islets including (1) Con (negative control): islets in RPMI-1640 medium alone for 24 h; (2) hydrogen peroxide (H2O2) (positive control): islets in RPMI-1640 medium for 24 h + H2O2 (50 μM) for 2 h; (3) islets in RPMI-1640 medium + SWCNTs nanoparticles (40 μg) for 24 h; (4) islets in RPMI-1640 medium + SWCNTs nanoparticles (80 μg) for 24 h; (5) islets in RPMI-1640 medium + SWCNTs nanoparticles (160 μg) for 24 h; and (6) islets in RPMI-1640 medium + SWCNTs nanoparticles (320 μg) for 24 h. Then, the viability assay was done on pretreated islets. This assay is based on the reduction of MTT, a yellow tetrazole to purple formazan by mitochondrial respiration in viable cells. After removing the medium, pretreated islets were washed twice with Krebs-HEPES buffer, and 20 mL of MTT solution was added and incubated for 4 h at 37°C. After washing, the formazan was resuspended in 100 mL of DMSO and the absorbance was measured at 570 nm by an ELISA reader, (State Fax® 2100, Awareness, USA). The viability of the groups was shown as the percentage of controls that is assumed to be 100%. 38
Insulin secretion measurement
Insulin secretion was evaluated in a glucose static incubation. The islets were preincubated overnight in RPMI medium. The isolated mouse islets were divided into seven groups, with each group containing10 islets: (1) control islets cultured with RPMI medium for 48 h; 39 (2) islets cultured for 24 h and then in same medium were exposed to SWCNTs nanoparticles (160 μM) for 24 h; (3) islets cultured for 24 h pretreated with naringin (40 μM), 40 and then were exposed to SWCNTs nanoparticles (160 μM) for 24 h; (4) islets cultured for 24 h pretreated with naringin (80 μM), and then were exposed to SWCNTs nanoparticles (160 μM) for 24 h; (5) islets cultured for 24 h pretreated with naringin (160 μM), and then were exposed to SWCNTs nanoparticles (160 μM) for 24 h; (6) islets cultured for 24 h pretreated with glibenclamide (Gli, 10 μM), 41 and then were exposed to SWCNTs nanoparticles (160 μM) for 24 h; and (7) islets cultured for 24 h pretreated with naringin (160 μM), and then were exposed to RPMI medium for 24 h. Then, islets were washed with HBSS, and 1 mL culture medium including 2.8 mM, 5.6 mM, or 16.7 mM concentration of glucose (basal and stimulatory) was added to them and incubated at 37°C for 1 h. 39 Then, the supernatants were collected and stored in separate micro tubes. Insulin concentration was determined using a Rat Insulin ELISA (Monobind, USA) according to the manufacturer’s protocol, and the results are displayed as micro units of insulin secreted per islet per hour. 42
Measurement of ROS
To measure ROS generation, a fluorometric assay using intracellular oxidation of DCFH-DA was performed. Briefly, the pretreated islets (seven groups similar to those previously mentioned in the insulin secretion measurement protocol) were washed with phosphate-buffered saline (PBS), and then incubated with 40 mM DCFH-DA for 30 min. At the end of incubation, the islets were washed with PBS, lysed with NaOH, and the fluorescence of dichlorofluorescein, which is the oxidized product of DCFH-DA, was measured by ELISA fluorimeter with excitation and emission wavelengths of 485 and 530 nm, respectively. The values were standardized by the amount of total protein in each well. 43
Measurement of islets viability
The mitochondrial viability in the islets was tested using MTT in order to investigate the protective effect of naringin on cell viability of islets that were exposed to SWCNTs nanoparticles (160 μM). Briefly, pretreated islets (seven groups similar to those previously mentioned in the insulin secretion measurement protocol) were washed twice by Krebs-HEPES buffer, and 20 mL of MTT solution was added and incubated for 4 h at 37°C. After washing, the formazan was resuspended in 100 mL of DMSO and the absorbance was measured at 570 nm by the ELISA reader. The viability of the groups was shown as the percentage of controls that is assumed to be 100%. 44
Preparation of the samples for biochemical analyses
For biochemical analysis, islets were isolated from overnight-fasted male NMRI mice and divided into seven groups similar to those previously mentioned in the insulin secretion measurement protocol in this study, but in this analysis, each group contained 50 islets. At the end, islets were incubated for 1 h in a medium containing 2.8 mM, 5.6 mM, or 16.7 mM concentration of glucose (basal and stimulatory), then the islets were washed three times with ice-cold PBS and lysed through sonication (Ultra sonic processor VCX-750 W) for 10 s and centrifuged for 10 min at 4°C. 39 Supernatants were used immediately for assaying SOD, CAT, and GSH-Px activities as well as MDA and GSH levels as follow:
MDA level measurement
Levels of MDA of islet tissues were measured by monitoring the thiobarbituric acid reactive substance formation. 45 Five hundred micro liter of islets tissue supernatant was added to 1.5 mL of TCA (10%). After centrifuging, the supernatant (1.5 mL) was blended with 2 mL of thiobarbituric acid (0.67%) and placed for 15 min in boiling water bath. After the sample has cooled, it was extracted with 2 mL of n-butanol followed by centrifuge (at 4000 × g for 15 min). The organic phase was removed and the absorbance was read at 535 nm (spectrophotometer, UV-1650PC SHIMADZU, Kyoto, Japan; Ex = 500 nm, Em = 520 nm) using a blank containing all the reagents except the sample. Values were expressed as nmol/mg of protein. The MDA standard curve was prepared using various dilutions of 1,1,3,3-tetramethoxypropan.
Determination of CAT activity
For measurement of CAT activity of islet tissues, we used the Claiborne’s method. 46 Initially, a mixture of 50 mM of potassium phosphate (pH 7.0), 19 mM of H2O2, and 20 mL supernatant of homogenate pancreas islets was prepared. The H2O2 was added to this mixture at the end, for beginning the reaction. The rate of H2O2 decomposition was assessed by measuring the absorbance changes at 240 nm for 60 s. One unit of CAT activity is defined as 1 μM of H2O2 that is consumed in 1 min. Ultimately, the specific activity of CAT was expressed as unit per milligram of protein.
Determination of SOD activity
The SOD activity in pancreatic islet tissues was estimated by applying the Suttle's method 47 with the SOD kit (Randox Labs, Crumlin, UK). The principle of this method is based on the ability of SOD to inhibit the reduction of nitro blue tetrazolium (NBT) and formation of red formazan, the concentration of which is measured by spectrophotometer (UV-1650PC SHIMADZU, Kyoto, Japan; Ex = 500 nm, Em = 520 nm) at 505 nm. Fifty percent inhibition of NBT reduction of the enzyme is defined as 1 unit SOD. The SOD activity was expressed as unit per milligram of protein.
Measurement of GSH-Px activities
The activities of GSH-Px were determined using assay kits (Jiancheng Bioengineering, China).
The GSH-Px activity was assayed by quantifying the rate of oxidation of the reduced GSH to the oxidized GSH by H2O2 catalyzed by GSH-Px. The GSH kit utilizes an enzymatic recycling method based on the reaction between GSH and 5,5-dithiobis(2-nitrobenzoic acid) (DTNB) that produces a yellow-colored compound (NBT). 48
GSH level measurement
The GSH content was measured according to the method described by Thomas and Skrinska. Islets tissue supernatants were incubated with 1 mL of 20% TCA and 1 mL of 1 mM EDTA for 5 min, which was used as protein precipitant. The total homogenate was centrifuged at10,000 × g for 30 min at 4°C; and 200 µL of supernatant was mixed with 1.8 mL of 0.1 mM DTNB. The GSH reacts with DTNB and forms a yellow-colored complex. The absorbance was read at 412 nm. The result was expressed as micromoles of GSH per milligrams of protein. 46
MMP measurement
Rhodamine 123 (Rh123) was used as an indicator of MMP. 49 –51 Rh123 is a lipophilic cation that partitions selectively into the negatively charged mitochondrial membrane. Hyperpolarization of the mitochondrial membrane causes the uptake of Rh123 into mitochondria and a decrease in fluorescence due to quenching. 49 Islets were incubated in Krebs-Ringer bicarbonate buffer containing 119 mM of NaCl, 4.7 mM of KCl, 2.5 mM of CaCl2, 1.2 mM of MgCl2, 1.2 mM of monopotassium phosphate (KH2PO4), 25 mM of NaHCO3, and 2 mM of glucose supplemented with 10 g/mL of Rh123 for 20 min at 37°C. Rh123 fluorescence was excited at 540 nm (to decrease phototoxicity) and measured at 590 nm.
Protein measurement
The concentration of protein was assayed according to Bradford method. 52 Briefly, a volume of 20 μL of prepared sample was added to 1 mL of Bradford reagent. Then, the light absorbance was measured after 5 min at 595 nm. BSA was used as standard.
Statistical analysis
All the results were statistically analyzed using GraphPad Prism (version 5.04) as mean ± standard error of mean with one-way analysis of variance (ANOVA) followed by post hoc Tukey test. Moreover, the differences were considered statistically significant at p < 0.05.
Results
Dose optimization for SWCNTs nanoparticles
Optimization of SWCNT nanoparticle doses was done through pretreating the islets with various concentrations of SWCNT nanoparticles (40, 80, 160, and 320 μg) for 24 h, and then the viability of islets was tested by MTT test. The viability of groups was shown by percentage of controls that was assumed to be equal to 100% compared with H2O2 50 μM. The cell viability decreased to approximately 50% when islets were treated with 160 or 320 μg of SWCNT nanoparticles which were comparable with islets treated by H2O2 50 μM (p < 0.001; Figure 1). We thus used 160 μg of SWCNTs nanoparticles in this experiment.

Effects of SWCNTs nanoparticles treatment on the viability of isolated mice islets. Islets were exposed to SWCNTs (40, 80, 160, and 320 μg) for 24 h or H2O2 50 μM for 2 h, and then cell viability was measured (seven mice in each group). Results are mean + SEM of five independent experiments performed in duplicate. The difference between control and other groups is significant at p < 0.01 (a*) and p < 0.05 (a). SWCNT: single-walled carbon nanotube; H2O2: hydrogen peroxide; SEM: standard error of mean.
Effect of naringin on insulin secretion in SWCNTs nanoparticles-treated islets
In the presence of 2.8 mM of glucose, islet cells did not exhibit a significant change in insulin secretion (Figure 2(a)). As shown in Figure 2(b) and (c), high level of glucose caused a significant increase in insulin secretion compared with the low glucose. Incubation with SWCNTs significantly decreased glucose-induced insulin secretion (p < 0.01). Although pretreatment with naringin (80 and 160 μM) indicated prevention of the SWCNTs-induced decrease in insulin secretion, the pretreatment with naringin 160 μM could individually increase insulin secretion compared with control after 24 h (p < 0.05).
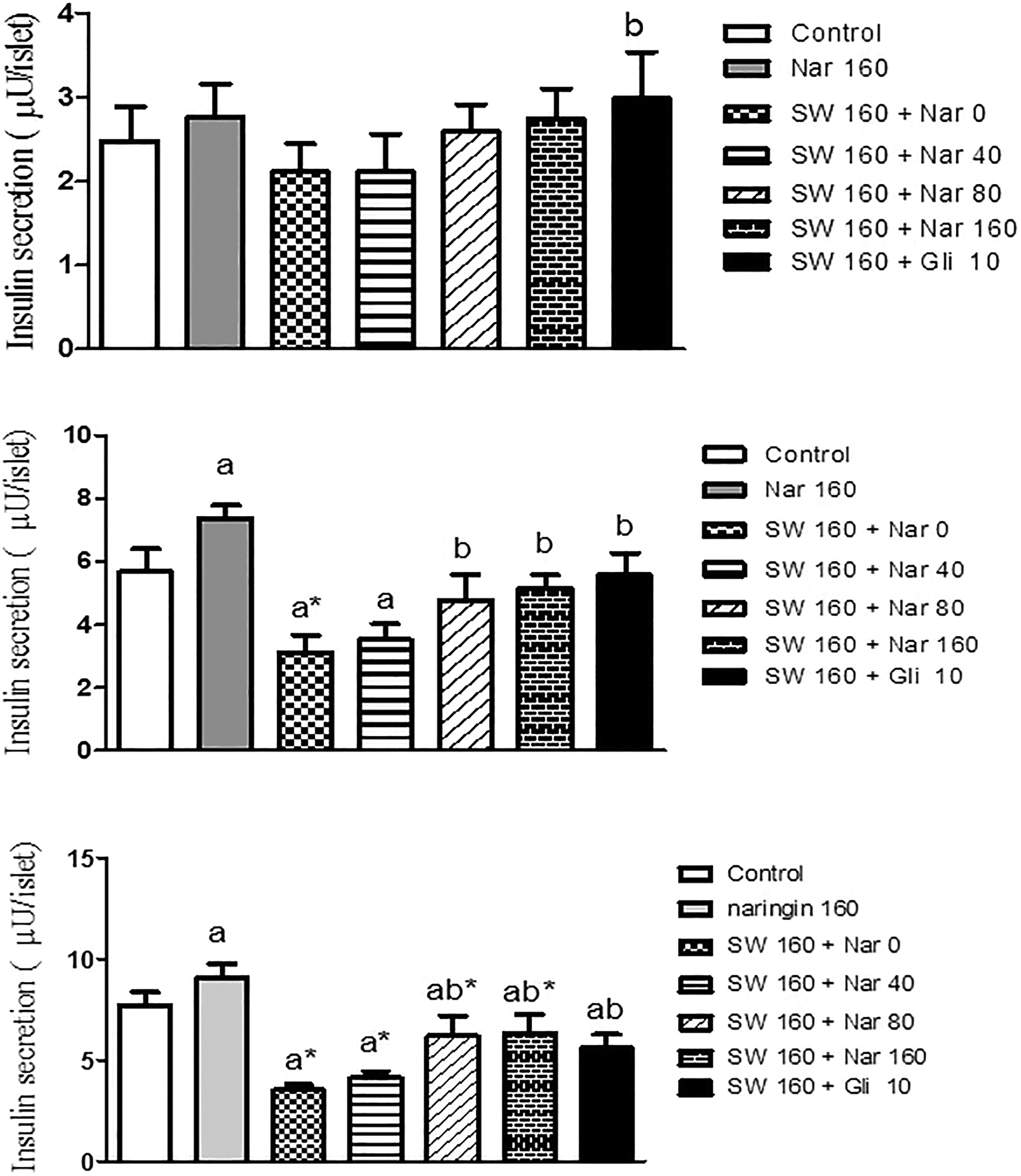
Pretreatment effects of naringin (0, 40, 80, and 160 μM) and glibenclamide (Gli, 10 μM) for 24 h on insulin secretion from isolated pancreatic mice islets after a 24-h exposure of islets to SWCNTs (160 μg) and a subsequent 1-h incubation with (a) 2.8 mM, (b) 5.6 mM, or (c) 16.7 mM glucose-containing medium (seven mice in each group). Results are expressed as mean ± SEM. The difference between control and other groups is significant at p < 0.01 (a*) and p < 0.05 (a). The difference between SWCNTs and other groups is significant at p < 0.01 (b*) and p < 0.05 (b). SWCNT: single-walled carbon nanotube; SEM: standard error of mean; NRG: naringin; Gli: glibenclamide.
Effect of naringin on oxidative stress in SWCNTs nanoparticles-treated islets
As shown in Figure 3, the exposure of islets to SWCNT significantly increased the production of ROS compared with controls (p < 0.01). However, the pretreatment with 80 and 160 μM of naringin significantly inhibited the SWCNTs-induced ROS production (p < 0.05 and 0.001, respectively). Pretreatment with naringin 160 μM did not produce any significant changes compared with controls.

Pretreatment effects of naringin (0, 40, 80, and 160 μM) for 24 h on cell viability from isolated pancreatic mice islets after a 24-h exposure of islets to SWCNTs (160 μg, seven mice in each group). Results are expressed as mean ± SEM. The difference between control and other groups is significant at p < 0.01 (a*) and p < 0.05 (a). The difference between SWCNTs and other groups is significant at p < 0.01 (b*) and p < 0.05 (b). SWCNT: single-walled carbon nanotube; SEM: standard error of mean; NRG: naringin.
Effect of naringin on cell viability in SWCNTs nanoparticles-treated islets
The cell viability decreased to approximately 50% when cells were treated with 160 μg of SWCNT nanoparticles for 24 h. We then investigated the effects of different doses of naringin on the oxidative stress-induced apoptosis. As shown in Figure 4, the MTT assay indicated that the pretreatment of islets with 80 and 160 μM of naringin significantly increased the oxidative stress-reduced cell viability (p < 0.05 and 0.01, respectively). However, there was no significant change in cell viability compared with control after 24 h of exposure to 160 μM of naringin.
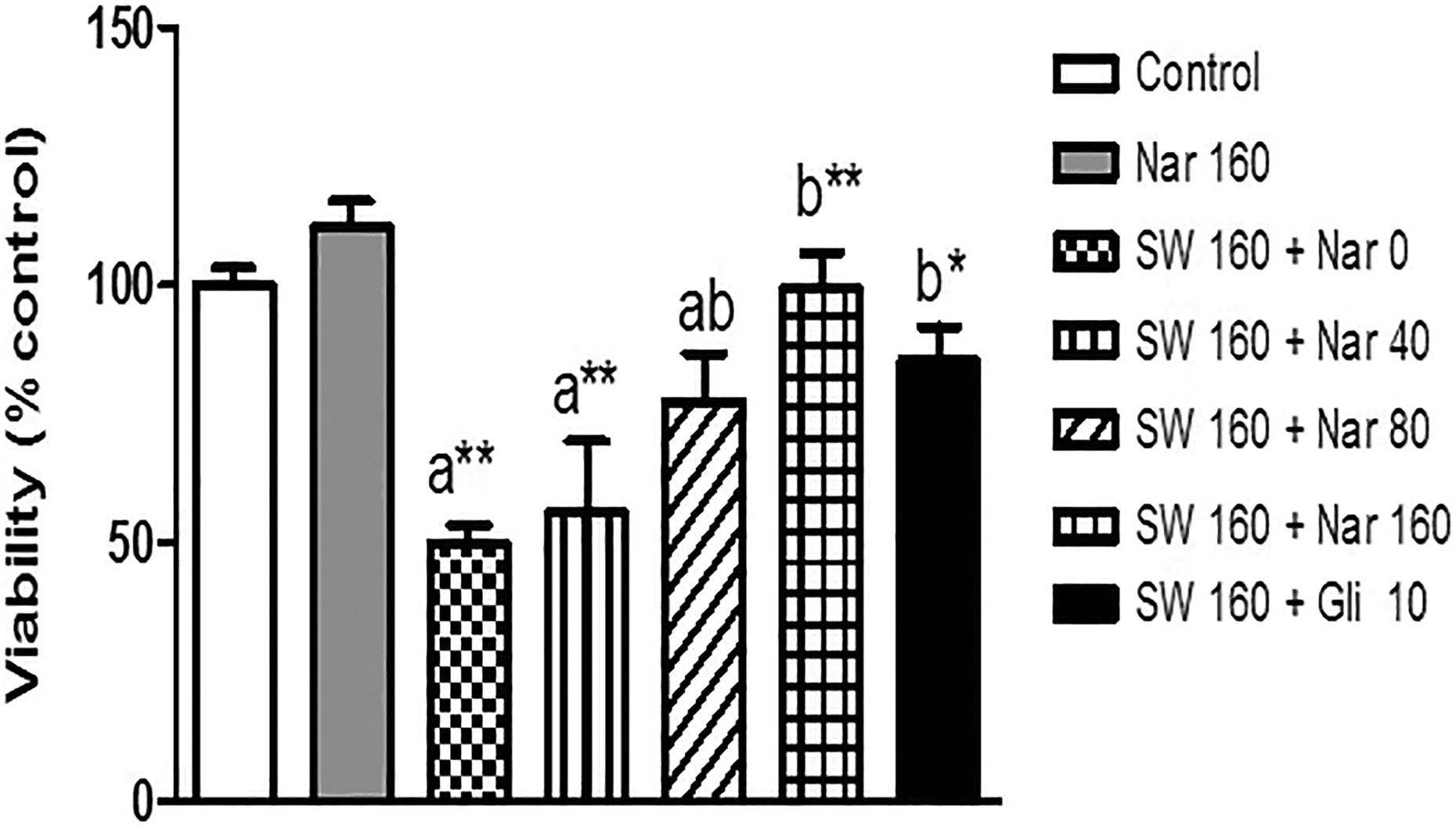
Pretreatment effects of naringin (0, 40, 80, and 160 μM) and glibenclamide (Gli, 10 μM) for 24 h on cell viability from isolated pancreatic mice islets after a 24-h exposure of islets to SWCNTs (160 μg, seven mice in each group). Results are expressed as mean ± SEM. The difference between control and other groups is significant at p < 0.001 (a**), p < 0.01 (a*), and p < 0.05 (a). The difference between SWCNTs and other groups is significant at p < 0.001 (b**), p < 0.01 (b*), and p < 0.05 (b). SWCNT: single-walled carbon nanotube; SEM: standard error of mean; NRG: naringin, Gli: glibenclamide.
Effect of naringin on MDA level in SWCNTs nanoparticles-treated islets
After incubation of islets with 160 μg of SWCNT nanoparticles, the MDA levels (nmol/mg protein) significantly increased compared to the control group (p < 0.01). Furthermore, a 24-h treatment of islets with 80 and 160 μM of naringin and glibenclamide decreased the MDA level compared to SWCNT group (p < 0.01, 0.01, and 0.001, respectively; Table 1).
The effects of naringin (0, 40, 80, and 160 μM) and glibenclamide (10 μM) pretreatment on the MDA and GSH levels; SOD, CAT, and GSH-Px activities; and MMP of isolated pancreatic mice islets following a 24-h exposure of islets to SWCNTs (160 μg) and a subsequent 1-h incubation with 16.7 mM glucose-containing medium.a

SWCNT: single-walled carbon nanotube; NRG: naringin; Gli: glibenclamide; MDA: malondialdehyde; GSH: glutathione; SOD: superoxide dismutase; CAT: catalase; GSH-Px: glutathione peroxidase; MMP: mitochondrial membrane potential; ANOVA: analysis of variance.
aData presented as mean ± SD; n = 7.
bDifference between control and other groups is significant at p < 0.01. The p value was from one-way ANOVA, followed by Tukey’s test for multiple comparisons.
cDifference between control and other groups is significant at p < 0.05. The p value was from one-way ANOVA, followed by Tukey’s test for multiple comparisons.
dDifference between SWCNTs and other groups is significant at p < 0.01. The p value was from one-way ANOVA, followed by Tukey’s test for multiple comparisons.
eDifference between SWCNTs and other groups is significant at p < 0.001. The p value was from one-way ANOVA, followed by Tukey’s test for multiple comparisons.
fDifference between SWCNTs and other groups is significant at p < 0.05. The p value was from one-way ANOVA, followed by Tukey’s test for multiple comparisons.
gDifference between control and other groups is significant at p < 0.001. The p value was from one-way ANOVA, followed by Tukey’s test for multiple comparisons.
Effect of naringin on GSH level in SWCNTs nanoparticles-treated islets
Results of GSH assessment indicated a significant decrease in 160 μg SWCNT nanoparticles group compared to the control group (p < 0.05). Furthermore, a 24-h treatment of islets with 160 μM of naringin and glibenclamide significantly prevented this decrease in GSH level in SWCNT-treated islets (p < 0.05; Table 1).
Effect of naringin on CAT activity in SWCNTs nanoparticles-treated islets
Due to the effects of naringin on CAT activity in SWCNT nanoparticles-treated islets according to the obtained results, after the addition of SWCNT nanoparticles to the islets, the CAT activity of islet tissues (unit/mg protein) decreased significantly compared to the control group (p < 0.01). A remarkable increase in CAT activity was observed in the pretreated naringin 80 μM (p < 0.05), and also naringin 160 μM (p < 0.01) compared with SWCNT group (Table 1).
Effect of naringin on SOD activity in SWCNTs nanoparticles-treated islets
After the addition of SWCNT nanoparticles (160 μg), the SOD activity of islet tissues decreased significantly compared to the control group (p < 0.001). Significant improvement of SOD activity was shown in naringin 80 μM (p < 0.01) and 160 μM (p < 0.001) groups compared to SWCNT group (Table 1).
Effect of naringin on GSH-Px activity in SWCNTs nanoparticles-treated islets
According to the obtained results, after the addition of SWCNT nanoparticles to the islets, the GSH-Px activity of islet tissues (unit/g protein) decreased significantly compared to the control group (p < 0.05). Furthermore, a significant improvement in GSH-Px activity was shown in naringine 80 and 160 μM groups compared to SWCNT group (p < 0.05; Table 1).
Effect of naringin on MMP in SWCNTs nanoparticles-treated islets
Cationic fluorescent dye and Rh123 were used for the measurement of MMP collapse. As shown in Table 1, SWCNT nanoparticles significantly decreased MMP in pancreatic islets (p < 0.001), but the 24-h treatment of islets with 80 and 160 μM of naringin and 10 μM of glibenclamide increased the membrane potential compared to SWCNT group (p < 0.05, 0.01, and 0.01, respectively; Table 1).
Discussion
Due to the wide application of CNTs, it is essential to investigate their toxic potentials. There is not any epidemiological study on the toxicity of these substances in humans, but studies on animals indicate that these materials can be collected in the liver and lung tissues and cause disruption of transmembrane electron transfer, penetration of cell envelope, oxidation of cell components, and production of secondary products such as ROS. 53,54 Studies on the potential risks of SWCNTs have mostly focused on the inhalation and epidermal contact because passing through the skin or respiratory tract is the most possible way of contact to nanomaterials. 55,56 Lam et al. conducted a research on mice, and they established that exposure of SWCNT in mice resulted in early granulomatous reaction, uncommon acute inflammatory response, and progressive fibrosis. 57 Another study revealed that inhaled nanoparticles accumulate in olfactory bulb of rats and reach the cerebral cortex, lung, and other organs such as tongue, esophagus, kidney, spleen, aorta, septum, heart, and blood. 58 SWCNTs can be simply suspended in the air and deposited by inhalation which is entered beyond the bronchus deep into the alveolar region of the lung and then translocated into the blood circulation. 59,13 The results recommend that aerosol exposure of raw CNTs in the workplace should be avoided to protect the human health. Most studies have focused on the effects of toxicity of nanoparticles on different cell lines such as inhibition of HEK 293 cell proliferation after exposure to SWCNTs, 11 cytotoxicity of SWCNT on a lung carcinoma cell line (A549), 6 and oxidative stress in rat lung cells. 60 Herzog reported the same oxidative stress which linked alterations in primary bronchial epithelial and A549 cells. 61 There are several results of various scientific tests on cells and some results indicate that it is highly toxic, but others have found no signs of toxicity. 9,13,62,63 Our results indicated that SWCNTs, which could induce the oxidative stress and ROS formation, were some of the main mechanisms in apoptosis of β-cells. Oxidative stress is a damaging situation which happens when there are additional ROS or a reduction in antioxidant levels. Unnecessary creation of ROS may induce cell impairment either by direct method through interaction and destruction of cellular proteins, lipids, and DNA or indirect methods by affecting normal cellular signaling ways and gene regulation. 64,65 Previous studies found that nanoparticles could induce intracellular oxidative stress by disturbing the balance between oxidant and antioxidant procedures. 66 Nel et al. 67 reported the ROS production and oxidative stress when they contacted keratinocytes and bronchial epithelial cells with SWCNTs. Similarly, we found that there was a significant increase at the levels of ROS and MDA in islets which were treated with SWCNTs. Cells also had a range of cellular protection systems including antioxidant enzymes such as SOD, GSH-Px and CAT, and low-molecular-weight antioxidants such as GSH in order to inhibit injury by ROS. SOD is generally considered as the main line of resistance against tissue and cellular impairment which is produced by ROS. It catalyzes the dismutation of superoxide anion to peroxide. CAT and GSH-Px deliver a second line of protection by dismutating peroxide into water and molecular oxygen. GSH can directly or indirectly scavenge ROS and play a key role in xenobiotic metabolism. 68 Because pancreatic islets have less antioxidant enzymes than other tissues, they are more at the risk of inducing oxidative stress. The present study investigated the activities of SOD, CAT, and GSH-Px and levels of GSH in order to determine the effects of SWCNTs on islets. Our results indicated that the activities of SOD, CAT, and GSH-Px and levels of GSH in islets were reduced after treatment with SWCNTs despite the fact that fewer influence was seen on GSH-Px activity and alteration of GSH content. Mitochondria are the main intracellular bases of ROS. Mitochondrial dysfunction can contribute to cell death by decreasing the production of ATP, increasing the production of ROS, and releasing death regulatory and signaling molecules from the intermembrane space. 69 In the present study, we also examined the effects of SWCNTs on MMP of islets. SWCNTs induced a decrease in MMP, as previous study by Grabinski et al. 70 indicated that HEL-30 cell, which was a mouse keratinocyte cell exposed to SWCNT, showed some dissipation of dye from mitochondria, but they did not determine any influence on MMP. In the present study, we indicated that SWCNTs significantly induced a decrease in MMP of islets. Therefore, the defeat of transmembrane potential of mitochondria may involve in SWCNTs-induced apoptosis of β-cells.
In the present study, we used H2O2 as a substitute for estimating the effects of ROS on islet cell function in order to compare the effects of SWCNTs on ROS. This model was also used in previous studies to evaluate the role of ROS. 71
In the present study, the exposure of mice islets to SWCNTs significantly decreased the insulin release after the addition of 5.6 and 16.7 mM of glucose mediums. Due to the vulnerability of pancreatic islets to oxidative damage, their contact to ROS can stimulate some cellular stress-sensitive ways which are associated with decreased insulin secretion. 72 A severe decrease in the concentration of decreasing equivalents is one of the first events which occur after the addition of SWCNTs. It seems that there is a relationship with KATP current and β-cell function even for mild oxidative stress. Low levels of ROS facilitate the regulation of insulin gene expression and β-cell function. 73 On the contrary, high concentration of ROS declines the insulin gene expression and insulin secretion leading to an impairment of islets. 74 Three causes namely glucose metabolism, ROS generation, and ATP production (and thus KATP channel activity) are closely related. An increase in the SWCNTs worsens ATP production and opens KATP channels which result in hyperpolarization of β-cells to limit calcium influx and glucose-induced insulin secretion. 75 ROS collection in mitochondria and ATP reduction are thoroughly associated with cell metabolism and apoptosis. 73
Our results indicated that SWCNTs as inducers of ROS decreased the insulin secretion of pancreatic islets by generating the oxidative stress. Administration of compounds with antioxidant properties could increase the protection ability of islet cells in order to deal with oxidative stress. 76
The present study also indicated that the pretreatment of SWCNTs-induced damaged islets with naringin increased the insulin secretion. Consistent with our study, Alam et al. indicated that naringin stimulated insulin secretion in MIN6 β-cells. 77 Murunga et al. also reported similar interesting effects on insulin secretion after incubation of rat islets with naringin. 78
Pretreatment of islets with naringin is likely to extent restored secretary function of islets through potentiating islet antioxidant defense system. Rates of lipid peroxidation and damage to the cell membrane were examined by evaluation of MDA and MMP levels. 79
Our findings indicated that pretreatment with naringin reduced the lipid peroxidation and MDA generation, but increased the MMP level in islet cells; and this might protect islets against SWCNTs-induced impairment.
Our results also indicated that antioxidant enzyme activities including SOD and CAT significantly increased in naringin groups compared to SWCNTs groups, and this indicated the antioxidative effect of naringin on islet cells.
It was also found that an increase in intracellular cyclic nucleotides such as cyclic adenosine monophosphate (cAMP) could decline the reactive oxygen production and oxidative stress, thereby improving the cellular dysfunction. 80 On the other hand, naringin could potentiate cell protection systems against oxidative stress probably by elevation of cAMP as well as stimulation of enzymes such as PKA. Reduction of damaging oxidative stress on islets function, viability, and ROS-induced cell death by increasing antioxidant capacity or decreasing ROS production are the promising strategies for inhibition of oxidative damage of pancreatic islets.
An important finding of this study indicated the antioxidant effect of NRG on damaged islets which are induced by SWCNTs. Its mechanism probably had two stages: NRG stimulated GSH production through an antioxidant protection mechanism against extreme ROS generation, which contributed to the inhibition of oxidative damage in addition to its effect on glycemic control 81 ; NRG protected the islets against SWCNT-induced impairment by potentiating the antioxidant defense system and suppressing the pro-inflammatory cytokine generation. 31,82
The main limitation of the present study was the loss of some intact islets at the end of isolation procedure due to the prolonged period needed for islet isolation.
Conclusion
In conclusion, the present study indicated the protective effects of naringin against SWCNT-induced oxidative damage in pancreatic islets. Based on the results of our study, this effect might be mediated by a reduction of lipid peroxidation, elevation of antioxidant enzyme activities, and increased GSH and MMP levels. However, there is a need for further studies on the detailed molecular mechanism of naringin against the effects of SWCNTs on pancreatic islets.
Footnotes
Authors’ note
The authors have seen and approved the study submitted. The manuscript has not been previously published in any language anywhere and it is not under simultaneous consideration by another journal.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This manuscript entitled “Naringin Protects Mice Pancreatic Islets from Oxidative Stress Induced by Single-Walled Carbon Nanotubes” is an investigation which was financially supported by the vice chancellor of research affairs of Ahvaz Jundishapur University of Medical Sciences. This study was labeled Student Research Project No. 95s105 and was also financially supported by the Student Research Committee of Ahvaz Jundishapur University of Medical Sciences, Ahvaz, Iran.
