Abstract
Hepatotoxicity is one of the major complications of methotrexate (MTX) therapy. This study was carried out to evaluate the possible protective effect of resveratrol (trans-3,5,4′-trihydroxystilbene, RVT) against MTX-induced hepatotoxicity. Rats were randomly divided into four groups as control, MTX treated (7 mg/kg/day, intraperitoneally (i.p.), once daily for 3 consecutive days), MTX + RVT treated (20 mg/kg/day, i.p.), and RVT treated. First dose of RVT was administrated 3 days before the MTX injection and continued for 3 days. Histopathology of liver was evaluated by light microscopy. Aspartate aminotransferase (AST), alanine aminotransferase (ALT), and alkaline phosphatase (ALP) were used as biochemical markers of MTX-induced hepatic injury. The levels of thiobarbituric acid reactive substances (TBARS, a marker of lipid peroxidation) and activities of hepatic antioxidant enzymes such as catalase (CAT) and glutathione-S-transferase (GST) were used to analyze the oxidative stress-mediated lipid peroxidation in liver sections. Our results showed that MTX administration significantly increased ALT, ASP, and ALP levels. TBARS, CAT, and GST levels were also markedly increased in liver after MTX administration. RVT treatment significantly prevented MTX-induced hepatotoxicity, as indicated by AST, ALT, and ALP levels and liver histopathology. Moreover, administration of RVT significantly decreased the elevated levels of TBARS and activities of CAT and GST in the liver compared to MTX-treated group. These results revealed that RVT may have a protective effect against MTX-induced hepatotoxicity by inhibiting oxidative stress-mediated lipid peroxidation. Consequently, RVT treatment might be a promising strategy against MTX-induced hepatotoxicity.
Introduction
Methotrexate (MTX), a folat antagonist, is commonly used in the treatment of many different types of cancer and inflammatory diseases. 1 –3 In spite of the widespread use of MTX, its efficacy is often limited by the occurrence of hepatotoxicity which is one of the major complications of MTX treatment. 4 Although the mechanism of hepatotoxicity of MTX is not yet completely unknown, considerable experimental and clinical evidence support the hypothesis that MTX-induced hepatic damage may be a consequence of oxidative stress. 5,6 MTX administration resulted in increased production of reactive oxygen species (ROS), leading to damage of macromolecules and trigger many pathological processes. 7,8 There are antioxidant defense mechanisms in the liver to neutralize the deleterious effects of free radicals. These antioxidant enzymes are the first line of defense systems against the cell damaging effects of oxidative stress. 9 Thus, the generation of ROS in liver tissue might be indirectly evaluated by measuring some antioxidant enzymes such as superoxide dismutase (SOD), catalase (CAT), and glutathione-S-transferase (GST). 10 –12
Natural antioxidants in foods are being used as a comprehensive supplement against drug-induced toxic effects, thereby enhancing the therapeutic efficacy of the drug. 13,14 Resveratrol (trans-3,5,4′-trihydroxystilbene, RVT) is a dietary polyphenol that has been shown to protect against nephrotoxicity, 15 cardiotoxicity, 16 and neurotoxicity 17 due to its antioxidant and anti-inflammatory features. Several studies have also highlighted the hepatoprotective properties of RVT against alcohol exposure and acetaminophen-induced hepatotoxicity in mice. 18,19 RVT provides protection against hepatic damage induced by free radicals and inflammatory cytokines and attenuates hepatic lipid peroxidation. 20,21
It has been shown that MTX-treated animals have exaggerated oxidative stress leading to cellular dysfunction in liver tissue. 5,6 However, it is unclear whether the hepatocellular toxicity induced by MTX can be prevented by RVT administration. Also, little is known about the mechanisms involved in the hepatoptotective effect of RVT in rat liver. Taking the above into account, RVT may attenuate MTX-induced hepatotoxicity due to its antioxidant properties. The aim of this study was to evaluate whether RVT is a promising treatment approach for preventing MTX-induced hepatotoxicity. To evaluate the mechanisms underlying its hepatoprotective effects, thiobarbituric acid reactive substances (TBARS) were measured as an indicator of lipid peroxidation. Additionally, activities of CAT and GST were evaluated to determine the antioxidant defense system response to RVT treatment.
Materials and methods
Experimental procedures
This study was approved by the Animal Ethics Committee of Akdeniz University Medical Faculty, Antalya, Turkey. Briefly, 24 male Wistar rats aged 3 months, weighing 250–300 g, were used in this study. All animals were housed under the standard animal laboratory conditions (12-h lighting cycle, controlled temperature, and humidity). Animals were divided into four groups consisting of six animals per group. Control group (C, n = 6) received only intraperitoneally (i.p.) physiological saline of 1 mL/kg per day. MTX-treated group (MTX, n = 6) received three consecutive daily i.p. injections of MTX at the dose of 7 mg/kg/day. MTX + RVT-treated group (MTX + RVT, n = 6) was treated with RVT (20 mg/kg per day, i.p.) alone for 3 days and then concurrently administered with MTX for 3 days. Finally, RVT group (n = 6) received only RVT at the same dose. Six sets of independent experiments (using one animal per treatment group per set) were performed in this study. Doses and route of administration of MTX and RVT were chosen on the basis of previously reported data. 22 –25 In this study, we administered RVT by i.p. injection to ensure that all animals received the exact same dose of the product.
All rats were weighed and anesthetized with a cocktail of ketamine hydrochloride (90 mg/kg) and xylazine (10 mg/kg) administered i.p. before killing each rat. Blood samples were obtained from the renal vein, 24 h after the last MTX injection. Thereafter, serum was separated by centrifugation at 2000g for 5 min at 4°C for the determination of aspartate aminotransferase (AST), alanine aminotransferase (ALT), and alkaline phosphatase (ALP) activities. The livers were quickly removed and weighed. All tissues were stored at −80°C until biochemical analysis.
Histopathological analysis
The livers were fixed in 10% formalin and processed routinely for paraffin embedding. Paraffin-embedded tissue samples were cut into 5-µm thick sections, mounted on SuperFrost Plus slides (Erie Scientific Company, Portsmouth, New Hampshire, USA), stained with hematoxylin and eosin. Microscopic scoring was done by experienced pathologists, who were unaware of the treatments. Severity of hepatic injury was semiquantitatively evaluated by Zeiss Axioplan microscope using the following alterations: (a) pyknotic nucleus and perinuclear vacuolization of hepatocytes, (b) vascular congestion in sinusoids and sinusoidal dilatation, and (c) number of activated Kupffer cells. Scores were given as 0 = absent, 1 = weak, 2 = moderate, and 3 = strong for each criteria. The microscopic score of each tissue was calculated as the sum of the scores given to each criterion, and at least five microscopic areas were examined under a 20× objective to score each specimen.
AST, ALT, and ALP activities
Plasma concentrations of the liver enzymes ALT and AST were determined using serum samples obtained from all groups of rats. Activities were expressed as unites per liter. The measurements were done in accordance with the methods of the diagnostic kits (Bt Products, Izmir, Turkey).
Preparations of tissue homogenates
Tissues were quickly removed and washed in ice-cold normal saline (0.9% NaCl), dried on filter paper, and weighed. Liver tissues were homogenized in cold potassium phosphate buffer (50 mM, pH 7.4). All tissues were rapidly sonicated using a thermally regulated sonicator for 1 min. Homogenate was divided into two portions. The first portion of the homogenate was centrifuged at 700xg for 20 min at 4°C in order to determine the CAT activity and TBARS level. The second portion of homogenate was centrifuged at 20,000xg for 30 min at 4°C in order to determine the GST activity. All assays were performed in duplicate.
CAT activity
CAT activity was measured by the method of Aebi, 26 using hydrogen peroxide (H2O2) as the substrate. The final volume of each enzyme assay was 1.5 mL containing 0.5 mL of 30 mM H2O2 and 1.0 mL supernatant. The decomposition of H2O2 can be correctly followed by monitoring the decrease in absorbance at 25°C and 240 nm for 30 s in spectrophotometer. Enzyme activity was expressed as k/g protein (k: rate constant of the first-order reaction).
TBARS level
The TBARS level in the liver tissue, an index of lipid peroxidation, was measured by a fluorometric method described by Wasowicz et al. 27 and Gümüşlü et al. 28 using 1,1,3,3-tetraethoxypropane as the standard. Supernatant (50 µL) was placed in tubes containing 1 mL distilled water. After addition of 1 mL of a solution containing 29 mmol/L 2-thiobarbituric acid in acetic acid (8.75 mol/L), samples were placed in a water bath and heated for 1 h at 100°C. After the samples had cooled, 25 µL of 5 mol/L HCl was added, and the reaction mixture was extracted by agitation for 5 min with 3.5 mL of n-butanol. After centrifugation at 3500g for 10 min, the butanol phase was separated and the fluorescence of the butanol extract was measured using a spectrofluorometer (Shimadzu RF-5000, Kyoto, Japan) set at wavelengths of 525 nm for excitation and 547 nm for emission. TBARS levels were expressed as nmol/mg protein in liver tissues. The protein content of samples was determined using the colorimetric method of Lowry, 29 using BSA as the standard.
GST activity
GST (EC 2.5.1.18) activity was measured at 30°C, using 1-chloro-2,4-dinitrobenzene (CDNB) as a substrate. 28,30 GST was assayed in 1 mL reaction volumes containing 100 µL of CDNB (10 mM), 100 µL of reduced glutathione (GSH; 10 mM), 725 µL buffer potassium phosphate (0.1 M, pH 7.5), and 75 µL supernatant. Reaction was initiated by the addition of CDNB and the formation of S-2,4-dinitrophenyl glutathione (DNPG) was monitored as the increase in absorbance. The ultraviolet absorbance of the conjugated product was measured at 340 nm. The specific activity of GST was calculated as 1 µmol DNPG formed per mg protein in 1 min. All enzyme activities are expressed per mg of tissue protein.
Drugs and chemicals
In the present study, MTX was administered as the clinical formulation. MTX was purchased from Sandoz, Turkey (Edicin 0.5 g) and prepared fresh daily during the experiments. RVT (Sigma Chemical Company, St Louis, Missouri, USA) was dissolved in ethanol (50%, v/v, Merck) before administration.
Statistical analysis
GraphPad Prism 3.0 (GraphPad Software, San Diego, California, USA) was used for statistical analysis. All values are expressed as mean ± standard error of the mean. Statistical analysis of the results was performed by one-way analysis of variance. Post hoc comparisons were done using Tukey’s multiple comparison test. A p value <0.05 was considered as significant.
Results
The initial and final body weights of all groups are shown in Table 1. Although MTX administration resulted in a slight decrease in body weight of rats, there were no significant differences between the initial and final body weights among all groups. Similarly, liver/body weight ratios did not significantly change after MTX administration when compared to controls (Table 1).
BW and liver/body weight ratio in control, MTX, MTX + RVT, and RVT groupsa

MTX: methotrexate; RVT: resveratrol; BW: body weight.
a Data are expressed as mean ± SEM; n = 6 for all groups.
Biochemical liver function tests
As shown in Table 2, serum ALT, AST, and ALP levels significantly increased in MTX-administered rats when compared to controls. RVT pretreatment significantly prevented the effect of MTX on these enzymes. In RVT-treated group, AST, ALT, and ALP levels were significantly lower than those of the MTX-treated group.
Serum AST, ALT, and ALP levels of control, MTX, MTX + RVT, and RVT groupsa

MTX: methotrexate; RVT: resveratrol; AST: aspartate aminotransferase; ALT: alanine aminotransferase; ALP: alkaline phosphatase.
a Data are expressed as mean ± SEM; n = 6 for all groups.
b p < 0.05 versus control group.
c p < 0.05 versus MTX group.
Histological evaluation
Control rats had a normal histological pattern of liver (Figure 1(a)), whereas MTX-treated rats showed various histological changes in liver injury. The degenerated hepatocytes, vascular congestion in sinusoids, dilatation of sinusoids, and increased number of activated Kupffer cells were seen in the liver sections of MTX-treated rats (Figure 1(b) and (c)). Treatment with RVT markedly diminished the tissue damage induced by MTX administration. The histopathological appearance of livers in MTX + RVT-treated rats was similar to those of the controls (Figure 1(d)). The histopathological scores of liver sections of all groups are presented in Table 3.

Liver histology: normal architecture in control (A) and resveratrol (D) administered rats. The vascular congestion and dilatation of sinusoids, degenerated hepatocytes and increased number of activated Kupffer cells in liver tissue of methotrexate-administered rats (B), partially protected by resveratrol treatment (C).
The total histopathological scores of the liver tissues in control, MTX, MTX + RVT, and RVT groupsa

MTX: methotrexate; RVT: resveratrol.
a Data are expressed as mean ± SEM; n = 6 for all groups.
b p < 0.05 versus control group.
c p < 0.05 versus MTX group.
TBARS levels and CAT and GST activities
Liver TBARS levels significantly increased in MTX-treated rats when compared with controls (1.84 ± 0.04 nmol/mg protein for control, 2.47 ± 0.03 for MTX, p < 0.05). CAT activities also significantly increased after MTX treatment. CAT activities were 48.15 ± 0.46 and 71.56 ± 0.37 k/g protein for control and MTX-treated group, respectively. Furthermore, GST activity in MTX-treated rats was higher than that of the control group (2.20 ± 0.02 U/mg protein for control, 3.49 ± 0.04 U/mg protein for MTX-treated group, p < 0.05). RVT administration caused a significant decrease in TBARS levels and in the activities of CAT and GST in MTX + RVT group (Figures 2 to 4).

Effect of RVT treatment on MTX-induced changes in liver TBARS levels. All values are expressed as mean � n = 6 for all groups, MTX, methotrexate; RVT, resveratrol. *p < 0.05 as compared with control, **p < 0.05 as compared with MTX.
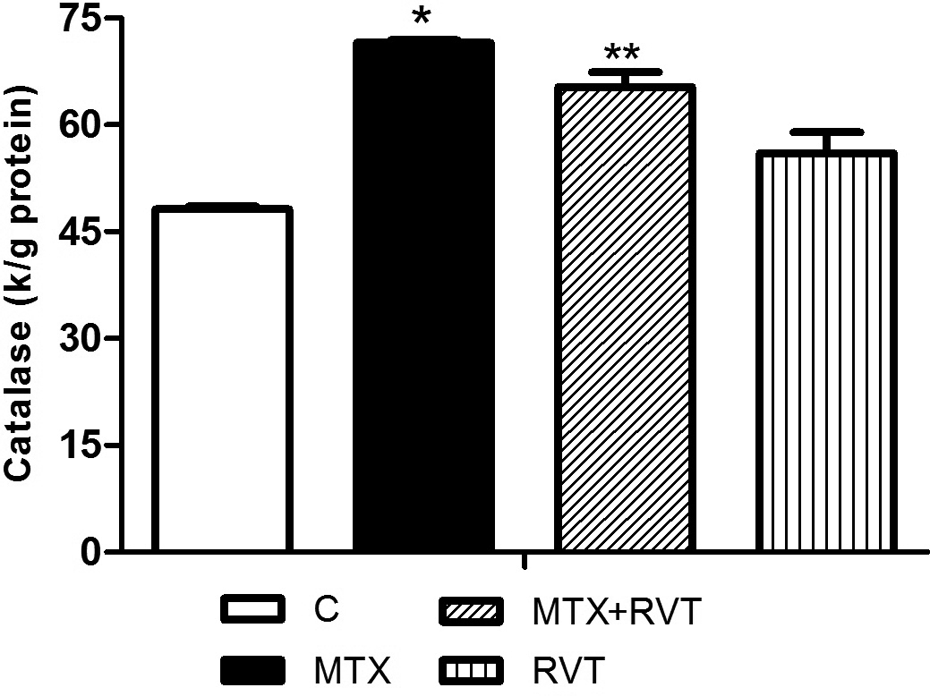
Effect of RVT treatment on MTX-induced changes in liver catalase activity. All values are expressed as mean � n = 6 for all groups, MTX, methotrexate; RVT, resveratrol. *p < 0.05 as compared with control, **p < 0.05 as compared with MTX.

Effect of RVT treatment on MTX-induced changes in liver GST activity. All values are expressed as mean � n = 6 for all groups, GST, glutathion S-transpherase; MTX, methotrexate; RVT, resveratrol. *p < 0.05 as compared with control, **p < 0.05 as compared with MTX.
Discussion
Oxidative stress and lipid peroxidation mediated by oxygen free radicals have been implicated as important cause of MTX-induced liver toxicity. 31 The present study was carried out to investigate the protective effect of RVT, a natural antioxidant that is widely consumed in the diet, against MTX-induced liver injury. The present investigation revealed that RVT pretreatment provides protection against MTX-induced hepatic damage by reducing TBARS content, as an index of lipid peroxidation.
Drugs used for cancer chemotherapy produce acute toxic side effects in multiple organ systems. MTX treatment can lead to serious adverse effects in the clinical setting. Liver is the major site of drug metabolism and the primary target for numerous xenobiotic toxicants including a number of anticancer drugs such as MTX. The present investigation demonstrated that MTX administration did not cause a significant decrease in liver/body weight ratios in rats. However, there is no doubt that both the histological appearance and the biochemical parameters supported a diagnosis of liver damage. The histopathological evidence of liver injury such as degenerated hepatocytes, vascular congestion in sinusoids, and dilatation of sinusoids, and increased number of activated Kupffer cells has been observed in rats treated with MTX. In addition to the histopathological evidence, biochemical markers of liver injury have been observed in rats treated with MTX. It is known that increased transaminase levels in the plasma correlate with the degree of hepatic damage. 32 A slight increase in these enzymes is indicative of hepatic injury. 33 Our results have clearly demonstrated that MTX treatment caused an increase in the levels of serum AST, ALT, and ALP. High levels of ALT and AST may be related with the release of these enzymes from the cytoplasm into the blood circulation after the cellular damage. In this study, we observed that adding RVT to MTX was found to produce a significant decrease in liver enzyme activities compared with MTX alone. The observed decrease in the serum activities of these enzymes showed that RVT preserved the structural integrity of the liver from the toxic effect of MTX.
MTX is one of the major cytotoxic agents suggested to increase the generation of ROS resulting in oxidative stress and cellular injury. 6 Because liver is the primary organ of MTX metabolism, increased ROS generation in the liver after MTX treatment seems to be involved in hepatotoxic effects. Membrane lipids are main targets of ROS, and the ROS formed might induce cellular damage by peroxidation of lipids. 34 High lipid content in liver is important for the high vulnerability of this tissue to oxidative stress. Lipid peroxidation, mediated by ROS, might be an important cause of hepatic damage. Therefore, the important consequences of oxidative stress induced by MTX could be attributed to stress-induced lipid peroxidation. TBARS levels, a lipid peroxidation marker, is known to be elevated in plasma and peripheral tissues in response to stress conditions. 35,36 The sensitivity of measuring TBARS has made the assay the method of choice for screening and monitoring lipid peroxidation, an important indicator of oxidative stress. 37 MTX treatment may initiate membrane damage by lipid peroxidation resulting in deleterious effects to the liver cells. The finding of elevated TBARS levels in our study suggests the presence of enhanced lipid peroxidation caused by MTX treatment in liver tissues. Membrane disintegration of hepatocytes with subsequent release of AST, ALT, and ALP is one of the consequences of lipid peroxidation. 38 These results indicate that MTX administration causes oxidative hepatic damage by increasing lipid peroxidation. Our observations are consistent with previous studies that showed that MTX-treated rats have an increased oxidative stress-mediated lipid peroxidation in tissues. 6,39
The mechanism of MTX-induced lipid peroxidation might be related with the direct toxic effects of MTX or with a possible enhancement of ROS production. Also, MTX may inhibit several antioxidant enzymes and thereby, increased lipid peroxidation may be due to reduction in the activities of antioxidant enzymes. 11 Mammalian cells have been equipped with several enzyme systems to detoxify ROS and their by-products, including SOD, CAT, and GST. 40,41 At the first level of cellular response, oxidative stress is encountered by increased antioxidant defense system. 24,41 These increases in antioxidant enzyme activities have been considered as the protective response of the cells against ROS-induced cellular damage. CAT is a major antioxidant enzyme that is mainly responsible for the detoxification of H2O2. 42 This enzyme works to eliminate ROS, and a possible defect in this defense system may lead to increased susceptibility of lipids, proteins, and DNA to oxidative damage. 43 GST is one of the main antioxidant defense systems with their maximal activity seen in the hepatocytes that eliminate lipid peroxides and ROS. 40,44,45 In the liver, GST conjugates GSH to hydrophobic electrophiles derived from the metabolism of ethanol, drugs, and other toxins. 46 In the present study, we observed that activities of CAT and GST increased after MTX administration. When we compared CAT activities between the groups, the higher CAT activity was measured in liver of MTX-treated rats. This result may suggest that H2O2 level is higher in liver tissues of MTX-administered group compared with control animals. Our results also demonstrated that GST activity significantly increased in hepatic tissue of MTX-administered rats. This increase may be due to an increased release of lipophilic substances during oxidative injury, which are substrates for GST. 47 The elevated activities of these antioxidant enzymes in the MTX-treated rats indirectly indicated an increase in the ROS generation in liver tissue. This suggests adaptive mechanism of the liver to struggle ROS-induced tissue damage. Hence, elevated TBARS levels in liver tissue of MTX-treated rats may not be due to the inhibitory effect of MTX in antioxidant enzymes.
In consistence with the previous reports, our findings indicate that RVT exerts a hepatoprotective effect against MTX-induced liver damage in rats. In the present study, prophylactic RVT treatment provided significant protection from the hepatotoxicity of MTX, as indicated by a decrease in AST, ALT, and ALP levels. RVT has been shown to have various physiological functions, such as inhibition of lipid peroxidation, scavenging of free-radicals, and anti-inflammatory activity. 48 Numerous studies have demonstrated the ability of RVT to scavenge both superoxide and hydroxyl radicals, which may initiate lipid peroxidation. 49,50 Therefore, it may decrease the concentration of lipid free radicals and terminate initiation and propagation of lipid peroxidation. In this study, we demonstrated that RVT administration causes a statistically significant reduction in lipid peroxidation of liver tissues induced by MTX. The reduced hepatic lipid peroxidation in RVT-treated rats can be attributed to its oxidant-scavenging ability. Moreover, the elevated CAT and GST activities were decreased by RVT treatment. This may be as a consequence of reduced free radical concentrations in liver tissue of RVT-administered rats.
Besides its ability to scavange free radicals, RVT may also have indirect antioxidant actions. RVT has been shown to enhance several antioxidant enzymes, including SOD, CAT, and glutathione peroxidase. 51 –54 Moreover, Upadhyay et al. 55 have clearly shown that RVT augments phase II xenobiotic metabolizing enzymes such as GST and restores antioxidant capacity of liver. The antioxidant activity of RVT has been confirmed by their ability to inhibit the oxidation of low-density lipoprotein and liver microsomes. 48,56 According to the previous information, the results of our study demonstrated that treatment of rats with RVT alone slightly increased the activities of CAT and GST in liver. The finding of increased activities of antioxidant enzymes in the liver tissues of rats treated with RVT alone suggest that RVT not only exhibits a direct scavenging effect on free radicals but also partly stimulates intracellular antioxidant enzymes. CAT requires nicotinamide adenine dinucleotide phosphate (NADPH) for its regeneration from its inactive form. 57 Enhanced glucose uptake by cells serves as a fuel for both the pentose phosphate shunt and oxidative phosphorylation, thus bringing up the cellular levels of NADPH and nicotinamide adenine dinucleotide and thereby also enhancing the activity of CAT in restraint stress. 57 Previous results indicate that RVT is able to increase glucose uptake in vitro. 58 The effect of RVT on glucose uptake may be associated with changes in the activity of CAT in liver tissue. Otherwise, MTX treatment is associated with a loss in the cellular levels of the antioxidant GSH. 59 GSH form adducts with the toxic metabolites and contributes to the detoxification of many xenobiotics. GST functionally binds with GSH and endogenous and exogenous substances. 60 Previous studies have demonstrated that RVT induces an increase in GSH intracellular content in cardiomyocytes and smooth muscle cells. 61,62 Recently, it has been reported that RVT induces GSH synthesis by activation of nuclear erythroid-related factor 2, a redox-sensitive transcription factor that is involved in transcriptional regulation of many antioxidant genes. 63 Thus, treatment with RVT might enhance the hepatic GST activity of liver by preventing hepatic GSH depletion. However, further studies are needed to clarify the exact nature of this effect.
In conclusion, this study reports that RVT has a hepatoprotective effect against MTX-induced liver damage by reducing lipid peroxidation. Moreover, RVT may partly stimulate the activities of intracellular antioxidant enzymes in liver. Based on the results of the present study, it is possible to suggest that RVT treatment may offer a novel therapeutic approach for the prevention of hepatotoxicity induced by MTX treatment.
Footnotes
Conflict of Interest
The authors declared no conflict of interest.
Funding
This study was supported by Akdeniz University Research Foundation.
