Abstract
This study aims to investigate the diagnostic and prognostic relevance of MMP-2 and MMP-9 as biomarkers for breast cancer, as well as their association with clinicopathological factors. Breast cancer is a leading contributor to cancer-related deaths among women worldwide. The discovery of biomarkers is crucial for early diagnosis, outcome prediction, and effective treatment. Matrix metalloproteinases (MMPs) play a significant role in various physiological and pathological activities, including development, tissue repair, inflammation, cancer spread, and metastasis. While the prognostic significance of MMP-2 and MMP-9 levels in breast cancer has been studied, the findings remain inconclusive. Participants were divided into three groups, with each group consisting of 62 individuals: Group I comprised healthy controls, Group II consisted of newly diagnosed breast cancer patients (stage I-III), and Group III included patients with metastatic breast cancer. Levels of MMP-2 and MMP-9 were evaluated in these groups using the ELISA method. An evident increase in MMP-2 and MMP-9 levels was noted when comparing the control group with both the breast cancer and metastatic groups. Furthermore, a notable correlation was identified between serum MMP-9 levels and the pathological diagnosis of breast cancer (
Introduction
Breast cancer is the most common cancer worldwide, with a rising incidence in recent years. In 2020, there were over 2.3 million new cases and 685,000 deaths attributed to breast cancer. Incidence rates differ by region, with certain Asian and African nations reporting less than 40 cases per 100,000 women. 1 In Egypt, breast cancer accounts for over 32% of cancer cases in women, with projections indicating a further increase by 2050. 2 Additionally, breast cancer contributes to 29.1% of cancer-related deaths in the country. 3
Despite progress in treatment and the introduction of screening initiatives for early detection, breast cancer continues to be a significant factor in female mortality. Ongoing endeavors are focused on discovering biomarkers that can improve the diagnosis and prognosis of breast cancer. 4 The advancement of cancer and its invasive nature, which are primary reasons for treatment challenges, are associated with a sequence of molecular alterations in cancer cells. 5
Matrix Metalloproteinases (MMPs) are a group of enzymes essential for breaking down the extracellular matrix (ECM), which affects tissue restructuring. Among these enzymes, MMP-9 is significant for breaking down ECM proteins such as collagen, which influences tissue organization and affects the invasion, movement, and advancement of cancer cells.6,7 The activity of MMP-9 is controlled by different biochemical substances, and its presence in breast cancer has been examined concerning patient prognosis.8,9
MMP-2, a member of the gelatinase subgroup, is another important matrix metalloproteinase to consider. It has the ability to break down type IV collagen found in the basement membrane. The activities of both MMP-2 and MMP-9 play a role in promoting cancer metastasis and angiogenesis by breaking down the extracellular matrix and activating pro-angiogenic factors. This association with unfavorable prognosis has been documented. 10
Prior research on MMP-2 and MMP-9 in breast cancer shows divergent findings, with certain studies indicating a link between increased expression and unfavorable outcomes, while others do not observe such a connection. 11 In light of these inconsistencies, the objective of this study is to evaluate the diagnostic and prognostic significance of MMP-2 and MMP-9 in Egyptian breast cancer patients, investigating their relationship with clinicopathological characteristics.
Subjects and methods
Characteristics of subjects
The study was conducted at the Baheya Foundation for Early Detection and Treatment of Breast Cancer in Giza, Egypt, and included 186 adult women recruited between March 2022 and December 2022. Participants were categorized into three groups: Group I (62 healthy women as controls), Group II (62 women with non-metastatic breast cancer), and Group III (62 women with metastatic breast cancer), with all groups carefully matched by age to ensure comparability. Ethical approval was granted by the Ethical Committee of the Baheya Research Center (IRB202204260015, 16, 19, 20), and the study adhered to the ethical principles outlined in the Declaration of Helsinki. Informed written consent was obtained from all participants prior to enrollment.
Breast cancer patients (metastatic or non-metastatic)
Measurement of serum MMP-2 and MMP-9
We used Sandwich Enzyme-Linked Immunosorbent Assay (ELISA) kits (SG10407 & SG10412, respectively; Sino Gene Clone Biotech Co., Ltd.) to measure human MMP-2 and MMP-9 serum levels as per the manufacturer’s instructions. Absorbance at 450 nm was determined with an automatic microplate spectrophotometer (Multiskan FC Microplate Photometer, Thermo Fisher Scientific). The average absorbance of the reference standards was used to generate a standard curve, and the corresponding concentration was calculated for each sample based on this curve.
Statistical analysis
Statistical analysis was conducted utilizing the SPSS software (version 20). The sample size for this study was calculated using Epi Info software, 12 based on the comparison of mean serum MMP-2 levels between the breast cancer group and the healthy control group. The mean serum MMP-2 level in the breast cancer group was estimated at 806.50, while in the healthy control group, it was 771.17, with a pooled standard deviation of 59.94. 13 To detect a statistically significant difference at a significance level of 0.05 and with 80% power, 45 participants per group are required. Additionally, this sample size is sufficient to detect a significant difference in serum MMP-9 levels, with a mean of 371.83 ± 47.10 in the breast cancer group and 272.50 ± 41.56 in the healthy control group. 13 This ensures that the study has adequate power to identify meaningful differences in both MMP-2 and MMP-9 levels between the groups.
Quantitative data estimates were presented as mean and standard deviation, while median and range were utilized when appropriate. Qualitative data was displayed in terms of frequency and percentage. The Mann-Whitney test was employed to compare non-normally distributed quantitative data between two independent groups, whereas the Kruskal-Wallis test was used for comparisons involving more than two groups. The Chi-square test was utilized to assess the association between qualitative variables, with Fisher’s exact test applied for 2 × 2 qualitative variables when more than 25% of cells had an expected count below 5. Correlation analysis was performed to determine the strength of association between numerical variables. Receiver Operator Characteristic (ROC) analysis was used to establish cut-off levels, and sensitivity, specificity, and predictive values were calculated. A
Results
Patient’s characteristics
The research involved 186 women aged between 22 and 83 years. The prevalence of hypertension, diabetes, and the use of hormonal contraception was notably higher in the groups of breast cancer patients compared to the healthy control group (
Levels of MMP-2 and MMP-9 in serum
The levels of both MMP-2 and MMP-9 were higher in the serum of both non-metastatic and metastatic breast cancer patients compared to that of healthy controls (
Patients’ general characteristics and clinicopathological features of the disease.

Comparison of MMP-2 and MMP-9 concentrations among the study groups.

Concentration was expressed in ng/ml. P1 = comparison between healthy controls & non-metastatic groups. P2 = comparison between healthy controls & metastatic groups. P3 = comparison between non-metastatic & metastatic group.
ROC curve analysis
In the current study, the analysis using ROC curves demonstrated that both MMP-9 and MMP-2 can serve as significant parameters for distinguishing between healthy females and those with non-metastatic breast cancer. The area under the curve for MMP-9 was found to be 0.935, and for MMP-2, it was 0.988. Using a cut-off value of 259.0 ng/mL, MMP-9 exhibited a sensitivity of 92.7% and a specificity of 87.1%, while MMP-2 at cut-off value 2.28 ng/mL, showed a sensitivity of 96.4% and a specificity of 91.1% (Figure 1(a)). Moreover, the AUC values for predicting metastatic breast cancer were 0.913 for MMP-9 and 0.99 for MMP-2, with a sensitivity of 90.2% and a specificity of 80.6% at a cut-off point of 229.0 ng/mL for MMP-9. Conversely, MMP-2 achieved a sensitivity of 95.1% and a specificity of 90.3% at a cut-off point of 2.25 ng/mL (Figure 1(b)). Additionally, both serum levels of MMP-9 and MMP-2 demonstrated limited predictive value in distinguishing between patients with metastatic and non-metastatic breast cancer, as indicated by the AUC values of 0.588 (95% CI, 0.484–0.691) and 0.506 (95% CI, 0.400–0.613), respectively (Figure 1(c)).
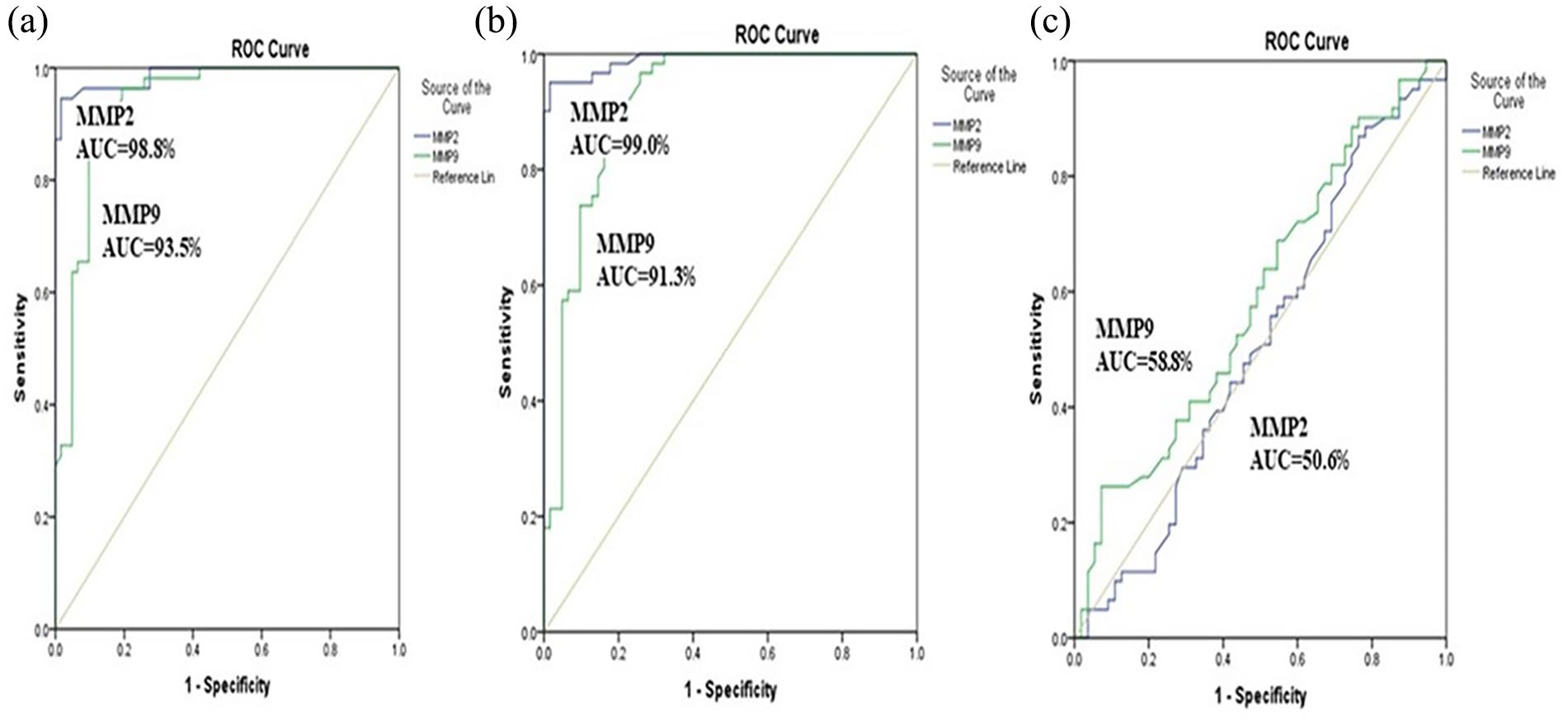
Receiver operating characteristic curves for prediction capacity of MMP-2 and MMP-9: (a) non-metastatic versus healthy controls, (b) metastatic versus healthy controls, (c) non-metastatic versus metastatic.
Association of MMP-9 and MMP-2 expression with clinicopathological characteristics
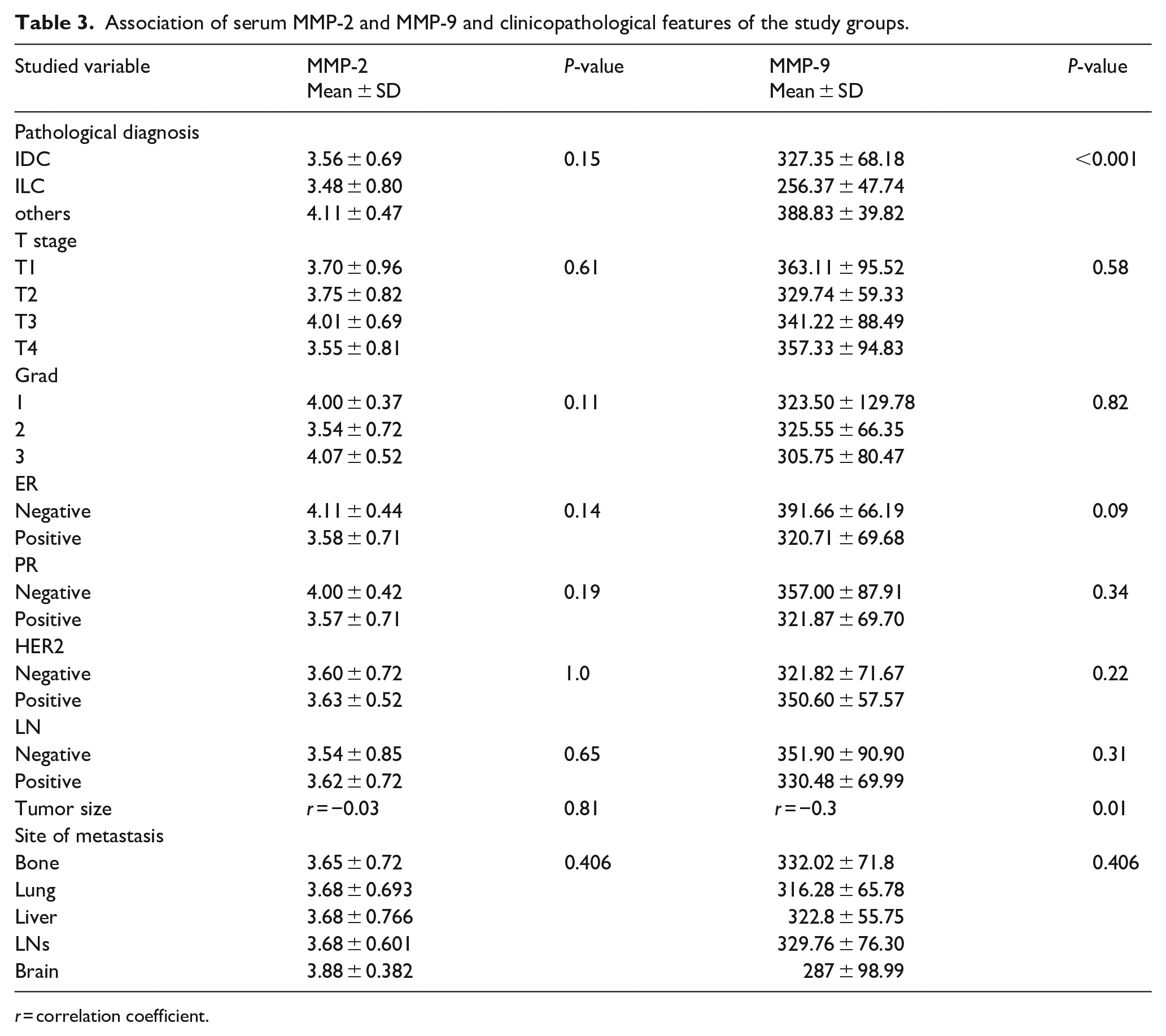
Analysis of MMP-2 and MMP-9 levels in relation to clinicopathological factors of breast cancer patients indicated a notable correlation between serum MMP-9 and both the pathological diagnosis of breast cancer (
Association of serum MMP-2 and MMP-9 and clinicopathological features of the study groups.
Discussion
Breast cancer comprises a varied range of tumors characterized by different morphological and molecular subtypes, which makes predicting disease progression and patient outcomes challenging. Discovering novel biomarkers is essential for customizing the most effective treatments for each patient. Recent research has investigated liquid biopsy and circulating proteins, obtainable from serum or plasma, as biomarkers for cost-effective, minimally invasive risk evaluation, early detection, prognosis, treatment modifications, and monitoring disease advancement. 14 Several experimental studies have shown the role of MMPs in the inception, progression, staging, and grading of tumors. 15
This research focused on examining the presence of human MMP-2 and MMP-9 in the blood samples of individuals with breast cancer, as blood is a readily obtainable bodily fluid. The enzyme-linked immunosorbent assay (ELISA) technique was employed to quantify the levels of total MMP-2 and MMP-9. Our findings demonstrate markedly higher levels of circulating MMP-9 and MMP-2 in all breast cancer patients when compared to the healthy control group (
In analogous research conducted among Egyptian individuals, notably higher levels of mRNA and protein expression of MMP-2 and MMP-9 were observed in cancerous breast tissue in comparison to healthy tissue. Additionally, a notable reduction in average plasma levels of MMP-2 and MMP-9 was observed after the removal of breast carcinoma, indicating their potential as indicators for successful tumor eradication. 21
These findings are in alignment with the expected increase in biomarker levels attributed to their specific involvement in tissue remodeling associated with cancer. MMP-2 (gelatinase-A) and MMP-9 (Gelatinase-B) play a role in collagen restructuring by breaking down the extracellular matrix (ECM), which includes elastin, fibronectin, and vitronectin. They also influence functions beyond the ECM, such as activating pro-TNF-α and transforming growth factor-beta (TGF-β). These gelatinases are predicted to be elevated because of their unique contribution to cancer-related tissue remodeling, affecting processes like tumor cell proliferation, migration, invasion, and metastasis. 22
Metastatic processes often involve the activation of a program that transforms tumor epithelial-mesenchymal cells (EMT), which is triggered by cytokines and factors released by various cells within the tumor microenvironment. Concurrently, there is a breakdown of the extracellular matrix (ECM) due to the production of matrix metalloproteinases (MMPs). This research did not find any significant differences in serum levels of both MMP-2 and MMP-9 between the group of malignant breast cancer (BC) patients and those with metastatic BC (
In terms of the relationship between serum MMP-2 and MMP-9 levels with clinicopathological variables and standard prognostic factors, no significant correlation was found between MMP-2 serum levels and any clinicopathological characteristics. Previous research has demonstrated conflicting associations with clinicopathological parameters such as patient age, tumor grade, tumor receptor status, and disease stage in breast cancer patients. Some studies have indicated elevated serum MMP-2 levels in patients with advanced tumor stages, while others have shown decreased MMP-2 levels in patients with unfavorable prognostic factors.16,24,25,26 Conversely, there is ongoing debate regarding whether MMP-2 positivity is associated with poorer overall survival in breast cancer patients. 27
Building on the results, there was a notable connection between serum MMP-9 levels and both the histological type of the tumor (
Furthermore, elevated mRNA levels of MMP-9 have been identified and specifically linked to Invasive Ductal Carcinoma (IDC) in breast cancer. 31 Research involving 113 individuals with non-palpable breast abnormalities revealed notably higher MMP-9 levels in women with invasive ductal carcinoma, indicating its potential as a biomarker. 32 Additional studies have explored the MMP-9 expression in IDC patients, showing differing levels of association with lymph node metastasis or tumor size. 33 This discrepancy could be attributed to variations in experimental techniques and sample sizes.
The debate regarding the link between metalloproteinase expression in blood or tissue and clinical results in different malignant tumors, such as breast cancer, is intricately tied to the regulation of MMPs. This includes factors like mRNA expression, the conversion of the pro-enzyme form into an active state, and the opposing effects of endogenous tissue inhibitors of metalloproteinases (TIMPs). Initially, MMPs are synthesized as zymogens (inactive Pro form) and are activated through various molecular processes. As a result, the levels of active MMP-9 in stromal cells and tumor cells can vary, leading to differences in clinical outcomes. 34
Genetic diversity plays a role in determining MMP expression levels, impacting the susceptibility of individuals to cancer. Research has explored the relationship between MMP-9 and MMP-2 genetic variations and the risk of breast cancer in different populations, yielding contradictory findings.33,35–40 Moreover, discrepancies may stem from variations in methodologies and sampling procedures prior to the quantitative assessment of the specific biomarkers.
Moreover, research has indicated that introducing the MMP-9 gene into existing breast cancer tumor cells can lead to tumor shrinkage by enhancing neutrophil infiltration and activating tumor-associated macrophages (TAM), demonstrating the potential antitumor properties of MMP-9. 41 The antitumor effects of MMPs offer a solid foundation for targeted therapy in clinical settings and hold promise for advancing clinical treatment and prognostic outcomes in the future. 14
Limitation
A key limitation of the study is the use of a case-control design instead of a prospective cohort approach to assess the prognostic impact of the biomarkers. While this design allows for efficient comparisons between metastatic and non-metastatic groups, it does not facilitate longitudinal follow-up, limiting our ability to establish causal relationships and evaluate how biomarker levels may influence progression to metastasis over time. Despite this limitation, our findings provide valuable preliminary insights that should be explored further in prospective studies to confirm their prognostic significance.
Conclusion
Elevated levels of MMP-9 and MMP-2 in the bloodstream of breast cancer (BC) patients indicate their involvement in the remodeling of cancer-associated tissues. The increased presence of MMP-9 in the serum of BC patients is linked to the type of cancer and the size of the tumor, impacting patient survival. This highlights MMP-9 as a potentially valuable prognostic indicator for advanced-stage BC in Egyptian individuals.
Recommendation
Nevertheless, additional extensive studies are required to validate these results. Furthermore, the simultaneous assessment of MMP-9 in both serum and tissue may offer a more effective indication of cancer aggressiveness compared to serum levels alone.
Supplemental Material
sj-docx-1-iji-10.1177_03946320241304911 – Supplemental material for Prognostic impact of matrix metalloproteinase 2 (MMP-2) and matrix metalloproteinase 9 (MMP-9) in Egyptian breast cancer patients
Supplemental material, sj-docx-1-iji-10.1177_03946320241304911 for Prognostic impact of matrix metalloproteinase 2 (MMP-2) and matrix metalloproteinase 9 (MMP-9) in Egyptian breast cancer patients by Fayrouz A Fouad, Mohamed A Khali, Inas Moaz, Hossam Elmasry, Nada Gheta, Asala Abdeen, Mariam Tantawi, Ganna Elkholy, Shaimaa Rihan, Mahmoud M Kamel, Ayman EL-Meghawry EL-Kenawy, Youssef AS Abdel-Moneim and Abdallah M Gameel in International Journal of Immunopathology and Pharmacology
Footnotes
Acknowledgements
We would like to extend our heartfelt thanks to Doaa Elsayed Abo Kresha for her invaluable assistance in data collection. Her dedication and support played a crucial role in the successful completion of this study. The authors extend their appreciation to Taif University, Saudi Arabia, for supporting this work through project number (TU-DSPP-2024-278).
List of abbreviation
Breast cancer (BC), Matrix metalloproteinases (MMPs), epithelial-mesenchymal transition (EMT), extracellular matrix (ECM), metalloproteinase-2 (MMP-2), Triple Negative Breast Cancer (TNBC), Enzyme-linked Immunosorbent Assay (ELISA), immunohistochemistry (IHC), transforming growth factor-beta (TGF-β), Invasive Ductal Carcinoma (IDC), epithelial-mesenchymal cells (EMT), tumor-associated macrophage (TAM), inhibitors of metalloproteinases (TIMPs).
Authors’ contributions
FF, MK, IM, MK, YA, AEE, & AG contributed to the project preparation, study design, submission for protocol and writing draft of the manuscript. HE, MK, & FF were responsible for the management of purchasing tasks and schedules. NG, MK, MT, & GI coordinated specimen collection and transport and implemented a quality policy throughout the laboratory analysis workflow. HE, NG, MK, MT, GI, AEE, and SR contributed to laboratory analysis. Appropriate patient selection and data collection were performed and supervised by FF, HE, YA, and MK. Statistical analysis of data and tabulation of results were accomplished by YA, IM, & SR. All authors have read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors have reviewed and approved the final version of the manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by Taif University, Taif, Saudi Arabia (TU-DSPP-2024- 278).
Ethical approval
The study was conducted in accordance with the ethical guidelines established in the Declaration of Helsinki and received approval from the ethical committee of the Baheya Foundation for Early Detection & Treatment of Breast Cancer (IRB202204260015,16,19,20). Prior to enrollment, all participants provided written informed consent after receiving a thorough explanation of the study’s objectives, procedures, and the potential risks and benefits of their involvement. Participants were assured of the voluntary nature of their participation, and confidentiality of their data was strictly maintained throughout the study.
Informed consent
Informed written consent was obtained from all participants after the study objectives were explained and before blood sampling. Confidentiality of patient data was guaranteed.
Trial registration
*Not applicable.
Availability of data and materials
All data and materials are available and can be submitted when needed, Corresponding Author is responsible person who should be contacted if someone wants to request the data from this study.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
