Abstract
An oligodeoxynucleotide with CCT repeats (CCT ODN) has been found in our previous study to selectively downregulate Toll-like receptor 7/9 (TLR7/9)-mediated immune responses both in vitro and in vivo. In this study, we unexpectedly found that CCT ODN induced severe patchy hair loss around the mouth in male F1 mice (female Balb/c × male C57BL/6) with lupus-like nephritis induced by injecting allogenic lymphocytes and also in male Balb/c mice, but not in female F1 mice and Balb/c mice and either gender of C57BL/6 mice. Increased infiltration of natural killer group 2, member D (NKG2D+) cells in hair loss skin and upregulated interferon-gamma (IFN-γ) messenger RNA expression in cultured splenocytes were observed in male Balb/c mice. The CCT ODN-conditioned supernatants of cultured mouse splenocytes caused catagen-like changes to hair follicles (HFs). We hypothesized that the CCT ODN could induce patchy hair loss in the male mice with certain genetic traits by mobilizing NKG2D+ cells to HFs and by inducing the production of IFN-γ from immune cells. Taken together these data indicated that a gender and genetic preference of immune-regulatory oligonucleotides is causing unexpected clinical situations such as hair loss.
Introduction
An oligodeoxynucleotide with CCT repeats (CCT ODN) has been found in our previous study to selectively downregulate Toll-like receptor 7/9 (TLR7/9)-mediated immune responses in cultured immune cells in vitro and in mice, showing the characteristics of inhibitory ODNs (IN-ODNs). 1 –3 The IN-ODNs with defined sequences have been demonstrated to inhibit innate immune response triggered by CpG ODNs, bacterial DNA, or cytosolic double stranded (ds) DNA. 4 Structurally, all of the IN-ODNs published contain G, 5 but the CCT ODN, in contrast, contains no G and could be classified as NO-G-IN-ODNs. The CCT ODN, like G-containing IN-ODNs, displayed immunosuppressive activities. It inhibited TLR7/9-activation, rescued mice from TLR9-activation-induced lethal shock, 2 and relieved symptoms of lupus-like nephritis in mice. 6 Up to now, no report exists regarding the effect of the IN-ODNs including the CCT ODN on nonimmune organs or cells, resulting in unexpected clinical situations, such as hair loss.
Hair loss is a common clinical disease that affects both men and women of all ages and can be divided into chemotherapy-induced alopecia (CIA), androgenic alopecia (AGA), and alopecia areata (AA) based on their causes and manifestations. 7 CIA is one of the most common side effects of cancer chemotherapy, with an approximate incidence of 65% in all cancer patients receiving chemotherapy. 8 AGA, also known as male-pattern baldness, is the most common form of hair loss in men and rarely affects women. 9 AA, with an equal incidence in male and female, 10 is thought as an autoimmune disease since the immune responses are found in the hair follicles (HFs) of AA patients. 11,12
HFs renew through the periodic hair cycle, which consists of anagen, catagen, and telogen. 13 Each phase of the hair cycle has its own unique immune milieu. In the anagen, HF is a relatively immune-privileged (IP) site where the expression of major histocompatibility complex (MHC) class I is downregulated, Langerhans cells are dysfunctional, and local immunosuppressants, such as transforming growth factor-beta (TGF-β), are increased. 14 The IP is linked to prevention of HF cells from attack driven by immune cells, and its collapse is thought to initiate hair loss as seen in patients with AA. 10,12 The collapse in the involved AA HFs, revealed by histological examination, is characterized by the infiltration of activated T-helper cells (TH cells) and cytotoxic T (TC cells) cells. 15, 16 The attribution of the TH and TC cells in the AA was confirmed in animal models. Depletion of either CD8+ or CD4+ T cells led to complete hair growth in the Dundee experimental bald rat (DEBR) model of AA. 17 The severe combined immunodeficient (SCID) mice, in the absence of CD4+ and CD8+ T cells, accepted the AA affected skin of C3H/HeJ mouse model and support the regrowth of the grafted skin. 18
Recently, natural killer (NK) cells have become a focus of AA research, and activation of NKG2D+ (natural killer group 2, member D) cells 14 and induction of NKG2D ligands (NKG2DLs) 11 including UL-16 binding proteins 1–5 (ULBPs) and MHC class I chain-related A and B (MICA and MICB) have been suspected of being involved in the pathogenesis of human AA. A massive infiltration of IFN-γ-secreting NKG2D+ cells including CD56+ NK cells and CD8+ T cells were observed in AA lesions of the AA patients. 14,15 NKG2DLs including ULBP3 and MICA were found overexpressed in the HFs of scalp biopsy specimens from AA patients with active disease, accompanying with the increased NKG2D+ cells. The overexpressed NKG2DLs attracted the NKG2D+ cells to infiltrate into the HFs where the NKG2D+ cells were activated to attack the HFs, resulting in hair loss. 11 The intradermal injection of NKG2D+ cells caused rapid focal hair loss in healthy human scalp skin transplanted onto mice. 19 However, few reports have been referred to how the NKG2DLs expression and NKG2D+ cells infiltration are induced in HFs by immune modulators, including IN-ODNs.
In this study, we unexpectedly found that CCT ODN could cause hair loss in male F1 mice (female Balb/c × male C57BL/6) and male Balb/c mice, not in female F1 mice, female Balb/c mice, and either gender of C57BL/6 mice. The finding hinted that the CCT ODN could impact HFs. Further, since our previous work revealed that the CCT ODN, when subcutaneously injected, caused massive infiltration of NK cells in drainage lymph nodes, 20 we suspected that NK cells or possibly other NKG2D+ cells could be mobilized or activated by the CCT ODN and therefore were involved with the development of hair loss in the male mice and tried to find whether the CCT ODN could modulate the immune cells in HFs by upregulating NKG2DLs and activating NKG2D+ cells in the process of hair loss. The study may provide a new understanding of clinical situations that happened unexpectedly in individuals with certain genetic background when using oligonucleotides as immune regulatory drugs and also a concern on the risk of hair loss when using this type of drugs.
Materials and methods
CCT ODN and primers
CCT ODN is a full phosphorothioate-modified ODN with the sequence of 5′-CCTCCTCCTCCTCCTCCTCCTCCT-3′. It was synthesized in Takara Biotechnology Company (Dalian, China) and diluted in phosphate-buffered saline (PBS) buffer with no detectable endotoxin. Primers used to amplify messenger RNA (mRNA) of glyceraldehyde-3-phosphate dehydrogenase (GAPDH) and NKG2D ligands (murine UL-16-binding protein-like transcript 1 (Mult1), retinoic acid early transcript 1 (RAE-1), and H60) are as follows: 5′-TGCTGAGTATGTCGTGGA-3′ and 5′-TTCAGCTCTGGGATGACCT-3′ for GAPDH, 5′-CCAAGGATAGAAGAGACTGC-3′ and 5′-GCTCCAGATGTTCTTTCCAGTG-3′ for Mult1, 5′-AAGGCAGCAGTGACCAAG-3′ and 5′-GAAGATGAGTCCCACAGAG-3′ for RAE-1, 5′-GGAACCAGCAATCAGGTAATG-3′ and 5′-AGAATGACTCTCTCTCCAGG-3′ for H60. Primers used to amply mRNA of IFN-γ and TGF-β are as follows: 5′-ATCGGCTGACCTAGAGAAGACA-3′ and 5′-TCGCCTTGCTGTTGCTGAAGAA-3′ for IFN-γ, and 5′-AAGGAGACGGAATACAGGG-3′ and 5′-CGGGTTGTGTTGGTTGTAG-3′ for TGF-β. All primers were synthesized in Takara Biotechnology Company (Dalian, China) and diluted in sterile distilled water.
Mice
Specific-pathogen-free C57BL/6 and Balb/c mice were purchased from Beijing Weitonglihua Laboratory Animal Co., Ltd. F1 mice were the hybrid offspring of the male C57BL/6 mice and female Balb/c mice. Mice were maintained at 22 ± 2°C with a 12-h light/dark cycle.
Animal experiments
Eight-week-old male F1 mice were used to induce lupus-like nephritis by intraperitoneal (i.p.) injection with the mixture cell suspension of thymus, spleen, and lymph node cells from Balb/c mice at a ratio of 3:2:1 for 4 times at 3-day intervals. 6 During the induction, F1 mice were treated with CCT ODN at 5 or 25 μg per mouse twice a week for 9 weeks from day 0 at 2 h after the first lymphocyte injection. Six-week-old Balb/c mice or C57BL/6 mice, both male and female, were injected with CCT ODN at 5 or 25 μg per mouse twice a week for 4 weeks. PBS-treated mice were used as controls. Each group composed of six mice for in vivo experiments and three mice for ex vivo experiments. All mice were photographed every week for recording the hair growth. To collect the whisker pads and isolate the splenocytes and peripheral blood mononuclear cells (PBMCs), mice were killed at indicated time points as shown in the results. During the animal experiments, the mice were treated in accordance with the National Institute of Health Guide for the Care and Use of Laboratory Animals and with the approval of the Scientific Investigation Board of Science and Technology of Jilin Province.
Evaluation of hair loss
The degree of hair loss in each mouse was evaluated according to the shortening of the long vibrissae and the loss of the short hairs. Based on the growth pattern of the vibrissae in mice, 21 the scores were given as 0, 1, 2, 3, or 4 when the percentage of the vibrissae with the length of 2–3 cm in all the vibrissae examined was >20%, 10–20%, 5–10%, <5%, or 0%, respectively. The length of the vibrissae was determined by measuring the distances from the left to the right end of the vibrissae with a ruler photographed with the mice.
Histological analysis and immunohistochemistry
The whisker pad tissues of Balb/c mice were fixed in 10% neutral-buffered formalin solution, embedded in paraffin and then cut into 5 μm sections. The sections were stained with hematoxylin and eosin (H&E) and examined under microscope. The catagen-like changes of the vibrissa HFs were analyzed by calculating the percentage of the vibrissa HFs at the stages of catagen and telogen in all the HFs. 22 HFs in catagen are characterized by thickening basal membrane or apoptotic cells, and HFs in telogen by wrinkling in the center of the inner root sheath. For immunohistochemistry (IHC) the tissue sections were dewaxed, rehydrated, and then incubated in citrate buffer at 95°C for 15 min, enabling antigen retrieval. After treating with hydrogen peroxide for 10 min at room temperature, the sections were incubated with 1:100 diluted rabbit anti-NKG2D polyclonal antibody (BoAoSen, Beijing, China) at 37°C for 90 min and then washed, followed by incubation with horseradish peroxidase (HRP)–streptavidin complex (Maixin, Fuzhou, China) for 30 min at 37°C. The sections were then stained with 3, 3′-diaminobenzidine tetrahydrochloride hydrate (DAB; Maixin) and counterstained with hematoxylin. All steps were performed at room temperature unless otherwise specified. NKG2D+ cells were scored as 1, 2, 3, 4, 5, or 6 when the percentage of the NKG2D+ cells in the totally counted 500 cells in each sample was <3%, 3–6%, 6–9%, 9–12%, 12–15%, or 15–18%, respectively.
Western blotting
Whisker pads from Balb/c mice were lysed by radioimmunoprecipitation assay (RIPA) buffer (150 mM NaCl, 50 mM Tris, pH 7.4, 1% NP-40, 0.5% sodium deoxycholate, and 0.1% sodium dodecyl sulfate (SDS)). Total protein was measured using a bicinchoninic acid (BCA) protein assay kit (Wanleibio, Shenyang, China). Samples were separated by 12% SDS polyacrylamide gel electrophoresis (SDS-PAGE). Gels were blotted using polyvinylidene difluoride membranes (Millipore, Billerica, Massachusetts, USA). Membranes were blocked overnight at 4°C with Tris-buffered saline containing 5% nonfat dried milk and then incubated with anti-H60 monoclonal antibody (mAb; R&D Systems, Minneapolis, Minnesota, USA) or anti-GAPDH mAb (Proteintech, Chicago, Illinois, USA). Blots were then incubated with HRP-conjugated goat anti-mouse IgG (Jackson Immunoresearch laboratories, West Grove, Pennsylvania, USA) for 1 h at room temperature. Immunoreactive bands were visualized with SuperSignal West Pico chemiluminescent substrate (Thermo, Hudson, New Hampshire, USA).
Flow cytometry
The splenocytes or PBMCs were isolated from spleen or peripheral blood of Balb/c mice and then stained with PE-labeled anti-NKG2D mAb (BD Biosciences, San Jose, California, USA) for 30 min on ice in dark. After washing with fluorescence-activated cell sorting (FACS) buffer (PBS containing 2 mmol/L ethylenediaminetetraacetic acid (EDTA) and 20 mL/L fetal bovine serum), the cells were analyzed by flow cytometry instrument FACS Canto (BD Biosciences).
Culture of mouse vibrissa HFs
The vibrissa HFs were isolated from whisker pads of 1-week-old normal Balb/c mice under a dissecting microscope. The part of the hair shaft that extended over the epidermal surface was cut off and the HFs at early or mid-anagen growth phase were selected for culturing using a modified procedure based on a published method.
23
Briefly, the HFs were maintained in 96-well plates with 0.2 mL incubation medium at 37°C and 5% CO2 in a humidified incubator. The basal medium was serum-free Williams’ E medium (GIBCO, Grand Island, New York, USA) supplemented with 100 U/mL penicillin, 100 μg/mL streptomycin, 10 μg/mL insulin, 10 ng/mL hydrocortisone, and 2 mmol/L
Total RNA isolation and polymerase chain reaction
Total RNA was isolated from splenocytes or cultured HFs of Balb/c mice using Trizol reagent (Invitrogen, Carlsbad, California, USA). Reverse transcription (RT) was carried out using TIANScript M-MLV (Tiangen Biotech, Beijing, China), according to the manufacture’s instruction. The mRNA expression of mouse NKG2D ligands (Mult1, RAE-1, and H60) in cultured HFs and IFN-γ and TGF-β in splenocytes was detected by RT-polymerase chain reaction (PCR). The cycle profile of PCR was as follows: 45 s at 94°C, 1 min at 55°C, and 1 min at 72°C for total 30 cycles, and then 10 min at 72°C for further extension. PCR products were analyzed on a 1.5% agarose gel. The mRNA of GAPDH was also amplified by RT-PCR as control for balancing the quantity of the amplified mRNA among different reactions.
Statistical analysis
Data were shown as mean ± standard error of the mean (SEM). Unpaired nonparametric test (Mann–Whitney) or unpaired t test was applied to analyze the data. p < 0.05 was considered statistically significant. Statistical analysis was performed using SPSS 13.0 software.
Results
CCT ODN-induced hair loss in male mice
An ODN with CCT repeats (CCT ODN) has been previously demonstrated in our study to selectively downregulate TLR7/9-mediated innate immune responses. 1 –3 To test whether the CCT ODN could treat autoimmune disease, we established a mouse model of lupus-like nephritis induced by injection of allogenic lymphocytes in F1 mice (female Balb/c mice × male C57BL/6 mice). 6 During the treatment with CCT ODN by i.p. injection at 25 μg per mouse twice a week for 9 weeks, the male F1 mice unexpectedly showed patchy hair loss around the mouth (Figure 1(a)).

CCT ODN induced hair loss in male F1 mice with lupus-like nephritis. The male F1 mice were injected i.p. with lymphocytes for 4 times in a 3-day interval and also injected with CCT ODN at 5 or 25 μg per mouse by i.p. twice a week for 9 weeks. (a) Photos of mice with hair loss on week 9 after the initial CCT ODN treatment in dose–effect observation. Hair loss is shown for six mice treated with CCT ODN at 25 μg. (b) Dose effects of CCT ODN. (c) Photos of mice with hair loss on week 9 after the initial CCT ODN treatment in dynamic effect observation. The boxed places were enlarged to show mouth without vibrissae. (d) Dynamic effect of CCT ODN. Each symbol represents one mouse. i.p.: intraperitoneal; ODN: oligodeoxynucleotide.
To determine whether the CCT ODN-induced hair loss is dose dependent, the CCT ODN at 5 and 25 μg per mouse was used to treat the male F1 mice with lupus-like nephritis based on the same procedure as discussed previously. The results showed that the CCT ODN at both 5 and 25 μg induced obvious hair loss in the male F1 mice (p < 0.05) (Figure 1(b)). In comparison, the CCT ODN at 25 μg induced more severe hair loss than at 5 μg (p < 0.05). Meanwhile, we also observed that the vibrissae of the CCT ODN-treated mice first became shortened and then disappeared prior to the short hair loss (Figure 1(c)). Kinetically, the hair loss induced by CCT ODN at 25 μg began on week 4 after the initial CCT ODN treatment and was gradually aggravated with the ongoing treatment (Figure 1(d)).
To understand whether genetic background could be attributed to the CCT ODN-induced hair loss, both gender of Balb/c mice and C57BL/6 mice, the ancestors of the F1 mice, were administered CCT ODN i.p. twice a week for 4 weeks and observed for their hair loss. The results showed that the hair loss around the mouth only happened in the male Balb/c mice, neither in the female Balb/c mice nor in either gender of C57BL/6 mice (Figure 2(a)), and that the degree of the hair loss tended to be in a dose-dependent manner, but no statistical significance (Figure 2(b)). Notably, no hair loss was observed in the female Balb/c mice (Figure S1). The results hinted that the CCT ODN-induced hair loss might depend on certain genetic traits in the Balb/c mice from which the traits could be transmitted to the F1 mice.

CCT ODN induced hair loss in male Balb/c mice, not in female Balb/c mice or both gender of C57BL/6 mice. Both gender of the Balb/c mice and C57BL/6 mice were injected i.p. with CCT ODN at 5 or 25 μg per mouse or PBS for 4 weeks in twice a week. (a) Photos of hair loss on week 4. (b) Hair loss scores. There are six mice in each group. i.p.: intraperitoneal; PBS: phosphate-buffered saline; ODN: oligodeoxynucleotide.
CCT ODN delayed the growth of HFs
To observe whether the CCT ODN could cause pathological changes on the vibrissa HFs, whisker pad tissues from the Balb/c mice treated with CCT ODN were sectioned and then stained with H&E. Under the microscope, the HFs in either anagen or catagen, rarely in telogen, were observed (Figure 3(a), left). In the male mice the percentage of regressive HFs of the total HFs was calculated, showing that no regressive HFs appeared in mice treated with PBS and those treated with 5 μg CCT ODN, while 11 and 15% regressive HFs happened in two of three mice treated with 25 μg CCT ODN (Figure 3(a), right). However, the CCT ODN treatment didn’t induce the regressive HFs in the female mice (Figure S2(a)).
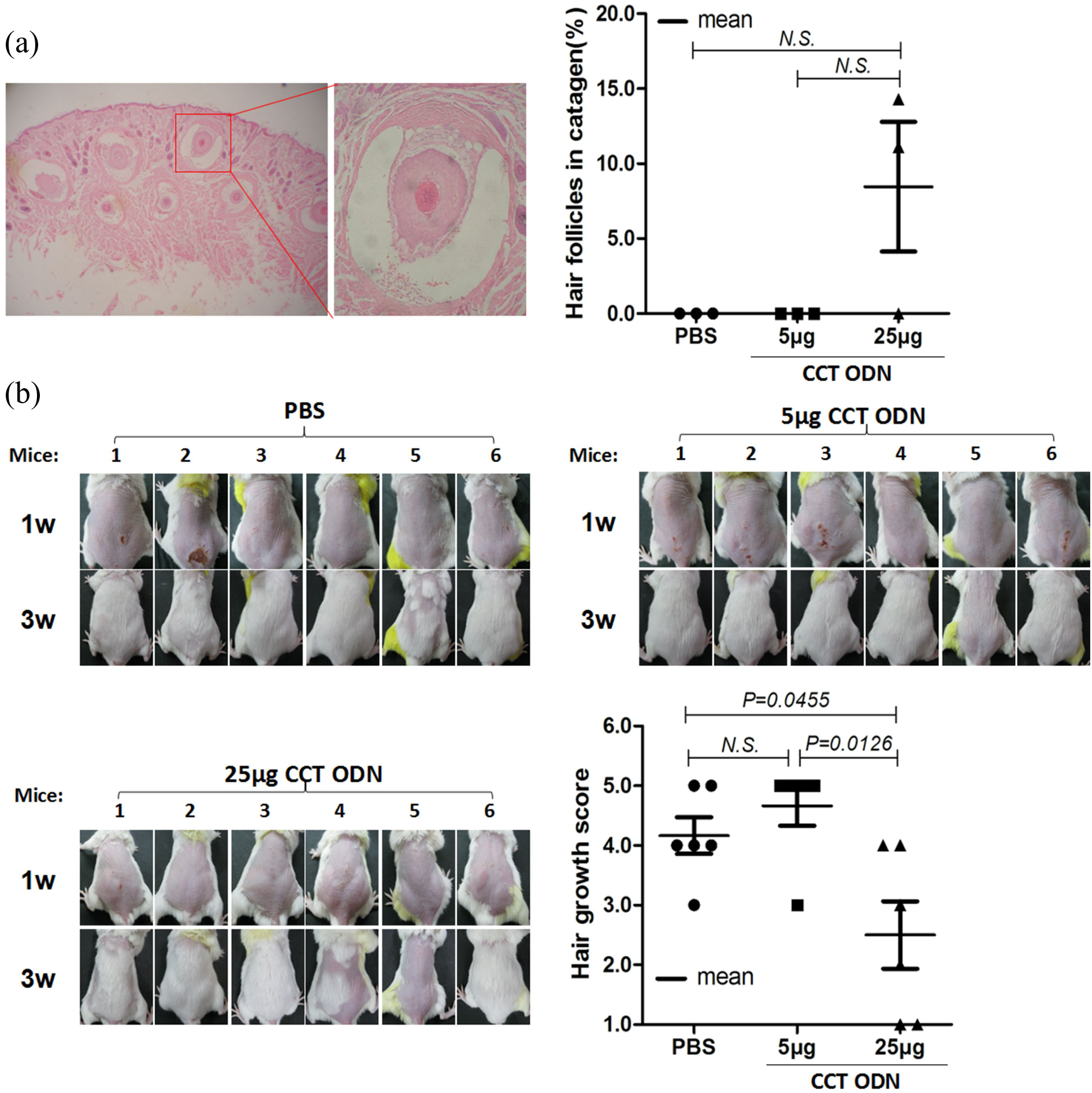
Effect of CCT ODN on morphological changes of vibrissa HFs and on hair growth of synchronized HFs. (a) Morphological changes of HFs. The male Balb/c mice were treated with CCT ODN at 5 or 25 μg per mouse or PBS twice a week for 4 weeks and then sacrificed for isolating whisker pad tissues. The skin tissues were sectioned, stained with H&E, and then observed under microscopy (40×) (left panel). The boxed places were enlarged to show the vibrissa in catagen (400×). The HFs at the stages of catagen and telogen were counted and calculated (right panel). (b) Hair growth from synchronized HFs. The back hairs of the Balb/c mice were removed using depilatory paste to synchronize the growth cycle of HFs. The regrowth of back hairs was evaluated using hair growth scores. HFs: hair follicles; PBS: phosphate-buffered saline; ODN: oligodeoxynucleotide.
Considering that the HFs from different mice are in different stages of life cycle, which will have influences on the evaluations of the HF pathological changes, we removed the back hairs of the Balb/c mice using depilatory paste before the CCT ODN treatment. The hair removal with depilatory paste can force HFs to renew and therefore synchronize the life cycle of the HFs. Using this method, we found that the CCT ODN at 25 μg resulted in delayed hair regrowth in the male mice. Notably, 3 weeks after the hair removal, two of the six mice treated with the CCT ODN at 25 μg had no noticeable hair growth on their back (Figure 3(b)). Compared with PBS and the CCT ODN at 5 μg, the CCT ODN at 25 μg significantly delayed hair regrowth (p < 0.05) (Figure 3(b)). However, the CCT ODN treatment didn’t delay the hair regrowth in the female mice with hair removal (Figure S2(b)).
CCT ODN induced infiltration of NKG2D+ cells in hair loss skin
To find whether the NKG2D+ cells are involved in the CCT ODN-induced hair loss, we first detected the NKG2D+ cells in the whisker pad tissue around the mouth of the Balb/c mice treated with CCT ODN or PBS for 8 times by IHC (Figure 4(a)). The detection showed significantly increased NKG2D+ cells in the skin from CCT ODN-treated male mice (Figure 4(b)), not in the female mice (Figure S3(a) and (b)), in comparison with that in mice treated with PBS (p < 0.05), suggesting that the CCT ODN could mobilize NKG2D+ cells into the HF tissues in the male mice. Then, we detected the NKG2D+ cells in mouse splenocytes and PBMCs by flow cytometry. The Balb/c mice were injected i.p. with CCT ODN at 25 μg per mouse or PBS once for 4 h and then sacrificed to isolate splenocytes and PBMCs. The cells were surface stained with PE-labeled anti-NKG2D mAb, followed by flow cytometry analysis. As shown in Figure 4(c), in the male mice with CCT ODN treatment, the percentage of NKG2D+ cells tended to decrease (no statistic difference) in the splenocytes or PBMCs. But the CCT ODN treatment didn’t induce significant increase in the NKG2D+ cells in the splenocytes or PBMCs of the female mice (Figure S3(c)). These results implied that the NKG2D+ cells might be mobilized to HFs, resulting in hair loss in the male mice.

Effect of CCT ODN on NKG2D+ cells in skin, splenocytes, and PBMCs. The male Balb/c mice were treated with CCT ODN at 5 or 25 μg per mouse or PBS twice a week for 4 weeks for isolating whisker pad tissues or once for 4 h for harvesting splenocytes and PBMCs. The skin tissues were sectioned and then stained with anti-NKG2D mAb followed by observation under microscopy (400×; (a)); splenocytes (b) or PBMCs (c) isolated from the spleen or peripheral blood were stained with PE-labeled anti-NKG2D mAb and then analyzed by flow cytometry. N.S.: no statistic difference; NKG2D+: natural killer group 2, member D; PBMCs: peripheral blood mononuclear cells; mAb: monoclonal antibody; PBS: phosphate-buffered saline; ODN: oligodeoxynucleotide.
Effect of CCT ODN on NKG2DLs expression
Since the NKG2D+ cells can target and kill the NKG2D ligand (NKG2DL) expressing cells, 24,25 we tried to find whether the CCT ODN could increase the NKG2DL expression on HFs of the Balb/c mice. Firstly, we detected the expression of H60, a murine NKG2DL, in the whisker pad tissue around the mouth of the mice treated with CCT ODN or PBS for 8 times, using Western blotting. H60 protein expression couldn’t be detected in the whisker pad tissues of either gender of the mice (data not shown). In contract, the H60 protein expression was readily detected in the mouse lymphoma (YAC-1) cells, used as positive control cells which constitutively express H60. Next, we cultured the vibrissa HFs isolated from both gender of the Balb/c mice with 8 μg/mL CCT ODN for 24 h and then detected the mRNA expression of three murine NKG2DLs including Mult1, retinoic acid early transcript 1 (RAE-1), and histocompatibility antigen 60 (H60) by RT-PCR. Of the three NKG2DLs, no mRNA expression was detected in the CCT ODN-treated vibrissa HFs from either gender of the mice (data not shown), whereas the mRNA expression of all the three NKG2DLs was readily detected in the YAC-1 cells. These results suggested that the CCT ODN couldn’t induce the HFs to express the NKG2DLs in mice.
CCT ODN induced IFN-γ that may modulate the growth of HFs
Since reports showing that IFN-γ produced by NKG2D+ cells and immunosuppressant TGF-β 26,27 were involved in the hair loss, we detected the mRNA expression of IFN-γ and TGF-β in the splenocytes of the Balb/c mice treated with CCT ODN or PBS for 24 h. The results showed that CCT ODN could upregulate the mRNA expression of IFN-γ (p < 0.05), not TGF-β, in the splenocytes of the male mice (Figure 5(a)) and failed to upregulate the mRNA expression of both IFN-γ and TGF-β in the female mice (Figure S4). These results implied that CCT ODN might cause hair loss in the male mice by inducing IFN-γ. To confirm this, the splenocytes of the male Balb/c mice were stimulated with CCT ODN for 24 h, and the supernatants were collected to culture the whole vibrissa HFs that were isolated from 1-week-old male Balb/c mice for 14 days. On day 7 and 14, the width of the cultured HFs was measured. The results showed that the HFs cultured with the CCT ODN-conditioned supernatants for 14 days were obviously smaller than those cultured with the supernatants without CCT ODN treatment (p < 0.05) (Figure 5(b)). The results revealed that CCT ODN-induced hair loss in the male mice could be related to the cytokines or other soluble factors.

Effect of CCT ODN on the mRNA expression of IFN-γ and TGF-β and on the growth of cultured vibrissa HFs. The splenocytes of the male Balb/c mice were cultured with CCT ODN at 8 μg/mL for 24 h to detect the mRNA expression of IFN-γ and TGF-β by RT-PCR (a) or harvest the supernatants. The supernatants were added to the cultured vibrissa HFs. The width of the HFs was measured on day 7 and 14 (b). HFs: hair follicles; IFN-γ: interferon gamma; mRNA: messenger RNA; TGF-β: transforming growth factor-beta; RT-PCR: reverse transcription polymerase chain reaction.
Discussion
In this study, we found that CCT ODN, a TLR7/9 selective immune inhibitor, could induce hair loss in male F1 mice (female Balb/c mice × male C57BL/6 mice) and male Balb/c mice, but not in female F1 and Balb/c mice and either gender of C57BL/6 mice. The selectivity in sex and mouse strain hints that other unidentified genetic traits are related to the CCT ODN-induced hair loss. The traits could be harbored on the X chromosome not on the Y chromosome in the Balb/c mice from which the traits could be transmitted to the F1 mice. On the X chromosome, the androgen-receptor (AR) gene was found to be associated with AGA. 9,28 In men with AGA, the CAG repeat numbers in the AR gene was found to be significantly decreased. 29 Thus, these findings hinted a possible association of the polymorphism in the AR gene of the male mice with CCT ODN-induced hair loss. However, when checked the exon-1 sequence of the AR gene, we failed to find the difference between the male mice with CCT ODN-induced hair loss and the mice without hair loss (Figure S5). Considering that AGA with progressive hair thinning could be resulted from the effects of the androgen on androgen-sensitive HFs, 30 we also detected blood testosterone levels in the male mice with CCT ODN-induced hair loss and found no obvious increase (Figure S5). Furthermore, we compared the sequences of U2HR, an inhibitory upstream open reading frame (ORF) in the 5′UTR of the HR homolog, in the human hairless (HR) gene of Balb/c mice and C57BL/6 mice. Mutations in the U2HR have recently been identified to be involved in the pathogenesis of Marie Unna hereditary hypotrichosis (MUHH), an autosomal-dominant form of genetic hair loss. 31,32 The comparison showed variation in single nucleotide polymorphism (SNP) between the Balb/c mice and C57BL/6 mice, but no variation between both gender of the Balb/c mice, and between both gender of the C57BL/6 mice (Figure S6). Taken together, we might deduce that other factors rather than the androgen and genetic elements could be involved in the development of the male preferential hair loss induced by CCT ODN.
In addition to AGA, AA is the second most common form of hair loss disease in human and presents as patchy hair loss. 15 Likely, CCT ODN-induced hair loss in mice appeared to be AA like, with the patchy hair loss mainly around the mouth. In recent years, AA has been documented as an autoimmune disease, and NKG2D+ cells and NKG2DLs are suspected of being involved in its pathogenesis. 11,14 Normally, very few NKG2D+ cells are observed in and around human anagen HFs which lack the expression of NKG2DLs, including MICA, MICB, and ULBPs. 12 However, in AA lesions, a massive infiltration of IFN-γ-secreting NKG2D+ cells including CD56+ NK cells and CD8+ T cells was found around the HFs. 12,14,15 In this study, the increased infiltration of NKG2D+ cells in the whisker pad tissues of the mice with CCT ODN-induced hair loss was also observed. The increased NKG2D+ cells in the hair loss skin might be mobilized from other lymphoid organs since the NKG2D+ cells in spleens of the mice treated with CCT ODN tended to be decreased. The NKG2D+ cells were reported to be attracted and activated to infiltrate into the HFs by NKG2DLs, such as ULBP3. 14 In scalp biopsy specimens from AA patients with active disease, accompanying with the NKG2D+ cells, NKG2D ligands including ULBP3 and MICA were found significantly upregulated in the HFs. The ULBP3 attracts the NKG2D+ cells to infiltrate into the HFs and the activated NKG2D+ cells in turn attack HFs, leading to hair loss. 11 However, in this study, the expression of NKG2DL H60 wasn’t detected in the whisker pad tissues of the mice with CCT ODN-induced hair loss, and the expression of the three NKG2DLs including Mult1, RAE-1, and H60 wasn’t detected in the cultured HFs stimulated with CCT ODN. These results implied that the CCT ODN-induced hair loss may not be correlated to the upregulation of NKG2DLs in the HF cells.
Although HFs are considered as an IP site under normal circumstances, few NKG2D+ cells are still found around healthy human anagen HFs. The IP status could be broken by IFN-γ produced by NKG2D+ cells. 14 In this study, we observed that CCT ODN induced increasingly infiltrated NKG2D+ cells in the whisker pad tissues of the male mice with hair loss, stimulated the splenocytes to express increased IFN-γ mRNA, and the CCT ODN-conditioned supernatants of cultured splenocytes caused the catagen-like changes in the cultured HFs isolated from the male Balb/c mice. Together with the observations, we might postulate that CCT ODN might mobilize NKG2D+ cells to HFs and induce IFN-γ to cause catagen-like changes in the HFs in the male mice, therefore leading to hair loss. The postulation could be partially supported by the accumulative evidence on the involvement of IFN-γ in hair loss: (1) IFN-γ transgenic mice exhibited hair loss, 33 while IFN-γ-deficient mice were resistant to the development of AA, 34 (2) IFN-γ potently induced catagen-like changes in cultured human anagen HFs, 35 (3) serum concentrations of IFN-γ in patients with AA were correlated with the duration of the disease, 36 and (4) IFN-γ antibody was tested in clinical trials for the treatment of AA. 37
Overall, in our previous study, CCT ODN displayed immunosuppressive activities by selectively downregulating TLR7/9-mediated immune responses, 1 –3 but its target and signal pathway were still unclear. In this study, CCT ODN-induced hair loss in male F1 mice and male Balb/c mice is likely through NKG2D+ cells and IFN-γ, suggesting that it might play immunostimulatory activities in an individual with certain genetic background. We don’t know the exact mechanisms of the CCT ODN-induced hair loss and need to further delineate the molecular mechanisms of the hair loss, such as to study the influence of CCT ODN on the in vivo expression of the genes of AR, NKG2D ligands, inhibitory receptors of NK cells, and other HF-IP-associated factors including CXC chemokine ligand-10 (CXCL10), adrenocorticotropic hormone, and α-melanocyte-stimulating hormone in the mice with hair loss.
Footnotes
Acknowledgments
We gratefully acknowledge Xiaoping Hu, Hongfei Wei, Xiuli Wu, and Youyou Yan for technical supports.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by National Nature Scientific Foundation of China [81172843 and 81471888].
