Abstract
In the current study, we evaluated the endocrine disruption effect and oxidative stress implication of therapeutic dose of artemether–lumefantrine combination therapy on the ovary and uterus of rats. In this respect, female rats were divided into four groups: animals were per orally treated with tween 80 (control), artemether (4 mg kg−1 body weight), lumefantrine (24 mg kg−1 body weight) and artemether–lumefantrine (artemether, 4 mg kg−1 body weight and lumefantrine, 24 mg kg−1 body weight). We found that therapeutic doses of the drugs did not change the levels of ovarian hydrogen peroxide (H2O2) and malondialdehyde (MDA), but increased uterine levels of H2O2 and MDA and reduced ovarian and uterine levels of reduced glutathione. In addition, whilst ovarian glutathione peroxidase (GPx) activity reduced in the lumefantrine monotherapy group, uterine GPx increased in the artemether monotherapy as well as the artemether–lumefantrine groups. Furthermore, the drugs reduced ovarian and uterine glutathione-S-transferase and uterine superoxide dismutase activities. The drugs reduced oestrogen level, whereas follicle-stimulating hormone was reduced by lumefantrine and artemether–lumefantrine therapies. Additionally, artemether and lumefantrine monotherapies significantly increased prolactin and progesterone levels compared with the control (p < 0.05). The results suggest that in the absence of malarial parasite infection, the drugs induced oxidative stress in the ovary and uterus and disrupt hormonal balance in the rats.
Keywords
Introduction
Malaria is a human parasitic disease transmitted in 108 countries of about 3 billion people. Lately, there have been decreased global malaria morbidity and mortality due to increased usage and accessibility of insecticide-treated bed nets and artemisinin-combination therapies (ACTs). In spite of this, malaria still kills about 2000 people per day, of which a large number are children less than 5 years old. 1 The World Health Organization recommends ACTs for the treatment of uncomplicated Plasmodium falciparum and Plasmodium vivax malaria. Among the ACTs, artemether–lumefantrine (Riamet®, Coartem®) is widely used because of its effectiveness against multidrug-resistant P. falciparum. 2,3
Artemether ((3 R, 5aS, 6 R, 8aS, 9 R, 10 S, 12 R, 12aR)-decahydro-10-methoxy-3,6,9-trimethyl-3,12-epoxy-12H-pyrano [4, 3-J]-1, 2-ben-zodioxepin) is a fat-soluble semi-synthetic derivative of artemisinin, a natural product isolated from Artemisia annua (Figure 1a). Artemether as an antimalarial drug has a better bioavailability than artemisinin. 4 In addition, it has rapid onset of activity after metabolism to dihydroartemisinin, the main active metabolite. Additionally, it is rapidly eliminated from the plasma due to a relatively low half-life of about 2–3 h. 5 At present, there are no confirmed neurotoxicity of artemether in humans, however, its neurotoxic effect has been reported in animal models. 6 Lumefantrine (2-(dibutylamino)-1-[(9E)-2,7-dichloro-9-[(4-chlorophenyl)methylidene]fluoren-4-yl]ethano) is a lipophilic antimalarial drug with a peak plasma concentration of about 6–8 h after administration (Figure 1b). Unlike artemether, lumefantrine has a slower antimalarial activity, 1 and a longer half-life of about 86 h, which makes it fit perfectly as a partner drug in combination therapy with artemether. 7 Some reported adverse effects of artemether–lumefantrine include headache, dizziness, gastrointestinal irritations, arthralgia, myalgia, fatigue, palpitations and weak evidence of cardiotoxicity. 6 It is believed that the toxicity of artemether is due to the generation of reactive free radicals that result from the reduction of its peroxide bridge. 2

Chemical structure of (a) artemether and (b) lumefantrine. 37
Reactive oxygen species (ROS) are highly reactive ions and free radicals containing oxygen molecules. Examples of ROS include nitric oxide, hydrogen peroxide (H2O2), singlet oxygen, peroxynitrite and superoxide free radicals. During normal physiological states in the ovary and the uterus, ROS and antioxidants are in balance because the ROS produced at low levels are scavenged by endogenous antioxidants such as catalase, superoxide dismutase (SOD), glutathione peroxidase (GPx), glutathione-S-transferase (GST), reduced glutathione (GSH) and vitamins C and E. However, oxidative stress occurs when ROS overwhelm the antioxidant buffering capacities of the tissues. 8 –10 In addition, oxidative stress could also result as a consequence of disturbance of the thiol redox circuits involved in cell signalling. The redox states of thiol are regulated by glutathione, thioredoxins and cysteine and they are very sensitive to two electrons oxidation by non-radical oxidants such as aldehydes, quinones, epoxides and peroxides produced from exogenous and endogenous precursors. 11
Based on the facts that artemisinin-induced hormonal imbalance and oxidative stress in the uterus, but not in the ovary of rats, 9 and that the generation of reactive free radicals from the reduction of the peroxide bridge of artemether is the mechanism responsible for its toxicity, 2 we sought to investigate the impacts of artemether–lumefantrine combination on the endocrine balance as well as oxidative stress and antioxidant status in the ovary and uterus of rats.
Materials and methods
Chemicals
Pure powdered artemether (99.5%) and lumefantrine (98.5%) were obtained from Emzor Pharmaceutical Industries (Lagos State, Nigeria). Glutathione, 5′,5′-dithio-bis-2-nitrobenzoic acid, H2O2, epinephrine, thiobarbituric acid and 1-chloro-2,4-dinitrobenzene (CDNB) were purchased from Sigma Chemical Co. (St. Louis, Missouri, USA). All the other reagents used in this study were of analytical grade, and they were purchased from British Drug Houses (Poole, Dorset, UK).
Animals treatment and sample collection
Female Wistar rats weighing between 150 g and 200 g were obtained from the Central Animal House Facility of the College of Medicine, University of Ibadan, Nigeria. They were kept in a well-ventilated room in plastic cages under controlled conditions of 12-h light/12-h dark cycle and temperature 23
Following treatment with the drugs, rats were allowed to fast overnight, killed by cervical dislocation and blood was collected through ocular vein into ethylenediaminetetraacetic acid anticoagulant bottles. Subsequently, blood was centrifuged at 3000g at room temperature for 15 min, and the plasma separated from blood sediments was used for hormonal analyses. Then, the ovaries and uteri were quickly excised, rinsed in cold 1.15% potassium chloride (KCl), weighed, and then homogenized in 50 mM of Tris-hydrochloride buffer (pH 7.4) containing 1.15% KCl, and centrifuged at 10,000g for 10 min to obtain the post mitochondrial supernatant fractions used for the determination of selected oxidative stress and antioxidant markers.
Analyses of oxidative stress and antioxidant markers
Determination of H2O2 generation
H2O2 generated following exposure to the drugs was determined by the method of Wolff. 14 The method is based on ferrous oxidation with xylenol orange reagent prepared with xylenol orange (100 μM L−1), ammonium ferrous sulfate (250 μM L−1), sorbitol (100 mmol L−1), and of sulfuric acid (25 mmol L−1). After this, 50 μL of the sample was added to the mixture, vortexed, incubated at room temperature for 30 min and the absorbance was measured spectrophotometrically at 560 nm and values expressed in μmol/mg of protein.
Determination of Lipid peroxidation status
Lipid peroxidation (LPO) status was determined based on MDA generated according to the method of Buege and Aust. 15 The sample (0.4 mL) was mixed with Tris-KCl buffer (1.6 ml) containing 30% TCA (0.5 mL). After adding 0.5 mL of 0.75% TBA to each of the tubes, they were incubated in a water bath at 80°C for 45 min, allowed to cool in ice and centrifuged at 3000g. Subsequently, the absorbance of the clear supernatant in each tube was read in a spectrophotometer against a reference blank at 532 nm. LPO was expressed in µUnits mg−1 protein using a molar extinction coefficient of 1.56 × 105 m−1cm−1.
Determination of GSH level
The GSH level was determined by Jollow et al.’s 16 method. After an aliquot of the sample was deproteinized by the addition of an equal volume of sulfosalicylic acid (4%) and centrifuged at 10,000g for 15 min at 4°C, 50 μL of the supernatant obtained was added to 4.5 mL of 10 mM 5,5′-dithiobis-(2-nitrobenzoic acid) (DTNB). The absorbance was then measured at 412 nm and the values were expressed in units/mg protein.
Determination of GPx activity
The GPx activity was determined using Rotruck et al.’s 17 method. The reaction containing 500 μL of sodium phosphate buffer, 100 μL of 10.0 mM of sodium azide, 200 μL of 4.0 mM of GSH, 100 μL of 2.5 mM of H2O2, and 50 μL of the sample was made up to 2.0 mL with distilled water and incubated for 3 min at 37°C. After the termination of the reaction with 0.5 mL of 10% trichloroacetic acid, it was centrifuged and the supernatant obtained was used for the determination of residual GSH content by the addition of 4.0 mL of disodium hydrogen phosphate (0.3 M) solution and 1 mL of DTNB reagent. The absorbance was read in a spectrophotometer at 412 nm and GPx activity expressed in µmol GSH consumed per minute per milligram protein.
Determination of GST activity
The GST activity was determined using the method of Habig et al. 18 This involves the pre-incubation of a mixture containing 1.7 mL of 100 mmol L−1 of phosphate buffer (pH 6.5) and 0.1 mL of 30 mmol L−1 of CDNB for 5 min at 37°C. Then, 20 μL of the sample was added and the absorbance of the solution was monitored for 5 min spectrophotometrically at 340 nm. The activity of GST was expressed in units/min/mg protein using an extinction coefficient of 9.6 mM−1 cm−1.
Determination of SOD activity
The activity of SOD was determined using the method of Misra and Fridovich. 19 This was based on inhibition of autoxidation of epinephrine (pH 10.2) at 30°C. The assay mixture was made up of 20 μL of the sample and 2.5 mL of carbonate buffer (0.05 M, pH 10.2). After equilibration in the spectrophotometer, freshly prepared 0.3 mL of 0.3 mM adrenaline was added and mixed by inversion. Thereafter, the increase in absorbance at 480 nm was monitored spectrophotometrically at 30 s intervals for 150 s. The activity of SOD was expressed in units/min/mg of protein.
Determination of plasma hormonal levels
Follicle-stimulating hormone (FSH), prolactin, progesterone and oestrogen levels were assessed using commercial enzyme immunoassay kits according to the manufacturers’ protocols. 9
Ovarian and uterine histological evaluations after exposure of rats to artemether–lumenfantrine combination therapy
The ovary and uterus histological evaluations were also carried out as previously reported (data not shown). 9
Statistical analyses
The data were expressed as mean
Results
Animal observation
After the 3 days exposure period, there was no visible clinical sign of toxicity in the rats exposed to artemether and lumefantrine monotherapies and their combination therapy.
Implications of artemether–lumefantrine combination therapy on ovarian and uterine oxidative stress markers of rats
The results of the ovarian and uterine oxidative stress markers (H2O2 and MDA) after the treatment of rats with artemether–lumefantrine combination therapy are shown in Figures 2 and 3. It appears that artemether and lumefantrine monotherapies as well as combined therapy do seem not to have any significant effect on the levels of ovarian H2O2 generated (Figure 2(a)) and MDA (Figure 3(a)) after the duration of treatment (p > 0.05). However, uterine levels of H2O2 (Figure 2(b)) and MDA (Figure 3(b)) increased significantly in the artemether-, lumefantrine- and artemther-lumefantrine-treated groups when compared with the control (p < 0.05). In addition, there were enhanced elevations of H2O2 in the lumefantrine group (Figure 2(b)) and MDA in the artemether–lumefantrine-treated group (Figure 3(b)) compared with the artemether monotherapy group (p < 0.05).
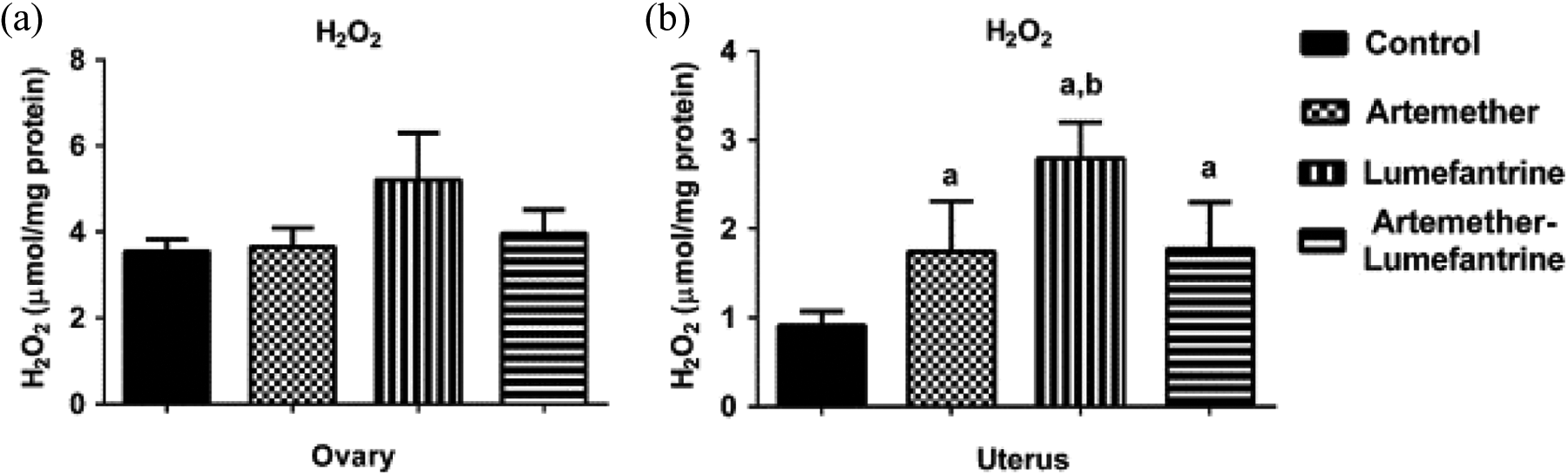
Ovarian and uterine H2O2 generation after exposure of rats to artemether, lumefantrine and artemether–lumefantrine combination for 3 days. Data are presented as mean
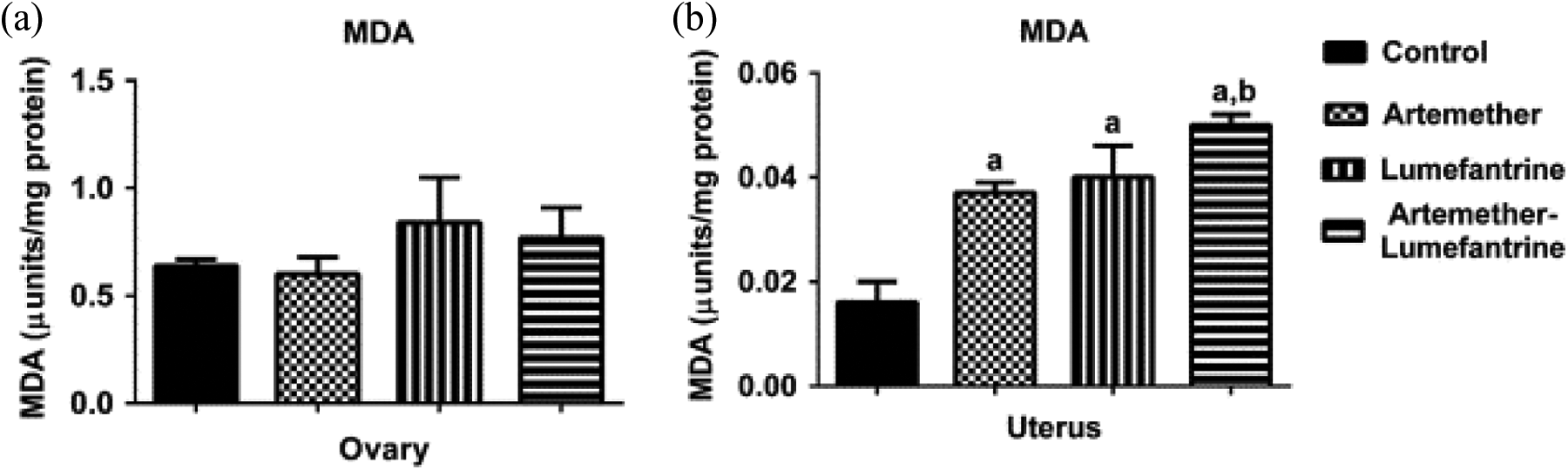
Ovarian and uterine LPO status after exposure rats to artemether, lumefantrine and artemether–lumefantrine combination for 3 days. Data are presented as mean
Implication of artemether–lumefantrine combination therapy on ovarian and uterine antioxidant status of rats
The effects of artemether–lumefantrine combination therapy on the ovarian and uterine antioxidants levels are shown in Figures 4 –7. Treatment of rats with artemether and lumefantrine both as monotherapies and combined therapy significantly reduced ovarian (Figure 4(a)) and uterine (Figure 4(b)) levels of GSH (a nonenzymatic antioxidant) compared with the control (Figure p < 0.05).

Ovarian and uterine GSH levels after exposure of rats to artemether, lumefantrine and artemether–lumefantrine combination for 3 days. Data are presented as mean

Ovarian and uterine GPx activity after exposure of rats to artemether, lumefantrine and artemether–lumefantrine combination for 3 days. Data are presented as mean

Ovarian and uterine GST activity after exposure of rats to artemether, lumefantrine and artemether–lumefantrine combination for 3 days. Data are presented as mean
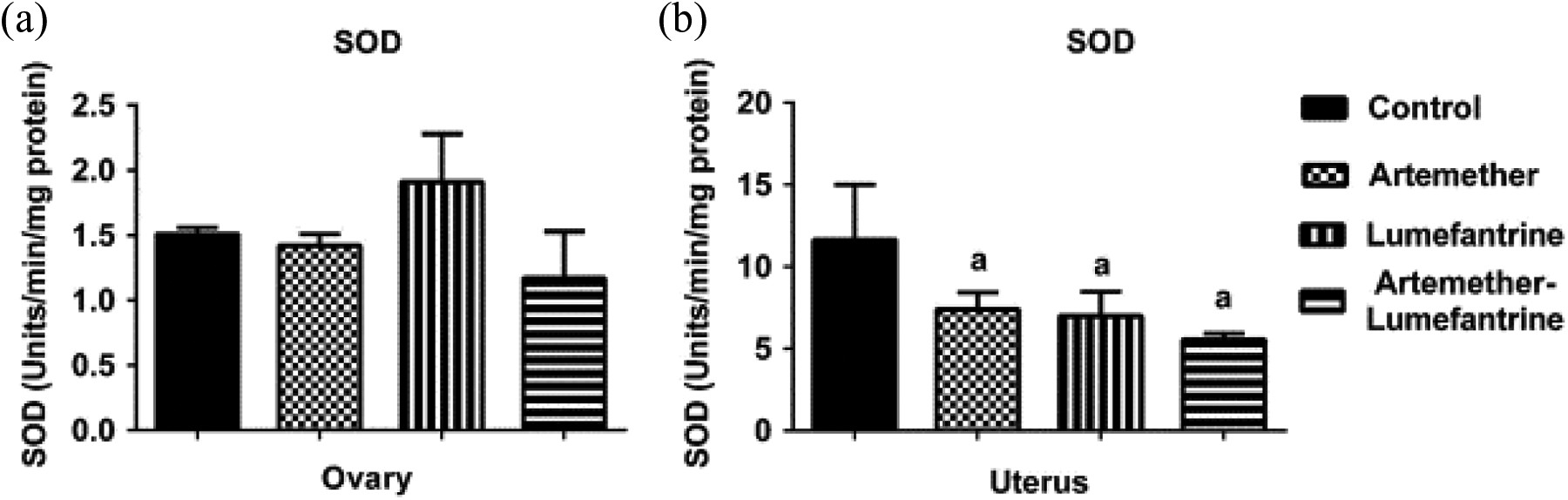
Ovarian and uterine SOD activity after exposure of rats to artemether, lumefantrine and artemether–lumefantrine combination for 3 days. Data are presented as mean
Whilst ovarian GPx activity reduced only in the lumefantrine monotherapy group (Figure 5(a)), uterine GPx (Figure 5(b)) increased in the artemether monotherapy as well as the artemether–lumefantrine groups compared with the control (p < 0.05). Further, whilst artemether and lumefantrine monotherapies and combined therapy significantly reduced the activity of GST (Figure 6(a)), uterine GST reduced in the artemether and lumefantrine monotherapy groups (Figure 6(b)) compared with the control (p < 0.05). Additionally, artemether–lumefantrine combination therapy reduced ovarian GST (Figure 6(a)), but increased uterine GST (Figure 6(b)) when compared with the artemether monotherapy group (p < 0.05).
Furthermore, there was no change in ovarian SOD activity in the artemether and lumefantrine monotherapies and combined therapy groups, whilst uterine SOD activity reduced in the artemether, lumefantrine and artemether–lumefantrine groups compared with the control (p < 0.05).
Implication of artemether–lumefantrine combination therapy on endocrine balance of female rats
In order to understand the influence of artemether–lumefantrine combination therapy on endocrine balance, we further determined selected levels of reproductive hormones after the 3 days of exposure (Figure 8(a–d)). Both lumefantrine and artemether–lumefantrine reduced the levels of FSH when compared with the control (Figure 8(a), p < 0.05). In Figure 8(b), although there was no change in prolactin level in the artemether–lumefantrine group (p > 0.05), artemether and lumefantrine monotherapies increased significantly prolactin levels compared with the control (p < 0.05). In addition, lumefantrine and artemether–lumefantrine groups reduced progesterone levels, whilst oestrogen levels reduced significantly in the artemether, lumefantrine and artemether–lumefantrine groups compared with the control (p < 0.05).
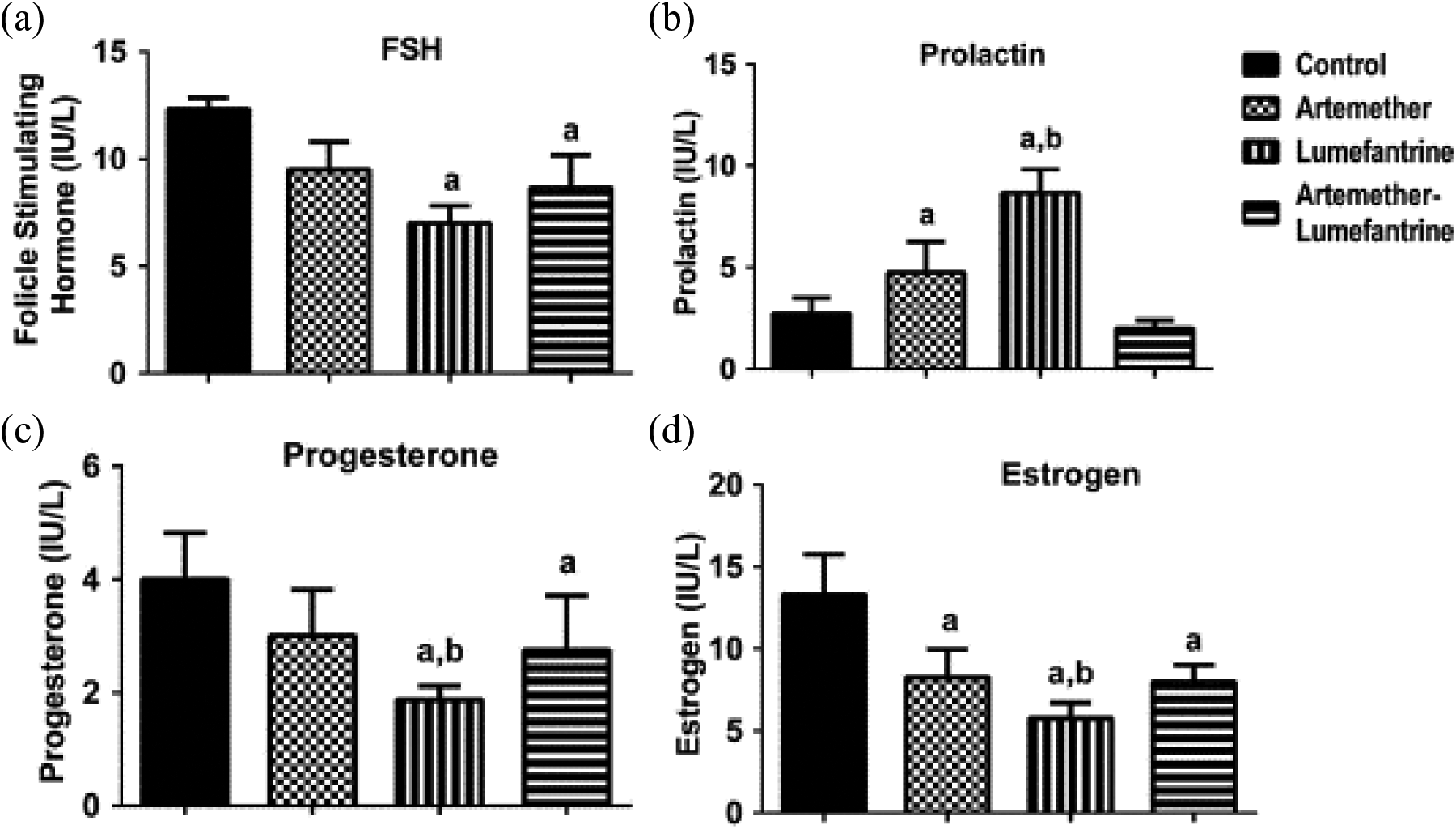
The levels of (a) FSH, (b) prolactin, (c) progesterone and (e) oestrogen in female rats exposed to artemether, lumefantrine and artemether–lumefantrine combination for 3 days. Data are presented as mean
Implication of artemether–lumefantrine combination therapy on the ovarian and uterine histology of rats
The histological results of the ovary and the uterus after exposure of rats to artemether–lumefantrine combination therapy (data not shown) revealed absence of visible lesions under the light microscope used.
Discussion
Previous studies on artemisinin and its derivatives revealed that these antimalarials are neurotoxic in different animal models. 20,21 Due to the hypothalamus–pituitary–ovarian axis connection, we investigated the implications of therapeutic doses of artemether and lumefantrine combination therapy on the endocrine balance and oxidative stress in the ovary and uterus of rats after 3 days of exposure. The results revealed that artemether–lumefantrine combination therapy disrupts hormonal balance and induced oxidative stress in the ovary and uterus of rats.
In the present study, artemether and lumefantrine monotherapies and combined therapy did not alter the levels of oxidative stress markers H2O2 and MDA in the ovary. However, uterine H2O2 and MDA levels were raised significantly in rats dosed with artemether and lumefantrine monotherapies and cotreatment groups. The fact that the therapeutic doses of the drugs did not increase ovarian H2O2 and MDA levels may suggest that the toxicity of the drugs in the ovary is not linked with lipid peroxidative pathway. On the contrary, the observed elevated levels of uterine MDA and H2O2 imply that artemether–lumefantrine combination therapy induces oxidative stress in the uterus due to enhanced LPO. This is due to the fact that increased production of MDA is a biomarker of oxidative stress in any system since this condition disrupts the physicochemical properties of the lipid bilayers of membrane, which leads to cellular dysfunction. 22 These data are in agreement with our recent study in which artemisinin significantly raised the levels of H2O2 and MDA in the uterus without affecting the ovary. 9 In addition, antimalarials such as mefloquine and halofantrine, 23 amodiaquine, 24 artesunate, 25 artemether–lumefantrine, 13,26 artesunate–amodiaquine 27 and artemisinin 28,9 have been reported to induce increased generation of H2O2 and/or MDA in different tissues such as erythrocytes, liver, brain, kidney, ovary and uterus.
Furthermore, we observed significant reductions in ovarian and uterine GSH levels, following exposure of rats to artemether–lumefantrine combination therapy. Glutathione, a tripeptide thiol molecule, is in abundance in the female reproductive tissues, where it is maintained in the reduced state by the NADPH-dependent reductases. 29 The reductions in ovarian and uterine GSH levels may be due to an attempt to maintain these tissues in the reduced state due to the formation of conjugates 30 with electrophilic metabolites of these drugs. Consequently, there is a disturbance in the redox homeostasis in the ovary and uterus of the rats. Due to the fact that GSH is important in several biochemical processes including signal transduction and transcription factor binding to DNA, the ovarian and uterine redox imbalance is an indication of the oxidative stress implication of these drugs and/or their metabolites in these tissues. Although, artemether–lumefantrine combination therapy did not alter ovarian MDA and H2O2 levels as presented here, the reduced level of ovarian GSH implies that the mechanism of toxicity of these drugs in the ovary is via the disruption of the redox balance. Whereas elevated H2O2 and MDA could cause damage to cellular components in the ovary and uterus, disruption of the redox homeostasis usually leads to the initiation of signalling response (stress response). 31
As a phase II metabolizing enzymes, GSTs play catalytic role in the conjugation of GSH with electrophilic centres of endogenous and exogenous molecules, which eventually leads to the detoxification of diverse toxic substances. Thus, the reduction in ovarian and uterine GST activity recorded here may imply that the phase II detoxification system of these tissues was impaired by the drugs. In this context, oxidative stress is implicated in the ovary and uterus of rats treated with artemether–lumefantrine combination therapy since GST also regulates several cellular processes involved in the survival of cells to oxidative stress. 32 This GST reduction might be connected with the reduction in ovarian and uterine GSH recorded in this study and/or a direct impact of the metabolites of the drugs on GST. The report of Farombi et al. 9 is partly in agreement with the present study. In this case, only uterine GST activity reduced, whilst ovarian GST activity was enhanced after 7 days of exposure of rats to artemisinin. Although, artemether is a derivative of artemisinin, one would not expect similar outcome in the two studies, since artemisinin was administered as a monotherapy, whilst artemether was exposed to the rats in combination with lumefantrine.
Furthermore, the drugs did not alter ovarian SOD activity, but significantly decreased uterine SOD activity. Superoxide radical (O2*−), H2O2 and hydroxyl radical (HO*) are usually elevated in tissues during the metabolism of drugs. The O2*− generated in the mitochondria is dismutated by SOD to H2O2. Then, the H2O2 reacts with Fe2+ (Fenton Reaction), to produce HO*, which is a strong reactive oxidant. 33 Since artemether–lumefantrine combination therapy did not alter the levels of ROS such as the ovarian H2O2, it is expected that the drugs would not alter the activity of ovarian SOD as noted in this study. Thus, the mechanism of artemether and lumefantrine toxicity in the ovary is not through the elevation of reactive oxygen species, but via the disruption of the redox homeostasis as explained earlier. In addition to the disturbance in redox status in the uterus, the drugs also decreased the activity of uterine SOD. Although, we did not investigate the activity of catalase in the present study, it is expected that its activity might be enhanced in the uterus, but possibly unaffected in the ovary as indicated here. The data in the present investigation further indicated that lumefantrine monotherapy decreased ovarian GPx, whereas uterine GPx was significantly enhanced by artemether monotherapy and artemether–lumefantrine combination therapy. GPx together with catalase metabolize H2O2 to water and molecular oxygen after the conversion of O2*− to H2O2. Thus, the stimulation of uterine GPx was an adaptive response to the elevated concentration of H2O2 generated after the exposure of rats to the drugs.
In order to assess the implication of artemether–lumefantrine combination therapy on the endocrine balance, we investigated the levels of selected female reproductive hormones routinely measured as markers of female reproductive-related disorders. FSH is among the glycoprotein hormone family that includes thyroid-stimulating hormone, luteinizing hormone and chorionic gonadotropin. FSH is produced in the gonadotroph of the pituitary and its receptors are located in the granulosa cells of the ovary. 34 The observed reduction in the level of FSH might have induced decreased production of oestrogenic level noted in this study, since FSH stimulates the ovarian follicles to synthesize oestrogen in order to promote the maturation of eggs. In this context, a decreased FSH level may impair reproductive function by blocking the process of folliculogenesis. 34 Since oestrogen plays critical role in female fertility, especially ovulation, artemether–lumefantrine combination therapy could hamper ovulation process in the absence of malaria. Additionally, oestrogen and progesterone are the two foremost ovarian hormones that specify uterine receptivity following fertilization. Thus, if ovulation occurs, the decreased oestrogen and progesterone levels could alter uterus lining making it unreceptive to pregnancy. 35,36
Although the drugs disrupt endocrine balance and induced oxidative stress in the ovary of rats, the histological results (data not shown) revealed the absence of visible lesions similar to what we noted in female rats exposed to artemisinin for 7 days. 9 One limitation of the present study is the fact that we did not carry out the levels of pro- and anti-apoptotic proteins in the ovary and uterus of the rats, which would have provided further insights on the mechanisms of toxicity of these drugs, nonetheless, the endpoint markers of oxidative stress and antioxidant status provided here clearly indicated that oxidative stress is implicated in the mechanism of toxicity of these antimalarials.
In conclusion, artemether–lumefantrine combination therapy induced ovarian and uterine toxicities via different mechanisms. Whereas, toxicity of the drugs in the ovary was via the disruption of the redox homeostasis without increasing the levels of reactive oxygen species, that of the uterus encompasses both. In both instances, oxidative stress was implicated following exposure of rats to artemether–lumefantrine combination therapy. Additionally, the drugs also disrupt the endocrine balance as evidenced by the changes in the reproductive hormonal levels of the rats. Although, artemether–lumefantrine combination therapy is efficacious against drug-resistant malarial parasite, however, in the absence of malarial parasite infection, the abuse of the drug should be avoided by women in malarial endemic regions of tropical Africa.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
