Abstract
The effect of 21 days of repeated oral administration of levofloxacin and enrofloxacin both alone and in combination with meloxicam, on the oxidative balance in blood was evaluated in rabbits. Rabbits were randomly allocated to six groups of four animals each. Control group was gavaged 5% dextrose and 2% benzyl alcohol. Three groups were exclusively gavaged meloxicam (0.2 mg/kg body weight o.d.), levofloxacin hemihydrate (10 mg/kg body weight b.i.d 12 h), and enrofloxacin (20 mg/kg body weight o.d.), respectively. Two other groups were co-gavaged meloxicam with levofloxacin hemihydrate and enrofloxacin, respectively. A reduction (p < 0.05) of reduced glutathione levels was observed in groups treated with meloxicam both alone and in combination with levofloxacin, whereas an increase (p < 0.01) in the levels of this antioxidant was observed in the groups treated with enrofloxacin. The activities of enzymes, glutathione peroxidase and superoxide dismutase, were induced (p < 0.05) in levofloxacin-alone treated group. Superoxide dismutase was also induced (p < 0.05) in meloxicam-alone treated group and inhibited (p < 0.05) in enrofloxacin-meloxicam co-treated group. The activity of catalase was non-significantly different between various groups. Enrofloxacin-treated groups had higher (p < 0.01) lipid peroxidation than control and levofloxacin-alone treated groups. Elevated lipid peroxidation was also observed in the groups treated with meloxicam both alone and in combination with levofloxacin (p < 0.05). In conclusion, these drugs have potential to induce oxidative imbalance, however, compared to levofloxacin, more oxidative damage is produced by enrofloxacin and meloxicam.
Introduction
Levofloxacin and enrofloxacin are fluoroquinolone (FQ) antimicrobial drugs with a broad spectrum of antibacterial activity. 1 These bactericidal drugs inhibit the bacterial enzymes DNA gyrase and topoisomerase IV. Non-steroidal anti-inflammatory drugs (NSAIDs) are widely used drugs in both veterinary and human medicine for various inflammatory conditions of infectious or noninfectious origin. 2 NSAIDs act by inhibiting the pro-prostaglandin enzymes, cyclooxygenase 2 (COX-2) and cyclooxygenase 1 (COX-1). The COX-2 inhibition is considered to mediate the therapeutic actions of NSAIDs, while the COX-1 inhibition usually results in unwanted side-effects, particularly those of gastrointestinal tract. 2 Meloxicam is a NSAID that has an apparently greater selectivity toward inhibition of inducible COX-2 isoform than the constitutive isoform COX-1. 2
Pharmaceutical use has been reported as one of the leading causes of hospitalizations and the third leading cause of deaths. 3 Although FQs are relatively safer drugs, their adverse effects such as gastrointestinal discomfort, hepatotoxic reactions, central nervous system effects, juvenile joint toxicity, and phototoxic and retinopathic effects, although rare, are also documented. 4,5 FQ-induced phototoxicity and cartilage damage and a part of their antibacterial activity have been attributed to the production of reactive oxygen species (ROS). 3 Although NSAIDs including meloxicam can improve oxidative imbalance, 2,6 alteration of antioxidant levels has been reported with several NSAIDs 7 demonstrating oxidative stress as the mechanism of their toxicity. 8
Treatments of complicated infections require weeks of antibacterial therapy and a protracted course of NSAIDs to manage the associated inflammatory conditions like pyrexia and pain. 9,10 Taking the above facts into consideration, the dubious nature of meloxicam on the oxidative balance and the lack of sufficient toxicological data regarding the simultaneous exposure of FQs and NSAIDs, we evaluated levofloxacin, enrofloxacin, and meloxicam for their effect on the antioxidant status/oxidative imbalance that could give some insight of their basic toxicological profile.
Materials and methods
Animal experimentation
Twenty-four male adult Soviet Chinchilla rabbits were kept under normal ambient conditions, with an average day length of 11.5 h, in a well-lightened experimental house. The animals were maintained on standard rabbit feed and ad libitum water. The experimental protocol was approved by the University Animal Ethics Committee vide order No. VMC/13/17/86-1806 dated April 4, 2013. Rabbits were randomly allocated to six groups of four animals each. As 5% dextrose and 2% benzyl alcohol are present as excepents in the drug preparations of meloxicam, levofloxacin, and enrofloxacin used in the treatment groups of this study, group A, serving as control, received these chemicals in prerequisite amounts. Group B received meloxicam (Melonex®, Intas, Ahmedabad, Gujrat, India) at 0.2 mg/kg body weight o.d. orally. Group C received levofloxacin hemihydrate (Levoflox®, Cipla, Mumbai, Maharashtra, India) at 10 mg/kg body weight b.i.d. orally for 12 h. Group D was co-gavaged levofloxacin and meloxicam at above dose rates. Group E received enrofloxacin (Enrocin™, Pfizer Animal Health, India) at 20 mg/kg body weight o.d. orally. Group F was co-gavaged enrofloxacin and meloxicam at the above dose rates. The dose of levofloxacin was extrapolated from the therapeutic dose in humans, 11 whereas meloxicam and enrofloxacin were used at their therapeutic dose in rabbits. 12 Meloxicam and enrofloxacin were administered every morning, whereas one dose of levofloxacin was administered at this same time, and the other dose was given at 12 h after the first dose. Each rabbit was dosed according to its individual body weight and weighed weekly so that necessary corrections in the total dosage of drugs or vehicle administered could be made. Rabbits were restrained manually on the morning of days 0, 7, 14, and 21 of treatment, and blood samples (1.5 ml) were collected from the ear vein using separate heparinized scalp vein needle (24 gauge) set. Time 0 d refers to the day prior to start of first dose of drugs, whereas the blood samples collected on days 7, 14, and 21 were obtained approximately 12 h after the last dose of levofloxacin or 24 h after the last dose of meloxicam or enrofloxacin. After obtaining the blood samples on days 7 and 14, the next corresponding dose was administered.
Blood biochemical analysis
Blood glutathione
To 0.2 ml of whole blood in 1.8 ml of distilled water, 3 ml of the precipitating solution (glacial metaphosphoric acid) was added and the mixture was allowed to stand for approximately 5 min and then centrifuged at 3000 rpm for 15 minutes. 13 To 2 ml of supernatant, 8 ml of 0.3 M phosphate solution (Na2HPO4·2H2O) and 1 ml of 5-5′-dithiobis-(2-nitrobenzoic acid) (DTNB) reagent were added. The absorbance was recorded at 412 nm and the concentration of glutathione (GSH) was determined from the standard curve of GSH in distilled water.
Superoxide dismutase
To a cuvette, 1.5 ml of 100 mM tris(hydroxymethyl)aminomethane–hydrochloride buffer, 0.5 ml of 6 mM ethylenediaminetetraacetic acid, and 1 ml of 0.6 mM pyrogallol solution were added. 14 The rate of auto-oxidation of pyrogallol was taken from the increase in absorbance at 420 nm, for 4 min after every 30 s lag. This constitutes the reading for blank. For the test, an appropriate amount of enzyme (in 20 µl of 10% erythrocyte lysate) was added to inhibit the auto-oxidation of pyrogallol to about 50%. A unit of enzyme activity is defined as the amount of enzyme producing 50% inhibition in the auto-oxidation of pyrogallol observed in blank.
Glutathione peroxidase
To 0.1 ml 10% erythrocyte lysate, 1 ml of 20 mM GSH, 1 ml of sodium phosphate buffer (0.4 M, pH 7), and 0.5 ml of sodium azide were added and the volume was made 4 ml with distilled water. 15 After 5 min preincubation, 1 ml of hydrogen peroxide (H2O2; pre-warmed to 37°C) was added. One milliliter aliquots of incubation mixture were removed after 1 min interval and added to 4 ml precipitation solution (m-phosphoric acid). GSH in protein-free filtrate was determined by mixing 2 ml of filtrate with 2 ml of 0.4 M disodium phosphate and 1 ml of DTNB reagent and the absorbance was recorded at 412 nm within 2 min after mixing. The zero time GSH concentration was determined in the same manner using an aliquot from a sample treated similarly, except that H2O2 was replaced with water.
where, C0 is the concentration of GSH at zero time and C is the concentration of GSH after 1 min incubation. The activity of GPx is expressed as EU/mg Hb
Catalase
In a cuvette, 20 µl of 10% erythrocyte lysate was mixed with 2 ml of phosphate buffer (50 mM, pH 7.0). 16 The reaction was started by the addition of 1 ml of 30 mM H2O2 and the decrease in absorbance was recorded at every 10 s interval for 1 min at 240 nm. The results of catalase (CAT) activity were expressed as micromoles of H2O2 decomposed per minute per gram protein using 36 as molar extinction coefficient (EC) of H2O2.
Lipid peroxidation
To 1 ml of 33% packed cell erythrocytes, 1 ml of 10% w/v trichloroacetic acid was added and vortexed. 17 The mixture was centrifuged at 3000 r/min for 10 min. The solution of 1 ml supernatant and 1 ml 0.67% w/v 2-thiobarbituric acid (TBA) was kept in boiling water bath for 10 min. The final reaction mixture was cooled and diluted with 1 ml of distilled water. The absorbance was read at 535 nm and the results were expressed as nanomolesmalondialdehyde (MDA) formed per milliliter erythrocytes. The molar EC of MDA–TBA complex at 535 nm is 1.56 × 108/M/cm.
Statistical analysis
All these assays were carried out in triplicate. The average value for each triplicate result for an animal represented the individual observation from the animal. The statistical significance of the difference between group means on 0, 7, 14 and 21st day, based on individual observations was determined by analysis of variance carried out in completely randomized design with Duncan’s multiple range test 18 using IBM SPSS Statistics® 20 software. The significance was assayed at 1% (p < 0.01) and 5% (p < 0.05) levels.
The results are expressed in bar graphs/figures. The comparative height of the bars on a particular day indicates the comparative magnitude/value of the parameter for various groups on that day. Bars on a particular day not sharing a common labeling symbol (π, ¥, and β) or letter (a, b, c, and d) differ significantly at p < 0.01 and p < 0.05, respectively. Numeric value of any parameter can be calculated from the graphs by marking the point at which straight line drawn from the maximum height of bar intercepts the Y-axis (parameter axis).
Results
Reduced glutathione
As recommended by the University’s Animal Ethics Committee, the two groups used in this study, control and meloxicam-alone group, were also common groups for our study on pazufloxacin mesylate since the two studies were conducted simultaneously. The study on pazufloxacin mesylate is published in Journal of the American Association for Laboratory Animal Science. 3 The permission for presenting data of these two groups in this manuscript has been granted by the Journal of the American Association for Laboratory Animal Science. The results for the effect of repeated oral administration of levofloxacin, enrofloxacin, and meloxicam on GSH level in rabbits are presented in Figure 1. On the 7th day of treatment, the GSH levels in all the treatment groups were non-significantly different from the control group. However, the levels in levofloxacin–meloxicam co-treated group and the group treated with enrofloxacin alone were higher (p < 0.05) than the meloxicam-alone treated group. On 14th day of treatment, the GSH levels in enrofloxacin-treated groups were higher (p < 0.01) than other groups. On the 21st day of treatment, the GSH levels of the groups treated with meloxicam, both alone and in combination with levofloxacin, were lower (p < 0.05) than control group; whereas the GSH levels in enrofloxacin treated groups remained high (p < 0.01).

Effect of repeated oral administration of levofloxacin, enrofloxacin, and meloxicam on the reduced GSH levels (nmol/l) in rabbits. Results represent mean ± SEM. Bars on a particular day not sharing a common labeling symbol (π and ¥) or letter (a, b, and c) differ significantly at p < 0.01 and p < 0.05, respectively. GSH: reduced glutathione.
Glutathione peroxidase
Figure 2 presents results for the effect of repeated oral administration of levofloxacin, enrofloxacin, and meloxicam on GPx activity in rabbits. Although the levofloxacin-alone treated group showed higher (p < 0.05) GPx activity than other groups on the 21st day of treatment, the GPx activity differed non-significantly between various groups throughout the study period.
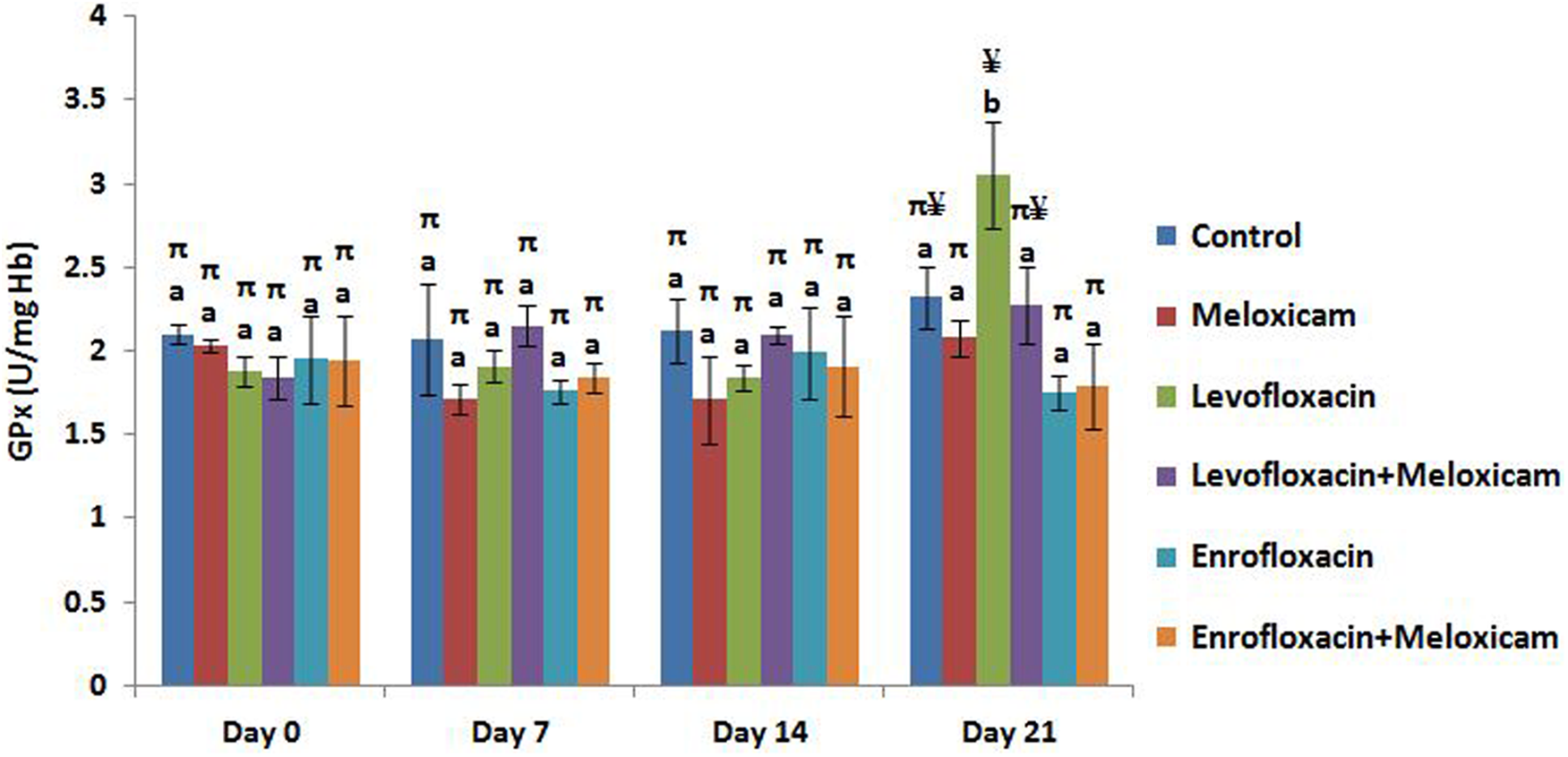
Effect of repeated oral administration of levofloxacin, enrofloxacin, and meloxicam on the activity of GPx (U/mg Hb) in rabbits. Results represent mean ± SEM. Bars on a particular day not sharing a common labeling symbol (π and ¥) or letter (a and b) differ significantly at p < 0.01 and p < 0.05, respectively. GPx: glutathione peroxidase; Hb: hemoglobin.
Superoxide dismutase
The results for the effect of repeated oral administration of levofloxacin, enrofloxacin, and meloxicam on superoxide dismutase (SOD) activity in rabbits are presented graphically in Figure 3. When compared with other groups, except the enrofloxacin–meloxicam co-treated group, the SOD activity in the levofloxacin-alone treated group was higher (p < 0.05) on the 7th day of treatment. On the 14th day of treatment, the SOD activity of the groups treated with meloxicam and levofloxacin, both alone and in combination, was higher (p < 0.05) than enrofloxacin–meloxicam co-treated group. On the 21st day of treatment, the SOD activity of levofloxacin-alone treated group was higher (p < 0.05) than control group and the groups treated with enrofloxacin, with the enrofloxacin-meloxicam co-treated group showing SOD activity statistically lower than all other groups (p < 0.01). Meloxicam-alone treated group had higher (p < 0.05) SOD activity than control and enrofloxacin–meloxicam co-treated group.

Effect of repeated oral administration of levofloxacin, enrofloxacin, and meloxicam on SOD activity (U/g Hb) in rabbits. Results represent mean ± SEM. Bars on a particular day not sharing a common labeling symbol (π and ¥) or letter (a, b, c, and d) differ significantly at p < 0.01 and p < 0.05, respectively. SOD: superoxide dismutase; Hb: hemoglobin.
Catalase
The results for the effect of repeated oral administration of levofloxacin, enrofloxacin, and meloxicam on CAT activity in rabbits are presented graphically in Figure 4. On the 14th day of treatment, CAT activity in the groups treated with meloxicam alone was higher than other groups (p < 0.05). Although CAT activity of enrofloxacin–meloxicam co-treated group was lower (p < 0.05) than meloxicam-alone treated group, its values for this enzyme were higher than levofloxacin-treated groups (p < 0.01) and enrofloxacin-alone treated group (p < 0.05). However, on the 21st day of treatment, the activity of this enzyme did not differ statistically between various groups.

Effect of repeated oral administration of levofloxacin, enrofloxacin, and meloxicam on catalase activity (micromoles of H2O2 decomposed per minute per milligram Hb) in rabbits. Results represent mean ± SEM. Bars on a particular day not sharing a common labeling symbol (π, ¥, and β) or letter (a, b, and c) differ significantly at p < 0.01 and p < 0.05, respectively. CAT: catalase; H2O2: hydrogen peroxide; Hb: hemoglobin.
Lipid peroxidation
Figure 5 presents the results for the effect of repeated oral administration of levofloxacin, enrofloxacin, and meloxicam on MDA levels in rabbits. Higher (p < 0.05) MDA level than control group was observed in the meloxicam-alone treated group on 14th day of treatment. On the 21st day of treatment, enrofloxacin-treated groups had higher (p < 0.01) MDA levels than control and levofloxacin-alone treated groups. Elevated (p < 0.05) lipid peroxidation was also observed in the groups treated with meloxicam both alone and in combination with levofloxacin.
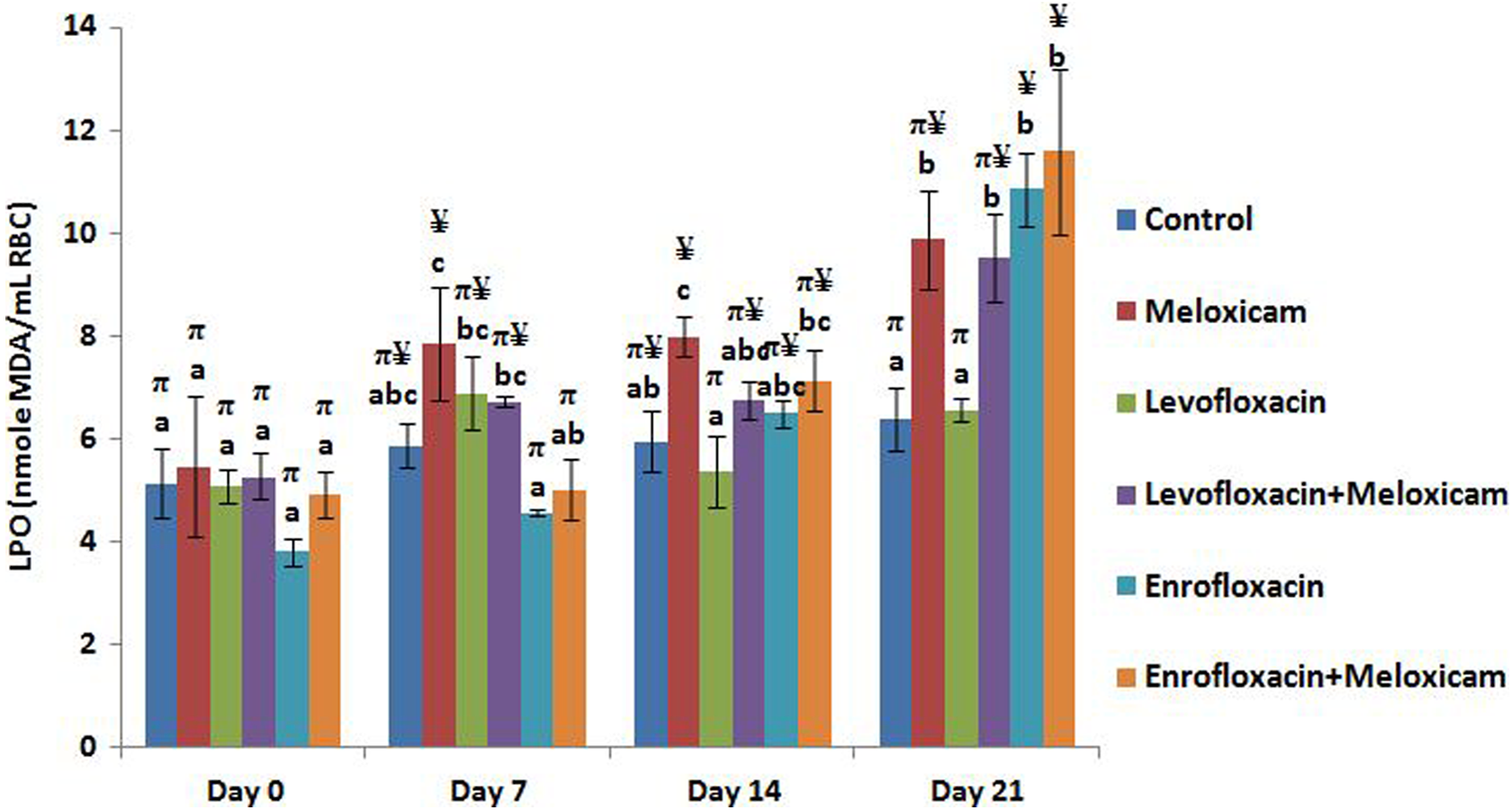
Effect of repeated oral administration of levofloxacin, enrofloxacin, and meloxicam on the MDA levels (nanomoles of MDA per milliliter RBC) in rabbits. Results represent mean ± SEM. Bars on a particular day not sharing a common labeling symbol (π and ¥) or letter (a, b, and c) differ significantly at p < 0.01 and p < 0.05, respectively. MDA: malondialdehyde; RBC: red blood cell.
Discussion
Irrespective of the source, oxidative stress has been implicated in the mechanism of several diseases. 2,4,19,20 Defining oxidative stress as an imbalance between pro- and antioxidant mechanisms has led to the use of indicators of each side of the balance with ROS/reactive nitrogen species as pro-oxidant markers, antioxidants as markers of antioxidant balance and lipid peroxidation, and protein and DNA oxidation as markers of oxidative damage. 21 However, due to non-specificity of MDA assay, 22 other markers like endogenous antioxidant GSH and various antioxidant enzymes are also considered to determine oxidative stress. 21
GSH, a potent reductant, acts as the major intracellular antioxidant that participates in the detoxification of toxic peroxides, maintenance of protein SH groups, and elimination of xenobiotics. Depleted GSH levels have been reported on exposures with several FQs 4,23 and meloxicam. 7 GSH depletion, considered an early hallmark in the progression of cell death from various apoptotic stimuli, renders cells or tissues more susceptible to oxidative injury. 21
SOD is the first and major line of defense against·O2 – and other ROS. 24,25 Superoxide is rapidly dismutated to H2O2 by manganese–SOD (SOD2), in the mitochondrial matrix, and CuZn–SOD (SOD1) in the intermembrane space and cytosol. 21 Final detoxification of mitochondrial superoxides occurs by conversion of H2O2 to H2O by CAT and GPx at high and low H2O2 concentrations, respectively. 21 GPx catalyzes the detoxification of a wide range of peroxides using GSH as reducing equivalent. 26 The induction of GPx activity has also been reported with ofloxacin, 4 whereas a non-significant change of GPx activity is reported with meloxicam. 7
Metabolism of xenobiotics is an energy-requiring process with energy being provided primarily by ATP synthesized from mitochondrial electron transport chain. During this energy transduction ROS are also produced with biological outcome governed by the direction of equilibrium for their production and detoxification. 27 An initial response of body to ROS is the expression of nuclear factor erythroid 2–related factor 2, a transcription factor that increases the expression of genes mediating response against stressors. 28 This might be a reason for the increased activity of SOD (7th and 21st day) and GPx (21st day) in the levofloxacin-alone treated group. Similar reasons could be ascribed to the increased activity of CAT (14th day) and SOD (21st day) in meloxicam-alone treated group and increased GSH levels from 14th day of treatment in enrofloxacin-treated groups.
At the end of the study period, the activity of SOD in levofloxacin–meloxicam co-treated group and enrofloxacin-alone treated group remained non-significantly different from control group. Similarly, the activity of GPx in all treatment groups, except levofloxacin-alone treated group, differed non-significantly from the control group. The CAT activity of all treatment groups was also at par with the control group. The probable reason for the non-significantly different values of antioxidant enzymes might be due to the simultaneous induction and excessive utilization of these enzymes in an exacerbated production of free radicals, 29 which could not provide a sufficient protection to these groups against lipid peroxidation. In levofloxacin-alone treated group, GSH levels and CAT activity remained statistically unaltered from the control group whereas the activity of antioxidant enzymes SOD and GPx was significantly induced in this group. This might have conferred an almost adequate protection to this group against oxidative damage as is evident from the non-significantly altered MDA values of this group from the control group value. Increased lipid peroxidation with FQs has been reported by several studies. 4,29,30 Rofecoxib, a COX-2-selective NSAID, has also been reported to predispose human low-density lipoprotein and cell membrane lipids to oxidative modification. 31 An in vitro study revealed that NSAIDs may increase cardiovascular risk by inducing oxidative stress in the vasculature, with nonselective NSAIDs demonstrating a greater oxidative stress than coxibs. 8 The extent of lipid peroxidation indirectly reflects the degree to which bio-membrane lipids are attacked by free radicals.
Both increase and decrease in the expression or activity of antioxidant enzymes can be rationalized to indicate oxidative stress. 21 However, consistently increasing exposure of pro-oxidative xenobiotics overwhelms the antioxidant status by inhibiting the activity or sufficient synthesis of antioxidants, which predisposes cells to oxidative damage. 32 Increased lipid peroxidation cross-links antioxidant enzymes with MDA that inactivates these enzymes, which results in an increased accumulation of ROS and aggravation of macromolecular damage. 33 Comparison of these parameters between different treatment groups indicates that the oxidative damage is more with enrofloxacin and meloxicam, when compared to levofloxacin. Since levofloxacin is eliminated, unchanged, by renal excretion compared to meloxicam and enrofloxacin that require hepatic metabolism, a more energy-requiring and ROS-generating process. 34,35 This becomes evident from the similar extent of peroxidative damage in the groups treated with enrofloxacin, meloxicam, and levofloxacin–meloxicam.
As antioxidants in every scenario are not the universal antidotes for oxidative stress, similar studies with therapeutic intervention of various antioxidants need to be carried out to formulate the ameliorative strategies against oxidative damage-mediated adversities produced by these drugs. The results from the current study indicate that repeated oral administration of levofloxacin, enrofloxacin, and meloxicam produces oxidative stress as indicated by alteration in some antioxidant parameters. However, enrofloxacin and meloxicam produces more lipid peroxidative damage compared to the exclusive administration of levofloxacin.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The current research has been carried out at the Department of Pharmacology and Toxicology, GADVASU, Ludhiana. The study is a part of the research project, “Evaluation of retinopathic potential of FQs” assigned by University Grants Commission, India, to the Department of Veterinary Pharmacology and Toxicology, GASVASU, Ludhiana, under letter no. FNo. 40-279/2011(SR). Financial assistance to the project has been made by University Grants Commission, India. As an ethical measure to reduce the overall number of animals used, the University Animal Ethics Committee recommended control and meloxicam-alone treated groups to be used in common for this study and our study on pazufloxacin mesylate that is published in Journal of the American Association for Laboratory Animal Science. 3 The two studies were conducted simultaneously and the permission for presenting data of these groups in this manuscript has been granted by the publishing journal. The authors declare that they have no competing interests.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors received financial support for the research from University Grants Commission, India vide letter no. FNo. 40-279/2011(SR).

