Abstract
Three hundred one-day-old avian broilers were divided into four equal groups of 75 animals that were fed for 42 days as follows: a control diet containing 23 mg fluorine (F)/kg and three high F diets containing 400, 800, and 1200 mg F/kg, respectively, for high F groups I, II, and III. The superoxide dismutase (SOD) and glutathione peroxidase (GSH-Px) activities were greatly decreased, while the malondialdehyde (MDA) contents were markedly increased in high F groups II and III. At the same time, mitochondrial injury and expanded endocytoplasmic reticulum were obviously observed in high F groups II and III, and the fluoride contents both in spleen and serum were significantly increased in the three high F groups when compared with those of control group. The results showed that excess dietary F in the range of 800−1200 mg/kg caused obvious oxidative stress, which provided a possible pathway for the apoptosis of splenocytes in chickens.
Introduction
Fluoride is ubiquitous in varying amounts in food and water and is an air pollutant from numerous industrial operations. 1 Fluoride is a chemically active ionized element. It can affect oxygen metabolism and induce the production of O2− free radicals. 2 Previous studies revealed that fluoride induced excessive production of oxygen free radicals and caused a decrease in biological activities of some antioxidant enzymes. 3 There have been reports about the fluoride-induced oxidative stress in brain, 4 liver, kidney, erythrocytes, 5 –7 and osteoblasts 8,9 in rats so far. However, there were no relevant researches about the impact of fluoride on the oxidative damage of splenocytes in chickens. The spleen represents the largest lymphoid tissue 10 and is widely considered to be of vital importance in the whole immune function of the body. In our recent study, 11 we found that excess dietary F in the range of 800−1200 mg/kg caused histological lesions and cellular apoptosis of the spleen in chickens. Moreover, oxidative stress has been demonstrated to cause apoptosis. 12 So, in order to reveal a possible reason for the increased apoptosis of splenocytes stated in our previous paper, the present study aimed to assess the antioxidant status of spleen by detecting the antioxidase (SOD, GSH-Px) activities and malondialdehyde (MDA) contents.
Materials and methods
Three hundred one-day-old healthy avian broilers were divided into four equal groups of 75 animals and fed on diets as follows: control group (23 mg fluorine [F]/kg) and high F group I (400 mg F/kg); high F group II (800 mg F/kg); and high F group III (1200 mg F/kg). The broilers were housed in electrically heated cages and were provided with drinking water (F ≤ 1 mg/L) as well as the aforementioned diets ad libitum for 42 days. Nutritional requirements were adequate according to the US National Research Council (NRC 1994).
At 14, 28, and 42 days of the experiment, five chickens in each group were phlebotomized from jugular vein to obtain serum. Then, euthanasia was used and the spleens were taken immediately. Spleens were then made into tissue homogenate and centrifuged. After determining the amount of total protein in the supernatant of the spleen-homogenate and the serum by the method of Bradford, 13 the SOD, GSH-Px activities, and MDA contents in the splenic supernatant and serum were measured by biochemical method following the instruction of reagent kits. The reagent kits for measurement of MDA contents and SOD and GSH-Px activities were provided by Nanjing Institute of Jiancheng Biological Engineering.
Moreover, fluoride in spleen and serum was measured with a CSB-F-I fluoride ion electrode at 42 days as described by Hall at al. 14 At the same time, spleens of four chickens in each group were dissected and then prefixed with a mixed solution of 4% paraformaldehyde and 2.5% glutaraldehyde, followed by softening in 3% EDTA solution for 20 min. The tissue was then fixed in 1% osmium tetroxide, dehydrated in series with acetone, infiltrated in Epox 812 for 2 hr, and then embedded in epoxy resin. Ultrathin sections were cut with glass knives, stained with uranyl acetate and lead citrate and examined with a TEM H-600 electron microscope (Hitachi, Japan).
Finally, data are expressed as mean ± standard deviation (SD). The significance of difference among four groups was analyzed by analysis of variance. The analysis was done with SPSS 12.0 for windows as described in our previous report. 11
Results
As showed in Table 1 and Table 2 , the splenic SOD and GSH-Px activities were significantly lower (p < 0.01) in high F group I than in control group at 28 days treatment. However, the splenic and serum SOD as well as GSH-Px activities were obviously decreased (p < 0.01) in high F groups II and III during the experiment. At the same time, the splenic and serum MDA contents were markedly increased in high F group I at 28 days treatment, and greatly increased in high F groups II and III compared with those of control group during the experiment, as described in Table 3 . The splenic and serum fluorine contents were increased in a dose-dependent manner in high F groups, as shown in Table 4 .
Change of the splenic and serum SOD activities a

Abbreviation: SOD: superoxide dismutase.
a Data are means ± SD (n = 5).
b Compared with the control group, p < 0.01, (t-test).
Change of the splenic and serum GSH-Px activities a
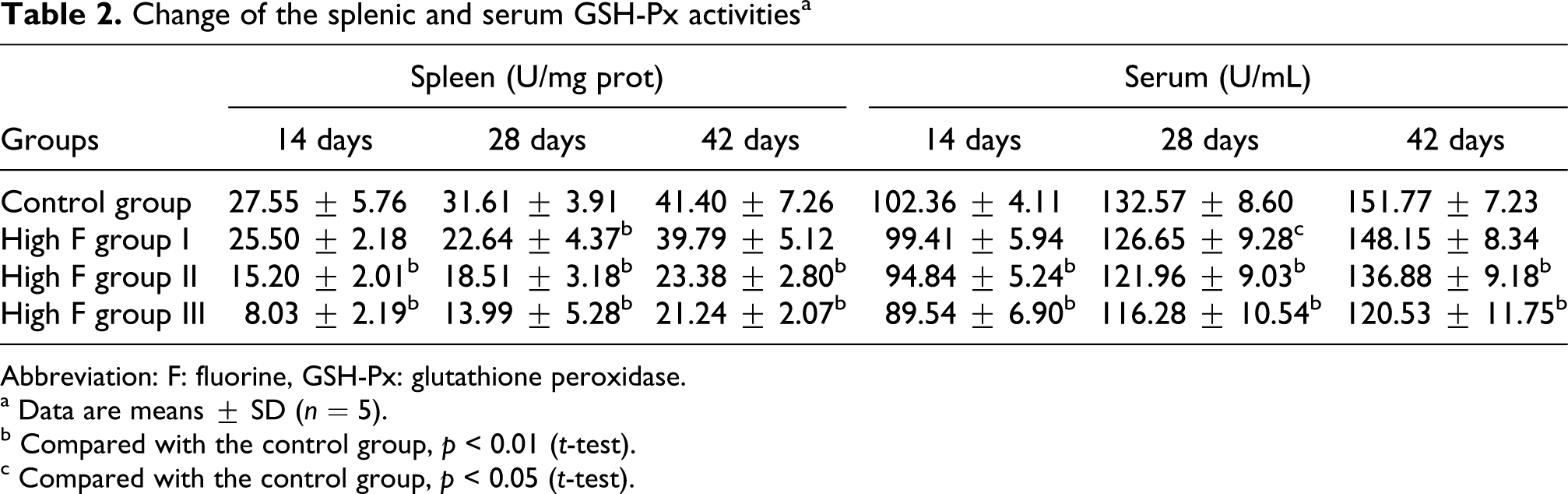
Abbreviation: F: fluorine, GSH-Px: glutathione peroxidase.
a Data are means ± SD (n = 5).
b Compared with the control group, p < 0.01 (t-test).
c Compared with the control group, p < 0.05 (t-test).
Change of the splenic and serum MDA contents a

Abbreviation: MDA: malondialdehyde.
a Data are means ± SD (n = 5).
b Compared with the control group, p < 0.01 (t-test).
c Compared with the control group, p < 0.05 (t-test).
Change of the splenic and serum fluorine contents a

a Data are means ± SD (n = 5).
b Compared with the control group, p < 0.01 (t-test).
Ultrastructurally, when compared with those of control group (Figure 1), dilated endoplasmic reticulum and enlarged mitochondria with its cristae broken/absent of splenocytes were obviously observed in high F groups II and III (Figures 2 and 3). Moreover, the occurrence frequencies of apoptotic splenocytes were much higher in high F groups II and III (Figure 4) than in control group.
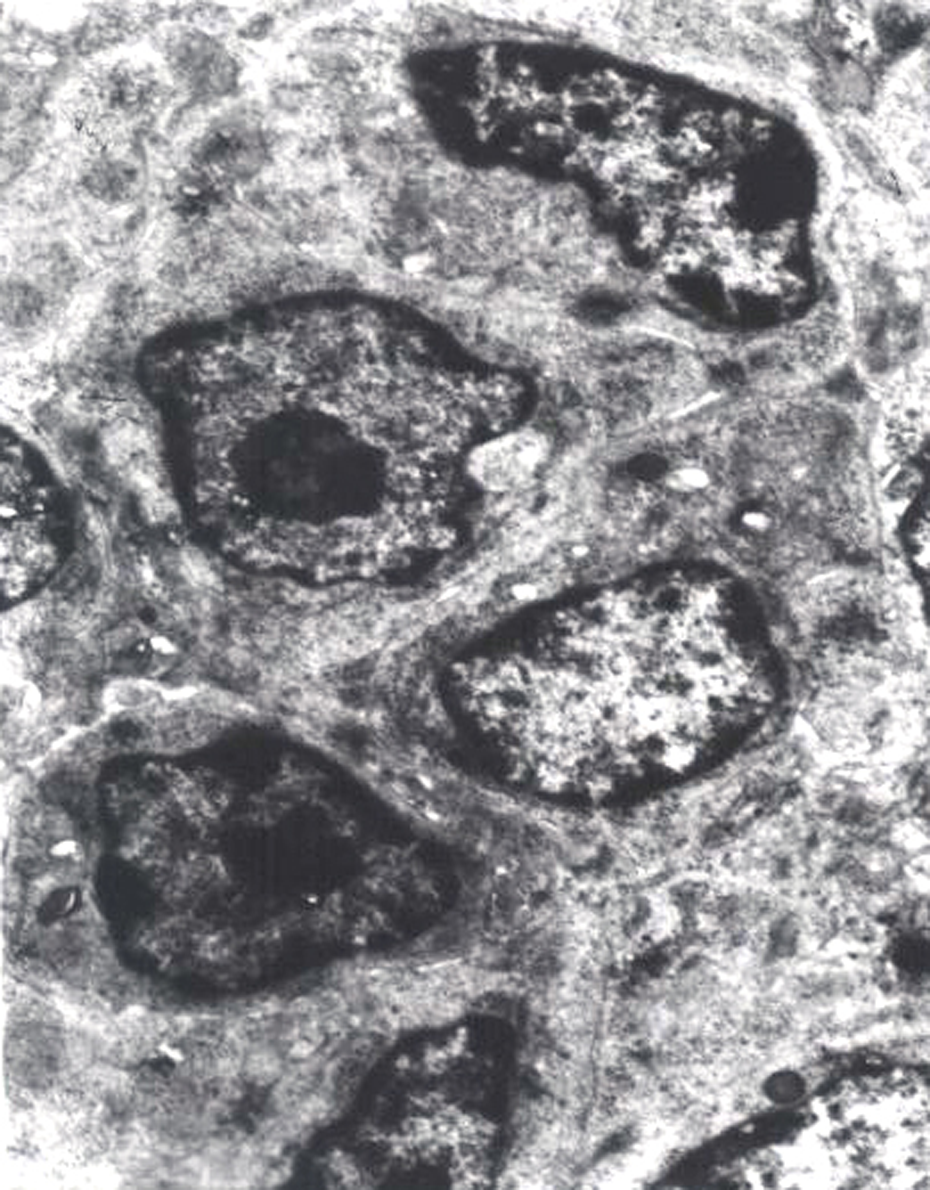
The splenocytes in the control group (×8000).

Dilated endoplasmic reticulum of splenocyte (Red arrow) in high fluorine (F) group II (×10000).

The mitochondria of lymphocytes were enlarged and their cristae were broken and absent (Red arrow) in the high fluorine (F) group III (×10000).

The apoptotic splenocytes were increased in the high fluorine (F) group III (×10000).
Discussion
As shown in Table 4, the contents of fluoride in spleen and serum were significantly higher in a dose-dependent manner in three high F groups when compared with those of control group, which indicate that the avain fluorosis model had been successfully built.
It is widely known that SOD and GSH-Px are antioxidase that are considered an important physiological antioxidant defense mechanism in removing excess oxygen negative ion. 15 Thus, the reduced SOD and GSH-Px activities could lead to the accumulation of free radical in the body. As reported previously, oxidant stress induced by free radicals and H2O2 is greater when fluoride impairs the production of free radical scavengers such as GSH, GSH-Px, SOD, and ascorbic acid. 16
Although a relationship in humans and animal fluorosis particularly between free radical generation, lipid peroxidation and antioxidant defence system has been investigated extensively, the results are mostly contradictory. Chlubek et al. found a 50% decrease in cytoplasmic SOD (Cu, Zn-SOD) activities and a tendency to reduce mitochondrial SOD (Mn-SOD) activities in the pancreas of rats receiving 50 and 100 mg F
In conclusion, excess dietary F in the range of 800−1200 mg/kg was found to cause inhibition of antioxidative enzymes, enhancement of lipid peroxidation, and finally induce excessive apoptosis in the spleen of chickens. Thus, oxidative stress induced by fluoride plays an important role in the pathogenesis of fluorosis.
Footnotes
The authors declare that there are no conflicts of interest.
This work was supported by the program for Changjiang scholars and the university innovative research team [IRT 0848], and the Education Department [09ZZ017] and Scientific Department [07JY027-022] of Sichuan Province.
