Abstract
Aim: Diazinon (DZN) is one of the most important organophosphorus compounds used to control pests in agriculture in many countries. Several studies have shown that exposure to DZN may alter protein expression in the liver. In order to further investigate the mechanism of DZN toxicity, differentially expressed ATP-interacting proteins, following subacute exposure to toxin, were separated and identified in rat liver. Main methods: Male rats were equally divided into four groups: control (corn oil) and DZN (15 mg/kg) by gavage once a day for 4 weeks. After homogenization of liver tissue, lysates were incubated ATP-sepharose beads. After several washes, ATP-interacting proteins were eluted and separated on 2-D polyacrylamide gels. Deferentially expressed proteins were cut and identified using matrix-assisted laser desorption/ionization/time-of-flight and Mascot database. Identified proteins were classified according to their biological process using protein analysis through evolutionary relationships (PANTHER) Web site. Key finding: In this work, we showed that several key proteins involved in biological processes such as antioxidant system, oxidative stress, apoptosis, and metabolism were differentially expressed after subacute exposure to DZN.
Introduction
Organophosphorus (OP) compounds, such as diazinon (O,O-diethyl-O-(2-isopropyl-6-methyl-4-pyrimidin-y)phosphorothionate; DZN), are extensively used as agricultural pesticides to control insect pests. 1 DZN has made great contributions to food production, but excessive use of this toxic chemical in agriculture has developed severe health hazards. 2,3 DZN is metabolized to diazoxon, which is an acetylcholinesterase inhibitor, and may contribute to the toxic effects of DZN. 4
An in vivo study showed that exposure to DZN increased activity of alanine transaminase, aspartate transaminase, and alkaline phosphatase in serum. 5 Moreover, DZN could interfere with lipid metabolism and increase the levels of phospholipids, triglycerides, and cholesterol. Furthermore, DZN may induce oxidative stress. 6
Subacute studies revealed that exposure to parathion, malathion, and DZN could significantly increase oxidative stress biomarkers such as lipid peroxidase, reduce antioxidant defense enzymes including glutathione, enzyme-chelating free radicals, glutathione peroxidase, catalase, superoxide dismutase, and glutathione reductase. Vitamins E and C could reduce DZN-induced oxidative stress. 7 In recent years, most of the studies about chronic exposure to OP poisoning showed that these toxins could induce apoptosis by activating caspases 9 and 3. 8 –10
Many reports have revealed that OPs induce hepatotoxicity. 11 –14 Liver has a significant role in activation, exertion, and detoxification of OP compounds. Previous studies have shown that OPs alter the expression pattern of different enzymes such as antioxidant enzymes in the liver. 15,16 DZN accelerates hepatocytes death and the loss of cellular ATP. 17
The aim of this study was to investigate differentially expressed proteins that could interact with purine group in adenine molecule in the liver tissues of rat after subacute exposure to DZN. This was achieved through pull down purification of proteins using ATP-sepharose, followed by two-dimensional (2-D) gel electrophoresis for protein separation and matrix-assisted laser desorption/ionization/time-of-flight (MALDI/TOF/TOF) for protein identification.
Materials and methods
Animals
Animal treatment
Adult male Wistar rats weighing 250–300 g were provided by Animal Center, School of Pharmacy, Mashhad University of Medical Sciences, Iran. Rats were maintained at 23 ± 1°C and 12 h-light/12 h-dark cycles with free access to food and water. All animal procedures were approved by the Ethical Committee Acts of Mashhad University of Medical Sciences for care and use of laboratory animals (permission # 89609). Twelve rats were equally divided (each group contains six rats) into control group (corn oil) and DZN-treated group (15 mg/kg). Both groups were administrated via gavage once a day for 4 weeks. After 4 weeks, rats were killed, and the liver tissues were removed and washed in normal saline, then stored at −80°C until further use.
Preparation of ATP-sepharose complex
According to manufacturer’s instruction, 0.5 g of ATP-sepharose beads (Sigma, St Louis, Missouri, USA) was suspended in sodium chloride (NaCl) solution (3 mM, 5 ml) and shaked for 5 min at 4°C to swell. Then the beads were centrifuged at 1000g for 1 min at 4°C. Supernatants were removed, and the beads were washed three times with double-distilled water (ddH2O; 1000 µl) and stored at 4°C until use.
Tissue preparation
Two hundred milligrams of liver tissues was homogenized in 1 ml of lysis buffer containing Tris (50 mM, pH 7.4), ethylene glycol tetraacetic acid (2 mM), ethylenediaminetetraacetic acid (1 mM), sodium orthovanadate (1 mM), sodium fluoride (10 mM), NaCl (150 mM), 2-mercaptoethanol (10 mM), complete protease inhibitor cocktail (2 µl, Sigma, USA), and phenylmethylsulfonyl fluoride (1 mM) using a polytron homogenizer (Kinematica, Switzerland). After sonication (UP100 H, Hielscher, Germany) for 40 s, homogenates were centrifuged (Hettich Universal 320 R, Germany) at 10,000g for 10 min at 4°C. Finally, total protein contents were determined using a Bradford protein assay kit (BioRad, Hercules, California, USA). Protein concentrations of lysates were adjusted to 1 mg/ml.
Affinity chromatography
One hundred microliters of tissue extracts, containing the same amount of protein for both groups were transferred to ATP-sepharose beads and shaked for 10 min at 4°C. After a brief centrifugation at 1000g for 1 min at 4°C, supernatants were removed and discarded. Then the beads were washed three times with ddH2O (1000 µl). NaCl solution (2 mM, 1 ml) was added to the beads and shaken for 5 min at 4°C. After a brief centrifugation, supernatants–containing ATP-interacting proteins were collected and transferred to clean microtubes. The latter step was repeated three times, and supernatants were pooled. For desalting, pooled samples were dialyzed against ddH2O for 3 days at 4°C using a semipermeable membrane with a 2000 Da cutoff Spectra/Por® (Rancho Dominguez, CA, USA). Finally, samples were freeze-dried and stored at −80°C until use.
2-D Gel electrophoresis
Briefly, isoelectric focusing was performed. Then strips were placed on top of 12% sodium dodecyl sulfate–polyacrylamide gels and ceiled with agarose gels. Electrophoresis was carried out at 120 V for 40 min per gel at 20°C. After silver staining, gels were scanned, and the stained gels were scanned on Image scanner III (Epson, Japan). All procedures were carried out according to procedure carried out in our laboratory. 18
Intensities of protein spots in control and DZN-treated rats were analyzed using Image Master Platinum 6.0 software (GE Healthcare, Cleveland, Ohio, USA). Protein spots normalization was performed against the total volumes of all the detected spots. To analyze statistically different spots in treatment and control groups, Student’s t-test was performed. The values of p ˂ 0.05 were considered as statistically different. Differentially expressed ATP-interacting protein spots were manually excised and transferred to microtubes-containing 1% acid acetic, which was then sent to the Center of Genomic Sciences at University of Hong Kong for in-gel digestion and mass analysis using MALDI/TOF/TOF analyzer.
Mass analysis and database searching for protein identification
Proteins were identified using MALDI/TOF/TOF analyzer (Applied Biosystems, Foster City, California, USA) and Web-based Mascot software (version 2.1.0, Matrix Science, London, UK). Mascot search parameters were set as follows: taxonomy, rattus (expressed sequence tag) or all entries; peptide charge: +1, monoisotopic; mass spectrometry (MS)/MS fragment tolerance: ± 0.2 Da; precursor mass tolerance: 75 ppm; cysteine carbamidomethylation for fixed modification; and methionine oxidation for variable modification. Proteins with confidence intervals (CIs) of 100% were accepted.
Classifications and network interaction analysis
For classification of altered proteins protein analysis through evolutionary relationships (PANTHER) online database (http://www.pantherdb.org) was used. This Web site categorizes proteins in different categories according to their biological process and biological pathways. 19
Results
To analyze the effect of DZN on expression pattern of ATP-interacting proteins, pull down assay was performed using ATP-sepharose beads. Then proteins were separated and identified by 2-D gel electrophoresis and MS analysis. In this project, 12 proteins were identified (Figures 1 and 2).

Two-dimensional gel electrophoresis of control targets in rat liver.
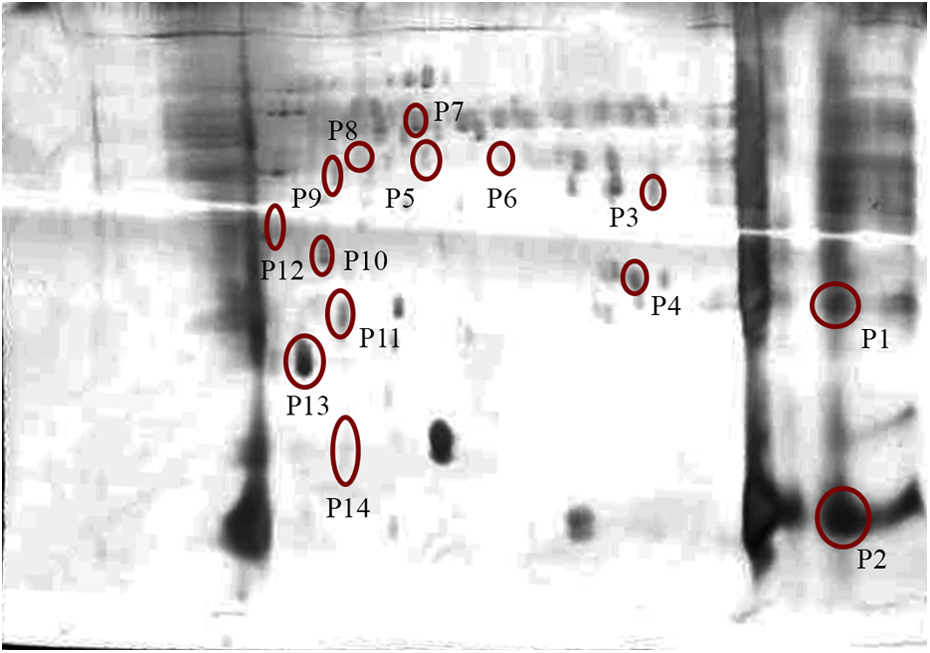
Two-dimensional gel electrophoresis of DZN targets in rat liver. DZN: diazinon.
To classify proteins according to biological processes, their SwissProt accession numbers were submitted to PANTHER database (Figure 3).

Biological process classification of DZN target proteins in rat liver by PANTHER classification system. DZN: diazinon; PANTHER: protein analysis through evolutionary relationships.
The proteins were divided in three classes including metabolic process, response to stimuli, immune system process, localization, cellular component organization, and cellular process. Most of the identified proteins were involved in metabolic process.
The information of identified proteins including protein name/gene names, size, Swiss Prot accession number, sample name, CI%, protein score, sequence coverage (%), theoretical isoelectric point, and sequence with the highest ion score are listed in Table 1.
Identified DZN target proteins in rat liver by MALDI/TOF/TOF.

GN: gene name; pI: isoelectric point; DZN: diazinon; CI: confidence interval; PANTHER: protein analysis through evolutionary relationships; MALDI: matrix-assisted laser desorption/ionization; TOF: time of flight.
Discussion
By combining affinity chromatography and proteomics, 12 ATP-interacting differentially expressed proteins were identified in the liver tissue of DZN-treated rats.
Previously, the effect of chronic exposure to DZN on total proteome was reported. 7,10,18,20 –23 In this study, we specifically studied the alteration of purine packet-containing proteins such as ATPases and kinases because these proteins are key proteins in many signal transduction pathways. In whole proteomics, proteins like kinases, cannot be detected because of low cellular concentration. Here we tried to concentrate these types of proteins prior to 2-D electrophoresis, so that we got a 2-D with less number of spots as compared to whole proteomics as we reported in Lari et al. 18
Here we introduced some novel proteins involved in DZN toxicity. Besides inhibition of neuronal acetylcholine sterase, 10,20,21 several reports have demonstrated that DZN could affect intracellular systems including apoptosis, oxidant, metabolic enzymes, and cell death. 7,10,20 –23
It has been shown that exposure to different concentration of DZN could induce oxidation of protein by decreasing the capacity for scavenging damaged proteins. 7,22,23
This study showed that DZN altered the levels of several ATP-interacting cellular proteins including metabolic enzymes, structural proteins, antioxidant, and apoptotic proteins.
Phosphatidyl ethanolamine-binding protein (PEBP), also called RAF-1 kinase inhibitor protein, decreased after chronic exposure to DZN. By inhibition of RAF1 and degradation of rapidly accelerated fibrosarcoma (RAF)–mitogen-activated protein kinase (MEK) complex, PEBP could inhibit RAF/MEK/extracellular signal-regulated kinase cascade 27 and affect the cell cycle fidelity. 28 Moreover, PEBP downregulation could promote cell proliferation and migration of human hepatoma cells. 29 It has been shown that low levels of PEBP may protect cancer cells against apoptosis and improve viability of metastatic cancer cells. 30 –32 It has been indicated that chronic stress condition decreased the level of PEBP. 33 PEBP downregulation after chronic exposure to DZN may induce cancer and metastasis in liver tissue.
DZN could reduce the level of catechol-o-methyl transferase (COMT). COMT involves in several detoxification pathways: (1) metabolism of intermediate indolic compounds in melanin synthesis, such as 5,6-dihydroxyindole-2-carboxylic acid, this compound could be readily oxidized to toxic quinone derivatives and (2) metabolism of catechol estrogens, such as 4-hydroxy estradiol, known as a hepatotoxin. 34 –37
Malate dehydrogenase (MDH) was also downregulated by DZN. Cytosolic MDH is a metabolic enzyme involved in the conversion of malate to oxaloacetate. 38 Previous study demonstrated that acute and chronic administration of DZN could downregulate MDH in liver tissue. Moreover, decrease of MDH has been accepted as a biomarker of damaged liver. 39 –41 Sastry and coworkers demonstrated that acute and chronic DZN administration resulted in the reduction of total MDH level.
This study showed that DZN increased proteasome activator subunit 1 (PSME). Proteasomes are distributed throughout eukaryotic cells at high concentration, and the main function of PSME is degradation of damaged or useless proteins in an ATP/ubiquitin-dependent process in a non-lysosomal pathway. 18 Proteasomal depredating pathway is essential for various intracellular processes including regulation of gene expression, cell cycle and division, stress response, elimination of damaged proteins, antigen process for immune system, oxidative stress, and apoptosis. 42 Previous studies revealed that DZN could increase cellular ubiquitinylated proteins and consequently led them to ATP/ubiquitin-dependent proteasomal degradation pathway. 43 It could be concluded that PSME increase could be related to increase in cellular ubiquitinylated protein levels.
DZN decreased the level of glutathione-S-transferase alpha3 (GSTα3). This enzyme involves in the cellular detoxification of both xenobiotic and endobiotic compounds. GST catalyzes conjugation of toxins with glutathione and produces less toxic and more hydrophilic products that can then be partially metabolized and excreted. 44 GSTα3 is well recognized as a serum biomarker of liver toxicity. 40 Previous reports showed that subchronic DZN administration could reduce the level of GSTα3 in liver. 41 Fujioka and coworkers demonstrated the important role of GSTα3 in OP detoxification. 45 Decreased level of GSTα3 using DZN showed that DZN could reduce the antioxidant capacity of cell and disturb cellular redox capacity.
Nitrilase homolog1 (NIT1) was also downregulated by DZN. NIT1 is one of the proteins involved in nitrogen compound metabolic process. Several studies revealed that NIT1 has tumor-suppressor properties that can enhance the apoptosis in cancer cells. 45 Downregulation of NIT1 by DZN may promote cell growth in tumor cells.
Our results showed that DZN increased annexin A3 (ANXA3), a calcium-dependent PEBP. ANXA3 plays a role in the regulation of cellular growth, inflammation, and signal transduction pathways. This protein inhibits phospholipase A2 (PLA2) and induces cleavage of inositol 1,2-cyclic phosphate to form inositol 1-phosphate and promotes inflammation recovery. 46 Previous studies showed that CCl4 increased the level of this protein in liver tissue. 47 Overexpression of ANXA3 promotes metastasis in different cancers. 48,49 ANXA3, using hypoxia-inducible factor-1 pathway by producing vascular endothelial growth factor, has angiogenesis effect. 50
Enoyl-CoA hydratase (ECH) was downregulated by DZN. ECH involves β-oxidation of fatty acids. 51 Some reports described that ECH dysfunction may produce acute liver encephalopathy, liver dysfunction, and sudden infant death syndrome. 52 DZN may play role in several metabolic dysfunction through reduction of ECH level.
3-Mercaptopyruvate sulfurtransferase (MPST) was also downregulated by DZN. This enzyme plays a role in cyanide detoxification. 53 MPST is an antioxidant enzyme with important role in cellular redox regulation. 54 This report showed that DZN may induce oxidative stress through decreasing total amount of MPST.
Besides MPST, DZN may induce oxidative stress by peroxiredoxin-6 (PRDX6) downregulation. 55,56 PRDX6 is an antioxidant enzyme that reduces cellular hydrogen peroxide (H2O2). This enzyme involves in the activities that are susceptible to cellular redox such as proliferation, signal transduction, and apoptosis. 56 PRDX6 is a bifunctional enzyme with both peroxidase and PLA2 activities. PRDX6 produces arachidonic acid (AA) using PLA2, which is essential in apoptosis-mediated tumor necrosis factor α (TNF-α). 56 Downregulation of PRDX6 by DZN may promote cellular susceptibility to H2O2-induced apoptosis and decrease apoptosis due to TNF-α in cells. 56
Conclusion
Using a pull down assay and proteomics approach, pattern of ATP-interacting proteins expression after exposure to DZN was studied. Our data provided some novel insights into the mechanism of DZN hepatotoxicity. We showed that DZN could alter the levels of proteins involved in antioxidant, apoptotic, and metabolic activities of the cells.
Footnotes
Acknowledgment
The authors are thankful to the Vice Chancellor of Research at the Mashhad University of Medical Sciences for the financial support of this project.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
