Abstract
Contrast-induced nephropathy (CIN) is an iatrogenic medical event in stable cardiology patients that may lead to acute renal failure. There is no current successful therapy to manage CIN. Increasing evidence in experimental models and humans has suggested that this disease is associated with renal tubular and vascular injury triggered by oxidative stress. Considering the importance of reactive oxygen species (ROS) generation in the pathogenesis of CIN, the goal of the present study was to evaluate the effects of sildenafil on CIN development. Male Wistar rats were divided into control, CIN, and CIN pretreated with sildenafil (50 mg/kg/day). CIN was induced by water deprivation, NG-nitro-L-arginine methyl ester + indomethacin injections (10 mg/kg, intraperitoneally) and intravenous iohexol administration (3 g/kg). Renal function was evaluated through glomerular filtration rate (GFR), renal blood flow (RBF), plasma creatinine, uremia, and proteinuria. Oxidative stress was assessed by flow cytometry for intracellular ROS. Treatment with sildenafil attenuated the marked reduction of GFR and RBF in the CIN group. Moreover, sildenafil treatment in CIN rats reduced plasma creatinine, uremia, and proteinuria. Flow cytometry demonstrated that sildenafil attenuated the ROS production in the CIN group. These data suggest that sildenafil may be a new therapeutic agent to prevent CIN through its ability to preserve renal function and attenuate oxidative stress.
Keywords
Introduction
Contrast-induced nephropathy (CIN), also termed as contrast-induced acute kidney injury (CI-AKI), is an iatrogenic disease 1 characterized by a sudden decline in renal function following intravascular administration of iodinated contrast medium in the absence of another nephrotoxic event. 2 Currently, CIN is a global clinical problem due to poor outcomes of at-risk populations (diabetic, cardiac, or kidney disease) of patients receiving contrast-enhanced imaging studies. 3,4,5 –8 Although no standard definition has been established for CIN, 4,9 most clinical studies define CIN as a rise in serum creatinine (absolute increase of ≥0.5 mg/dL or a relative increase ≥25% above baseline) or a decrease of estimated glomerular filtration rate (GFR; ≤60 mL/min) within 24–72 after contrast exposure. 1,9 –11
The mechanisms involved in the development of CIN include two major pathways: (1) altered renal hemodynamics and (2) cytotoxic effects of contrast media (CM). After contrast administration, renal vessels present prolonged vasoconstriction due to an imbalance between vasodilation and vasoconstriction. 3 This leads to increased renal vascular resistance (RVR) and decreased renal blood flow (RBF) and GFR. The resultant medullary hypoxia increases the production of reactive oxygen species (ROS), further aggravating renal vasoconstriction and endothelial and tubular cell damage. 12 CM has additionally been shown to increase tubular flow by volume expansion, leading to a rise in tubular reabsorption in Henle’s loop and higher oxygen consumption, exacerbating the hypoxia. 1 Current therapeutic interventions attempt to counteract these pathophysiological cascades to reduce the risk of CIN. However, intravenous hydration, avoidance of high osmolar agents, and reduction of CM volumes have not shown a sufficient reduction in the risk of CI-AKI in large-scale studies. 13
Among the potential therapeutic interventions, the use of phosphodiesterase-5 (PDE-5) inhibitors, such as sildenafil, are promising due to their actions on nitric oxide (NO)/cyclic guanosine monophosphate (cGMP)-mediated endothelium-dependent vascular responses. 14 –16 Recent data from our group have demonstrated that sildenafil ameliorates oxidative damage in the stenotic kidney 17 and in resistance arteries 14,18 in renovascular hypertensive mice. In accordance with these findings, studies have shown that sildenafil reduced kidney injury during cardiac bypass in swines 19 and ameliorates gentamicin-induced nephrotoxicity in rats. 20 We hypothesized that sildenafil will ameliorate kidney dysfunction in a rat model of CIN by improvement of renal hemodynamic and oxidative stress. Our results indicate that sildenafil may offer a novel approach to prevent CIN.
Methods
Animals
Experiments were conducted in male Wistar rats (290–390 g), maintained in the animal care facility at the Federal University of Espirito Santo, Brazil. The animals were housed in individual cages with a controlled temperature (22–23°C) and humidity (60%) and were exposed to a 12-h light:12-h dark cycle. All of the experimental procedures were performed in accordance with the National Institutes of Health guidelines, and the experimental protocols were previously approved by the Institutional Animal Care and Use Committee (CEUA-UFES Protocol no. 040-2013).
Contrast-induced nephropathy
The CIN procedure was adapted from protocols previously validated in rats by Agmon et al. 21 and Yokomaku et al. 22 After 24-h water deprivation, rats were administered with NG-nitro-L-arginine methyl ester (L-NAME; 10 mg/kg, intraperitoneally (i.p.)) and indomethacin (10 mg/kg, i.p.). After 15 min, CIN was induced by intravenous injection of iohexol (3 g/kg). 21,22 Animals were then given access to food and water, and 24 h later renal function was determined. Control animals received saline injections instead of indomethacin, L-NAME, and iohexol, and did not endure water deprivation. To determine whether sildenafil treatment could have a protective effect, rats received either a 7-day-treatment of sildenafil citrate (50 mg/kg/day) or vehicle (distilled water, 2 mL/kg/day) by gavage, starting 5 days before iohexol administration. The sildenafil dose and duration of the treatment used in the present study was based on previous studies showing the effects of sildenafil on pulmonary hypertension, placental ischemia-induced hypertension, or renovascular hypertension. 23 –26 Experimental design and groups are illustrated in Figure 1.
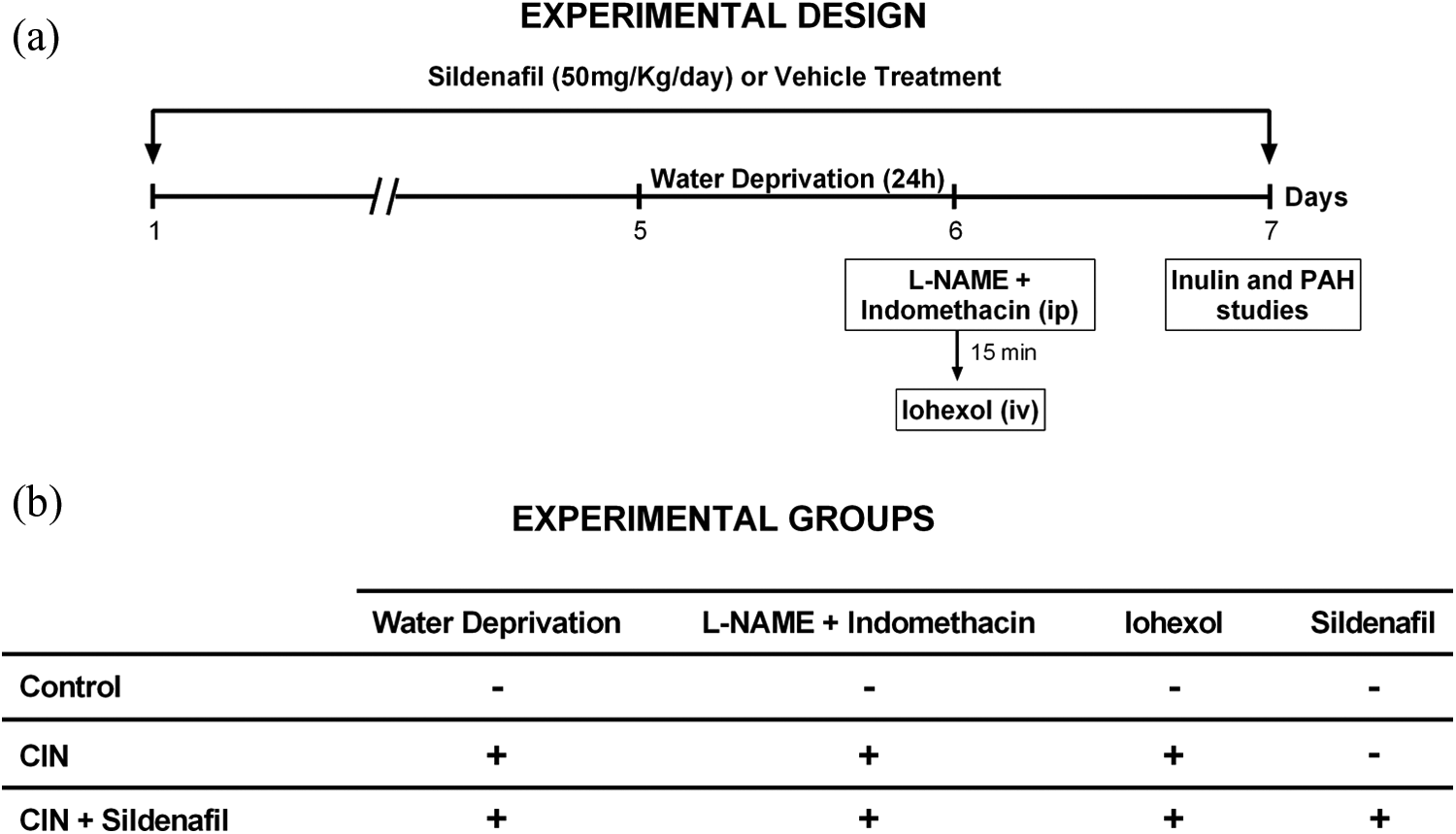
Experimental design (a) and experimental groups (b) of the present study. CIN induction began 5 days after sildenafil treatment started. CIN was induced by water deprivation for 24 h followed by L-NAME + indomethacin and iohexol administration at day 6. IN and PAH clearances were performed following CIN induction (a). The detailed procedures of each group are illustrated in (b). CIN: contrast-induced nephropathy; L-NAME: NG-nitro-L-arginine methyl ester; IN: inulin; PAH: sodium para-aminohippurate.
Renal function evaluation
Renal function was assessed by two different methods: (a) using inulin (IN) and sodium para-aminohippurate (PAH) clearance and (b) using repeated measures of plasma creatinine, urea, and urinary protein excretion. Renal hemodynamic was determined using IN and PAH clearance to estimate the GFR and renal plasma flow (RPF), respectively. 27
The animals were anesthetized with sodium thiopental (50 mg/kg ip.) and then the trachea was catheterized with a polyethylene tube (PE-90) to facilitate breathing while a catheter (PE-240) was introduced into the bladder for urine sampling. The arterial catheter was connected to a pressure transducer (Cobe Laboratories, Pico Rivera, California, USA) plugged into a pressure processor amplifier and data acquisition system (MP100, Biopac Systems, Goleta, California, USA) for continuous monitoring of mean arterial pressure (MAP) and heart rate (HR). The venous catheter was connected to an infusion pump (0.1 mL/min) with a saline solution (0.9%) containing 3% of mannitol. The solution was infused over a period of 30 min. Following infusion, the animals received an intravenous injection of prime solution containing IN (300 mg/kg) and PAH (6.66 mg/kg) and were maintained on a continuous infusion of saline (0.9%) containing IN (15 mg/mL), PAH (4 mg/mL), and mannitol (3%) until the end of the experiment. At 30-min intervals, urine and blood samples were taken, for up to 2 h. Haematocrit was measured using a heparinized capillary tube. Plasma and urinary IN and PAH concentrations were measured using a colorimetric assay. 28 Blood samples were also used for plasma urea quantification through spectrophotometry.
Renal blood flow (RBF) and RVR were determined as previously described. 29 Briefly, RBF was calculated by the equation RBF = RPF/(1 − hematocrit), and RVR was calculated using the equation RVR = MAP/RBF.. Plasma creatinine, urea, and proteinuria were quantified at two different time-points. Repeated blood and urine samples were collected before L-NAME and indomethacin injections and after a 24-h period, and the concentrations of these substances in the samples were determined by spectrophotometry. Data are expressed in %changes on plasma creatinine, urea, or protein excretion in urine.
Cardiac and kidney hypertrophy quantification
At the end of the experiments, the animals were euthanized with an overdose of sodium thiopental perfused via the left ventricle with Krebs-Hepes buffer (pH 7.4). The left kidney and the heart were removed, cleaned of connective tissue, and weighed. The tibia length was also used as an index to determine the ratio of kidney or heart weight according to Yin et al. (1982). 30
Measurement of intracellular ROS production
ROS analysis was achieved by flow cytometry as previously described. 31 Dihydroethidium (DHE) and 2′,7′-dichlorofluorescein diacetate (DCF-DA) were used to detect intracellular •O2 − and hydrogen peroxide, respectively. DHE (160 mM) and DCF-DA (20 mM) were added to cell suspension (106 cells) and incubated at 37°C for 30 min. 32 For positive control, samples were treated for 5 min with 10 μM doxorubicin or 50 mM H2O2 to create an oxidative stress without being toxic to the cells, whereas for negative control, the cells were incubated with ethanol. Cells were then washed, resuspended in PBS, and kept on ice for an immediate detection by a flow cytometer (FACSCanto II, Becton Dickinson, San Jose, California, USA). The measurement of intracellular NO was performed as previously described. 33 Briefly, the NO-sensitive fluorescent probe 4,5-diaminofluorescein-2/DA (DAF-2/DA: 2 μM) was added to cell suspension (106 cells) and incubated at 37°C for 180 min in the dark. For positive control, samples were treated with 100 μM sodium nitroprusside. Cells were then washed, resuspended in PBS, and immediately analyzed by flow cytometry (FACSCanto II, Becton Dickinson Bioscience).
Highly reactive oxygen species, such as hydroxyl radical and peroxynitrite, were selectively detected by 2-[6-(4′-hydroxy)phenoxy-3H-xanthen-3-on-9-yl]benzoic acid (HPF) as previously described. 34 Briefly, renal cells (106 cells) were incubated with HPF (10 μM) at 37°C for 30 min in the dark. For positive control, samples were treated with 100 μM sodium nitroprusside and 10 μM doxorubicin. Cells were then washed, resuspended in PBS, and analyzed by flow cytometry (FACSCanto II, Becton Dickinson Bioscience).
Data were analyzed using the FACSDiva software (Becton Dickinson). Quantification of DHE, DCF, DAF-2T, and HPF fluorescence was achieved by analysis of samples acquired in duplicate and 10,000 events were used for each measurement. Cells were excited at 488 nm and fluorescence was detected using a 530/30 bandpass filter and data expressed as geometric mean fluorescence intensity.
Statistical analysis
Values are expressed as means ± SEM. Statistical comparisons between the different groups were performed by one-way analysis of variance (ANOVA) followed by Tukey’s post hoc test. The statistical analyses were performed using Prism software (Prism 6, GraphPad Software, Inc., San Diego, California, USA). A value of p < 0.05 was regarded as statistically significant.
Results
Table 1 summarizes the average values of body weight, kidney, and heart weight ratio. At the end of the experiment, our data show a reduction in body weight between CIN groups when compared to control (CIN: ∼20% vs. CIN + sildenafil: ∼27%, p < 0.05). On the other hand, the administration of CM caused hypertrophy in kidney and heart, which was not affected by sildenafil treatment.
Cardiac and kidney hypertrophy in CIN rats treated with sildenafil (50 mg/kg) or vehicle.a

CIN: contrast-induced nephropathy; ANOVA: analysis of variance.
All values are expressed as means ± SEM. The number in parentheses represents the number of animals in each group.
ap < 0.05 vs. control group. One-way ANOVA followed by Tukey’s post hoc test.
Figure 2 summarizes the results from renal hemodynamic evaluation, including GFR (a), RPF (b), RBF (c) and RVR (d), as determined by IN and PAH clearance. CIN vehicle animals presented a marked reduction in GFR when compared to control group (3.8 ± 0.3 vs. 8.5 ± 0.6 mL/min/kg, respectively, p < 0.05). Interestingly, sildenafil treatment attenuated the reduction in GFR (6.8 ± 0.4 mL/min/kg, p < 0.05). This coincided with a decrease of RPF (20.0 ± 0.9 mL/min/kg vs. 35 ± 2.0 mL/min/kg, respectively, p < 0.05). Similarly, sildenafil treatment ameliorated this parameter (13.2 ± 0.3 vs. 22.6 ± 0.8 mL/min/kg, respectively, p < 0.05). Finally, we observed an increase in RVR in CIN animals (115%, p < 0.05) compared to absolute values from control group (3.6 ± 0.5 a.u.). This alteration in RVR was prevented by administration of the sildenafil (4.9 ± 0.17 a.u., p < 0.05).

Effects of sildenafil treatment on renal hemodynamic, evaluated through determination of GFR, as assessed by IN clearance (a) and RPF, as assessed by PAH clearance, as well as RBF (c) and renal vascular resistance (d) in control (white bars), CIN (black bars) and CIN + sildenafil (grey bars). GFR, RPF, and RBF were significantly decreased CIN animals. Sildenafil treatment prior to CIN induction resulted in an amelioration of these parameters. RVR was increased in CIN animals and sildenafil treatment blocked the rise in RVR. N = 7–13. Values are means ± SEMs. *p < 0.05 vs. control group; #p < 0.05 vs. CIN. One-way ANOVA followed by Tukey’s post hoc test. GFR: glomerular filtration rate; IN: inulin; RBF: renal blood flow; PAH: sodium para-aminohippurate; CIN: contrast-induced nephropathy; RPF: renal plasma flow; RVR: renal vascular resistance; ANOVA: analysis of variance.
In addition to hemodynamic renal parameters, we evaluated renal function, using repeated measurements of plasma creatinine and urea, as well as proteinuria, before L-NAME and indomethacin and CM administration and 24-h after this procedure. Figure 3 summarizes the changes in plasma creatinine and urine, as well as proteinuria among all groups. Plasma creatinine change (a) was significantly higher in CIN group (207 ± 19%), which was blocked by sildenafil treatment (57 ± 4%, p < 0.05), reducing the values close to those observed in the control group (37% ± 1%). Moreover, plasma urea change (b) was also higher in CIN vehicle animals (280 ± 19%, p < 0.05) and the administration of sildenafil prior CIN prevented the rise in plasma urea (57 ± 4%) similarly to control (55% ± 3%). The CIN group demonstrated an augment in protein urinary excretion (130 ± 4%, p < 0.05) when compared to control animals (29 ± 1%). Interestingly, sildenafil treatment also effectively attenuated proteinuria (65 ± 3%, p < 0.05).

Effects of sildenafil treatment on renal biochemical parameters. Control (white bars), CIN (black bars), and CIN + sildenafil (grey bars) animals were analyzed through repeated measurements of plasma creatinine (a), plasma urea (b), and urinary protein excretion (c) before and after L-NAME + indomethacin and iohexol injections. All parameters were elevated in CIN animals compared to control groups, and the administration of sildenafil prior to CIN induction prevented the rise on plasma creatinine and urea, as well as the increase in proteinuria. N = 5–9. Values are means ± SEMs. *p < 0.05 vs. control group; #p < 0.05 vs. CIN. One-way ANOVA followed by Tukey’s post hoc test. CIN: contrast-induced nephropathy; L-NAME: NG-nitro-L-arginine methyl ester; ANOVA: analysis of variance.
Renal production of •O2 −, H2O2, NO and •ONOO−/•OH− were assessed, as shown in Figure 4. We detected an increase in three parameters of ROS production in the CIN group (DHE: 2106 ± 103, DCF: 2649 ± 239, HPF: 3353 ± 160 a.u., p < 0.05) when compared to control rats (DHE: 1605 ± 91, DCF: 1673 ± 55, HPF: 1534 ± 85 a.u.). Surprisingly, the NO was the unique parameter that was not modified in CIN group in relation to control rats (DAF: 1532 ± 19 vs. 1590 ± 51 a.u., respectively). Treatment with sildenafil attenuated the increase in H2O2 and •ONOO−/•OH− bioavailability (DCF: 1973 ± 118 and HPF: 2890 ± 101 a.u., p < 0.05) without modifying the amount of •O2 − and NO (DHE: 2033 ± 39 and DAF: 1589 ± 42 a.u.).

Effects of sildenafil treatment on intracellular ROS production. DHE production (superoxide anions, black box) was increased in both CIN and CIN + sildenafil groups when compared to control animals. DCF (hydrogen peroxide production, grey box) and HPF (peroxynitrite and hydroxyl production, white box) were also augmented in CIN animals when compared to control group. Sildenafil treatment decreased CIN-induced overproduction of these ROS. Nitric oxide (DAF, dotted box) bioavailability was not affected by CIN or sildenafil treatment. N = 10–12. Values are means ± SEMs. *p < 0.05 vs. control group; #p < 0.05 vs. CIN. One-way ANOVA followed by Tukey’s post hoc test. ROS: reactive oxygen species; DHE: dihydroethidium; CIN: contrast-induced nephropathy; DCF: 2′,7′-dichlorofluorescein diacetate; HPF: 2-[6-(4′-hydroxy)phenoxy-3H-xanthen-3-on-9-yl]benzoic acid; DAF: 4,5-diaminofluorescein-2; ANOVA: analysis of variance.
Discussion
Although previous studies have demonstrated that PDE5 inhibitors prevent renal injury in many conditions, 35 –37 our study is the first report to investigate the effects of sildenafil in renal hemodynamic parameters and oxidative stress in an innovative radiocontrast-induced nephrotoxicity in Wistar rats. Interestingly, we found that the sildenafil at 50 mg/kg prevented the development of CIN by preserving renal function and attenuating renal oxidative stress.
It is known that murine animals are classically unaffected by CIN, 38 thus the search for novel drugs to prevent CIN has been met with difficulty. To overcome this obstacle, we modeled a male Wistar rat-CIN model based on a previous report by Yokomaku and colleagues (2008) 22 that investigated the susceptibility of Sprague-Dawley rats to iodinated CM after inducing hypoxic stress by acute inhibition of NO and prostaglandin production. 22,39 In a recent parallel experiment by Ozbek et al. (2015), 40 another successful Wistar rat-CIN model was developed. However, their model required an extensive period of water deprivation before contrast administration (48 h, not being required previous NO and prostaglandin blockades), and only a discrete increase of serum creatinine and urea when compared to our model.
It is known that CM induces endothelial lesion of renal vessels causing hypoperfusion and hypoxia accompanied by oxidative stress and low NO bioavailability, 4,41 compromising renal structure and function. The renal and cardiac hypertrophy observed in CIN group and CIN+ sildenafil groups suggests that tissue edema may develop as a consequence of renal acute dysfunction. Interestingly, our finding suggests that the sildenafil may act, in part, by improving the function of the remaining nephrons rather than preventing direct renal injury. The efficacy of CIN experimental model is commonly assessed by renal biomarkers such as urea, creatinine, 40,41,42 –45 and more recently, the sensitive parameter cystatin C. 40 Our data extend this observation by the description of loss of normal intrinsic physiological renal parameters that consolidate the injury in CIN model, as demonstrated by IN and PAH clearance. Furthermore, our results exhibit an increased uremia as CIN develops. Thus, our physiological data support and clarify previous studies with CIN models that have only hypothesized the impairment of renal function by increment of vascular renal resistance through renal biomarkers 4,39 –41,46,47 and reinforce the protective effects of sildenafil in other renal experimental models. 18,25,35,48
Our study has demonstrated that sildenafil exerts nephroprotective effects on CIN. Although the pathogenesis of CIN has been proposed to involve disturbances to renal blood flow mediated by hydration or NO signaling, 4,49 conventional strategies with antioxidants (scavengers) to increase the bioavailability of NO are not sufficient to attenuate CIN. 4,50 Interestingly, the improvement of renal parameters by sildenafil probably occurs by sensitization of the NO/cGMP signaling. 14,17,40,46,51 We hypothesized that this may be mediated by a total reduction in kidney ROS production and potentiated by known preservation of cGMP. It should be emphasized that the main contribution of sildenafil against ROS production was observed by its stability in comparison with decomposition of other ROS. 51 –53 Therefore, our data reinforce the antioxidant properties of sildenafil as previously demonstrated recently by both direct 14,17 and indirect methods. 54 –57 Moreover, although Liu et al. (2014) 4 have shown that the prevention of oxidative stress and renal hypoperfusion are secondary factors in CIN experimental model, our data suggest that attenuation of CIN may occur through reduction of oxidative stress.
However, alternative mechanisms of CIN progression have been proposed. Aside from oxidative stress, the renin–angiotensin (Ang) system has been proposed to be important in nephrotoxicity induced by CM. 44,58 Previous data from our lab have demonstrated that sildenafil was able to reduce Ang II intrarenal levels and to increase its physiological antagonist serum Ang 1-7. 17,18 Thus, we cannot discard the possibility that sildenafil may act through RBF improvement and antagonism of Ang II signaling. Furthermore, other studies have shown that renal ischemia may mobilize endothelial progenitor cells (EPC) to participate in kidney repair. 17,59,60 Chiang et al. (2014) 61 had also demonstrated that decreased EPC level is associated with a greater risk of CIN, also probably contributing to the poor prognosis in CIN patients. Therefore, we may speculate that EPC could contribute to renoprotective effects against CM, due to recent evidences that sildenafil increases the number of EPC in experimental model of oxidative stress 62 and in healthy patients. 63 Thus, sildenafil may exert a nephroprotective effect in CIN through antioxidant actions, as well as its actions on renin–Ang system and EPCs.
In summary, our results indicate for the first time in Wistar rats, the renal protective role of sildenafil in CIN through a vasodilator and antioxidant activity. Sildenafil may be a promising therapeutic agent in the prevention and treatment of CIN. Confirmation by additional experimental investigations and clinical trials is required to confirm these preliminary findings and to optimize the time of sildenafil administration as well as its ideal dosage.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by the National Council for the Development of Science and Technology-CNPq (473177/2013-7, 302582/2011-8, 476525/2012-8, 305188/2012-7, and 445080/2014-0 Grants) and the State Agency for the Development of Science and Technology-FAPES (012/2011 – 54674166, 012/2011 – 54498465, 012/2009-48473677 Grants).
