Abstract
Intestinal mucositis is a serious toxic side effect of 5-fluorouracil (5-FU) treatment. Bu-Zhong-Yi-Qi decoction (BZYQD), a water extract of Chinese traditional herbal medicine, is widely used in chemotherapy in Asia as an alternative treatment to reduce the side effects of chemotherapy. However, the mechanism is unknown. To evaluate its mechanism, we investigated the effect of BZYQD on 5-FU-induced intestinal mucositis in mice, especially with regard to apoptosis in the intestinal mucosal epithelia. In the present study, mice were divided into three groups: control, 5-FU, and 5-FU + BZYQD. Mice in the 5-FU and 5-FU + BZYQD groups were administered 5-FU (100 mg/kg/day, intraperitoneally) for 6 days, and the mice in the latter group were given BZYQD (8 g/kg/day, intragastrically) beginning 4 days before 5-FU and continuing until the termination of the experiment. Loss in body weight and diarrhea during the 5-FU treatment were significantly attenuated by administration of BZYQD. The morphological signs of intestinal damage, including shortened villi height, crypt destruction, apoptosis, and necrosis, in intestinal mucosal epithelia were also reversed, accompanied by reduced neutrophil infiltration, nitrite levels, and inflammatory factors (tumor necrosis factor α and interleukin 1β) and increased levels of reduced glutathione. These results suggest that BZYQD inhibits 5-FU-induced intestinal mucositis, and this effect may be due to the reduction in apoptosis and necrosis in intestinal mucosal epithelia via the suppression of inflammatory cytokine upregulation. In conclusion, inhibiting cytokine-mediated apoptosis or necrosis can be the molecular mechanism by which BZYQD reduces the gastrointestinal side effects of cancer chemotherapy.
Introduction
Gastrointestinal toxicity is one of the most common side effects experienced during chemotherapy treatment for cancer. 1 In the gastrointestinal tract, the mucosal epithelium, the most important component of the mucosa in tissues, is involved in the defense against pathogens, the intake of nutrients, and the secretion of hormones. 2 Being the outermost layer of the mucosa, mucosal epithelia are susceptible to external stimuli, such as cytotoxic drugs, which damage mucous membranes and result in chemotherapy-induced mucositis. 3 5-Fluorouracil (5-FU), an antimetabolite antineoplasm agent, is widely used for the treatment of malignant tumors, but it frequently causes intestinal mucositis. Several pathogenic elements are involved in 5-FU-induced intestinal mucositis, including direct toxicity, oxidative stress, apoptosis, hypoproliferation, and abnormal inflammation. 4,5 Cytokine-mediated apoptosis plays a critical role in the pathogenesis of 5-FU-induced intestinal mucositis, in which the upregulation of inflammatory cytokines, such as tumor necrosis factor (TNF)-α and interleukin (IL)-1β, is observed. 6
Bu-Zhong-Yi-Qi decoction (BZYQD; also called Bu-Zhong-Yi-Qi-Tang in China and Bojungikki-tang or Hochu-ekki-to in Japan), which comprises crude ingredients that are extracted from eight herbs (Table 1), is a well-known formula in traditional Chinese medicine that has been used in Asia for over 1000 years. 7 –10 Based on the traditional Chinese medicine theory, BZYQD is a basic prescription as an energy tonic (Chinese medical concept, reinforcing the middle-jiao and replenishing qi), 11 which has been reported to retard the progression of chronic renal failure, improve the disability of refractory polymyositis patient, relieve the epigastrium disorders such as fullness and distension, and improve nutritional status in elderly patients with chronic obstructive pulmonary disease. 11 –14 In addition, BZYQD is used as an anti-inflammatory drug in perennial allergic rhinitis and chronic obstructive pulmonary disease patients 14,15 and as an alternative treatment in antineoplaston treatment to increase food intake, reduce side effects, and improve cancer-related fatigue and quality of lives in postsurgery and chemotherapy patients. 16 –19 In recent decades, the anti-inflammatory mechanism of BZYQD is being focused on, such as the stimulation of peripheral blood mononuclear cells to produce granulocyte colony-stimulating factor (G-CSF) and TNF-α in hepatocellular carcinoma, the reduction in antiasthmatic effects by inhibiting T-helper 2 responses in ovalbumin-sensitized mice and the inhibition of COX-2 messenger RNA expression in IL-4-stimulated polymorphonuclear neutrophils in perennial allergic rhinitis patients. 10,20,21 But the mechanism of BZYQD in reducing the side effects of chemotherapy is still unknown.
Herbal constituents of BZYQD.

BZYQD: Bu-Zhong-Yi-Qi decoction.
In the present work, we investigated the effect of BZYQD on 5-FU-induced intestinal mucositis in mice, especially focusing on its antiapoptotic and anti-inflammatory effects on jejunum tissue.
Materials and methods
Preparation and administration of BZYQD
Eight herbs (Table 1) were placed in a 20-fold volume of distilled water, decocted from 80°C to 100°C, filtered, concentrated at 40–80°C, and stored in a refrigerator until use. 7
Animals and treatment
Thirty pathogen-free Kunming mice weighing 20–25 g (aged 8 weeks) were obtained from the Experimental Animal Center of Liaoning University of Traditional Chinese Medicine (Shenyang, China). The animals were acclimatized to standard laboratory conditions (temperature 22–25°C, relative humidity 50–60%, and a 12-h photoperiods with lights on from 07:00 to 19:00). This study was approved by the Institutional Animal Ethics Committee of Liaoning University of Traditional Chinese Medicine and was conducted in accordance with the “Guide for the Care and Use of Laboratory Animals.”
The mice were randomly divided into three groups (n = 10/group, half male and half female), and the treatments of the animals in different groups are shown in Figure 1. Control group: The mice were pretreated with normal saline (N.S.) intragastrically (i.g.) with N.S. at the same volume as BZYQD on day 1 and administered daily until the termination of the animal experiment and intraperitoneally (i.p.) with N.S. at the same volume as 5-FU daily from day 5 to day 10.
7
5-FU group: The mice were pretreated with N.S. at the same volume as BZYQD, i.g., for 10 days and with 5-FU 100 mg/kg, i.p., for 6 days (from day 5 to day 10).
22
5-FU + BZYQD group: The mice were pretreated with 8 g/kg BZYQD, i.g., for 10 days (according to the traditional Chinese medicine decoction Jianfu method, the water extraction of eight herbs in Table 1 is defined as one dose, which is usually a patient’s daily dose of the decoction.
23
In our study, the concentration of 8 g/kg BZYQD for the mice was the most commonly used dosage in clinical treatments as an alternative to reduce gastrointestinal side effects and confirmed protective effect in diabetic rats in previous study
7,18,19
) and with 5-FU 100 mg/kg, i.p., for 6 days.

Mouse treatments. The mice were divided into three groups: control, 5-FU, and 5-FU + BZYQD. BZYQD or normal saline (N.S.) was administered by intragastrically (i.g.) from day 1 to day 10, and 5-FU or N.S. was given intraperitoneally (i.p.) from day 5 to day 10. 5-FU: 5-fluorouracil; BZYQD: Bu-Zhong-Yi-Qi decoction; N.S.: normal saline.
Weight and diarrhea measurement
The weight of mice was recorded three times: day 1 before BZYQD or N.S. i.g. treatment, day 5 before 5-FU or N.S. i.p. treatment, and 6 h after the termination of the animal experiment at day 10.
The mice were inspected twice daily for signs of diarrhea from days 5 to 10. Scores were recorded as 0, 1, 2, or 3, where 0 refers to normal stool consistency, 1 refers to loose stools, 2 refers to overt diarrhea with perianal soilage, and 3 refers to severe/bloody diarrhea with substantial tail soilage. The daily scores were then summed to provide a cumulative score over the course of the study. 24
Intestinal histopathology and apoptosis assay
Segments of the jejunum (3-cm segment immediately distal to the ligament of Treitz) were collected, fixed, and stained with hematoxylin and eosin (H&E) for histopathological evaluation. Cell apoptosis in the segments was detected using an immunohistochemical assay with cleaved caspase 3 antibody (Abcam, Cambridge, Massachusetts, USA). The results were reviewed by two independent researchers and analyzed using the MetaMorph/DP10/BX41 image analysis system (UIC/Olympus, Chicago, IL, USA/ Tokyo, JP). 25
Transmission electron microscopy
Segments of the jejunum were fixed, postfixed, and embedded as described previously. 25 Ultrasections were observed under a transmission electron microscope (model RILI H-7500, Hitachi, Japan) at 80 kV.
Determination of the intestinal myeloperoxidase activity
A myeloperoxidase (MPO) enzyme-linked immunosorbent assay (ELISA) kit (R&D Systems, Minneapolis, Minnesota, USA) was used to quantify the extent of granulocytes infiltration into the intestinal mucosa. Briefly, the fresh jejunum tissues samples (50 mg) were homogenized in 2 mL of phosphate-buffered saline (PBS; pH 7.4) and then centrifuged for 20 min at 3000g. According to the manufacturer’s instructions, the supernatant samples were added to ELISA plates after being incubated and colored and then measured at 450 nm. The results are expressed as nanograms per milliliter. Each experiment was performed in triplicate.
Determination of the sulfhydryl group concentrations
The concentrations of nonprotein sulfhydryl groups (mainly reduced glutathione (GSH)) in the fresh jejunum tissues were assessed using the GSH Assay Kit (NanJing JianCheng Bioengineering Institute, China). The tissue was homogenized and centrifuged, and the supernatant was diluted with 10 volumes of PBS (pH 7.4). The diluted supernatant was reacted with 5, 5-dithiobis-(2-nitrobenzoic acid) and measured at 405 nm. The results are expressed as micromoles per liter. Each experiment was performed in triplicate. 26
Determination of the nitrite concentrations
Intestinal tissue nitrite levels were measured using the Total Nitric Oxide Assay (R&D Systems). In this assay, the determination of nitrite concentration in supernatant of fresh jejunum tissues provides a quantitative measurement of the nitric oxide (NO) that is generated by NO synthesis. After being homogenized and centrifuged, the supernatant of the tissue was added to ELISA plates, incubated, and colored, and then the optical density (OD) was measured at 540 nm. 25 The results are expressed as micromoles per liter. Each experiment was performed in triplicate.
Detection of cytokine (TNF-α and IL-1β) concentrations
The concentrations of cytokines (TNF-α and IL-1β) in the supernatant of the fresh jejunum tissues were determined by ELISA using protocols that were supplied by the manufacturer (R&D Systems). The OD was measured at 540 nm. The results are expressed as nanograms per liter. Each experiment was performed in triplicate. 10
Statistical analyses
Each experiment was performed separately at least three times. The data are expressed as the mean ± standard deviation, and a statistical comparison between different groups was performed by an analysis of variance test. The value p < 0.05 indicates significance, and NS indicates no significant difference (p > 0.05). Statistical analyses were conducted using SPSS 15.0.
Results
Effects of BZYQD on the weight loss and diarrhea score of mice that were treated with 5-FU
There was no significant difference in the weight of mice from different groups at the baseline (data not shown). After 4 days of treatment, there was no significant difference in the rate of weight gain in any treatment group (Figure 2(a)). On day 10, the weight of the mice in the control group increased continuously, while the weight of mice in the 5-FU group was significantly decreased (control vs. 5-FU, p < 0.05). However, the weight of the mice in the 5-FU + BZYQD group was significantly increased (5-FU vs. 5-FU + BZYQD, p < 0.05; Figure 2(b)). The onset of diarrhea occurred on day 5 after 5-FU treatment (Figure 3). BZYQD treatment effectively changed the frequency of diarrhea onset. The diarrhea score for the 5-FU and 5-FU + BZYQD groups began to show significance on day 6 and remained until the end of the experiment (5-FU vs. 5-FU + BZYQD, p < 0.05 on days 6 and 7; p < 0.01 from days 8 to 10).
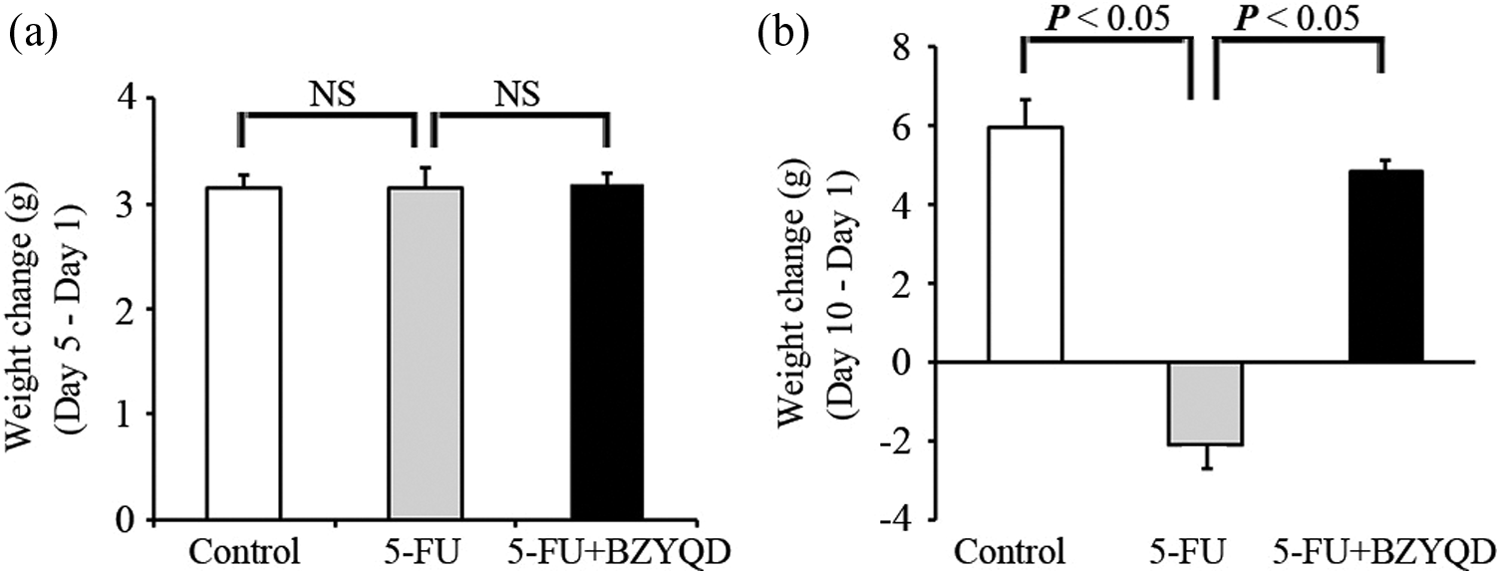
Effects of BZYQD treatment on weight changes after 5-FU induction. Weight changes in the mice in the three groups were measured at day 5 (a) and day 10 (b). The difference was analyzed by ANOVA. NS indicates no significant difference (p > 0.05). BZYQD: Bu-Zhong-Yi-Qi decoction; 5-FU: 5-fluorouracil; ANOVA, analysis of variance.

Effects of BZYQD treatment on the diarrhea severity of the 5-FU-induced mice. The cumulative diarrhea score increased after 5-FU induction. The 5-FU and 5-FU + BZYQD groups exhibited diarrhea. BZYQD significantly inhibited the cumulative diarrhea score as induced by 5-FU (5-FU vs. 5-FU + BZYQD, *p < 0.05, #p < 0.01). BZYQD: Bu-Zhong-Yi-Qi decoction; 5-FU: 5-fluorouracil.
Effects of BZYQD on the intestinal morphology of mice that were treated with 5-FU
We analyzed the intestinal structure of different groups by microscopic examination. In the control group, the intestinal villi were arranged orderly, and the intestinal crypts had uniform depth (Figure 4(a)-A). In addition, the surface mucosal epithelia were continuous and intact, without any morphological changes (Figure 4(b)-A). In contrast, the intestine of the mice in the 5-FU group was characterized by shortened villi height and crypt destruction (Figure 4(a)-B). The surface mucosal epithelia showed obvious apoptosis with nuclear condensation; in addition, necrosis with hypochromatosis, vacuolization, and edema was observed (Figure 4(b)-B). After administration of BZYQD, the mucosa morphology significantly improved compared to that of the 5-FU group, with villi arranged orderly, the damage of crypts better repaired, and epithelia apoptosis and necrosis decreased (Figure 4(a)-C and 4(b)-C).

Effects of BZYQD on the intestinal morphology of mice treated with 5-FU. (a) and (b) Jejunum sections stained with H&E. The normal morphology of the jejunum of mice is showed in the control group ((a)-A and (b)-A); 5-FU induced shortened villi, crypt destruction (→in (a)-B), nuclear condensation (→in (b)-B), hypochromatosis ( in (b)-B), vacuolization, and edema in mucosal epithelia (
in (b)-B), vacuolization, and edema in mucosal epithelia ( in (b)-B); BZYQD restored the morphological changes as induced by 5-FU ((a)-C and (b)-C). (c) The immunoreactivity for cleaved caspase 3 in jejunum. “→” indicates the positive results of immunohistochemistry and “
in (b)-B); BZYQD restored the morphological changes as induced by 5-FU ((a)-C and (b)-C). (c) The immunoreactivity for cleaved caspase 3 in jejunum. “→” indicates the positive results of immunohistochemistry and “ ” indicates the epithelia with hypochromatosis. (d) The ultrastructure of mucosal epithelia in the jejunum. The normal ultrastructural morphology of the epithelia is shown in the control group ((d)-A); 5-FU induced epithelium structural destructions including cell shrinkage, nucleus deformity, chromatin marginalization and lumpiness, and cytoplasm vacuolization (4(d)-B); BZYQD restored the morphological changes as induced by 5-FU (4(d)-C) (×6000). The “*” indicates cytoplasm vacuolization. BZYQD: Bu-Zhong-Yi-Qi decoction; 5-FU: 5-fluorouracil; H&E: hematoxylin and eosin.
” indicates the epithelia with hypochromatosis. (d) The ultrastructure of mucosal epithelia in the jejunum. The normal ultrastructural morphology of the epithelia is shown in the control group ((d)-A); 5-FU induced epithelium structural destructions including cell shrinkage, nucleus deformity, chromatin marginalization and lumpiness, and cytoplasm vacuolization (4(d)-B); BZYQD restored the morphological changes as induced by 5-FU (4(d)-C) (×6000). The “*” indicates cytoplasm vacuolization. BZYQD: Bu-Zhong-Yi-Qi decoction; 5-FU: 5-fluorouracil; H&E: hematoxylin and eosin.
To further examine this pathological effect, the expression of caspase 3, an apoptosis-related gene, 25 was detected in intestine sections. Cleaved caspase 3 was dramatically increased after 5-FU treatment, while BZYQD treatment significantly reduced its expression (cleaved caspase 3 positive rate, 5-FU vs. 5-FU + BZYQD, p < 0.01; Figure 4(c)). We further investigated the ultrastructural pathological changes in apoptosis by electron microscopic studies. The mucosal epithelium in the control group had an oval nucleus and well-distributed chromatin (Figure 4(d)-A). In contrast, the epithelium in the 5-FU group showed obvious structural destructions in that cell volume was shrunk with a deformed nucleus, and the chromatin was marginalized and clumped. Moreover, cytoplasm vacuolization was also observed in the cells with a deformed nucleus (Figure 4(d)-B). The intestinal structure in the 5-FU + BZYQD group was markedly restored (Figure 4(d)-C), with an intact and well-organized epithelium.
Effects of BZYQD on MPO activity, GSH, and nitrite concentrations
Following 5-FU administration, we observed significantly increased MPO in the intestine compared to that of the control group. In contrast, the increasing MPO as induced by 5-FU was markedly reversed by BZYQD in the 5-FU + BZYQD group (Figure 5(a)). In addition, we detected significantly reduced levels of GSH in the 5-FU-treated mice compared to the control mice, but the effect was reversed after treatment with BZYQD (Figure 5(b)). Finally, 5-FU significantly increased nitrite concentrations in the intestine compared to the control. Similar to the results of MPO and GSH, treatment with BZYQD reversed the nitrite concentrations induced by 5-FU in the intestine (Figure 5(c)).

Effects of BZYQD on the MPO, GSH, and nitrite concentrations in the jejunum. (a) MPO levels. (b) GSH levels. (c) Nitrite levels. (ANOVA followed by Bonferroni’s test.) MPO: myeloperoxidase; GSH: reduced glutathione; BZYQD: Bu-Zhong-Yi-Qi decoction; ANOVA, analysis of variance.
Effects of BZYQD on the production of TNF-α and IL-1β
We investigated TNF-α and IL-1β, two important inflammatory factors, to observe the effect of BZYQD on the inflammatory reaction as induced by 5-FU in the intestinal tissue. A significant elevation in TNF-α was observed in the 5-FU group compared to the control group, but the TNF-α levels in the 5-FU + BZYQD group were approximately similar to that of the control group (Figure 6(a)). For IL-1β, there was a significant elevation in the 5-FU group compared to the control, and treatment with BZYQD reduced IL-1β levels as well (Figure 6(b)).
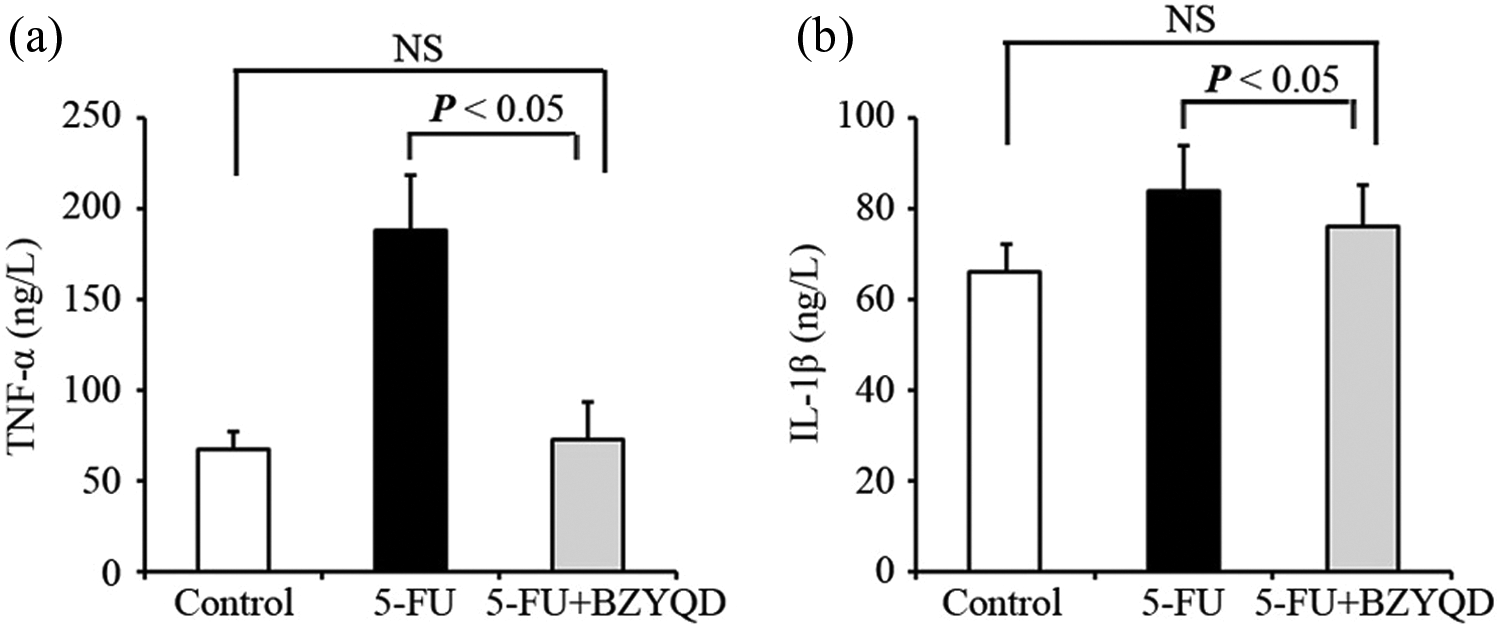
Effects of BZYQD on TNF-α and IL-1β production in the jejunum. (a) TNF-α levels. (b) IL-1β levels. (ANOVA followed by Bonferroni’s test.) TNF-α: tumor necrosis factor α; IL-1β: interleukin 1β; BZYQD: Bu-Zhong-Yi-Qi decoction; ANOVA, analysis of variance.
Discussion
Mucositis is a major oncological problem that is associated with the cytotoxicity of chemotherapy and radiotherapy. The development of intestinal mucositis can be divided into the following stages: the inflammatory stage, the epithelial degradation stage, the ulceration/bacterial stage, and the reestablishment of functional epithelia. 4 5-FU is usually administered intravenously in chemotherapy treatment; however, i.p. infusion of 5-FU has been suggested as an alternative route of administration, with the purpose exposing peritoneal and local tumor remnants to high cytotoxic drug levels. 27 In addition to the side effects such as abdominal pain or discomfort, 5-FU frequently causes intestinal mucositis accompanied by severe diarrhea, which involves several pathogenic elements, including mucosal epithelia apoptosis and inflammation. 4 –6 In the current study, typical pathogenic changes in 5-FU-induced intestinal mucositis, including physiological symptoms (weight loss, diarrhea, and inflammation) and morphological damages (shortened villi height, crypts destruction, and apoptosis), were observed in the mice given 5-FU. In addition, typical necrosis morphological changes, such as hypochromatosis, vacuolization, and edema, were observed in the intestinal mucosal epithelia in 5-FU-treated mice.
There have been reports that some methods can prevent intestinal mucositis, such as probiotics, by promoting crypt cell proliferation and regulating the intestinal immune system, and some herbal medicines, by inhibiting intestinal mucosal epithelia apoptosis. 4 –6 BZYQD, the water extract of herbal medicine, has been identified as an effective drug for improving function of the digestive system, for example, against alcohol-induced gastric injury in rat. 28 In our study, oral pretreatment of mice with BZYQD before 5-FU administration and continuously until the end of the 5-FU administration was able to prevent diarrhea and weight loss induced by 5-FU. In addition, treatment with BZYQD significantly reversed 5-FU-induced morphological intestinal damage, including shortened villi height, crypt destruction, apoptosis, and necrosis in intestinal mucosal epithelia. Our findings that BZYQD administered by i.g. to the mice are somewhat consistent with the previous study that BZYQD administered by i.p. could protect intestinal crypts by reducing the frequency of intestinal crypt apoptosis as induced by γ-rays in mice. 27 Thus, these results indicate that BZYQD can partly protect intestinal mucosa from the damage of radiotherapy and chemotherapy.
To further clarify the functions of BZYQD in alternative treatment in cancer chemotherapy, we performed assays of intestinal inflammatory parameters related to apoptosis, such as MPO, a biochemical marker of the infiltration of granulocytes, GSH, related to tissue damage and inflammation reaction, and nitrite, a biomarker that is correlated with mucosal epithelium function. In our experiments, BZYQD reduced neutrophil infiltration, nitrite levels, and inflammatory factors (TNF-α and IL-1β) and increased GSH, reversing the effects of 5-FU. These results are consistent with the previous studies that BZYQD was beneficial to patients with perennial allergic rhinitis and virus infection via suppressed production of inflammatory factors by an anti-inflammatory effect. 15,29,30 Previous studies have reported that the inflammatory stage precedes epithelial degradation, 4 by which we thought that BZYQD could reverse 5-FU-induced morphological intestinal damage, such as shortened villi height, crypt destruction, mucosal epithelium apoptosis, and necrosis, by inhibiting the produced intestinal inflammatory parameters. These protective effects of BZYQD are considered to be closely related to pharmacological effects of its component herbs, which possess various pharmacological effects such as anti-inflammatory effect. 28 By this role, BZYQD could protect intestinal mucosa from gastrointestinal toxicity during chemotherapy treatment for cancer.
In conclusion, our data confirmed the protective mechanism in BZYQD can protect intestinal mucosa from cytokine-mediated apoptosis or necrosis in 5-FU-induced intestinal mucositis in mice and BZYQD is a useful alternative treatment for patients suffering from gastrointestinal side effects induced by cancer chemotherapy.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (81573856); the Science and Technology Foundation of Liaoning Province (2015020379); the Xinglin scholars Project of Liaoning University of Traditional Chinese Medicine; the Innovation and Entrepreneurship Program for College Students of Liaoning University of Traditional Chinese Medicine.
