Abstract
The hair follicle (HF) has a wide range of functions including thermoregulation, physical and immunological protection against external insults, sensory perception, social interactions, and camouflage. One of the most characteristic features of HFs is that they self-renew during hair cycle (HC) throughout the entire life of an individual to continuously produce new hair. HC disturbances are common in humans and comparable to some alopecic disorders in dogs. A normal HC is maintained by follicular stem cells (SCs), which are predominately found in an area known as the bulge. Due to similar morphological characteristics of the human and canine bulge area, the particularity of compound HFs in humans and dogs as well as similarities in follicular biomarker expression, the dog might be a promising model to study human HC and SC disorders. In this review, we give an overview of normal follicular anatomy, the HC, and follicular SCs and discuss the possible pathogenetic mechanisms of noninflammatory alopecia.
Introduction
The base of each hair shaft (HS) resides in a multicellular miniorgan called hair follicle (HF) that is invaginated under the surface of the skin. The HF has a wide range of functions including thermoregulation, physical and immunological protection against external insults, sensory perception, social interactions, and camouflage. The HF also serves as a repository for multipotent stem cells (SCs) that are capable of regenerating all skin lineages. One of the most characteristic features of HFs is that they self-renew during hair cycle (HC) throughout the entire life of an individual to continuously produce new HSs. In the life span of a human, HFs normally regenerate 8 to 10 times. The regulation of HF development in the embryo and the HC during postnatal life are highly conserved processes that involve a series of interactive signals between epithelial and mesenchymal cells. Despite the importance of an intact skin barrier and functional HFs, our knowledge about the factors that regulate the innate HF formation and the postnatal HC as well as the hair diversity is still not completely understood in rodents, and only rudimentarily known in humans, and knowledge is poor in our companion animals.
Hair loss (hypotrichosis or alopecia) is a common problem, and it is known that hair loss affects approximately 60 million men, women, and children in the United States, and the costs for medical consults and treatment sum up to US$1.3 billion per year (Hodgson et al. 2014).
Hair growth disorders are attributed, at large, to a decreased and/or abnormal formation, a decreased regeneration, or an increased destruction of HFs, and the consequence is that either the quality or the total density of HSs is altered (Stenn and Paus 1999; Al-Nuaimi et al. 2010). Whereas in mice knowledge about HF biology has rapidly progressed and numerous genes and signaling events have been identified as cause of various alopecic disorders, the understanding of human and to a greater extent canine HF biology is still very limited and data from the mouse cannot be transferred easily to humans and dogs. One example of this is that in pregnant or lactating mice, the telogen phase of the follicle is prolonged and a new anagen phase is only initiated after lactation is finished. This is in sharp contrast to the findings in the human scalp, where pregnancy results in an increased proportion of anagen HFs. The simultaneous entry of these anagen HFs into telogen during the postpartum period then results in increased hair loss and sometimes even in alopecia (Hodgson et al. 2014; Gizlenti and Ekmekci 2014).
In order to manage alopecia, it is essential to increase our knowledge about the microenvironmental and macroenvironmental control of the HF morphogenesis and the HC in humans and dogs. Until now, unfortunately, we could only speculate about some of the underlying causes of alopecic disorders based on the knowledge gained from studies in mice and few in vitro studies performed with human plucked HFs or dermal papilla (DP) cells.
HF Morphogenesis
The mammalian skin and its appendages are derived from ectoderm and mesoderm during embryogenesis. The embryo surface emerges as a single layer of epithelial cells which gives rise to the epidermis. The dermis is formed from the underlying mesoderm composed of mesenchymal cells. Subsequently, the epidermal–dermal interaction results in HF morphogenesis (Fuchs 2007; Figure 1). This occurs in early embryonal life. The stages of HF morphogenesis are broadly classified into induction, organogenesis, and cytodifferentiation (Rishikaysh et al. 2014). Morphologically, HF induction is characterized by a local thickening of the epidermis known as hair placode. The SC that gives rise to the placode formation is leucine-rich repeat containing G protein-coupled receptor (LGR)6 positive (Biggs and Mikkola 2014; Snippert et al. 2010). Upon successful initiation of the epithelial placode, organogenesis starts with a condensation of mesenchymal cells underneath the placode, leading to the formation of the hair germ, also named the follicular bud. The hair germ proliferates and invaginates into the dermis to form the hair peg and the bulbous peg (Fuchs 2007). A complex interplay of signals sent between the mesenchymal condensate and the overlying epithelial placode is orchestrating the proliferation of the mesenchymal condensate and the down-growth of the epithelial cells into the dermis. Numerous studies have shown that once a morphologically distinct dermal condensate has formed, this condensate can induce HF formation even in the epithelium that does not produce HF under normal circumstances (Yang and Cotsarelis 2010).
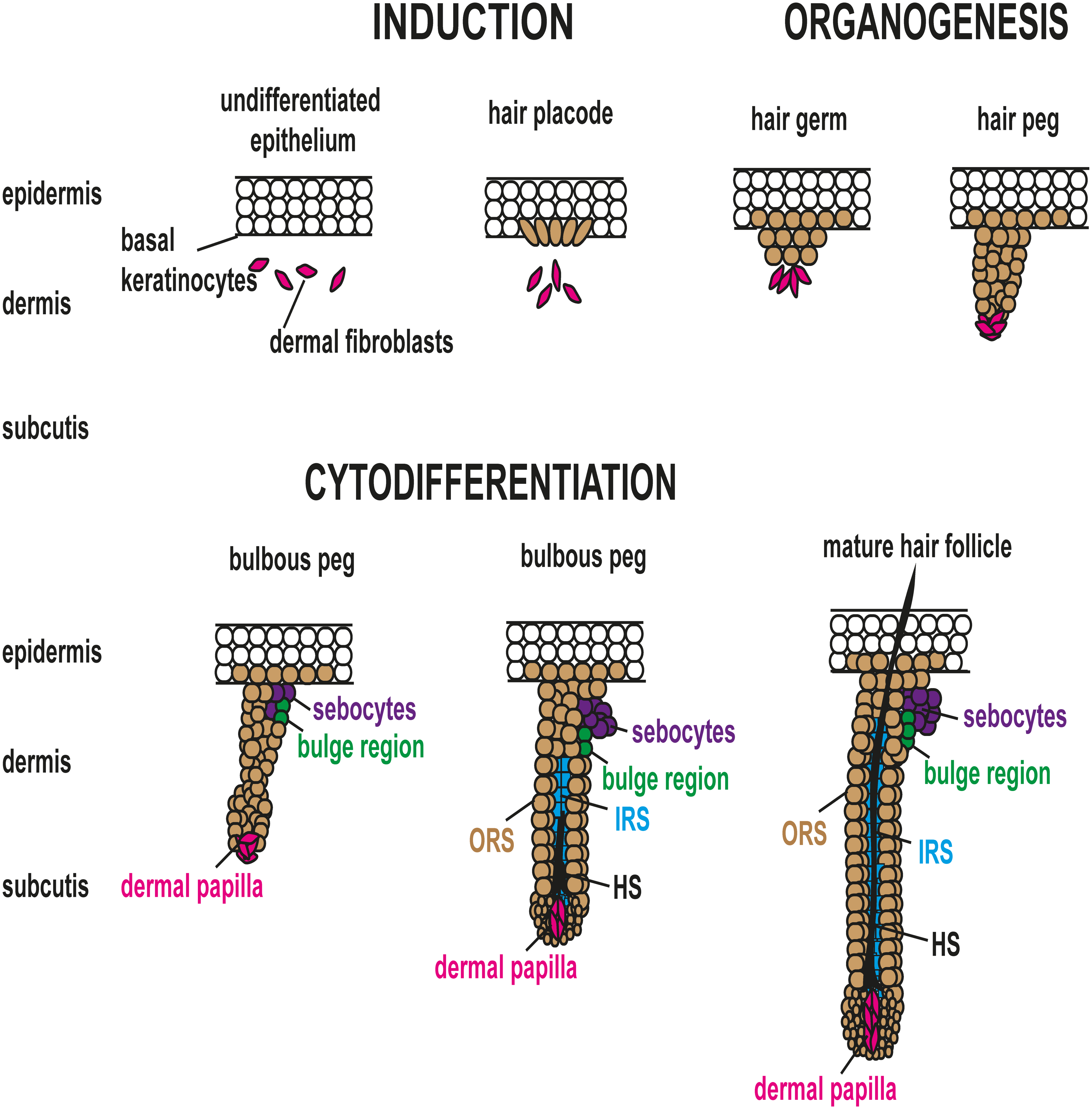
Hair follicle morphogenesis. HF morphogenesis is induced by a local thickening of the epidermis named the placode. Thereafter, mesenchymal cells (pink color) condense underneath the placode, leading to the formation of the hair germ, also called the follicular hair bud. The hair germ proliferates and invaginates into the dermis to form the hair peg and the bulbous peg. The superficial portions of the hair follicles subsequently develop 2 distinct, asymmetrical accumulations of cells that form the sebaceous gland (purple color) and the bulge representing the location of the presumptive follicular stem cells (green color). During cytodifferentiation, the edges of the down-growing follicular epithelium gradually engulf the dermal condensate, which from thereon is called the dermal papilla (DP). Signals from the DP to the adjacent epithelial cells induce their differentiation into the IRS (blue color) in which the future HS will develop. When the IRS is formed, the epithelial cells surrounding the DP start to differentiate into the different components of the HS (black color). IRS, inner root sheath; ORS, outer root sheath; HF, hair follicle; HS, hair shaft.
In cytodifferentiation, the edges of the down-growing follicular epithelium gradually surround the dermal condensate which from thereon is called the DP. Signals from the DP to the adjacent epithelial cells induce their differentiation into the inner root sheath (IRS) in which the future HS will develop. When the IRS is formed, the epithelial cells surrounding the DP (also known as matrix cells) start to differentiate into distinct lineages to form the different components of the HS that will grow inside the IRS and eventually protrude through the epidermis. Several families of secreted signaling molecules (e.g., wingless type (Wnt)/βcatenin, sonic hedgehog (Shh), Notch, bone morphogenetic protein [BMP], ectodysplasin-A/ectodysplasin receptor (EDA/EDAR)) have been implicated in the communication between epidermis and dermis during HF development (reviewed in Biggs and Mikkola 2014). Among them, the dermal Wnt/βcatenin signaling and the epithelial EDA/EDAR signaling appear to be the earliest signaling events required for placode formation (Fu and Hsu 2013; reviewed in Biggs and Mikkola 2014). Shh is involved in morphogenesis and late stage differentiation, Notch signaling determines SC fate, while BMP is involved in cellular differentiation. The cellular mechanisms and signaling pathways instructing the ectoderm to shape the entire HF have been recently summarized (reviewed in Duverger and Morasso 2013). Nevertheless, the puzzle is not yet complete and especially the mesenchymal events are not well understood. It is known, however, that dysregulation of these pathways leads to HF or HS abnormalities and in some cases to tumor development.
Follicular Anatomy
Most omnivores and herbivores (e.g., horses, cattle, pigs, rats, and mice) have simple follicles, in which each infundibulum contains 1 HS. Carnivores (e.g., dogs and cats) and also rabbits have compound follicles, in which each infundibulum contains multiple HSs. Some HSs in the compound follicle have a large diameter (primary or guard hairs), whereas the majority of the HSs have a small diameter (secondary or undercoat hairs; Meyer 2009). Humans and sheep have, depending on the body location, simple or compound follicles (Meyer 2009). In the human scalp, most follicular units are composed of 2 to 4 hairs, whereas on the body, simple HFs are present (Pinkus 1951).
Extending vertically from the epidermis to the base of the HF, the anagen HF can be divided into 3 major anatomic regions (Figure 2):
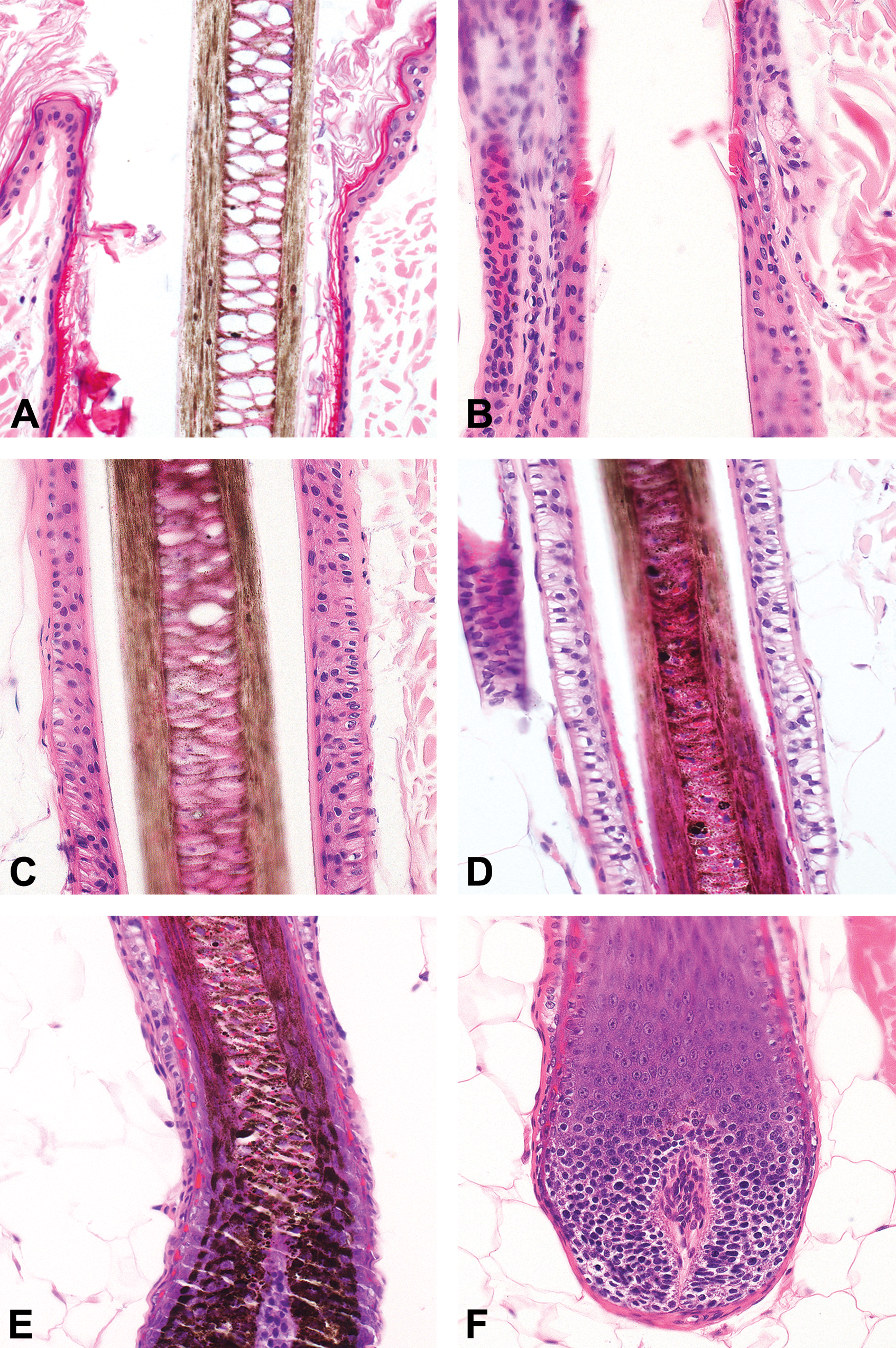
Schematic drawing of an anagen hair follicle illustrating major anatomic regions, namely, the infundibulum, the isthmus, and the inferior portion with its suprabular and bulbar region. In addition, sebaceous glands, and the arrector pili muscle are depicted in this image. The infundibulum, which extends from the opening of the HF to the point at which trichilemmal cornification begins (approximately the opening of the sebaceous gland duct; Figure 3A and B). The isthmus, which extends from the distal end of the infundibulum to the interface between the completely cornified IRS and the first noncornified cell of Huxley’s layer (Adamson’s fringe; approximately the insertion of the arrector pili muscle; Figure 3B–D). The inferior portion is composed of a suprabulbar and a bulbar region and extends from the last cell in which red trichohyalin granules can be seen to the base of the HF. The suprabulbar region is characterized by the noncornified IRS surrounded by the outer root sheath (ORS). The bulbar region is composed of matrix cells with intermingled melanocytes and the DP (Figure 3D–F). Histological image depicting the infundibulum with the ostium to the outside (A), the transition between infundibulum and isthmus (B), the isthmus (C), the transition between isthmus and inferior portion (D), the inferior portion (E), and the bulbar region of the inferior portion (F). Note the cuticule (thin arrow), the cortex (star), the medulla (asterisk), and the infundibular keratinocytes (thick arrow) in A; the small area of trichilemmal keratinization (thick arrow) close to the sebaceous gland (asterisk) at the transition between infundibulum and isthmus and the fully cornified inner root sheath of the isthmus (thin arrow) in B; the layers of the follicular wall of the isthmus characterized by the fully cornified inner root sheath (thin arrow), the companion layer (thick arrow), the outer root sheath (asterisk), and the fibrous sheath (star) in C. Note that, below the transition between the isthmus and the inferior portion, the Huxley layer is not cornified (thick arrow), whereas the Henley layer is already cornified (black and white arrow). The thin arrow marks the fully cornified inner sheath of the isthmus and the asterisk marks the outer root sheath (D). The suprabulbar region of the inferior portion is characterized at the distal end by the already cornified Henley layer and the noncornified Huxley layer (white and black arrow). At the proximal end, Henley and Huxley layers are not yet cornified and are characterized by trichohyalin granules (black arrow). The asterisk marks the outer root sheath (E). Note the fibrous sheath (thin arrow), the noncornified inner root sheath (thick arrow), the germinative cells (star), and the dermal papilla (asterisk) of the bulbar region (F). Hematoxylin and eosin.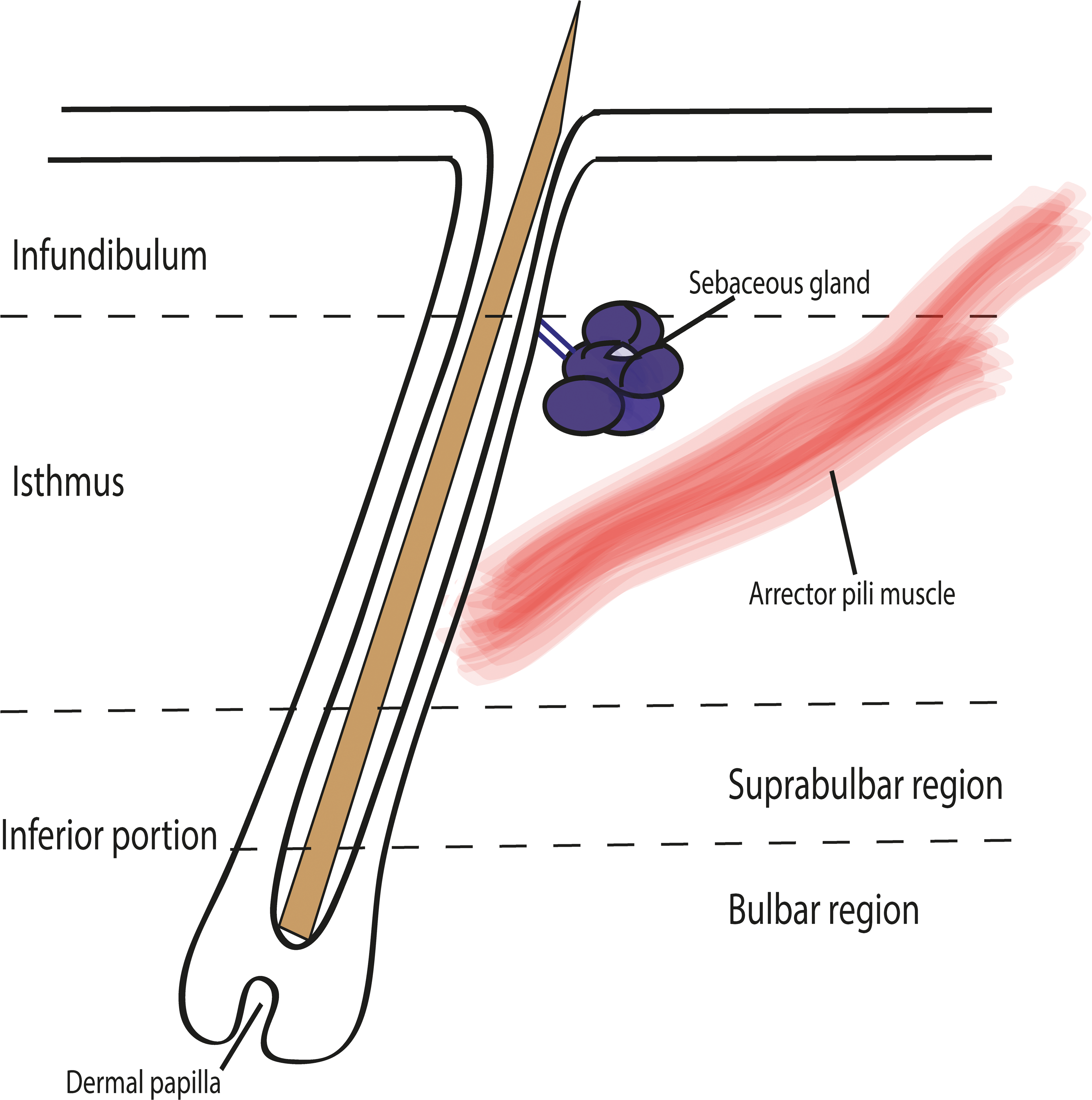
The infundibulum and the isthmus constitute the permanent portion of the HF, whereas the inferior segment is transitory and undergoes regression during catagen and is absent during telogen. From the outside to the inside, the anagen HF can be conceptualized as 8 concentrical layers (Alcolea and Jones 2014; Figure 4).
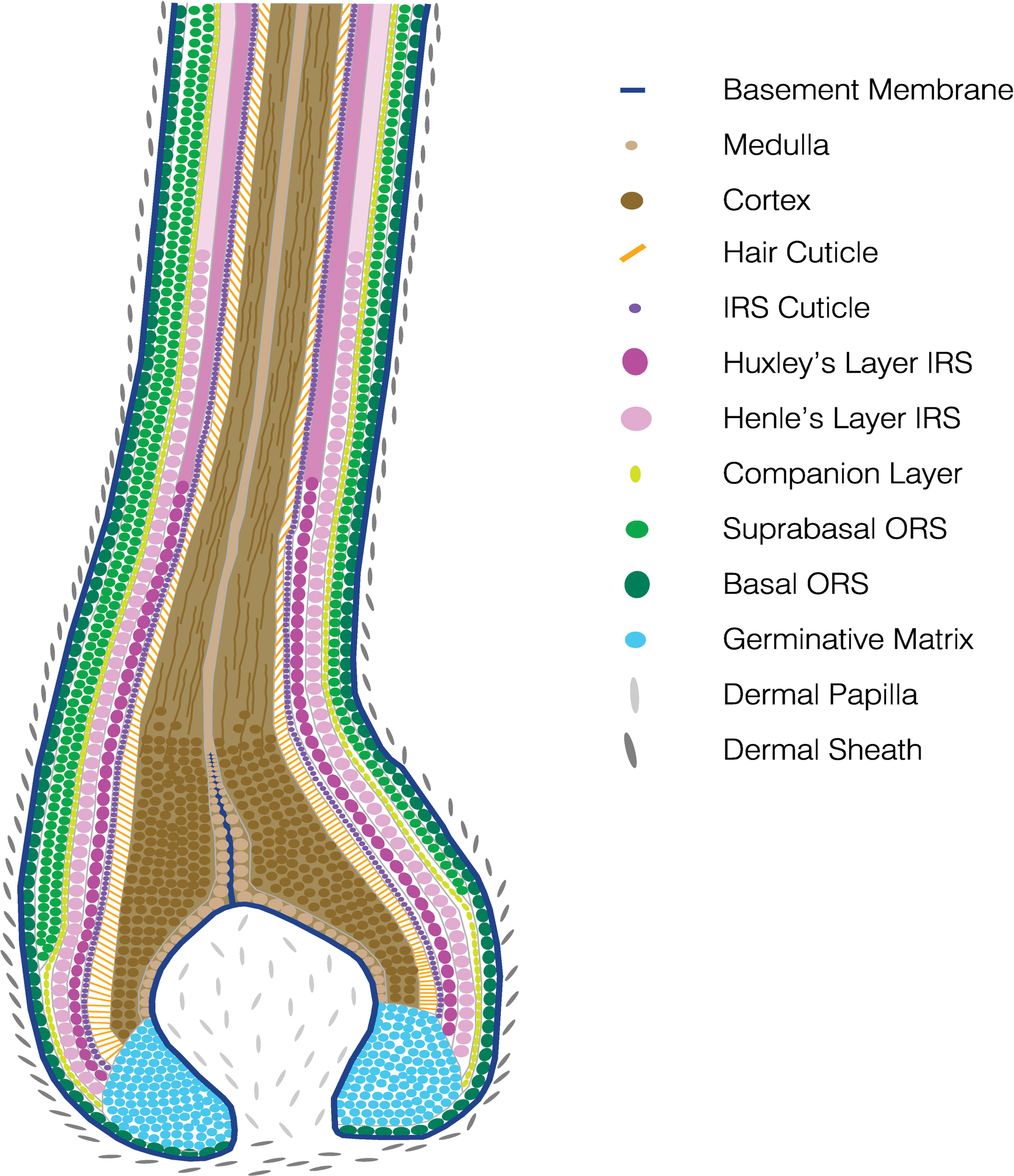
Schematic drawing illustrating the concentric layers of the anagen hair follicle. IRS, inner root sheath; ORS, outer root sheath.
The outermost layer is the ORS. The ORS is contiguous with the basal layer of the interfollicular epidermis. In the infundibulum, it undergoes keratinization in the same fashion as in the interfollicular epidermis (infundibular cornification). In the isthmus of a telogen follicle, the ORS undergoes trichilemmal cornification (a telogen follicle has no IRS; Figure 5).
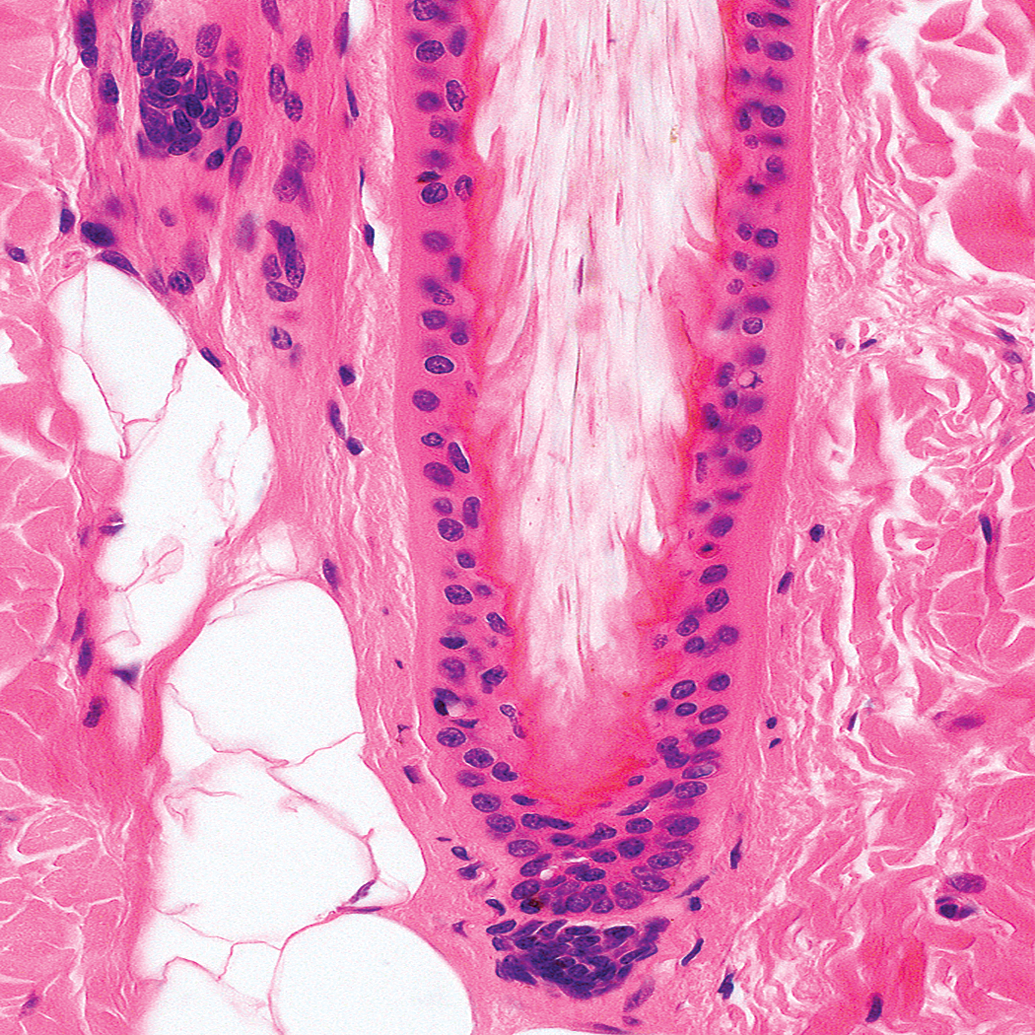
Histological picture of a telogen hair follicle. Note that the club hair (asterisk) is anchored by trichilemmal keratin to the outer root sheath (thin arrow) which is surrounded by the fibrous sheath (star). The dermal papilla is situated at the lower end of the isthmus (thick arrow). Hematoxylin and eosin.
Between the ORS and the IRS, histologically barely visible cells called the companion layer are located. The IRS is composed of 3 concentrical layers. The innermost layer is the cuticle and interlocks with the cells of the hair cuticle. The outer 2 layers contain trichohyalin granules and produce the IRS keratins. The inner layer of these 2 layers is called Huxley’s layer. The outer layer is called Henle’s layer. IRS cornification is complete at the interface inferior portion/isthmus. This region is also called Adamson’s fringe. The ORS, the companion layer, and the IRS surround the HS. The HS consists of the hair cuticle (forms the hair surface), the cortex (forms the bulk of the hair and is composed of keratinized cells which contain pigment from the melanocytes of the bulb), and the innermost medulla.
Together with the IRS and the companion layer, the HS arises from the germinative cells of the bulb known as matrix cells (Sequeira and Nicolas 2012). Within a concavity of the matrix cells, the DP is located surrounded by a thin basement membrane.
The HF is surrounded by a basement membrane and a connective tissue sheath containing dermal sheath cells. Some of these are capable of regenerating the DP (reviewed in Rompolas and Greco 2014).
The HC
Hair growth is needed to renew the protective covering of the body, and it enables the seasonal change in coat quality and color. Therefore, after the initial follicular morphogenesis, the HF is maintained by cycling through periodic stages which include a growth phase (anagen), a regression phase (catagen), and a quiescent phase (telogen; Figure 6). Thereafter, a new cycle starts again (Hsu, Pasolli, and Fuchs 2011; Zhang et al. 2009). During this next cycle phase, the old club hair is shed in a process called exogen (Milner et al. 2002). The initiation of this phase coincides with the end of telogen and is completed when the club hair has been shed. Depending on the hair type and the species, the shedding of the hair fiber can occur at any time of the subsequent HC and thus is independent of the other cycle phases (Higgins, Westgate, and Jahoda 2009; Stenn 2005). Mice, for example, shed their overfur hair in the subsequent anagen, while their underfur hair is shed either in the following anagen or telogen phase. The actual mechanisms involved in the release of the club hair are not yet fully understood, but it seems that it is driven by activation and inhibition of proteases (Higgins, Westgate, and Jahoda 2009, 2011; Tobin et al. 2002). Another term that has been introduced recently in HC terminology is “kenogen”: It applies to HFs that have passed the telogen stage, lost their hair fiber (exogen), and remain empty for a certain time before a new anagen phase is initiated (Higgins, Westgate, and Jahoda 2009). Each of these HC phases has several subphases. The subphases of anagen, catagen, and telogen have been described morphologically in the mouse and in the dog (Muller-Rover et al. 2001; Muntener et al. 2011).
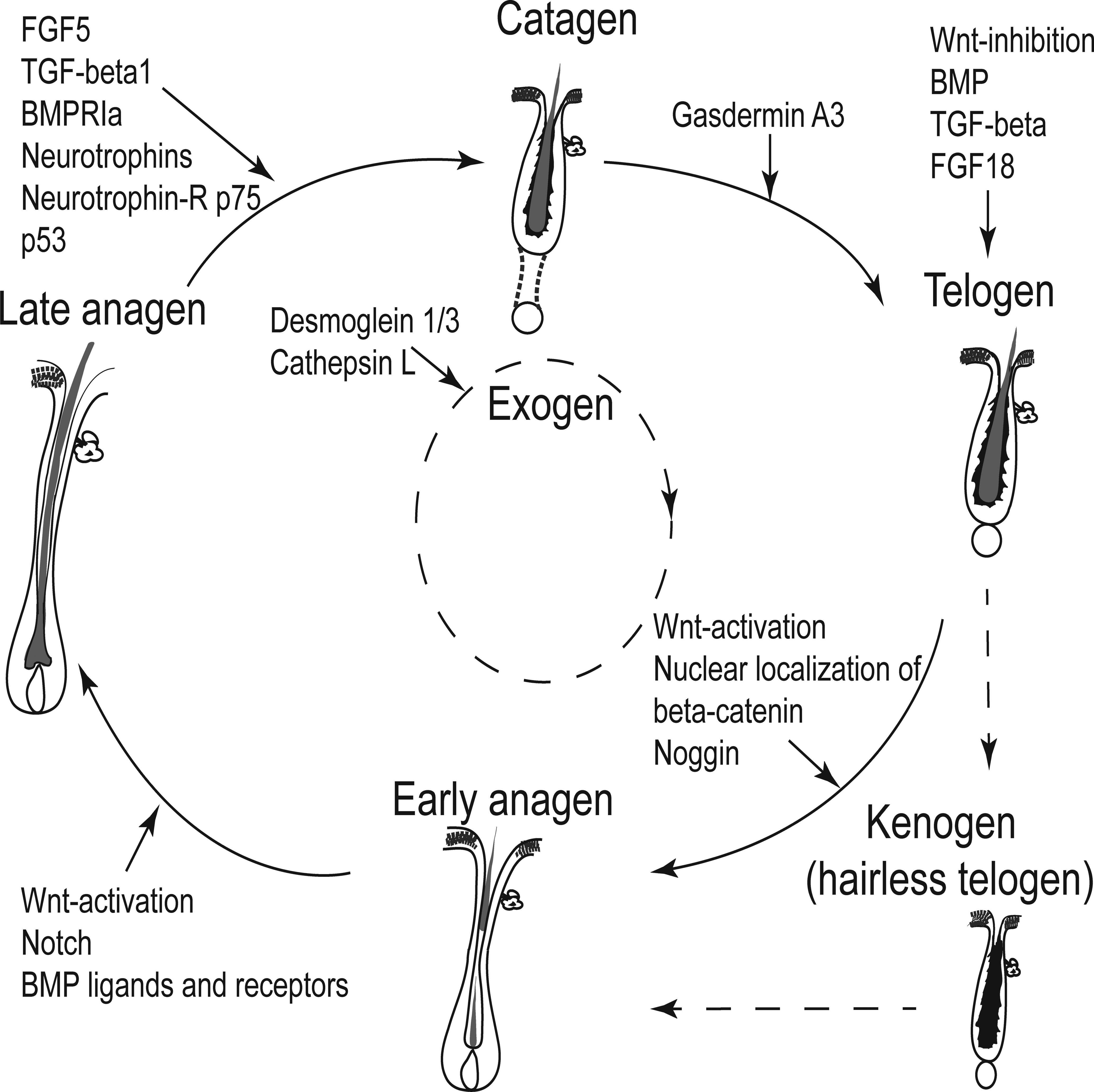
Schematic drawing illustrating the different hair cycle stages. The hair follicle cycles through periodic stages, which include a growth phase (anagen), a regression phase (catagen), and a quiescent phase (telogen). Thereafter, a new cycle starts. During the next cycle phase, the old club hair is shed in a process called exogen. A fifth hair cycle stage is called kenogen. Kenogen is a lag phase and applies to hair follicles that have passed the telogen stage, lost their hair fiber, and remain empty for a certain time before a new anagen phase is initiated.
The length of the HC phases determines the length of the HS and its replacement rate during shedding (Westgate, Botchkareva, and Tobin 2013). The duration and length of the different cycle phases vary depending on the genetic background, the age, sex, body region, hormonal influence, neurogenic stimulation, composition of the extracellular matrix, nutrition, status of health, numerous environmental factors (day length or photoperiod, grooming, ambient temperature, friction, and trauma), intrinsic factors (growth factors and cytokines), and drug therapy.
For example, it has been shown that mouse HFs and human eyebrow HF stay in anagen for only 2 to 4 weeks, while the anagen phase of human scalp HFs lasts 2 to 6 years (Duverger and Morasso 2013). The catagen phase lasts only for 2 to 3 weeks and the duration of telogen is for an average of 3 months. In humans, it is also known that about 1% of the telogen HFs are shed each day. These data are not available for our domestic animals. Hair growth in humans, as well as in companion animals, is not synchronized. Therefore, distinct stages occur simultaneously in different follicles. Nevertheless in the human scalp, in sheep, and in canine breeds that need clipping (such as poodles), the anagen stage of the HC predominates in the HFs. In humans, about 80% of the scalp HFs are in anagen, and in poodles, even 98% of the follicles are in anagen. In most mammalian species, however, the frequency of the telogen stage is significantly higher and the percentage of telogen follicles in most dog breeds is up to 34% (Muntener et al. 2011, 2012). In dogs, a difference can be observed in regard to the ratio of telogen and kenogen follicles between breeds which have a more synchronous HC to prepare for seasonal changes (arctic breeds) and breeds whose HC is asynchronous and therefore hair is shed during the whole year (R. W. Dunstan, personal communication, 28.09.2002). It can be assumed, although it has not been demonstrated, that the kenogen follicles enter into new anagen if a thicker hair coat is needed.
HF SC Dynamics
To sustain cyclic regeneration, each HF relies on its epithelial SCs. They act as a regeneration pool for the cells needed to maintain and remodel the epithelium of the continuously cycling HF throughout lifetime. SCs reside in niches that provide spatially distinct microenvironments for their maintenance and function. It is important to appreciate that HF-SCs are not a single multipotent entity, but that numerous SC populations and subpopulations characterized by different markers exist (Alcolea and Jones 2014; Purba et al. 2014; Rompolas and Greco 2014). Roughly HF-SCs can be subdivided into populations that are more quiescent and primed populations which are more prone to proliferation (Greco et al. 2009). These SCs have differing abilities to contribute to structures including the HF, the interfollicular epidermis, and the sebaceous glands (Goldstein and Horsley 2012). The isthmus is the SC bearing region of the HF and contains the bulge. It is identified in the anagen HC by the attachment of the arrector pili muscle and in the telogen stage by its location at the lower end of the HF (Kobayashi et al. 2009; Cotsarelis, Sun, and Lavker 1990; Lyle et al. 1998). It has to be noted that some authors define the isthmus as the area above the bulge and refer to the bulge and the secondary germ as separate compartments within the mid portion of the HF.
At late telogen, cells of the hair germ (which is located directly above the DP) get activated and at anagen onset the first cells start to proliferate (Greco et al. 2009; Lien et al. 2014). The cells located in the hair germ are considered to be “primed,” short-term HF-SCs and are LGR5 positive (Greco et al. 2009). The HF-SCs from the hair germ divide rapidly and give rise to transit amplifying cells that populate the germinative layer of the matrix that is surrounding the DP before they terminally differentiate to form the HS, its surrounding channel, the IRS, and the companion layer (Sequeira and Nicolas 2012). It has been shown by clonal analyses that the cells of the germinative layer are clonally related to each other (Legue, Sequeira, and Nicolas 2010).
Several days after the activation of the hair germ (late telogen/early anagen), the HF-SCs in the mid and upper bulge proliferate briefly to self-renew (Rompolas and Greco 2014). The HF-SCs of the lower bulge form the upper ORS. A third clonally distinctive cell population is named the lower proximal cup. These cells abut the DP at the proximal end of the HF and surround as a single cell layer the germinative matrix cells forming the outermost layer of the bulb. They extend distally and give rise to the lower ORS (Sequeira and Nicolas 2012; Rompolas and Greco 2014). As the cells derived from the hair germ and those derived from the bulge grow downward, the DP is pushed further away from the bulge, thus prompting the HF-SCs to return to quiescence. In contrast, the matrix cells still surround the DP and thus maintain contact. This is important because signals from the DP stimulate the matrix cells to continuously proliferate and to finally expand as a column upward, differentiate, and generate the concentric layers of the IRS and the HS (reviewed in Rompolas and Greco 2014). Each cell lineage induces the expression of distinct keratins.
Interspersed with the matrix cells are also melanocytes, which produce melanin granules that are transferred to the matrix cells, thus pigmenting the HS (Slominski et al. 2005). The SCs that support the production of new melanocytes in each HC are neural crest-derived melanocyte SCs. They also reside in the bulge (Nishimura et al. 2002) and if they are not maintained, graying of hair results (Nishimura, Granter, and Fisher 2005).
Once a new HS grows out to a certain length, catagen is induced. During catagen, matrix cells and the cells of the lower ORS undergo apoptosis and push the HF into the involution phase of the HC. As regression proceeds, the bulb loses its volume due to the apoptosis and forms a narrow epithelial strand that retracts upward. At its end the DP is trailing. Simultaneously to the apoptosis of the bulb, the ORS cells of the upper isthmus repopulate a new SC niche. Thereby the upper ORS cells that divided the least form the new bulge, whereas those with slightly more activity become the new hair germ. The ORS cells that proliferated the most differentiate to form an inner bulge layer of terminally differentiated cells that secrete inhibitory BMP6 and fibroblast growth factor (FGF)18 signals (Hsu, Pasolli, and Fuchs 2011). By the end of catagen, the HF is remodeled back into its resting phase (telogen) and the HC has been completed. At this stage, the HS is named club hair which is anchored by trichilemmal keratin composed of keratin (Krt)6 which is produced by the inner layer of the ORS. These ORS cells may play a critical role in maintaining SC quiescence in the niche during the resting phase (Hsu, Pasolli, and Fuchs 2011, reviewed in Rompolas and Greco 2014). It has to be noted that all studies investigating SC dynamics have been performed in different mouse models and up to now no functional studies are available from humans and dogs. In these species, only experiments labeling the different SC populations and clonality assays have been performed. However, since the HC stages are morphologically very similar between different species and some SC markers are shared, it can be assumed that the SC dynamics are similar. The SC dynamics during the HC and their contribution to the HF regeneration are summarized in Figure 7.
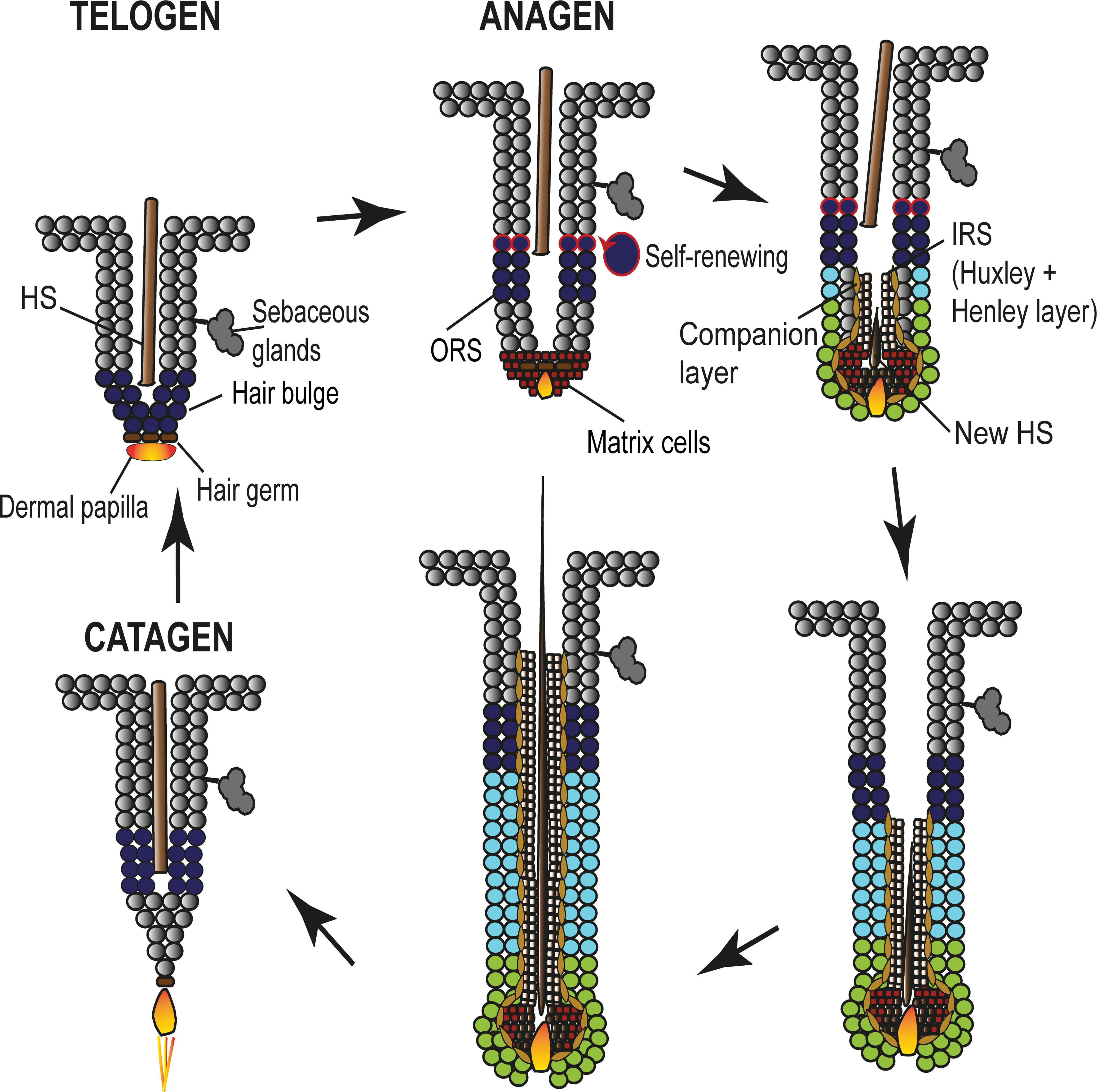
Schematic drawing illustrating stem cell–initiated differentiation of the hair follicle. During early anagen, hair follicle stem cells from the hair germ (brown) divide rapidly and give rise to transit amplifying cells (red brown) that populate the germinative layer of the matrix surrounding the dermal papilla. The matrix cells terminally differentiate to form the HS (dark brown), the IRS (light-brown), and the companion layer (yellow brown). The stem cells of the upper bulge (dark-blue with red rim) proliferate briefly to self-renew, whereas the stem cells of the lower bulge (dark-blue) proliferate to form the distal 2/3 of the ORS (light blue). The outer layer of the bulb is formed by a distinct lineage of cells named the lower proximal cup, which abut the dermal papilla in continuity with the germinative layer (green) and give rise to the proximal ORS. IRS, inner root sheath; ORS, outer root sheath; HS, hair shaft.
SC-associated Marker Expression
As mentioned already above, HF-SCs are not a single multipotent entity, but extensive cellular heterogeneity exists within the mature pilosebaceous unit. The SC-associated marker expression has been the topic of a number of excellent recent reviews, and differences between human and murine SC markers have been outlined (Arwert, Hoste, and Watt 2012; Rompolas and Greco 2014; Solanas and Benitah 2013; Purba et al. 2014). Thus, SC marker expression in mice and humans will be discussed only briefly in the review and new data about canine SC-associated biomarker expression will be presented.
In the murine adult HFs, the most primitive epidermal SCs are restricted to the central isthmus directly above the bulge. These cells express LGR6 and it has been shown that these LGR6-positive cells renew epidermal cells and sebocytes. Furthermore, LGR6-positive cells can exit their niche during the HC, give rise to LGR5-positive SCs, and contribute to the formation of the hair germ (Rishikaysh et al. 2014; Snippert et al. 2010).
In vivo lineage tracing experiments in mice showed that the progeny of Krt15-positive SCs participates in the formation of all epithelial lineages of the HF. It has also been shown that CD34+/integrin α6+ bulge cells contribute to the formation of all epithelial skin layers and thus confirm their identity as SCs. In addition to these markers, bulge SCs in the mouse HF can also be identified based on the expression of Krt19 as well as several transcription factors (Alcolea and Jones 2014, reviewed in Rompolas and Greco 2014).
In human studies, the SC niche has not been investigated as detailed as in the mouse, but it has been shown that at least an upper and a lower reservoir of SCs exists. Both reservoirs are Krt19 positive and can be distinguished by the differential expression of Krt15 and CD200. Both are expressed higher in the upper reservoir, whereas CD271, CD29, and CD34 are expressed stronger in the lower reservoir (Inoue et al. 2009; Rathman-Josserand et al. 2013). In addition, it has been shown that LGR5-positive cells reside in the hair germ (Jaks et al. 2008).
In the canine HF, CD34-, Krt15-, SRY-box (Sox)9-, and LGR5-positive cells are present in the isthmus and in the hair germ. Although CD34 is present in the entire isthmus and the suprabulbar region of the anagen HC stage and in the lower ORS of the telogen HC stage, Krt15 is present only in the ORS cells of the lower half of the isthmus of telogen HFs and in the outermost layer of the ORS of the upper isthmus during anagen. Sox9-positive cells are present in the innermost layer of the ORS in anagen only and LGR5-positive cells are present only in the hair germ of telogen HFs. Nestin is expressed in the DP and the lower fibrous sheath of anagen HFs (Gerhards et al. 2016; Kobayashi et al. 2009). In addition, messenger RNA expression for LGR6 and CD200 was shown in the canine HF (Kobayashi et al. 2010). Summarizing the data gathered from dogs so far, canine SC-associated biomarker expression pattern resembles in many aspects the human HF SC compartment (CD34, K15, Sox9, and Nestin). It also shares however features with the murine SC reservoir (K15, Sox9, and LGR5). Disagreement of marker expression occurs in the lack of Sox9+ cells in canine telogen HFs and, compared to mice, the restriction of LGR5 expression to telogen and early anagen HC stages.
Expression of SC markers in mice, humans, and dogs is summarized in Figure 8 (Li et al. 2003; Wang et al. 2006; Alcolea and Jones 2014; Kobayashi et al. 2009; Solanas and Benitah 2013; Rishikaysh et al. 2014; Gerhards et al. 2016, reviewed in Purba et al. 2014).
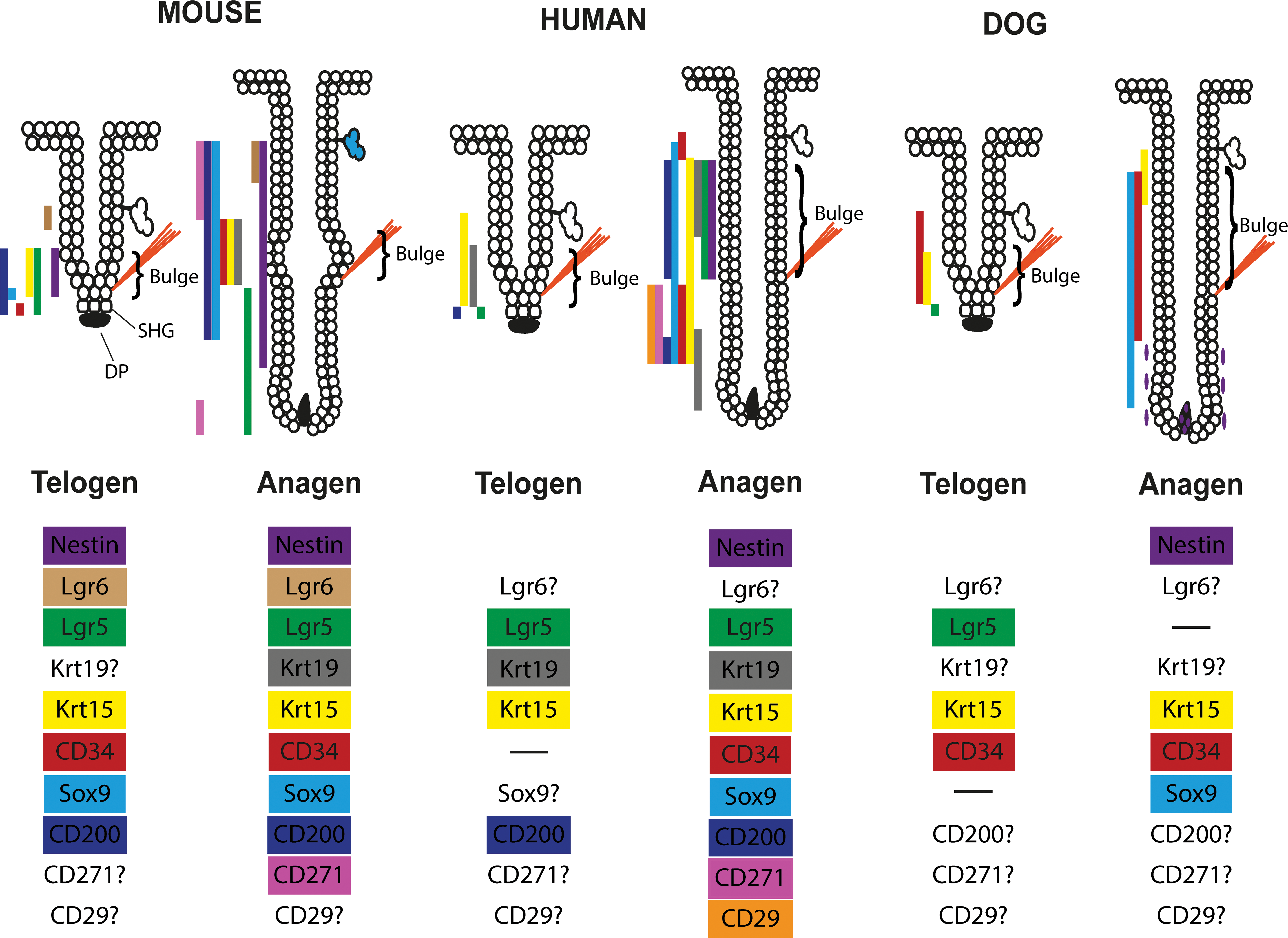
Summary of the differential stem cell–associated biomarker expression in mice, humans, and dogs. The information are gathered from Li et al. (2003), Wang et al. (2006), Alcolea and Jones (2014), Kobayashi et al. (2009), Solanas and Benitah (2013), Rishikaysh et al. (2014), Gerhards et al. (2016), and Purba et al. (2014). The ? indicates that no literature has been found on this marker for this species.
HC Control
The coordination of the HC phases and the SC activity is dependent on complex interactions between signals of the follicular (niche components) and dermal microenvironment (e.g., keratinocytes, fibroblasts, adipocytes, immune cells, and nerve fibers), macroenvironmental factors (e.g. hormones, genetics, and age), and environmental factors (e.g. daylight, nutrition, and circadian rhythm) (Chen et al. 2015; Chen and Chuong 2012). By now, the molecular and cellular composition of the HF-SC niche in the mouse has been well characterized. The state of the murine HF is mediated by a complex interplay between transcription factors implicated in, as well as proteins belonging to, several signaling pathways, including the Hedgehog, Wnt/β-Catenin, transforming growth factor (TGF)-β, BMP, and Notch pathways (reviewed in Lee and Tumbar 2012). However, the understanding how the niche elements work together and how they interact with macroenvironmental and environmental factors to initiate and promote a new HC and thus to regenerate a new HF is still not complete. Signaling pathways that regulate SC quiescence, proliferation, and differentiation include Wnt, BMP, Notch, and FGF18 (reviewed in Rompolas and Greco 2014). It has been demonstrated in various studies that various signals from these pathways constantly compete, and it is a delicate balance of these signals that keeps SCs and their progenies either in a stage of rest or in a stage of proliferation. In general, BMP signals derived from dermal, adipose, and epithelial tissue repress cell proliferation, while Wnt signals promote SC activation and growth.
In more detail, bulge SCs maintain a slow-cycling behavior due to an overabundance of BMP, TGF-β, FGF18, as well as Wnt inhibitors that are secreted from various sources including the DP, the inner Krt6-positive bulge layer but also the bulge SCs themselves during the resting phase of the HF (telogen). If the balance of inhibiting signals is tipped toward more activating signals, new anagen is initiated. It has been shown that activation of canonical Wnt signaling and subsequent stabilization and nuclear localization of β-catenin is critical for the entry into anagen. Wnt signaling is also important for the proliferation of matrix cells and thus anagen progression. BMP ligands and receptors regulate the terminal differentiation program of the IRS and HS precursors. In addition, Notch signaling is critical for differentiation into the mature HF (reviewed in Rompolas and Greco 2014). The induction of catagen is promoted by molecules such as neurotrophins (Botchkarev et al. 1998, 1999), FGF5, p75, p53, TGFβ1, and BMPRIa (reviewed in Rishikaysh et al. 2014). Gasdermin A3 has been recently shown to be important for the transition of catagen to telogen by balancing the Wnt-signaling pathway (Bai et al. 2015). The signaling events contributing to different HC stages are summarized in Figure 9.
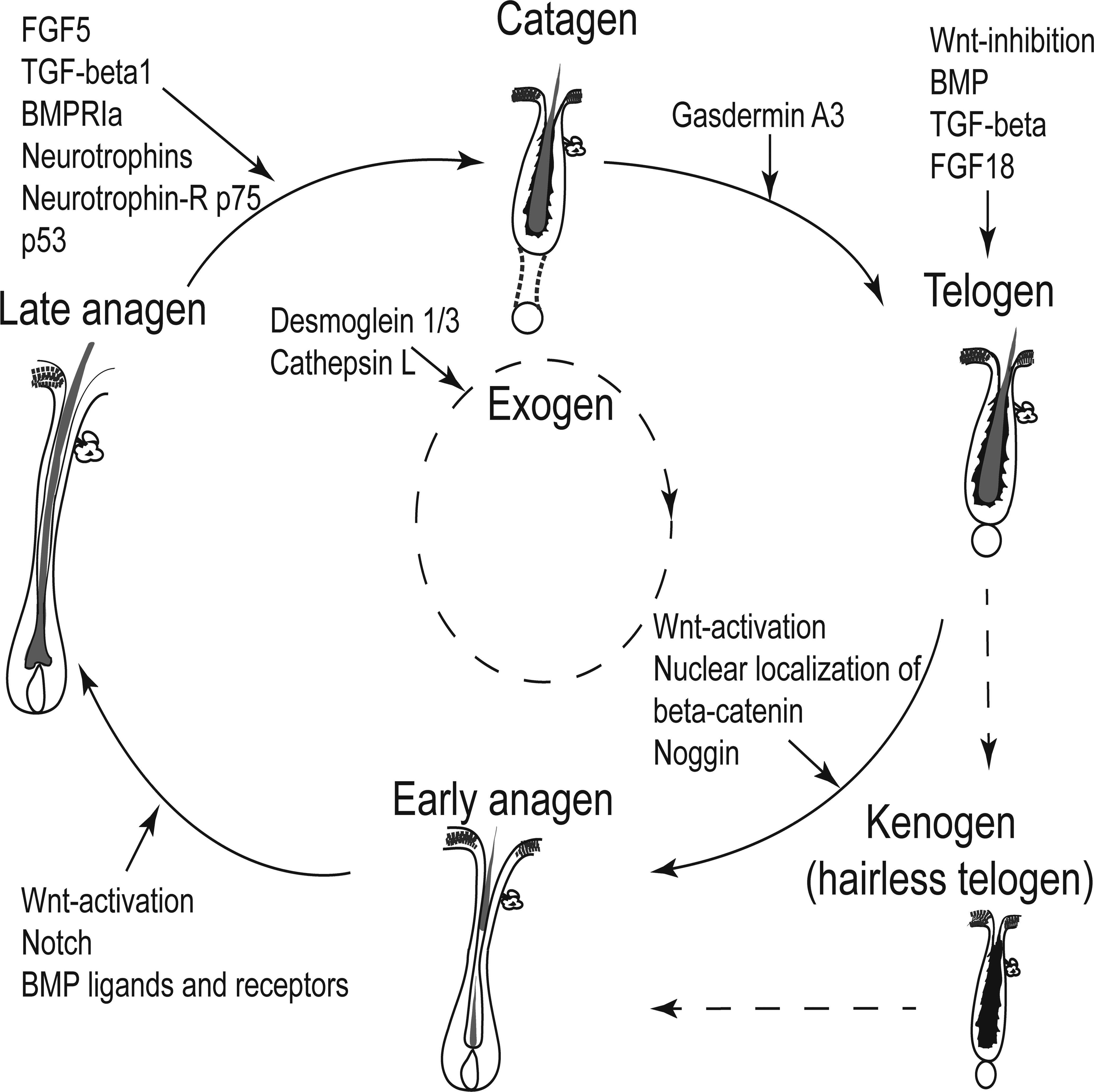
Schematic drawing illustrating some known factors associated with the maintenance of the different physiological hair cycle stages. The coordination of the hair cycle phases is dependent on complex interactions between signals from the follicular and dermal microenvironment, macroenvironmental factors, and environmental factors.
Acquired Noninflammatory Alopecia
Hair loss, hypotrichosis, or alopecia in humans or dogs is a regular cause for consulting a medical doctor or a veterinarian, and the possible underlying causes are numerous. It would exceed the scope of this review to discuss all possible etiologies for alopecia.
Since we propose the dog with some of its natural occurring noninflammatory disorders as a possible model to study human alopecia, we will outline in this chapter some basic principles that may be relevant for canine and human HC disorders. In all canine hair cycle arrest disorders (such as hyperadrenocorticism, hypothyroidism, hyperestrogenism, alopecias of unknown origin and pattern baldness) the number of kenogen follicles is increased 3- to 4-fold, whereas the number of anagen follicles decreases dramatically (Muntener et al. 2012). An increased amount of kenogen follicles in alopecia is also observed on the human scalp (Rebora and Guarrera 2002, 2004).
The increased amount of kenogen follicles indicates that either exogen has occurred prematurely and the empty telogen follicle is not yet competent to enter new anagen or the induction of a new anagen phase does not occur after the completion of telogen phase and the old club hair has been shed during exogen. A third possible cause may be that signals which are maintaining the anagen phase (anagen promotion) are missing, the HFs enter catagen prematurely and thus enter the subsequent telogen phase earlier. At the same time, the initiation of new anagen is not more effective, which results in a higher number of telogen follicles which lose the HS and thus stay in kenogen until adequate signals initiate new anagen. If a HF stays in kenogen for a prolonged time, this follicle may also get atrophic.
Conclusion
As a summary, this review outlines the general knowledge about HF morphogenesis, HF anatomy, the HC, follicular SC dynamics, and some possible pathomechanisms for noninflammatory alopecia. Data about HF physiology and SC dynamics mostly generated from rodents have helped in understanding the physiology of HF development and the HC. This, in combination with newly available techniques, may help in the future to fully understand alopecic disorders in both humans and companion animals. Since some of the naturally occurring canine diseases can be explained by pathomechanisms generated by data from humans and mice, the dog might be a promising model for alopecia in humans.
Footnotes
Author Contribution
Authors contributed to conception or design (MW, DW); data acquisition, analysis, or interpretation (MW, DW); drafting the article (MW). All authors critically revised the manuscript, gave final approval, and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
