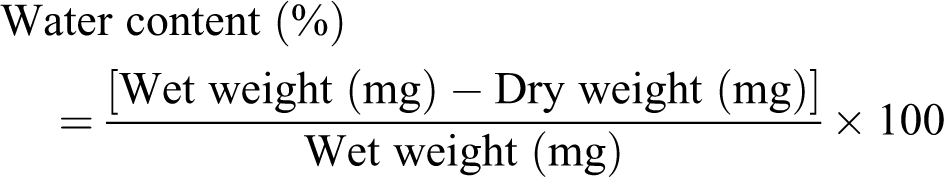Abstract
This study was designed to evaluate the hepato and neuroprotective activity of Daflon and low-dose γ radiation on thioacetamide (TAA)-induced liver damage and hepatic encephalopathy (HE) in rats. Effect of daily Daflon treatment (100 mg/kg body weight, Per OS (p.o.) for consecutive 3 days) and/or fractionated low-dose γ-radiation (LDR; 0.25 Gy, twice the total dose of 0.5 Gy at the 1st and 3rd day, respectively) was evaluated against TAA (300 mg/kg, intraperitoneal × 3) induced liver damage and HE in rats. Serum aspartate transaminase, alanine transaminase, γ-glutamyltransferase, total bilirubin, ammonia, and manganese were estimated to evaluate liver function. In addition, malondialdehyde (MDA) as well as reduced glutathione (GSH), glutathione peroxidase (GPX), superoxide dismutase (SOD), and catalase (CAT) were determined to assess antioxidant capacity in liver tissue. Moreover, hepatic apoptotic markers (cysteine-dependent aspartate-directed proteases 3, 8 (caspase-3, 8) and cytochrome C) were estimated to indicate hepatic apoptosis. HE was evaluated through the determination of whole brain ammonia, manganese, MDA, GSH, GPX, SOD, CAT, and caspase-3. The cognitive and locomotor deficits were assessed via step through passive avoidance test, activity cage (actophotometer), γ-aminobutyric acid, and N-methyl-
Keywords
Introduction
Hepatic encephalopathy (HE) is a common complication and one of the most incapacitating signs of liver disease, rigorously disturbing the lives of patients and their families. Furthermore, mental impairment accompanied with cirrhosis consequences than other signs of liver disease. HE can be defined as a brain dysfunction caused by liver ineffectuality and exhibits as a wide range of neurological abnormalities ranging from subclinical changes to coma. 1
HE is a progressed condition in liver caused by many factors like viral infections, cirrhosis caused by many toxins, and congenital diseases. These factors play a major role in the pathophysiology of HE through increased level of ammonia in brain astrocytes and glutamine accumulation, leading to oxidative stress and energy failure, which is caused by inflammation, infections, and sepsis, which cause alteration in cytokines that leads to changes in the permeability in blood brain barrier (BBB) with consequent alteration in glutamate uptake and expressions of γ-aminobutyric acid (GABA) receptors. 2
Moreover, patients with liver diseases suffer from zinc deficiency, which is important in the activities of ornithine transcarbamylase that leads to excretion of ammonia ions. In addition, cirrhotic individuals suffer from increased manganese in their basal ganglia. 3
Diabetes mellitus and insulin resistance were recently shown to be more commonly associated with the incidence of HE. 4 Due to amplified glutaminase activity in the liver, kidney, and small intestine, elevated pro-inflammatory cytokines such as tumor necrosis factor (TNF)-α and interleukin-6 result in systemic inflammatory response and upshot of protein catabolism and ammonia release. 5 All these factors leading to the progression of HE.
Daflon is a drug in the form of micronized purified flavonoids containing 90% diosmin and 10% hesperidin, which is used mainly as venotonic and a vasculoprotector and also is suggested for treating chronic venous insufficiency, hemorrhoidal diseases, and in the reduction of postoperational symptoms for hemorrhoidal disease. 6
Daflon acts by downregulation of lysosomal enzymes activities and hindering with enzymes tangled in the formation of arachidonic acid, which causes inflammation, and also proven as an antioxidant drug, which counteracts with free radicals as well as declining the synthesis of prostaglandin E-2 and thromboxane A-2 by the macrophage. 7 All these effects may lead to partial improvement in liver and brain in our HE model.
Radiation hazards present an enormous challenge for the biological and medical safety. The deleterious effects of ionizing radiation in biological systems are mainly mediated through the generation of reactive oxygen species (ROS) in cells as a result of water radiolysis 8 Nevertheless, the possibility that low doses of radiation may have beneficial effects has been the subject of considerable debate. 9 Cellular stimulatory effects are observed following low-dose γ-irradiation (LDR) at dose level range of 0.01–0.5 Gy. These effects include adaptive responses, 10 activation of immune functions, 11 enhancement of resistance to high-dose radiation, 12 regulation of mitochondrial dysfunction caused by a higher dosage of radiation, 13 and induction of endogenous antioxidant defense in the liver, 14 the latter effect could be beneficial in protecting the liver cells from oxidative stress.
In the light of the abovementioned beneficial effects of Daflon and LDR, the current study aimed to evaluate the possible ameliorative effect of Daflon and/or LDR in modulating thioacetamide (TAA)-induced acute liver failure (ALF) and HE in rats.
Material and methods
Drugs and chemicals
TAA was purchased from Sigma-Aldrich (St Louis, Missouri, USA) and Daflon from SERVIER Co. (Almaza, Cairo, Egypt), and other chemicals used were of analytical grades. Daflon was administered orally, which is suspended in isotonic saline (0.9% sodium chloride) immediately before use. Before administration, TAA was dissolved in saline at a concentration of 100 mg/mL.
Animals
Adult male Wistar albino rats weighing 150–160 g were purchased from the National Research Centre Laboratory (Dokki, Giza, Egypt) and were housed in standard polypropylene cages and kept under persistent environmental conditions with equal light–dark cycles.
Ethics statement
This experiment was carried out according to recommendations in the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health (NIH no. 85:23, revised 1996) and in compliance with regulations of the National Research Centre. All efforts were made to minimize suffering of animals.
Experimental design
Seventy five rats were divided into five main groups (n = 15), where in the first group, rats were served as the normal control group (C) and received 5 mL/kg of vehicle. In the second group, animals served as positive control (T) and administered three injections of TAA (300 mg/kg, intraperitoneal (i.p.)) at 24 h interval. 15 In the third group (Daflon-treated rats, TD), rats were administered Daflon (100 mg/kg body weight (b.w.), orally) 16 for successive 3 days; 2 h prior to TAA administration (300 mg/kg, i.p. at 24 h interval). In the fourth group (radiation-treated rats, TR), rats were exposed to fractionated LDR (0.25 Gy, twice the total dose of 0.5 Gy 17 at the 1st and 3rd day, respectively) 2 h before TAA administration (300 mg/kg) at 24 h interval. While in the fifth group (thioacetamide+daflon+radiation (TDR), rats were administered Daflon (100 mg/kg b.w., orally) for successive 3 days and exposed to fractionated LDR (0.25 Gy, twice the total dose of 0.5 Gy at the 1st and 3rd day, respectively) followed by three doses of TAA (300 mg/kg) at 24 h interval.
Exposure of rats to fractionated LDR
Whole-body γ-irradiation of rats was performed with a Canadian gamma cell-40 (137Cs) at the National Center for Radiation Research and Technology, Cairo, Egypt, at a dose rate of 0.45 Gy/min.
Behavioral experiments
Locomotor activity detection
Spontaneous motor activity was recorded by actophotometer (M 7420; Ugo Basile, Italy) for all tested groups. On the day of the test sessions, each rat was allowed to adapt the room temperature for 1 h in the polypropylene cage and then in the activity cage for half an hour. The activity measured was based on the traditional infrared photocell principle where interruption of infrared beam was recorded. A summation of photocell interferences, for each 5-min interval period, throughout the 60-min period was recorded. 18 Test sessions were held at the 1st and 3rd day (3 h post-TAA dosing) of the experiment and after 18 h of the last dose of TAA.
Step through passive avoidance paradigm
This test was done using apparatus (Ugo Basile) composed of a Plexiglas box divided into two chambers. One chamber is white and illuminated with a light fixture, containing a 24-V, 10-W bulb, attached to the chamber lid. The second chamber made up of black Perspex boards. The two chambers are divided by an automatically operated glided door. The apparatus included a steel bar grid floor, which consisted of parallel bars (0.3 cm in diameter, set 1.2 cm apart). The bars of the dark chamber floor are wired to a persistent current high accuracy eight-pole scrambling circuit linked to a controller. During the adaptation period of the test, each rat was located in the lightened chamber then when it walked into the dark chamber and settled on the grid floor, the door automatically closed and an electric shock was applied for 2 s. The exclusion of rats was based on the locomotor profile (immobility) and failing to step through within a time of 90 s. Test sessions were held at the 1st and 3rd day (4-h post-TAA dosing) of the experiment, and after 18 h of the last dose of TAA, the test was performed and the latency to step through the dark compartment was recorded as a passive avoidance behavior indicating memory acquisition. 19 Maximal cutoff time of 300 s was applied and no electric current was delivered during test sessions.
Collection of serum samples for analysis
Animals were sacrificed 24 h after treatment, and blood was collected and centrifuged (700g at 4°C for 20 min) to separate serum. Sera were used to estimate liver enzymes (aspartate transaminase (AST), alanine transaminase (ALT), and γ-glutamyltransferase (GGT)) and total bilirubin (T Bil) using colorimetric reagent kits Quimica Clinica Aplicada (Amposta, Spain) and XpressBio (Thurmont, Maryland, USA), respectively. Enzyme-linked immunosorbent assay (ELISA) technique was performed for the assessment of ammonia (Abnova, Taipei, Taiwan) according to the manufacturer’s instructions.
Tissue extracts
Animals were decapitated without anesthesia to potential influence of anesthetic drug on the brain neurotransmitters and oxidative status. After blood collection for serum parameters estimation, both liver and brain were separated out and weighed.
Assessment of liver parameters
The dissected livers were washed and homogenized in phosphate-buffered saline (PBS; 10%). Tissue homogenate was centrifuged at 5000g at 4°C for 15 min and the supernatant was stored at −20°C in aliquots. These aliquots used by ELISA kits in the estimation of cysteine-dependent aspartate-directed proteases-3 and -8 (caspase-3 and -8; Chemicon, Billerica, Massachusetts, USA), cytochrome C (Cyt C) protein (Abcam, Cambridge, Massachusetts, USA), reduced glutathione (GSH; Cayman Chemical, Ann Arbor, Michigan, USA), glutathione peroxidase (GPX; Sigma-Aldrich), malondialdehyde (MDA; Biovision, Milpitas, California, USA), superoxide dismutase (SOD) enzyme (CUSABIO, Wuhan, China), and catalase (CAT) enzyme estimated after two freeze–thaw cycles were performed to break the cell membranes, the aliquots were centrifuged for 5 min at 5000g, and then the supernatant was removed and assayed immediately (EIA ab, Wuhan, China) as described by the manufacturer’s procedures.
Assessment of brain parameters
Brains were quickly removed and cut along the medial longitudinal fissure and separated into two pieces containing brain and cerebellum hemispheres. Hippocampi were obtained by the microdissection of the medial temporal lobe. Cerebella and hippocampi were homogenized immediately in cold-buffered sucrose medium (0.25 M sucrose, 10 mM K/sodium phosphate (NaPO4), 1 mM ethylene diamino tetraacetic acid, pH 7.0). After homogenization, the crude synaptosomal fraction was obtained according to the method of Whittaker and Barker. 20 For cyclic guanosine monophosphate (cGMP) assessment, a part of the isolated cerebella and hippocampi were homogenized in a chilled solution of 0.1 N hydrochloride followed by sonication for 15 s and subsequently centrifuged at 15,000g for 15 min. One of the cerebral hemispheres (half of the brain/rat) was homogenized in cold-buffered sucrose medium and this homogenate was centrifuged at 5000g at 4°C for 15 min and the supernatant was stored at −20°C in aliquots. These aliquots were used by ELISA kits for estimation of ammonia (Abnova), aquaporin-4 (AQP-4; Uscn; Houston, USA), hypoxia-inducible factor-1α (HIF-1α; Kamiya, Seattle, Washington, USA), GSH (Cayman Chemical), GPX (Sigma-Aldrich), MDA (Biovision), matrix metalloproteinase 9 (MMP-9; CUSABIO), and SOD (CUSABIO). CAT enzyme (CUSABIO) was estimated after two freeze–thaw cycles were performed to break the cell membranes, the aliquots were centrifuged for 5 min at 5000g, and then the supernatant was removed and assayed immediately.
Assessment of cerebellar and hippocampal parameters
Supernatants obtained after cerebella and hippocampi homogenization were used to estimate GABA (EIA ab) and N-methyl-
Assessment of brain water content (cerebral edema index)
To indicate cerebral edema, the wet and dry weight method of the brain was used. After animal’s decapitation, brains were removed immediately and half of the brain was weighed before and after 48 h incubation in 100°C oven. Water content of the brain samples was measured by the wet and dry method 22 as follows:
Protein assay
The protein content in the liver and brain tissue homogenates was analyzed using the technique designated by Bradford 23 and the bovine serum albumin was used as a standard.
Determination of manganese in (serum/brain tissue) samples by flame atomic absorption
Apparatus
A Philips PU 9285 model (Philips, Germany) flame atomic absorption spectrometer equipped with deuterium lamp background correction, a hallow cathode lamp (HCL), and an air acetylene burner was used for the determination of the manganese. The instrumental parameters were as follows: wavelength, 279.5 nm; band pass, 1.0 nm; lamp current, 9.0 mA; and fuel flow rate, 1.0 L/min. All pH measurements were performed with a Jenway 3010 model digital pH meter (UK).
Reagents
Doubly distilled deionized water and analytical reagent grade chemicals were used unless otherwise specified. Manganese stock solution (1000 mg/mL) was prepared by dissolving the appropriate amount of manganese (II) sulfate. The working solutions were prepared by dilution from the stock solution. Amberlite XAD-4 (20–40 mesh, 780 m2/g; Sigma-Aldrich) was used as a substrate for the immobilization of Saccharomyces carlsbergensis. 24
Histopathological examination
Autopsy samples were taken from the liver of rats in different groups and fixed in 10% formalin saline for 24 h. Washing was done in tap water and then serial dilutions of alcohol (methyl, ethyl, and absolute ethyl) were used for dehydration. Specimens were cleared in xylene and embedded in paraffin at 56°C in hot air oven for 24 h. Paraffin bees wax tissue blocks were prepared for sectioning at 4 μm thickness by slide microtome. The obtained tissue sections were collected on glass slides, deparaffinized, and stained by hematoxylin & eosin stain for routine examination through the light electric microscope. 25
PCNA immunohistochemistry examination
All solutions according to manufacturer’s recommendation were prepared, and serial unstained paraffin slides from histology were obtained and paraffin sections cleared and rehydrated according to the steps described by Campbell-Thompson et al. 26
Then antigen retrieval using citrate buffer in a steamer at 95°C for 30 min was performed and the slides were washed with buffer for ≤5 min. Next, the slides were loaded on mouse monoclonal anti-proliferating cell nuclear antigen (PCNA) antibody PC 10Dakoautostainer (Dako, Santa Barbara, California, USA) for 15 min and rinsed. After that, the slides were incubated with the first primary anti-rabbit immunoglobulin G for 30 min and rinsed twice, then incubated with the secondary antibody (Mach 2 HRP polymer) for 30 min and rinsed twice. Diaminobenzidinetetrahydrochloride was used as chromogen for 4 min and rinsed twice. Then, slides were washed with PBS and counterstained in hematoxylin (1 min) and washed with water and allowed to air dry for 2 h. Finally, coverslips were kept over the slides using aqueous mounting media and scanned using Aperio CS scanner (Aperio, Vista, California, USA). 26
Statistical analysis
Values were stated as mean ± standard error of mean (SEM) of six to eight rats and the variances between groups were tested for significance using analysis of variance (ANOVA), followed by Tukey–Kramer post hoc test estimated by SPSS software, version 21. Correlation coefficient (r) was carried out by Pearson’s correlation test. The level of statistical significance was at p < 0.05. For locomotor activity and passive avoidance task, data were expressed as mean ± standard error and analyzed by nonparametric (passive avoidance data were expressed as medians and quartiles) Kruskal–Wallis test followed by Dunn’s test as a post hoc test.
Results
Effect of Daflon and/or LDR on locomotor activity of TAA-treated rats
Spontaneous motor activities in control and different treated groups are illustrated in Figure 1. TAA administration to rats exhibited a significant reduction (p < 0.05) in motor activity counts as revealed by nonparametric Kruskal–Wallis test followed by Dunn’s test after 51 and 69 h of treatment initiation. Pretreatment of TAA-administered rats with Daflon and/or fractionated LDR 2 h prior to TAA administration significantly alleviated (p < 0.05) locomotor activity impairment and remarkably restored motor counts when compared to TAA alone treated rats. Combination of Daflon with fractionated LDR (TDR group) achieved the superior amelioration in motor activity among all treated groups (TR and TD).
Effect of TAA and daily oral administration of Daflon and/or fractionated LDR on locomotor activity. Values are means of six to eight animals ± SEM. As compared with normal control (*) and T (#) groups (nonparametric Kruskal–Wallis test followed by Dunn’s post hoc test, p < 0.05. TAA: thioacetamide; LDR: low-dose γ-irradiation; SEM: standard error of mean; T: positive control.
Effect of Daflon and/or LDR on step through passive avoidance paradigm of TAA-treated rats
As presented in Figure 2, cognitive function of TAA-treated rats was analyzed by Kruskal–Wallis test and further by Dunn’s test revealed that administration of TAA (300 mg/kg) for three successive days resulted in a significant reduction (p < 0.05) in latency to step through as compared to control group after 52 and 70 h of treatment. Pretreatment of TAA-administered rats with Daflon and/or fractionated LDR 2 h prior to TAA administration resulted in a significantly prolonged latencies (p < 0.05) as compared to TAA alone treated rats. A substantial improvement in cognitive function was noticed in Daflon and LDR combination group (TDR) as represented by restored comparable latency with normal control group.

Effect of TAA and daily oral administration of Daflon and/or fractionated LDR on step through passive avoidance paradigm. Values are means of six to eight animals ± SEM. As compared with normal control (*) and T (#) groups (nonparametric Kruskal–Wallis test followed by Dunn’s post hoc test), p < 0.05. TAA: thioacetamide; LDR: low-dose γ-irradiation; SEM: standard error of mean; T: positive control.
Effect of Daflon and/or LDR on serum AST, ALT, GGT, ammonia, and manganese of TAA-treated rats
Administration of TAA to rats induced a significant increase in all investigated serum parameters compared to the control group. The levels of serum T Bil, manganese, and ammonia as well as GGT activity were powerfully reversed by treatment with Daflon and/or LDR prior to TAA administration. Meanwhile, AST and ALT activities significantly ameliorated in groups that treated with Daflon and/or LDR prior to TAA administration as compared to TAA alone group (Table 1; Figure 3 (a) and (b)).
The level of AST, ALT, GGT, and

AST: aspartate transaminase; ALT: alanine transaminase; GGT: γ-glutamyltransferase; T Bil: total bilirubin; C: negative control; T: positive control; TR: radiation-treated rats; TD: Daflon-treated rats; ANOVA: analysis of variance; SEM: standard error of mean; TAA: thioacetamide.
aData are expressed as means ± SEM (n = 6–8 rats). One-way ANOVA followed by Tukey–Kramer post hoc test, p < 0.05.
bSignificant difference versus control (C).
cSignificant difference versus TDR group.
dSignificant difference versus TAA (T) group.

Effect of TAA (300 mg/kg, i.p. × 3) and daily oral administration of Daflon (100 mg/kg b.w., orally) and/or fractionated LDR (0.25 Gy, twice the total dose of 0.5 Gy at the 1st and 3rd day, respectively) on the serum (a and b) levels of ammonia and manganese. Values are means of six to eight animals ± SEM. As compared with normal control (*), T (#), and TDR (@) groups (one-way ANOVA followed by Tukey–Kramer post hoc test), p < 0.05. TAA: thioacetamide; LDR: low-dose γ-irradiation; SEM: standard error of mean; i.p.: intraperitoneal; ANOVA: analysis of variance; T: positive control; b.w.: body weight.
Effect of Daflon and/or LDR on brain ammonia and manganese level of TAA-treated rats
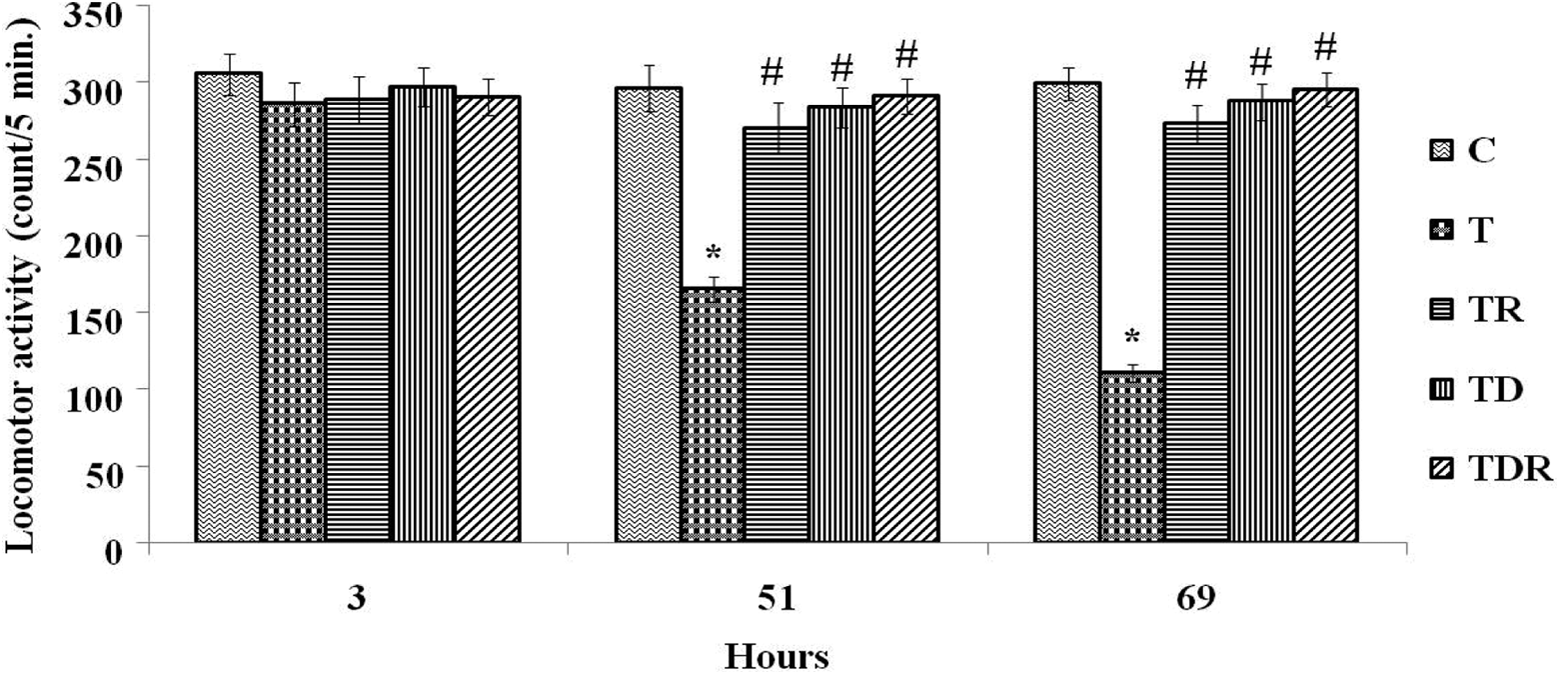
As illustrated in Figure 4, the administration of TAA to rats induced a significant increase (p < 0.05) in brain ammonia and manganese level. Pretreatment of rats with Daflon and/or fractionated LDR produced a significant reduction (p < 0.05) in brain ammonia and manganese level. Combination of Daflon and LDR (TDR group) was the most efficient treatment and exhibited the greater effect on brain ammonia and manganese level as compared to TR and TD groups.
Effect of TAA (300 mg/kg, i.p. × 3) and daily oral administration of Daflon (100 mg/kg b.w., orally) and/or fractionated LDR (0.25 Gy, twice the total dose of 0.5 Gy at the 1st and 3rd day, respectively) brain tissue (a and b) levels of ammonia and manganese. Values are means of six to eight animals ± SEM. As compared with normal control (*), T (#), and TDR (@) groups (one-way ANOVA followed by Tukey–Kramer post hoc test), p < 0.05. TAA: thioacetamide; LDR: low-dose γ-irradiation; SEM: standard error of mean; i.p.: intraperitoneal; ANOVA: analysis of variance; T: positive control; b.w.: body weight.
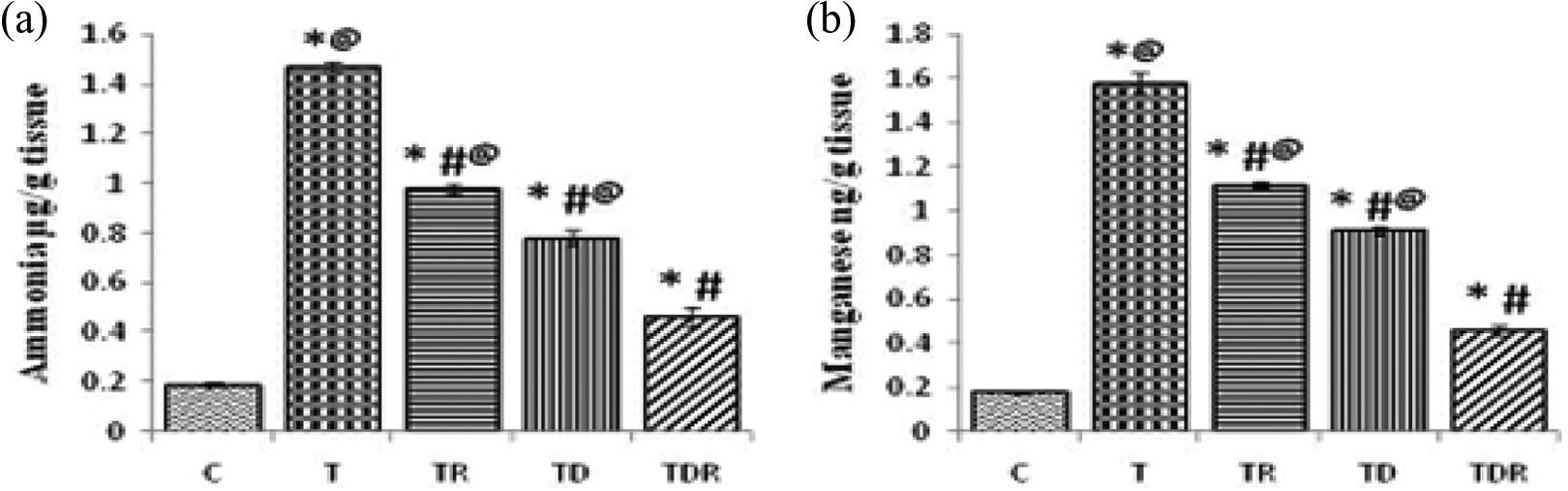
Effect of Daflon and/or LDR on liver and brain oxidative status (MDA, GSH, CAT, SOD, and GPX) and apoptotic markers of TAA-treated rats
The levels of liver tissue
The level of CAT, GSH, SOD, MDA, and GPX in liver tissue of different animal groups.a

C: negative control; T: positive control; TR: radiation-treated rats; TD: Daflon-treated rats; ANOVA: analysis of variance; SEM: standard error of mean; ANOVA: analysis of variance; CAT: catalase; GSH: reduced glutathione; SOD: superoxide dismutase; MDA: malondialdehyde; GPX: glutathione peroxidase; TAA: thioacetamide.
aData are expressed as means ± SEM (n = 6–8 rats). One-way ANOVA followed by Tukey–Kramer post hoc test, p < 0.05.
cSignificant difference versus TDR group.
dSignificant difference versus TAA (T) group.
Effect of TAA and daily oral administration of Daflon and/or fractionated LDR on the liver tissue (a, b, and c) levels of caspase-3, caspase-8, and Cyt C. Values are means of six to eight animals ± SEM. As compared with normal control (*), T (#), and TDR (@) groups (one-way ANOVA followed by Tukey–Kramer post hoc test), p < 0.05. TAA: thioacetamide; LDR: low-dose γ-irradiation; SEM: standard error of mean; ANOVA: analysis of variance; Cyt C: cytochrome C; T: positive control.
The level of CAT, GSH, SOD, MDA, and GPX in brain tissue of different animal groups.a

CAT: catalase; GSH: reduced glutathione; SOD: superoxide dismutase; MDA: malondialdehyde; GPX: glutathione peroxidase; C: negative control; T: positive control; TR: radiation-treated rats; TD: Daflon-treated rats; ANOVA: analysis of variance; SEM: standard error of mean; TAA: thioacetamide.
aData are expressed as means ± SE (n = 6–8 rats). One-way ANOVA followed by Tukey–Kramer post hoc test, p < 0.05.
bSignificant difference versus control (C).
cSignificant difference versus TDR group.
dSignificant difference versus TAA (T) group

Effect of TAA and daily oral administration of Daflon and/or fractionated LDR on the brain tissue levels of caspase-3. Values are means of six to eight animals ± SEM. As compared with normal control (*), T (#), and TDR (@) groups (one-way ANOVA followed by Tukey–Kramer post hoc test), p < 0.05. TAA: thioacetamide; LDR: low-dose γ-irradiation; SEM: standard error of mean; ANOVA: analysis of variance; T: positive control.
Effect of Daflon and/or LDR on brain HIF-1α, AQP-4, and MMP-9 of TAA-treated rats
One-way ANOVA showed a significant rise (p < 0.05) in brain HIF-1α, AQP-4, and MMP-9 of TAA-treated rats. Treatment of rats with Daflon and/or fractionated LDR significantly diminished (p < 0.05) the mentioned parameters in brain tissue as compared to TAA-treated rats. Combination of Daflon with fractionated LDR revealed the greater effect on reducing brain HIF-1α, AQP-4, and MMP-9 level when compared to TR- and TD-treated groups as shown in Figure 7.
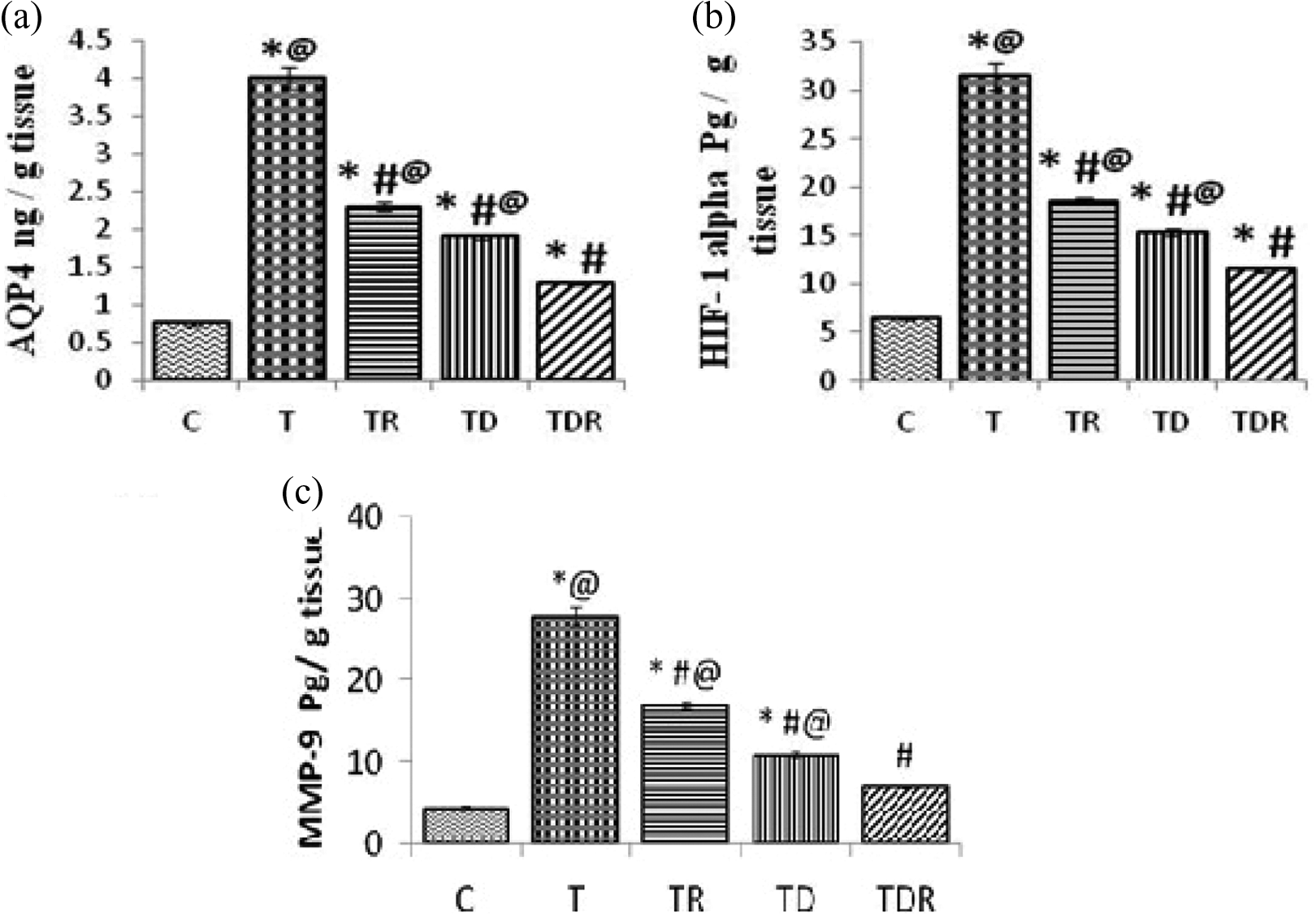
Effect of TAA and daily oral administration of Daflon and/or fractionated LDR on the brain tissue levels of AQP-4, HIF-1α, and MMP-9 (a, b, and c). Values are means of six to eight animals ± SEM. As compared with normal control (*), T (#), and TDR (@) groups (one-way ANOVA followed by Tukey–Kramer post hoc test), p < 0.05. TAA: thioacetamide; LDR: low-dose γ-irradiation; AQP-4: aquaporine-4; HIF-1α: hypoxia inducible factor-1; SEM: standard error of mean; MMP-9: matrix metalloproteinase 9; ANOVA: analysis of variance; T: positive control.
Effect of Daflon and/or LDR on brain water content
The brain water content was calculated as an index of cerebral edema. One-way ANOVA revealed a significant rise in brain water content in TAA-treated group as compared with their corresponding control. Treatment of rats with Daflon and/or fractionated LDR markedly reduced (p < 0.05) brain water content as compared to TAA-treated rats. Combination of Daflon with LDR (TDR group) indicated the superior reduction in brain water content when compared to TR and TD groups as clarified in Figure 8.
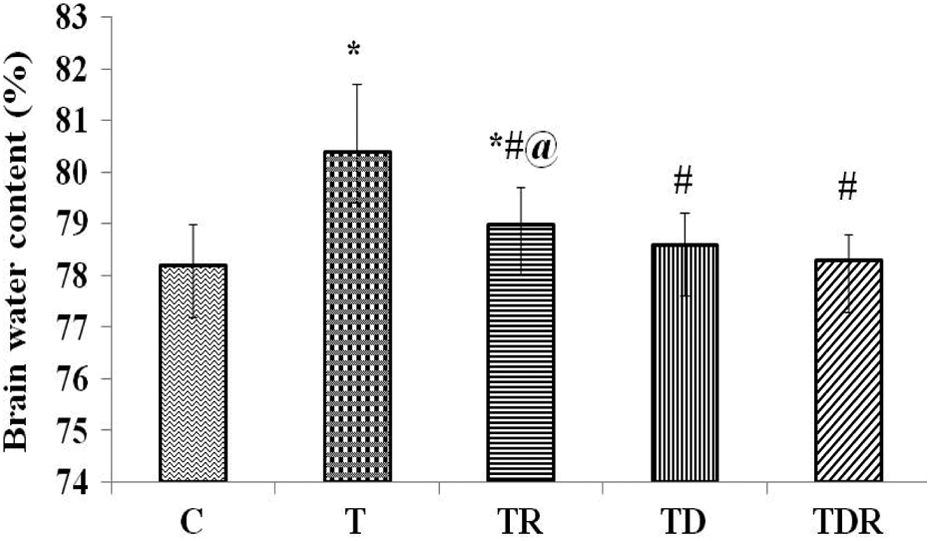
Effect of TAA and daily oral administration of Daflon and/or fractionated LDR on brain water content (%). Values are means of six to eight animals ± SEM. As compared with normal control (*), T (#), and TDR (@) groups (one-way ANOVA followed by Tukey–Kramer post hoc test), p < 0.05. TAA: thioacetamide; LDR: low-dose γ-irradiation; SEM: standard error of mean; ANOVA: analysis of variance; T: positive control.
Effect of Daflon and/or LDR on cerebellar and hippocampal GABA, NMDA, ATP, nNOS, NO, and cGMP of TAA-treated rats
TAA administration to rats showed a significant elevation (p < 0.05) in cerebellar and hippocampal GABA and NMDA when compared to their corresponding control. Meanwhile, TAA-administered rats revealed a significant reduction (p < 0.05) in cerebellar and hippocampal ATP, nNOS, NO, and cGMP level as compared to control rats. Pretreatment of TAA-administered rats with Daflon and/or fractionated LDR significantly attenuated (p < 0.05) elevated GABA and NMDA levels and remarkably restored cerebellar and hippocampal ATP, nNOS, NO, and cGMP levels as compared to TAA alone treated rats. It is obvious from the results that effect of Daflon and fractionated LDR combination is more prominent when compared to TR and TD groups as depicted in Figure 9.
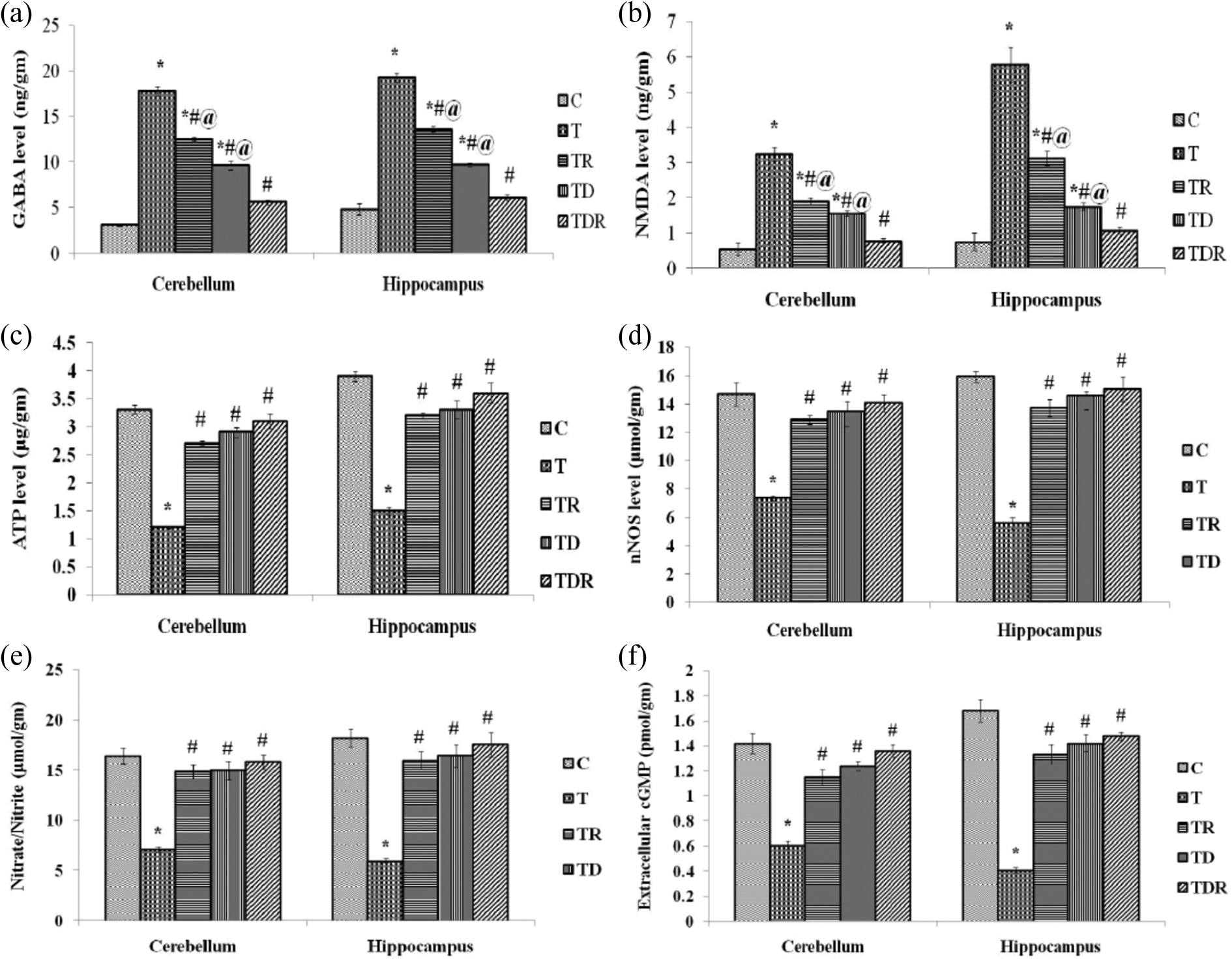
Effect of TAA and daily oral administration of Daflon and/or fractionated LDR on cerebellar and hippocampal GABA, NMDA, ATP, nNOS, NO, and cGMP (a, b, c, d, e, and f). Values are means of six to eight animals ± SEM. As compared with normal control (*), T (#), and TDR (@) groups (one-way ANOVA followed by Tukey–Kramer post hoc test), p < 0.05. TAA: thioacetamide; LDR: low-dose gamma irradiation; GABA: γ-aminobutyric acid; NMDA: N-methyl-
Correlation between HIF-1α, AQP-4, and MMP-9 with brain water content of TAA-treated rats
As depicted in Figure 10, the correlations between HIF-1α, AQP-4, and MMP-9 with brain water content as an index of cerebral edema were indicated. Using Pearson’s correlation method, brain water content was found to correlate positively with HIF-1α, AQP-4, and MMP-9 brain levels indicating the increase in brain water content as HIF-1α, AQP-4, and MMP-9 brain levels increase.

The correlation between HIF-1α, AQP-4, and MMP-9 with brain water content of TAA-treated rats. The significance of correlation was estimated by Pearson’s correlation test (significant level at p < 0.01). HIF-1α: hypoxia inducible factor-1; AQP-4: aquaporine-4; MMP-9: matrix metalloproteinase 9; TAA: thioacetamide.
Correlation between cerebellar and hippocampal NMDA level with cerebellar and hippocampal ATP and cGMP level of TAA-treated rats
As presented in Figure 11, cerebellar and hippocampal NMDA level was found to correlate negatively with cerebellar and hippocampal ATP and cGMP levels indicating cerebellar and hippocampal ATP and cGMP depletion with the increase in cerebellar and hippocampal NMDA level.

The correlation between ATP and cGMP level with NMDA level in cerebellum and hippocampus of TAA-treated rats. The significance of correlation was estimated by Pearson’s correlation test (significant level at p < 0.01). ATP: adenosine triphosphate; cGMP: cyclic guanosine monophosphate; NMDA: N-methyl-
Histopathological analysis of liver sections
Normal control livers (C) were clear of any pathological abnormality having normal histological structure of hepatic lobule which consists of central vein and concentrically arranged hepatocytes. In contrast, TAA-treated rats (T) showed loss of normal histological architecture, as represented by centrilobular necrosis with hemorrhage as well as inflammatory cell infiltration in portal area (Figure 12, T), while the livers of rats administered TAA and exposed to fractionated LDR (TR) showed dilation of central vein with ballooning degeneration and few necrosis in surrounding centrilobular zone (Figure 12, TR). In addition, liver of rats intoxicated with TAA and treated with Daflon (TD) showed ballooning degeneration with rupture of the centrilobular hepatocytes and few necrosis as well as few hemorrhage (Figure 12, TD). Interestingly, liver of rats of the combination group (TDR) revealed a marked attenuation of the histological alterations elicited by TAA administration as indicated by preserved normal architecture but with mild dilation of central vein with hemorrhage (Figure 12, TDR).

The histological examination of the liver obtained from control group (C) showed the normal histological structure of hepatic lobule which consists of CV and concentrically arranged hepatocytes (C). However, the liver of TAA (T)-treated rats revealed centrilobular necrosis (arrow) with hemorrhage (double-headed arrow) as well as inflammatory cell infiltration in portal area (T). While the liver of (TR) rats showed dilation of central vein with ballooning degeneration (arrow head) and few necrosis in surrounding centrilobular zone (TR). The liver of TD group showed ballooning degeneration (arrow head) with rupture of the centrilobular hepatocytes with few necrosis (arrow) as well as few hemorrhage (double-headed arrow; TD). The liver of (TDR) group showed preserved normal architecture but with mild dilation of central vein with hemorrhage (double-headed arrow; TDR). CV: central vein; TAA: thioacetamide; T: positive control; TR: radiation-treated rats; TD: Daflon-treated rats.
Immunohistochemical staining of liver sections
The effect of Daflon and/or fractionated LDR on cell proliferation following TAA-induced liver damage was examined by immunohistochemical analysis of PCNA expression in the liver parenchyma using anti-PCNA antibody (Figure 13). Hepatocytes of the normal control group (C) showed no PCNA staining, indicating that no cell regeneration was occurring. In contrast, hepatocytes of TAA-treated group (T) had upregulated PCNA expression with sever regenerative index, indicating proliferation to repair the severe liver tissue damage induced by TAA. Liver tissues treated with TAA and administered Daflon (TD) or exposed to fractionated LDR (TR) showed moderate regenerative index as indicated by PCNA expression, indicating reduced hepatic damage comparable to TAA alone treated group (T). Moreover, hepatocytes of rats treated with TAA and administered Daflon combined with fractionated LDR showed mild PCNA expression, indicating the marked reduction in hepatocellular damage when compared to T group as well as TR and TD groups.

Liver obtained from control group (C) showed negative immunoreactions using PCNA. However, the liver of T group showed sever positive immunoreactions. While the liver of TR and TD group showed moderate positive immunoreactions. The liver of TDR group showed mild positive immunoreactions using PCNA. PCNA: proliferating cell nuclear antigen; T: positive control; TR: radiation-treated rats; TD: Daflon-treated rats.
Discussion
The hepatotoxic ability of TAA is well described in the literature and this drug has been extensively used in rat models of ALF and HE in sequential repeated dose administration. 27 TAA administration to rats caused a significant elevation in the serum AST, ALT, and GGT activities in parallel with significant increase in serum T Bil, ammonia, and manganese level as compared to their corresponding controls. The recorded rise in liver injury biomarkers in TAA-treated rats indicates the substantial hepatic damage produced by TAA. Once in circulation, bioactivation and metabolism of TAA take place in hepatocytes by microsomal CYP2E1, which result in generation of a highly toxic metabolites. 28 The binding of these metabolites to tissue macromolecules may be responsible for the production of hepatic necrosis 29 and oxidative stress. 30 The release of hepatic enzymes (AST, ALT, and GGT) as well as bilirubin after TAA administration indicating necrotic changes that characterize liver injury. 31 Moreover, the recorded rise in serum and brain ammonia and manganese as indicated by our findings was confirmed by the results obtained by Dhorajiya and Galani 32 and Ahmed et al. 33 As a result of liver injury, the number of functional hepatocytes decreases and liver loses its capacity to remove toxic substances from the blood. Blood that bypasses the liver is not detoxified and toxic substances including ammonia, manganese, and mercaptans, which are neurotoxic, accumulated in circulation and enter the brain. 33,34 The major reason for elevated ammonia level is the disturbance in the urea cycle due to the deactivation of urea cycle enzymes. 35 In parallel with hyperammonemia, the factors responsible for increased level of manganese in blood and brain could be related to the reduced uptake of the absorbed manganese by liver as a result of impaired microcirculation and intrahepatic shunting; also, decreased biliary excretion plays a role in manganese overload. 36
According to the obtained results, pretreatment of rats with Daflon and/or irradiation of rats with fractionated LDR ameliorated liver injury elicited by TAA and recorded a significant decrease in serum AST, ALT, and GGT activities as well as markedly reduced T Bil, ammonia, and manganese serum and brain levels, suggesting that it offers protection by preserving the structural integrity of the hepatocellular membrane against hepatotoxins due to its flavonoids constituents. 37 These results are in harmony with those obtained by Abdel-Salam et al., 38 Hozayen et al., 39 and Fouad et al. 40 In addition to its membrane-stabilizing efficacy, Germoush 41 reported that hesperidin supplementation significantly reduced ammonia level in hyperammonemic rats. This might be attributed to the improvement of hepatic microcirculation by diosmin. 42 Therefore, diosmin–hesperidin complex maintains hepatic blood flow via preserving hepatic microcirculation, and consequently improves the hepatic detoxifying capacity. Concerning the hepatoprotective efficacy of LDR, the obtained results are in agreement with previous studies. 17,43,44 The ameliorative effect of LDR might be correlated to LDR-induced hormetic response in cells 45 and the ability of LDR (up to 0.5 Gy) to stimulate resistance of cells to oxygen radicals’ toxicity, which is reflected on improved liver enzymes.
A disruption in redox balance in liver and brain tissues was observed in TAA-challenged rats when compared to control as indicated by a significant rise in lipid peroxidation marker (MDA) associated with marked suppression in SOD, GPX, and CAT activities and GSH level. The provoked oxidative stress by TAA through generation of ROS and reactive nitrogen species which stimulates lipid peroxidation was confirmed by the studies of Murthy et al. 46 and Túnez et al. 47 Generation of these toxic metabolites after TAA metabolism resulted in the denaturation of cellular biomolecules such as lipids, resulting in lipid peroxidation and its by-product. 48 The significant decrease in liver and brain SOD, GPX, and CAT activities and GSH level after TAA challenge as revealed by our data is in accordance with the findings of Ahmed et al. 33 Pretreatment of rats with Daflon and/or fractionated LDR reduced MDA and boosted antioxidant defense mechanisms (GSH, GPX, SOD, and CAT) in liver and brain of treated rats. The exhibited effect of Daflon suggests its role in scavenging free radicals generated by TAA, which might be attributed to its flavonoids content. 38 The radical scavenging and antioxidant enhancement properties of hesperidin were reported in several studies. 40,49 The antioxidant activity of hesperidin seems to be correlated to its structure. 50 The antioxidant activity of LDR as revealed in our study is in consistence with those obtained by Mansour, 43 Fahmy et al., 51 Rashed et al., 17 and El-Ghazaly et al. 52 who reported that exposure of rats to whole-body LDR restored endogenous GSH, SOD, and CAT and reduced MDA to a significant level. These effects may be due to the enhancement of antioxidant enzymes activities resulting from induction of their synthesis following LDR. 53,54 The proposed mechanism by which LDR restores GSH liver and brain level might be attributed to the ability of LDR to stimulate thioredoxin (TRX) activity on cellular level. 55 TRX is not only a potential endogenous antioxidant but also contributes to the biosynthesis of GSH by promoting cystine transport into cells and is also a key protein for the control of cellular redox status. 56
TAA administration to rats induced a significant rise in hepatic caspase-3 and -8 activities and Cyt C level as compared to control suggesting its proapoptotic activity. The proapoptotic activity of TAA could result from a combination of both pathways: intrinsic apoptotic pathway by generation of oxidative stress and the extrinsic apoptotic pathway by activation of kupffer cells that can secrete TNF-α 57 which was evident by hepatocellular degenerative changes as represented by centrilobular necrosis as indicated by histopathological examination of liver sections. This effect was confirmed and supported by Ahmed et al. 33 In the same line, PCNA, a prominent marker for TAA-induced hepatotoxicity, 58 showed a marked upregulation in TAA-treated group, indicating extensive proliferation, likely in an attempt to repair TAA-induced tissue damage. 59 The anti-apoptotic efficacy of Daflon as revealed by the obtained results might results from antioxidant and anti-inflammatory effect of diosmin–hesperidin complex. These effects are compatible with the results obtained by Tamilselvam et al. 60 and Arab et al. 61 The mechanism that might explains the suppressive effect of hesperidin on caspase-3, caspase-8, and Cyt C is associated with the upregulation of peroxisome proliferator-activated receptor-γ and downregulation of nuclear factor-κB. 62 Also, diosmin might augment the anti-apoptotic effect of hesperidin through downregulation of p53, Bax, and caspase-3 expression 63 and upregulation of Bcl-2 protein. 61 Interestingly, exposure of rats to LDR attenuated TAA-induced hepatocellular apoptosis as revealed by downregulation of caspase-3, caspase-8, and Cyt C. These findings are in harmony with the observations of Bogdándi et al. 64 and Rashed et al. 17 LDR can increase Bcl-2/ BAX ratio 65 and maintain mitochondrial membrane potential (Δψm) via stimulation of cyclin D1/BAX complex formation, which results in inhibition of apoptosis by LDR. 66 The anti-apoptotic effect of Daflon and LDR was confirmed by histopathological examination of liver sections. Moreover, downregulation of PCNA expression by Daflon and/or LDR treatment may be attributed to reduced damage comparable to TAA alone treated group.
The current data revealed that TAA administration to rats elevated cerebellar and hippocampal GABA and NMDA levels as compared to control rats. This effect of TAA on brain GABA level was reported by Najmi et al. 67 In ALF-associated HE, ammonia may indirectly increase GABA-ergic neurotransmission by the increased access of GABA in the synaptic clefts and loss of presynaptic feedback inhibition of the GABA release, which is the result of decreased GABAB receptor function. 68 Moreover, hyperammonemia can modify the function of NMDA receptors and of some associated signal transduction pathways. 69 The stimulation of ammonia-induced NMDA receptors reduces antioxidant enzyme activity and increases the production of superoxide anions. 70 Excessive activation of NMDA receptors leads to neuronal degeneration and death. 69 The enhanced NMDA receptors stimulation might explain the reduced antioxidant enzymes activities and increased caspase-3 activity in the brain as indicated by our findings. The impairment of the cognitive and locomotor function in TAA-challenged rats in our study as indicated by reduced motor activity counts and step through latency in the behavioral tests was proved by different studies. 32,71 This effect might be correlated to excessive GABA and NMDA activation in cerebellum and hippocampus, since they are involved in motor performance and learning functions that are dominantly impaired in HE. 72 Ammonia-induced activation of NMDA receptors would lead to glutamate release due to sodium-dependent uptake of glutamate reversion as a result of ATP depletion, 73 which results in impaired glutamate–NO–cGMP necessary for learning and cognitive function. 74 The improvement exhibited by Daflon and/or LDR in behavioral tests as revealed by our findings might be due to restoration of NMDA–ATP–nNOS/NO–cGMP axis in cerebellum and hippocampus. This effect owing to the enhanced liver detoxifying capacity which is reflected on decreased blood and brain ammonia. The mechanism might be involved in restoring NMDA–NO–cGMP pathway by Daflon is related to the ability of hesperidin–diosmin complex to restore neuronal ATP level. 75,76 In addition, LDR stimulates ATP release and restores the redox balance via activation of purinergic (P2X and P2Y) receptors 77 that are located in cerebellum and hippocampus. 78 Moreover, constitutive NOS activation is an early cytoprotective response to radiation that results in enhanced cGMP formation. 79
Our findings showed a significant increase in HIF-1α, MMP-9, and AQP-4 brain levels in positive correlation with brain water content of TAA-administered rats as compared to their respective controls, suggesting that TAA-induced cerebral edema is mediated via HIF-1α/MMP-9/AQP-4 pathway and disturbance of the BBB in the peri-astrocytic capillaries. Higashida et al. 80 reported that HIF-1α plays a role in brain edema formation and BBB disruption via a molecular pathway cascade involving MMP-9 and AQP-4 after a traumatic brain injury. Administration of Daflon and/or exposure of rats to fractionated LDR markedly mitigated HIF-1α/ MMP-9/AQP-4 pathway cascade as implied by the significant decrease in their brain level suggesting their beneficial effects in treating vasogenic brain edema. This effect is attributed to the inhibition of HIF-1α by Daflon (diosmin–hesperidin) 81,82 and downregulation of MMP-9 expression by hesperidin. 83 The ameliorative effect of LDR on HIF-1α/MMP-9/AQP-4 pathway is attributed to ability of LDR to enhance endogenous antioxidants 84 and downregulation of MMP-9. 85 These effects might contribute to the protective effect of LDR against ROS-induced endothelial dysfunction and inhibition of proteolytic cleavage of tight junction proteins by MMP-9. In addition, pre-exposure of aged rats to LDR suppressed AQP-4 expression and prevented brain edema induced by cerebral ischemia. 86
Conclusions
Taken together, Daflon and/or LDR treatment exhibited a hepatoprotective and neuroprotective activity against TAA-induced ALF and HE. These effects might be attributed to antioxidant and anti-apoptotic effects. The neuroprotective effect of Daflon and LDR against TAA-induced HE is due to the modulatory effect of both treatments on the inhibitory neurotransmitter (GABA) as well as restoration of NMDA–ATP–nNOS/NO–cGMP axis in cerebellum and hippocampus, which is reflected on the recovery of cognitive and locomotor function. In addition, inhibition of HIF-1α/MMP-9/AQP-4 pathway cascade might contribute to brain edema. The emergence of pharmacological blockade of this pathway in patients with ALF-associated HE may provide a novel therapeutic strategy in the future and alleviate brain edema that complicates HE.
Footnotes
Acknowledgments
The authors are thankful to Prof. Adel Kholoussy, Department of Pathology, Faculty of Veterinary Medicine, Cairo University, Egypt, for the kind help in histopathology and immunohistochemistry.
Author Contributions
All authors of the current manuscript contributed equally to accomplish different parts of this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.