Abstract
Objective
The study aimed to investigate the role of luteolin in alleviating POF and its underlying molecular mechanisms.
Methods
POF model was established in rats by intraperitoneal injection of 100 mg/kg cyclophosphamide. Then, rats in the treatment group received intragastric administration of with luteolin at doses of 25 mg/kg, 50 mg/kg and 100 mg/kg, respectively. Rats in POF model group were administered the same volume of saline intragastrically. The concentrations of E2, FSH, AMH, P, LH, SOD and MDA in serum were quantified using ELISA kits. H&E and TUNEL staining were employed to assess pathological alterations and apoptosis. The cellular localizations of 4-HNE, 8-OHdG and NTY were detected by immunohistochemistry staining. The PharmMapper database and UbiBrowser prediction were used for the prediction of luteolin interaction with RNF8/HDAC2.
Results
Luteolin treatment significantly increased serum levels of estradiol (E2) (P < 0.01) and anti-Müllerian hormone (AMH) (P < 0.01) while reducing follicle-stimulating hormone (FSH) and luteinizing hormone (LH) levels (P < 0.05), restoring ovarian function. Additionally, luteolin could significantly improve ovarian tissue morphology and reduce apoptosis. ELISA kit results indicated luteolin significantly reduced the levels of SOD and MDA. Immunohistochemical staining results revealed a significant decrease in the expressions of 4-HNE, 8-OHdG and NTY in luteolin treatment group. Combining The PharmMapper database and UbiBrowser prediction, we found luteolin could bind RNF8 and inhibit HDAC2 expression.
Discussion
Luteolin could mitigate cyclophosphamide-induced ovarian senescence, with its molecular mechanisms involving the regulation of RNF8/HDAC2 signaling axis and the inhibition of oxidative stress.
Introduction
Premature ovarian failure (POF) is a gynecological disease characterized by amenorrhea, infertility, diminished estrogen levels, and excessive gonadotropin secretion, leading to the main cause of infertility, and the incidence of women under 40 years of age is about 5%. 1 Premature failure of the follicles due to accelerated atresia of the original follicles, a lack of follicles that leads to reduced ovarian function. And, the onset of POF is directly linked to the functional status of ovarian granulosa cells. It has been reported2,3 that the pathological mechanism of premature ovarian failure is related to chromosome mutation, viral infection, drug chemotherapy and other effects. Patients with premature ovarian failure are often associated with infertility, osteoporosis, cardiovascular disease, and even an increased risk of death.4,5 Hormone replacement therapy (HCT) can improve symptoms in patients with POF to some extent, but it could increase the risk of endometrial cancer and breast cancer. 6 Hence, discovering novel therapeutic agents that could enhance efficacy while minimizing adverse effects is of paramount importance in the clinical management of POF.
During recent decades, traditional Chinese medicine has emerged as an irreplaceable modality in treating a myriad of illness, notably cancer, diabetes, cardiovascular and cerebrovascular diseases and respiratory diseases.7,8 Luteolin is a common flavonoid monomer, which mainly exists in a variety of plants. 9 In traditional Chinese medicine, plants rich in luteolin have been used to treat diseases such as cardiovascular disease, inflammatory diseases and cancer. Luteolin exhibits a diverse range of pharmacological effects, including anti-inflammatory, anti-tumor, antiviral. Clinically, luteolin exhibits a multifaceted profile, including promoting blood circulation to alleviate stagnation, eliminating phlegm and alleviating cough, as well as exhibiting anti-tumor properties. Wu et al. reported 10 that luteotein inhibits the expression of MMP9 and prevents the proliferation and metastasis of triple-negative breast cancer (TNBC) by down-regulating the levels of H3K27Ac and H3K56Ac induced by AKT/mTOR. Yi et al. demonstrated 11 that luteolin can play a potential role as an inhibitor of Musash1 in the treatment of glioma. Luteolin alleviated ischemia reperfusion-induced myocardial infarction by reducing ROS and inhibiting ferroptosis. 12 However, its therapeutic effect and molecular mechanism of luteolin in POF have not been reported.
Oxidative stress plays a crucial role in the onset and progression of POF, particularly through oxidative damage to granulosa cells and oocytes. 13 Oxidative stress could promote cell apoptosis and mitochondrial dysfunction, accelerating follicular depletion and ovarian dysfunction. RNF8, an E3 ubiquitin ligase, plays an important role in responding to oxidative stress by regulating protein ubiquitination and degradation, potentially mitigating oxidative damage. We hypothesized that RNF8 may serve as a potential molecular target for treating POF.
Although luteolin has demonstrated antioxidant properties in cardiovascular and neurodegenerative diseases, its role in premature ovarian failure remains unexplored. Given that RNF8 plays a key role in oxidative stress regulation, we hypothesized that luteolin may mitigate ovarian damage by modulating the RNF8/HDAC2 signaling axis. In the study, we investigated the role of luteolin in cyclophosphamide-induced POF and its molecular mechanism. In a cyclophosphamide-induced rat model, we determined the therapeutic effect of luteolin. In addition, we explored the effects of luteolin on oxidative stress in POF rats and its potential drug targets.
Methods
Cell culture and treatment
The 8-week-old female SD rats (n = 24) were obtained from Beijing Vital River Laboratory Animal Technology Co., Ltd. The rats were killed by cervical dislocation, and their ovarian tissue was isolated under sterile conditions and placed in PBS at 4°C. It was chopped, 2.0 ml hyaluronidase was added for digestion at 37°C for 1 min. Digestion was terminated by adding 200 μL fetal bovine serum, mixed with 5 mL PBS, and centrifuged at 1500 rpm for 5 min. The supernatant was discarded, the supernatant was discarded, and the cell particles were re-suspended by adding DMEM: F12 (1:1) and mixed evenly. The cell suspension was inoculated into a 6-well cell culture plate and maintained at 37°C with 5% CO2. When the cell fusion reached 80%, and they were used in the subsequent cell experiments.
Premature ovarian failure model and groups
A total of 30 female SD rats (6 rats per group, aged 8 weeks, 220 ± 10 g) were housed in cages maintained at a humidity of 55%, a temperature of 20°C, and a 12-h light/12-h dark cycle. They had free access to food and water. Premature ovarian failure model was established in rats by intraperitoneal injection of 100 mg/kg cyclophosphamide on the first day and then with 8 mg/kg/day cyclophosphamide for 14 days.14,15 After the success of the model, the rats in the treatment group were intragastric administration with 25 mg/kg luteolin, 50 mg/kg luteolin and 100 mg/kg luteolin, respectively. Rats in POF model group were intragastric administration the same amount of saline. After 28 days of treatment, the rats were euthanized without pain using intraperitoneal lethal doses of pentobarbital sodium. Ovarian tissue and blood were harvested for follow-up experiments. All animal procedures complied with the ethical standards of the Animal Protection and Ethics Committee of Guizhou Provincial People’s Hospital (Approval No. GPH-DWLL-20231209) and followed the internationally accepted guidelines for the care and use of laboratory animals. All efforts were made to minimize animal suffering, and euthanasia was performed using an intraperitoneal overdose of pentobarbital sodium to ensure a humane endpoint.
Western blotting assay
As previously reported, 16 western blot was performed to detect protein expression levels in ovarian tissue and ovarian granulosa cells. Proteins (40 μg) was subjected to SDS-PAGE. The isolated protein was then transferred to PVDF membrane and sealed with 5% skimmed milk at room temperature for 1 h. Subsequent incubation with primary antibodies (RNF8, ab128872, 1:1000; HDAC2, ab32117, 1:1000; and GAPDH, ab8245, 1:1000) at 4°C overnight. After washing the membranes with TBST, secondary antibodies were incubated at room temperature. Finally, visualization was performed by enhanced chemiluminescence reagent. The relative expression of the protein was assessed by calculating its gray value, with GAPDH serving as the internal reference.
RT-qPCR analysis
Total RNA was extracted from ovarian granulosa cells and ovarian tissue with TRIzol reagent (Invitrogen, USA). Total RNA was reverse transcripted to cDNA by using cDNA Reverse Transcription Kit (Applied Biosystems, NO.4368813) according to the manufacturer’s instructions. Quantitative RT-PCR was performed with the qRT-PCR Kit (Clontech, NO.639201). GAPDH was used as the internal reference for mRNA. The target genes relative levels were analyzed by the 2-ΔΔCt method. The primers used in this study were as follows: RNF8 (Forward: 5′-GGTTACCACTCCATAACCATCTGTACG-3′; Reverse: 5′-CAGAAGGTAGCAACAGAACACGACG-3′), HDAC2 (Forward: 5′- ATGGCGTACAGTCAAGGAGG-3’; Reverse 5′-TGCGGATTCTATGAGGCTTCA-3′), GAPDH (Forward: 5′-GGTTGTCTCCTGCGACTTCA-3′; Reverse: 5′-CCA CCACCCTGTTGCTGTAG-3′).
Co-immunoprecipitation assay
Co-immunoprecipitation (Co-IP) assay was conducted according to the method as previously reported. 17 In brief, whole-cell protein lysates were obtained using IP lysis buffer (1.0% NP-40 lysis buffer, 0.2 mM EDTA, 20 mM pH 8.0 Tris-HCl, 180 mM NaCl), phosphatase inhibitors (Thermo), and protease inhibitor cocktail (Roche) at 4°C. After centrifugation at 12000 rpm for 10 min at 4°C, the supernatants were prepared for endogenous IP or exogenous IP. For endogenous IP, the supernatants were incubated with corresponding antibodies and Protein A/G Magnetic Beads (MCE) at 4°C overnight. For exogeneous IP, supernatants were incubated with anti-FLAG or anti-HA antibodies and Protein A/G Magnetic Beads (MCE) at 4°C overnight. Subsequently, the samples were separated by a magnetic separator and denatured by SDS loading buffer, and examined by western blotting.
Hematoxylin and eosin(HE) staining
Simply, the ovarian tissues were then dewaxed by ethanol gradient dehydration, paraffin embedding, section, and xylene immersion. The sections were stained with haematoxin eosin, permeated with xylene and sealed with neutral gum. Finally, the slices are viewed under a microscope and photographed.
Terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) staining
As previously reported, 18 the apoptosis was detected using the TUNEL kit. TUNEL staining was performed following the kit instructions. Briefly, paraffin-embedded ovarian tissue sections (4 μm) were dewaxed in xylene and rehydrated in a gradient of ethanol. Then, they were incubated with TUNEL mixture for 60 min at 37°C. DAPI counterstaining was performed for 5 min at room temperature. Finally, the sections were observed and photographed under a fluorescence microscope. The apoptosis rate was calculated as the percentage of TUNEL-positive signals to DAPI-positive signals (%).
Immumohistochemical staining
Immunohistochemical staining was performed according to previously reported methods. 19 After the paraffin section was dewaxed with xylene and dehydrated with alcohol, 3% H2O2 was applied to the slides to block endogenous peroxidase activity. The slides were then washed with PBS and incubated with blocking serum to prevent nonspecific binding. Then, the sections were incubated with primary antibodies specific to the target proteins(HDAC2, ab219053, 1:200; 4-HNE, ab125066, 1:200; 8-OHdG, NTY) overnight at 4°C. After washing, the sections were incubated with appropriate secondary antibodies conjugated to HRP for 1 h at room temperature. Finally, the sections were developed with DAB, counterstained with hematoxylin, and observed under a light microscope.
ELISA kit assay
After 28 days of treatment, the blood was collected and serum was obtained by centrifugation. The levels of E2, FSH, AMH, P, LH, SOD and MDA in serum were determined according to the instructions of the ELISA kits (HB-PD5774S, JL13251, SPS-31220, ER1880, ER1123, S0101S, S0131S).
UbiBrowser web services
UbiBrowser (https://ubibrowser.ncpsb.org/) is an integration of bioinformatics platform, can be used to predict RNF8 downstream substrates of molecular targets. 20
Target prediction of active ingredients
PharmMapper server were used to predict potential targets for all the predicted targets of luteolin. 21
Statistical analysis
Data normality was assessed using the Shapiro-Wilk test. For normally distributed data, one-way ANOVA followed by Tukey’s post-hoc test was performed using SPSS 22.0 software package. Non-parametric data were analyzed using the Kruskal-Wallis test followed by Dunn’s post-hoc test. A P value below 0.05 was deemed statistically significant.
Results
Protective effects of luteolin on the HPG axis and ovarian AMH in cyclophosphamide induced-premature ovarian failure
First, POF rats were treated with luteolin for 28 days, serum was collected for ELISA analysis. As depicted in Figure 1, the POF group exhibited lower levels of E2, P, and AMH compared to the control group. However, luteolin treatment significantly upregulated these hormone levels in the POF group. Additionally, the POF model group showed significantly elevated serum FSH and LH levels compared to the control group. While the levels of serum FSH and LH were decreased significantly in luteolin treatment groups compared with POF group. The findings indicated that luteolin had a dose-dependent effect on significantly enhancing the recovery of ovarian function. The serum levels of E2, FSH, LH, P and AMH in different groups E2 (A), FSH (B), LH (C), P (D) and AMH (E). FSH: follicle-stimulating hormone; LH: luteinizing hormone; AMH: anti-Mullerian hormone. For normally distributed data, one-way ANOVA followed by Tukey’s post-hoc test was performed. versus control group, **p < 0.01, ***p < 0.001; versus POF group, #p < 0.05, ##p < 0.01, n = 6.
Effect of luteolin on follicular development and apoptosis in the POF model
Next, the histopathological changes of ovarian tissue were observed by H&E staining. It (Figure 2(a)) showed all levels of follicles in the control group. There were fewer primary and secondary follicles in the POF model group. Compared with the POF model group, the number of follicles in the luteolin treatment group increased significantly and the number of atretic follicles decreased. TUNEL staining results (Figure 2(b)) showed the apoptosis was significantly increased in the POF model group and significantly decreased in the luteolin treatment group. The above results suggested that luteolin could inhibit cell apoptosis and improve follicle development in POF rats. Effect of luteolin on follicular development and apoptosis in the POF rats. H&E staining (a) was used to detect the histopathological changes of ovarian tissue in different groups, ×40. TUNEL staining (b) was performed to detect the apoptosis in ovarian tissue, ×40. For normally distributed data, one-way ANOVA followed by Tukey’s post-hoc test was performed. versus control group, *p < 0.05, **p < 0.01; versus POF group, #p < 0.05, ##p < 0.01, n = 3.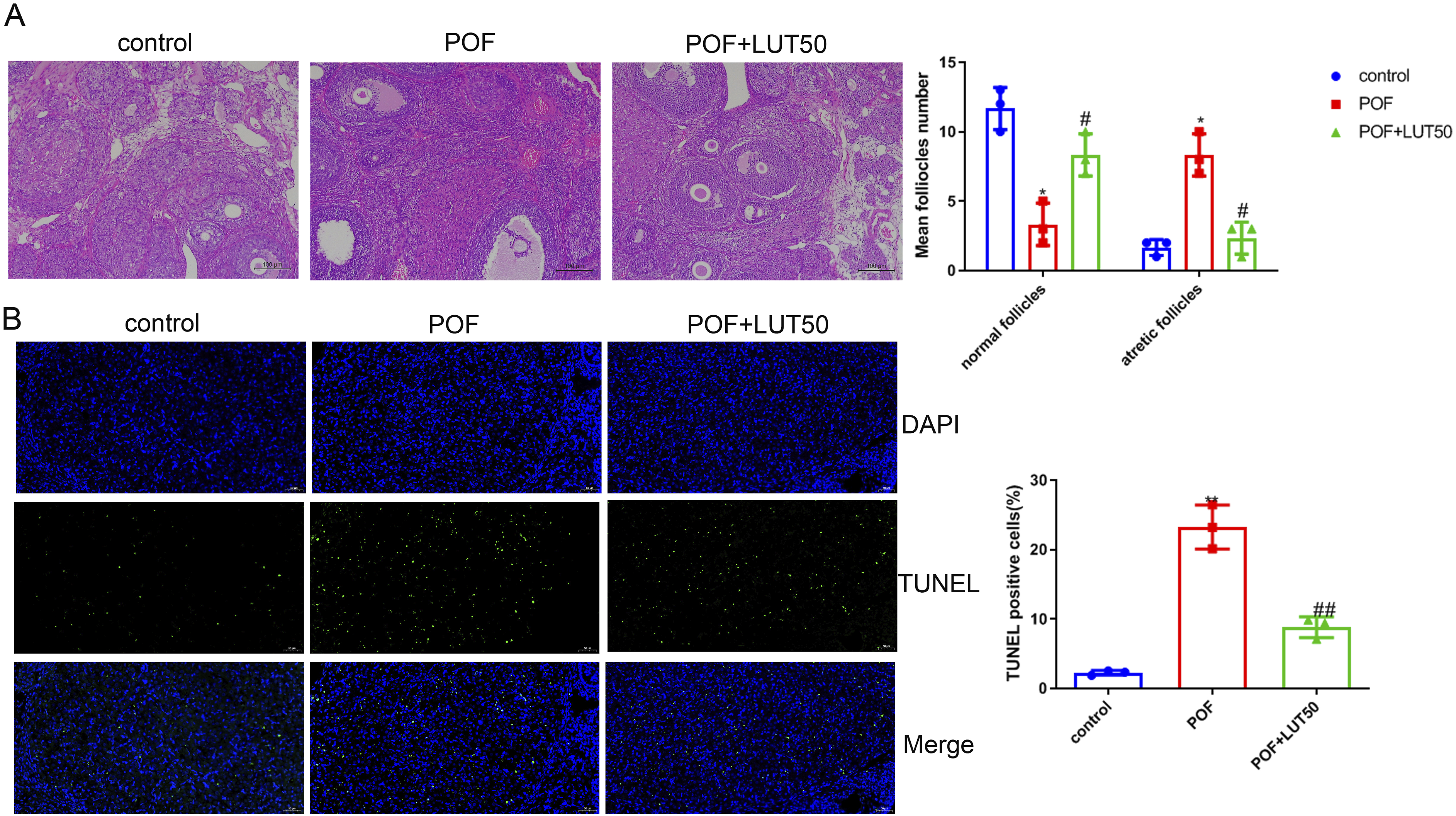
Effect of luteolin on oxidative stress in the POF rat
The results (Figure 3(a) and (b)) showed that SOD activity in POF group was significantly lower than that in control group, and MDA level was higher than that in control group. After luteolin treatment, SOD activity increased, MDA level decreased significantly. The cell locations of 4-HNE, 8-OHdG and NTY proteins were detected by immunohistochemistry staining. 4-HNE, NTY and 8-OHdG proteins are mainly located in ovarian stromal cells. The protein levels of 4-HNE, 8-OHdG and NTY were notably elevated in POF group compared to the control group. Concersely, the luteolin treatment group exhibited a significant reduction in the protein expression of these same markers, namely 4-HNE, 8-OHdG, and NTY (Figure 3(c)–(e)). In summary, luteolin could significantly inhibit oxidative stress in POF rats. Effect of luteolin on oxidative stress in the POF model. Serum levels of SOD (a) and MDA (b). Immunohistochemical staining was performed to detect the expressions of 4-Hydroxynonenal (4-HNE) (c), 8-hydroxy-2 deoxyguanosine (8-OHdG) (d) and nitrotyrosine(NTY) (e). For normally distributed data, one-way ANOVA followed by Tukey’s post-hoc test was performed. versus control group, *p < 0.05, **p < 0.01; versus POF group, #pP < 0.05, n = 6&3.
Luteolin targeted RNF8 and activated RNF8 expression in POF rats
PharmMapper database predicted that RNF8 might be a target binding molecule of luteolin (Figure 4(a)). RT-qPCR findings (Figure 4(b)) revealed a marked decrease in RNF8 mRNA expression in the POF group compared to the control group. Notably, luteolin treatment reversed this trend, restoring RNF8 mRNA levels. This pattern was corroborated by the western blot results (Figure 4(c)), which exhibited a similar trend in RNF8 protein expression. Luteolin targeted RNF8 and activated RNF8 expression in premature ovarian failure rats (a) The PharmMapper database was employed to predict the target proteins. (b) RT-qPCR analysis was used to quantify the mRNA expression of RNF8 in ovarian tissues. (c) Western blotting assay was utlized to detect the protein expression of RNF8 in the ovarian tissue. For normally distributed data, one-way ANOVA followed by Tukey’s post-hoc test was performed. versus control group, *p < 0.05, **p < 0.01; versus POF group, #p < 0.05, n = 3.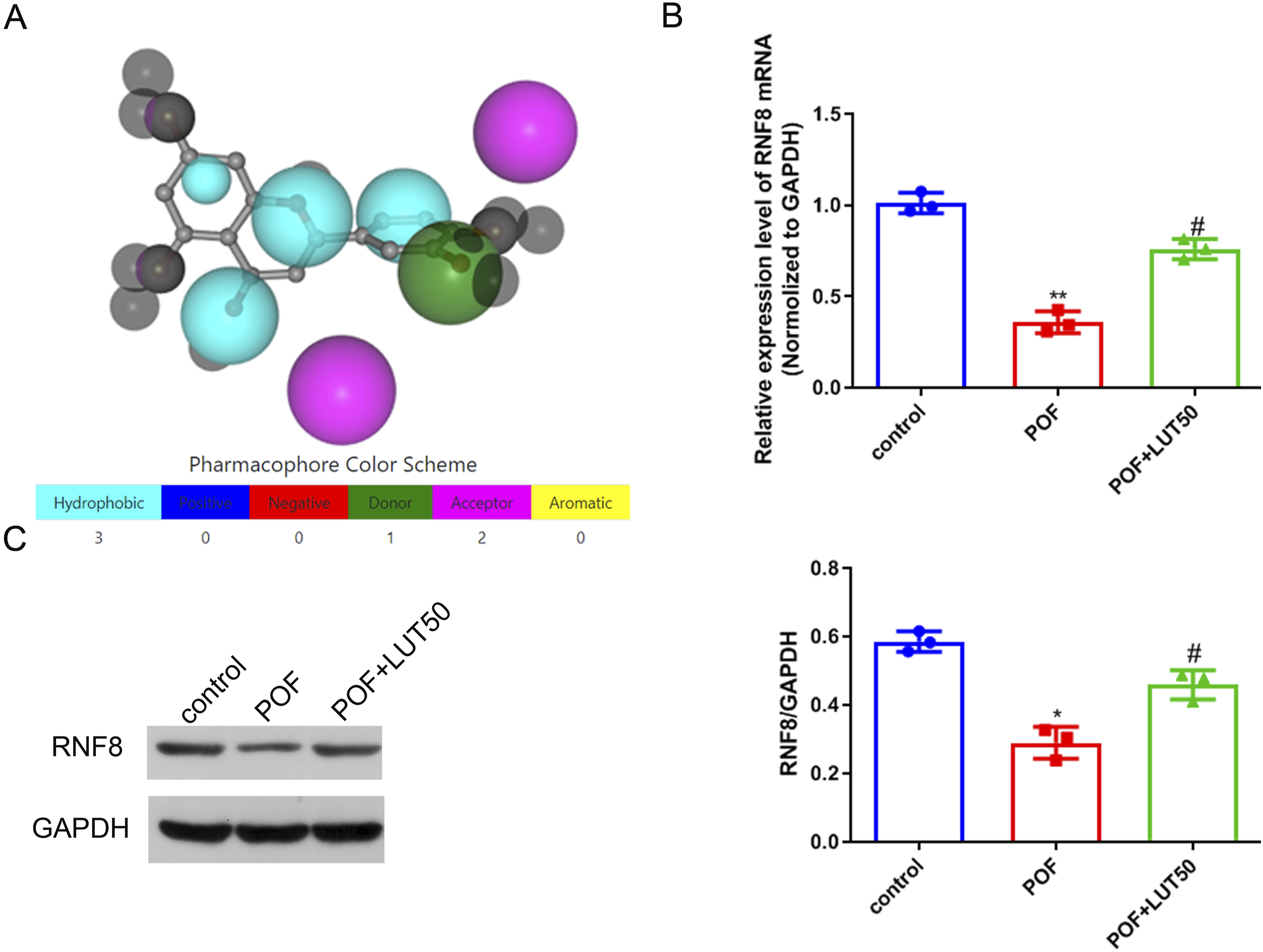
RNF8 induces HDAC2 ubiquitination and degradation in cyclophosphamide induced ovarian granulosa cells
Next, we investigated the molecular mechanism of ubiquitination regulation by RNF8 in cyclophosphamide induced ovarian granulocyte cells. Considering that RNF8 is a well-known E3 ubiquitination ligase and combining with UbiBrowser predictions, we speculate that HDAC2 may be the target substrate for RNF8 ubiquitination(Figure 5(a)). To verify that HDAC2 is indeed a substrate for RNF8, we first performed co-immunoprecipitation experiments to verify whether the two molecules can bind to each other. As shown in Figure 5(b), Co-IP results confirmed the interaction between HDAC2 and RNF8. Co-IP results (Figure 5(c)) showed that Myc-RNF8 treatment significantly enhanced the ubiquitination level of HDAC2 and accelerated the degradation of HDAC2 protein. Furthermore, western blot results (Figure 5(d)) revealed a notable downregulation of RNF8 protein expression in the CTX group compared to the control group, accompanied by a significant upregulation of HDAC2 protein levels. Interestingly, luteolin treatment reversed these trends, significantly enhancing RNF8 protein expression while concurrently diminishing HDAC2 protein levels. This finding underscores the potential regulatory role of luteolin in modulating the expression of RNF8 and HDAC2 in the context of ovarian tissue. RNF8 induces HDAC2 ubiquitination and degradation in cyclophosphamide induced ovarian granulosa cells (a) UbiBrowser prediction, (b) co-immunoprecipitation assay (Co-IP) was used to verify the interaction between RNF8 and HDAC2. (c) Co-IP was used to verify that RNF8 regulates HDAC2 ubiquitination. (d) Western blotting assay was used to detect the protein expression of RNF8 and HDAC2 in different groups. For normally distributed data, one-way ANOVA followed by Tukey’s post-hoc test was performed.versus control group, **p < 0.01; versus CTX group, #p < 0.05, n = 3.
Effect of luteolin on expression of HDAC2 in the POF model
Finally, we further studied the effect of luteolin on HDAC2 expression level in premature ovarian failure rats. RT-qPCR results (Figure 6(a)) demonstrated a significant increase in HDAC2 mRNA expression in the POF group compared to the control group, with suppression observed in the treatment group. Consistently, western blot results (Figure 6(b)) exhibited a similar trend. Immunohistochemical staining results (Figure 6(c)) further revealed a marked augmentation in the positive signal of HDAC2 in the premature ovarian failure group, as compared to the control group. The positive signal decreased in the luteolin treatment group. Taken together, could significantly inhibit the mRNA and protein expression of HDAC2 in ovarian tissue of POF rats. Effect of Luteolin on expression of HDAC2 in the POF model (a) The mRNA expression of HDAC2 in different groups were determined by RT-qPCR analysis. (b) The protein expression of HDAC2 in each group was detected through western blotting assay. (c) Immunohistochemical staining was performed to detect the expression of HDAC2. For normally distributed data, one-way ANOVA followed by Tukey’s post-hoc test was performed. versus control group, *p < 0.05, **p < 0.01; versus POF group, #p < 0.05, n = 3.
Discussion
Premature ovarian failure brings great psychological burden and distress to patients, and even increases the risk of death in serious cases. Although there is a large amount of basic researches focusing on the pathological mechanism underlying premature ovarian failure, no efficacious drug has been approved for clinical application to date. Our study revealed notable alterations in hormonal profiles among POF rats following luteolin treatment, specifically, a marked elevation in serum levels of E2, FSH and LH, alongside a decrease in P and AMH levels. Histological staining results also confirmed that ovarian follicles increased and apoptosis decreased in the luteolin treatment group. Our study suggested that luteolin effectively relieved premature ovarian failure and improved ovarian function.
During the aging process, the accumulation of endogenous reactive oxygen species disrupts the internal environmental balance, thereby exacerbating oxidative damage to cells. The reproductive capacity of granulosa cells in the ovary is particularly sensitive to oxidative stress. It is reported 22 that ROS elevation regulates the expression level of SIAH1 and accelerate the aging of ovarian granulosa cells. Consistently, our study also found significant increases in ROS levels in the POF model. Luteolin treatment reversed these changes, restoring normal SOD activity and reducing MDA levels, thereby mitigating oxidative stress. The immunohistochemical staining results further corroborated these findings, showing reduced expression of oxidative stress markers such as 4-hydroxynonenal (4-HNE), 8-hydroxy-2′-deoxyguanosine (8-OHdG), and nitrotyrosine (NTY) in luteolin-treated ovaries. Our study suggested that luteolin could inhibit oxidative stress and delay premature ovarian failure, which is crucial for preventing follicular depletion and maintaining ovarian function.
One of the most intriguing findings of this study is the role of RNF8 in luteolin’s action. RNF8 is an E3 ubiquitin ligase that plays a critical role in regulating the ubiquitination and degradation of key proteins involved in cellular stress responses. Subsequent studies showed that luteolin could target and regulate the expression of RNF8 and promote the upregulation of RNF8. The results of UbiBrowser database showed that HDAC2 is the substrate of RNF8, and RNF8 could negatively regulate the expression level of HDAC2. RING finger protein 8 (RNF8) is a member of the RING finger family that was initially identified by Seki in 1998. 23 Li et al. reported 24 that RNF8, through the enhancement of K48 polyubiquitination and subsequent targeting of the degradation of N1ICD molecules, exerts a negative regulatory effect on the Notch signaling pathway, thereby inhibiting the proliferation of breast progenitor cells. This mechanism ensures a refined control over cellular expansion in the breast tissue. As a co-activator of the transcription factors Twist and ERα, RNF8 also upregulates transcription factors to promote the growth and metastasis of breast cancer cells. 25 Zhu et al. found 26 that RNF8 plays a pivotal protective role in cerebral ischemic injury treatment by down-regulating the expression of HDAC2 and inhibiting the activity of GSK3β. HDAC family, functioning as negative regulators of histone acetylation, has emerged as a pivotal player in a diverse array of human diseases closely associated with oxidative stress. FoxF1 regulates the miR-342/KLF5/NF-κB p65 signaling axis to protect paraquat induced lung injury in rats following HDAC inhibition. 27 In the study, we discovered luteolin may decrease the level of HDAC2, thereby inhibiting oxidative stress and exerting a therapeutic role in POF rats. The molecular dynamics between RNF8 and HDAC2 offer a novel mechanistic insight into the therapeutic action of luteolin in POF.
Overall, our study confirmed that luteolin inhibited oxidative stress and played protective effects in POF rats. In addition, we revealed that its regulatory molecular mechanism may be closely related to the target molecule RNF8 and its downstream molecule HDAC2. Although this study establishes a protective role for luteolin against premature ovarian failure via RNF8/HDAC2 modulation, future research should explore its broader metabolic effects and validate findings in vitro granulosa cell models. Considering that the sample size in this study was insufficient (n = 6), the sample size will be expanded in subsequent animal experiments to further consolidate the efficacy of luteolin. Besides, clinical studies are needed to further explore the therapeutic potential of luteolin in human ovarian dysfunction and to validate the translational relevance of these findings in treating POF patients.
Footnotes
Acknowledgments
We are grateful to all participants for their contributions to the present study.
Ethical considerations
All animal experiments were performed in accordance with the ARRIVE guidelines to ensure transparency and reproducibility in preclinical research. All animal care and experimental procedures were approved by the Animal Care and Use Committee of the Institute of Guizhou Provincial People’s Hospital (Approval No. GPH-DWLL-20231209).
Consent to publish
The authors gave their consent for publication.
Authors contributions
Y. P. : Writing, Conceptualization, Methodology, Software
X. Z. : Data curation, Writing, Original draft preparation
X. R. : Visualization, Investigation, Software
Y. X: Supervision, Methodology, Investigation, Reviewing and Editing
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Correction (September 2025):
The affiliation of author “Yang Xu” was incorrect and has been corrected from “Obstetrics and Gynecology Department, Guizhou Provincial People’s Hospital, Guiyang, China” to “The First School of Clinical Medicine, Guizhou University of Traditional Chinese Medicine, China.”
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Raw data and experimental details are available upon request to ensure transparency and reproducibility in accordance with ethical research practices.
