Abstract
Lethal cardiac complications leading to death and various arrhythmias have been reported after organophosphate and/or carbamate poisonings. The present study focuses on the long-term effects of repeated low-level exposure to diazinon, propoxur, and chlorpyrifos (CPF) on cardiac function in rabbits. The yearly based experimental scheme of exposure consisted of two oral administration periods, lasting 3 months and 1 month each, interrupted by an 8-month washout period (total duration 12 months). At the end of the experimental scheme, the rabbits underwent an echocardiographic evaluation under sedation, after which they were killed and the tissue and serum samples were collected. A mild localized cardiotoxic effect was established by echocardiography for the three pesticides tested. Severe histological alterations were identified, especially in the diazinon-treated animals in agreement with increased persistence of this pesticide established in the cardiac tissue. In addition, all pesticides tested increased the oxidative stress and oxidative modifications in the genomic DNA content of the cardiac tissues, each one following a distinct mechanism.
Introduction
Organophosphates (OPs) and carbamates constitute two classes of pesticides, widely used in the last decades on a variety of crops and as indoor protection management products. Exposure of the general population to these compounds has been documented through occupation-related pathways, through the domestic use of pesticides and food consumption. These two classes of pesticides have been associated with various acute and chronic effects on human health. 1
The OP, diazinon (O,O-diethyl O-[4-methyl-6-(propan-2-yl)pyrimidin-2-yl] phosphorothioate), is a synthetic chemical compound used worldwide for its insecticidal action, primarily used for the control of plaques, in crops, domestic flocks, and against pests infecting pets. Its action is mediated mainly through the inhibition of acetylcholinesterase (AChE), a serine protease that hydrolyzes the neurotransmitter acetylcholine in the synaptic space. 2 The activity of this compound is also associated with oxidative stress, as demonstrated by in vivo studies. 3 –5 Diazinon causes toxic effects to organs and tissues, reduces sperm motility and viability, increases sperm morphological abnormalities, 6 and has been described as a mutagenic compound for somatic cells in vivo and in vitro. 7 –9 Interestingly, the oxon metabolite of diazinon is reported to be more toxic than its parent compound. 10
Chlorpyrifos (CPF) [O,O-diethyl-o-(3,5,6-trichloro-2-pyridyl)phosphorothionate] is among the leading OP pesticides widely used throughout the world for agricultural and domestic applications. CPF has been registered as a potential groundwater contaminant by the US Environmental Protection Agency (US EPA), as a chemical of special concern by the Pesticide Action Network (PAN), and classified by the World Health Organization (WHO) as a class II toxic and moderately hazardous compound. Notably, in 2002, the US EPA eliminated all residential uses of CPF due to its high potential health risk. 11 Although the principal action mechanism of CPF is acetylcholinesterase inhibition, other mechanisms, including induction of oxidative stress, have also been implicated. Studies on the genotoxicity of CPF are controversial. The effects of CPF on DNA and on chromosomes of mammals 9 have been reported to cause dose-related DNA damage in mice lymphocytes. 12 Moreover, CPF has been reported to elicit a number of effects including hepatic dysfunction, embryotoxicity, teratogenicity, neurobehavioral, and neurochemical changes. 4 Acute and chronic exposures to CPF have resulted in liver damage, as evidenced by histopathological changes and increases in several liver enzymes. 13,14 Others, on the other hand, have classified CPF as nonteratogenic 15 and noncarcinogenic. 16
Propoxur, an N-methylcarbamate ester (2-isopropoxyphenyl N-methylcarbamate), is likewise endowed with the ability to inhibit the catalytic action of AChE in insects. It is used for the control of insects in crops and pets, while its inhibitory activity appears to be less evident in mammals. Indeed, in mammals, N-methylcarbamate esters metabolized into N-nitroso metabolites by N-nitrosation during the digestion process, especially in the stomach region of the digestive tract. N-Nitrosomethylcarbamate retains inhibiting activity of AChE but is less toxic than the parent compound. N-nitroso N-methylcarbamate insecticides mainly target the forestomach, where tumors have been reported. 17 Furthermore, propoxur and nitroso-propoxur increase the frequency of sister chromatid exchanges (SCEs) and micronuclei (MN) in human lymphocytes in vitro. 18 The toxicological reactions produced by AChE inhibitors are associated with severe toxic effects caused by accumulation of acetylcholine in the synaptic space. 19 In acute poisonings, lethal doses of AChE inhibitors were shown to induce increased blood pressure and respiratory arrest. 20 Respiratory failure may occur due to extreme cholinergic stimulation of the respiratory centers and/or peripheral respiratory musculature. 21,22 In addition, cardiovascular complications may occur as a result of the obstruction of vasomotor neuron depolarization through excessive cholinergic stimulation. 23 Cardiac complications leading to various arrhythmias 24,25 and even to death 26,27 have been reported after OP and/or carbamate poisonings. Other researchers have reported hypertension–hypotension alterations and direct myocardial damage. 14,28 –30
Although a significant amount of information exists on the short term, single-dose effects of OPs and carbamates from numerous cases of acute poisonings in humans, the effects of prolonged, low-level exposure on human cardiac functions are still unclear. The aim of our study, therefore, was to investigate the long-term effects of repeated low-level exposure to diazinon, propoxur, and CPF on cardiac function in healthy New Zealand white female rabbits.
Methods
Animals
Eight healthy New Zealand white female rabbits (weighing between 3200 and 3500 g each) were housed in individual metal cages and kept on a 12-hour dark/light cycle, at a temperature of between 20 and 23°C, in the animal facilities of the University Hospital of Heraklion, Crete. They were fed with commercial rabbit pellets ad libitum (Bio Zo. El Spyridakis, Margarites Milopotamou, Rethimno, Crete) and provided with drinking (tap) water. As confirmed by daily inspection, animals were consuming all water made available to them (250 ml of tap water per day). The rabbits were acclimatized under laboratory conditions for 2 weeks, whereupon the treatment period began.
The animals were divided into 4 groups consisting of 2 animals each. The limited number of animals per group constitutes the main limitation of the study. 11 Group 1 received diazinon at 2 different doses, 2.6 (low-dose diazinon, LDD) and 5.2 mg/kg/day (high-dose diazinon, HDD). Group 2 received propoxur at 2 different doses, 8.8 (low-dose propoxur, LDP) and 18 mg/kg/day (high-dose propoxur, HDP) respectively. Group 3 received CPF at 2 different doses, 8.7 (low-dose CPF, LD CHLO) and 18 mg kg/day (high-dose CPF, HD CHLO). The animal in the LD CHLO group did not survive due to pathophysiological conditions independent of CPF administration. This rabbit suffered from pasteurellosis and it developed subcutaneous abscesses, which were observed during necrotomy, as the animal during its life did not develop any clinical symptoms. Group 4 served as the control group (C) and received only tap water. The administered dose of diazinon, propoxur and CPF corresponded to 1/30 and 1/15 of the respective LD50 for LDD and HDD treatment groups, 31 in order to avoid any overt toxicity. Originally, the appropriate amounts of pesticides were diluted in 500 ml tap water. The yearly based experimental scheme of exposure, selected in continuation of previous studies and conducted in order to mimic seasonal human exposure to pesticides 11 consisted of two oral administration periods, lasting 3 months and 1 month, respectively. During the administration period, the pesticides were administered daily by gavage for 7 days per week. The administration period was interrupted by an 8-month washout period (total duration of 12 months). All rabbits, at the age of 15 months (weight 3.6–4.2 kg) and after the end of the second administration period, underwent an echocardiographic evaluation under sedation. The animals were then terminated by an injection of 5 ml pentothal (thiopental sodium solution, 25 mg/ml, by vetoquinol), according to the bioethical rules of the University of Crete. During the study period, the animals were weighed every month and their feed consumption was recorded daily. All rabbits were regularly observed (on a weekly basis) and their condition was closely monitored. The LD CHLO animal suffered from pasteurellosis with abscesses in the lungs and adhesions to the chest wall as observed during autopsy. No pathological/clinical signs were observed in the other animals at any point. At the end of both administration periods, urine and serum samples were collected. Following termination, heart tissue samples were collected and stored at −20°C.
Ethics
This study was carried out in strict accordance with the recommendations in the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health. The protocol was approved by the Committee of Ethics of the Medical School of the University of Crete. All efforts were taken to minimize suffering.
Determination of propoxur, diazinon, and CPF in cardiac tissues at the end of administration period
Rabbit cardiac tissue samples were collected and stored at −20°C. Samples were thawed and 250 mg of tissue was placed in 0.5 ml of nanopure water and homogenized using a Heidolph RZR2000 DIG (USA) homogenizer. For the extraction of pesticides from the homogenized tissue, 2 ml of methanol (LC-MS grade) were added and after vigorous mechanical shaking, the supernatant fraction was isolated and the final volume was reduced to 0.5 ml under a nitrogen steam. Samples from animals that received propoxur and diazinon were analyzed using a liquid chromatography–mass spectrometry (LCMS-2010 EV Shimadzu-Kyoto, Japan) using an atmospheric-pressure chemical ionization (APCI) source for the ionization of the analytes in positive mode. For chromatographic analysis, a Discovery 250 mm column (Sigma-Aldrich, USA) was used. Gradients of water (A) and methanol (B) of LC-MS grade were selected as mobile phases at a flow rate of 0.5 ml/min for the detection of propoxur (m/z 210.0, 211.0 for propoxur and 201.0 for carbaryl as the internal standard). Gradients of formic acid 10 mM in methanol (A) and water (B) of LC-MS grade were selected as the mobile phases at 0.65 ml/min for diazinon (m/z 305.1 for diazinon and 302.2 for methidathion as the internal standard). For the evaluation of the extraction procedure, blank tissue samples were spiked at concentration levels ranging from 0.1 to 10 μg/kg. Recoveries were calculated to be 80.1% and 82.3% for propoxur and diazinon, respectively. All standard and spiked solutions tested exhibited good linearity (r 2 > 0.99). The samples from animals that received CPF were analyzed by gas chromatography–mass spectrometry (GC-MS) as previously described. 32
Oxidative stress
Total antioxidant activity (TAC) expressed in mmol diphenyl-1-picrylhydrazyl (DPPH)/L reduced to DPPH:H was determined by the DPPH spectrophotometric assay using stable DPPH radical as the reagent. The plasma was mixed with phosphate-buffered saline (PBS) and DPPH, then incubated in the dark for 60 min, and centrifuged for 3 min at 20,000g at 25°C. The absorbance was read at 520 nm. Thiobarbituric acid-reactive species (TBARS), expressed in μmol/L, were measured in blood plasma using a previously described method. 24 Briefly, plasma was mixed with trichloroacetic acid (TCA), Tris–hydrochloric acid (Tris-HCl), sodium sulfate (Na2SO4), and thiobarbituric acid and incubated at 95°C. TCA was added again, centrifuged (speed/time), and the absorbance was read at 530 nm. Catalase, expressed in U/mgHb, was determined in red blood cell lysates. Protein carbonyls, expressed in nmol/mg protein, were determined in plasma, as previously reported. 33
Echocardiographic protocol
The echocardiography protocol used has been previously described. 34 Following the subcutaneous administration of ketamine (17 mg/kg) and xylazine (7 mg/kg), the sedated rabbits, moved to the animal-keeping lab special exam room, having their anterior chest and upper abdomen hair removed, were placed in the supine position and evaluated using a VIVID3 Expert, GE Systemand (USA) a 10-MHz phased array cardiac ultrasound probe. Echocardiography was performed in all cases; images of appropriate quality were available allowing appropriate measurements. Echocardiographic views included left ventricular (LV) long and short axes and four-chamber views. M-Mode, 2D imaging, pulsed-wave (PW) Doppler and tissue Doppler imaging (TDI) recorded video loops and still frames were digitally stored and transferred as raw data to an external server while a designated software (Echo-Pac PC, GE-Healthcare, USA) allowed for off-line measurements on a personal computer. Measurements were performed by a single observer with certified expertise in echocardiography.
M-Mode was used for the documentation of radial LV dimensions: intraventricular septum (IVS), LV cavity dimension (LVD), posterior wall, both in diastole (d) and systole (s). From these measurements further M-Mode-derived variables included LV fractional shortening (FS) and ejection fraction (EF-M-Mode), stroke volume (SV), and cardiac output (CO) (absolute values and indexed to the body weight) as measures of LV contractility and systolic radial performance. The myocardial mass (absolute and indexed) was also estimated using LV end-diastolic dimensions.
The availability of raw data information allowed for the placement of a virtual M-Mode line on the stored 2D four-chamber views (anatomic M-Mode), perfectly aligned with the axis of longitudinal displacement of the lateral mitral and tricuspid valve annuli. 35 The maximum longitudinal displacement of the basal lateral segments toward the heart apex was documented as mitral annulus peak systolic excursion (MAPSE) and tricuspid valve annulus peak systolic excursion (TAPSE), as a measure of systolic longitudinal myocardial performance.
PW Doppler was used for documentation of the mitral valve (MV) early (E) and late (A) peak diastolic flow velocities and for the estimation of the E/A ratio, as measures of LV diastolic performance. PW myocardial TDI was used for documentation of peak systolic (s), early (e) and late (a) peak myocardial velocities of basal lateral LV wall segments as measures of LV longitudinal function. For each measured variable, the average value of three measurements corresponding to consecutive cardiac cycles was documented.
Histopathological lesions
Myocardial tissue block samples, embedded in paraffin and sectioned at 4 μm, were stained with eosin–hematoxylin and were subsequently examined under light microscopy by a histopathologist without knowing the source of the tissues.
Telomerase activity
The telomerase activity in cardiac tissue samples and peripheral blood monocytes (PBMCs) was measured using a commercial telomerase polymerase chain reaction–enzyme linked immune sorbent assay (PCR-ELISA) (Roche Diagnostics Corp., Indianapolis, IN, USA), based on the telomeric repeat amplification protocol. 36 The method for the isolation of PBMCs is described elsewhere. 37 PBMC isolation with density gravity centrifugation from diazinon-treated animals did not result in enough DNA to proceed with the PCR elongation/amplification step of the commercial telomerase activity kit due to technical errors.
OxiSelect oxidative DNA damage quantitation kit (apurinic/apyrimidinic—AP or abasic sites)
Genomic DNA was isolated from heart using the Quiagen FlexiGene DNA kit (Qiagen, Germany). Oxidative DNA damage was determined in the isolated DNA using the OxiSelect Oxidative DNA Damage Quantitation kit (Cell Biolabs, INC, San Diego CA, USA) (AP sites), which uses an aldehyde reactive probe (ARP) to react specifically with an aldehyde group on the open ring form of AP sites. In this way, AP sites are tagged with biotin, which is later detected with a streptavidin–enzyme conjugate. The quantity of AP sites in the DNA sample (expressed as the number of AP sites per 100,000 base pairs) is determined by comparing its absorbance with a standard curve generated from the provided DNA standard containing predetermined AP sites.
Results
Diazinon, propoxur, and CPF levels in cardiac tissues
The significant persistence of various amounts of both propoxur and diazinon in cardiac tissues was detected, suggesting that the cardiac muscle cells were directly exposed to both pesticides. However, diazinon was preferentially retained in the cardiac tissue demonstrating amounts equal to 0.121–0.159% of the original dose. In contrast, propoxur was more readily metabolized and excreted, since only 0.0029–0.033% of the original dose was retained in the cardiac tissue. CPF was not detected in the cardiac tissue.
Morphological and functional cardiac parameters monitored by echocardiographic monitoring after exposure to diazinon, propoxur, and CPF
Due to the presence of a single animal in each group, nonparametric tests (Mann–Whitney U test) failed to demonstrate any significant differences among the pesticide groups and the control and the pesticide subgroups, based on either the pesticide type or the level of dosage. However, the borderline differences (p < 0.1) along with the visual inspection of the bar charts suggested a trend for a higher heart rate, impaired longitudinal systolic myocardial function (expressed as lower MAPSE and TDI peak systolic velocities), impaired diastolic function (expressed as the lower E/A mitral inflow velocity ratio), impaired cardiac index, as well as reduced myocardial mass in the pesticide-treated animals compared to controls. These differences were more pronounced in the diazinon-treated animals. Animals exposed at higher cumulative doses of pesticides (for each pesticide) also showed similar trends (Figure 1(a)—(d)).

Echocardiographic indices of impaired LV systolic and diastolic performances in the pesticide group compared to the control group. Reduction in the peak myocardial systolic velocity at the lateral basal LV wall as assessed by TDI (a). Reduction in the longitudinal systolic function as assessed by mitral annulus peak systolic excursion (MAPSE) (b), impaired diastolic function as expressed by lower E/A MV inflow velocity ratios (c) and reduction in indexed myocardial mass (d). Values are the average of three measurements of each variable. TDI: tissue Doppler imaging; MAPSE: mitral annulus peak systolic excursion; MV: mitral valve; LV: left ventricular.
Diazinon-, propoxur-, and CPF-mediated histopathological alterations of the cardiac muscle tissue
The main histopathologic findings were fibrosis, hemorrhagic infiltration of myocardial tissues and degeneration of muscle cells, with no signs of inflammation (Figure 2). The myocardial tissues from HDD-exposed animals showed areas with hemorrhagic infiltration and foci of fibrosis, while the myocardial tissues obtained from HDP-treated animals showed only areas of fibrosis. These findings are consistent with the observed higher excretion rate of propoxur from the cardiac tissues, which resulted in lower continuous exposure of the cardiac muscle cells, when compared to the diazinon treatment. For CPF -exposed animals neither hemorrhagic impregnation nor signs of inflammation were observed in the cardiac tissue.
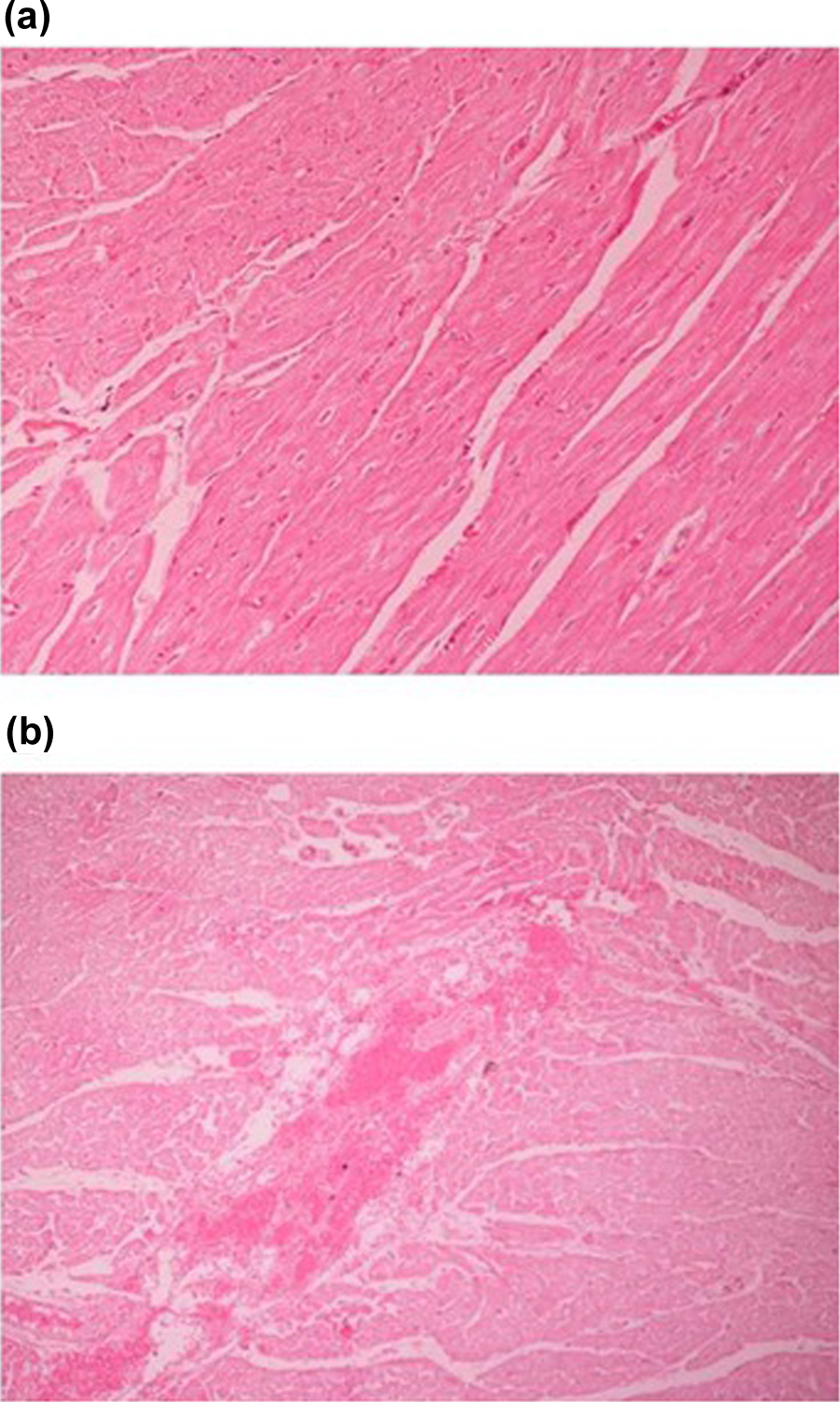
Representative histological sections (HE). (a) Normal myocardial tissue (HE × 100). (b) Myocardial tissue with hemorrhagic infiltration (HE × 100). HE: Hematoxylin Eosin.
Localized and systemic oxidative stress parameters were increased upon exposure to diazinon, propoxur, and CPF
Levels of systemic oxidative stress parameters are presented in Figure 3 (for diazinon and propoxur, data have previously been published). 38 All the tested pesticides induced oxidative stress. In particular, diazinon at both doses increased TBARS levels by 20 and 70%, respectively. Propoxur treatment increased TBARS levels by about 50% at both doses, while CPF induced a 28% increase in TBARS levels compared to controls (Figure 3(a)). Regarding protein carbonyls, the markers of protein oxidation, diazinon and propoxur, did not affect their levels, while CPF exposure caused a 50% increase (Figure 3(b)). Diazinon and propoxur had no effect on the catalase activity at low concentrations, but they decreased the enzyme activity at high concentrations by 20 and 25%, respectively. Similarly, CPF decreased the catalase activity by 39% (Figure 3(c)). The effects of diazinon and propoxur on TAC levels were intriguing, since both pesticides at low concentrations increased TAC levels but had no effect at high concentrations. On the contrary, CPF decreased TAC levels by 55% (Figure 3(d)).

Parameters related to oxidative stress in rabbit serum and red blood cell lysate.
A prominent effect of the oxidative environment on the cell function is the creation and accumulation of oxidative modifications in the DNA. These modifications, if they are not repaired, accumulate and could lead to impairment of cell function and cell death. To investigate if diazinon, propoxur, and CPF can induce oxidative DNA modifications, the genomic DNA fraction of the cardiac muscle tissues was analyzed (Figure 4). All the pesticides tested induced significant amounts of modified genomic DNA occurrence. No dose-dependent mode of action was evident for CPF, which seemed to be more DNA toxic. Propoxur (high dose 1.30 ± 0.02; low dose 0.57 ± 0.05) and diazinon (high dose 1.16 ± 0.01; low dose 0.18 ± 0.07) worked in a dose-dependent manner.

Quantification of the oxidative modifications produced in the genomic DNA in the cardiac muscle tissue after diazinon, propoxur, and CPF exposures. Values are the average of triplicate measurements and the error bars represent the standard deviation. CPF: chlorpyrifos.
Telomerase activity in PMBCs and cardiac tissues
Telomerase activity in PBMCs increased nonsignificantly in a dose-dependent manner for propoxur-treated animals compared to controls (Figure 5). The relative telomerase activity in high doses of propoxur reached 129%, while low doses of both propoxur and CPF did not exceed a 40% increase in circulating telomerase activity (38.6% and 32.5%, respectively). Telomerase activity measurements in the cardiac muscle tissues demonstrated no significant changes after exposure to the three pesticides studied, although an increase is observed for all pesticides tested, varying from 43 to 56%.

The effect of diazinon, propoxur, and CPF on the telomerase activity in cardiac tissues (a) and PMBCs (b). CPF: chlorpyrifos; PBMCs: peripheral blood mononuclear cells.
Discussion
The aim of the present study was to assess the long-term effects on heart function of a repeated transient low-level exposure to diazinon, propoxur, and CPF, simulating the actual situation faced yearly by the agricultural workers. Several echocardiographic techniques and indices were used for the assessment of systolic, diastolic, and global myocardial performances of rabbits exposed to the pesticides studied, some of them being conventional (M-, PW doppler), 39,40 others being cutting edge including TDI, Myocardial Performance Index (MPI), 41,42 and MAPSE assessments by anatomic M-Mode rarely applied in animal models. 35 Compared to control animals, the diazinon-/propoxur-treated groups were characterized by thin-walled left ventricles with reduced myocardial mass and reduced systolic and diastolic performances. The most pronounced negative effects on the myocardial size and function were observed in the diazinon group, while higher doses of both pesticides were generally associated with further impairment of the LV myocardial performance. CPF administration had a similar detrimental effect to rabbit myocardial mass and function. These findings are in accordance with the findings of Cetin et al. who observed that CPF-treated rabbits exhibited diminished CO and reduced LV EFs. 43 The observed effect was evident in the dose of approximately 28.12 mg/kg/body weight per day, while the dose of 18.75 mg/kg/body weight per day that is similar to the dose of our study did not produce equal effects. Furthermore, the augmented heart rate of pesticide-treated animals is a very interesting finding due to the knowledge that organophosphorus insecticides elicit toxicity via acetylcholinesterase inhibition, allowing acetylcholine accumulation and excessive stimulation of the cholinergic receptor. 19 The augmented heart rate could be attributed to the compromised heart function and is probably a sign of severity of heart dysfunction. Since pesticide administration was performed on rabbits of a relatively young age, not having reached their maximum myocardial mass, our findings might represent a mild myocardial cardiotoxicity combined with a probable inhibition of normal myocardial heart growth. This is in agreement with echocardiographic findings of late cardiotoxicity following anthracycline administration in young children. 44 However, further echocardiographic studies evaluating cardiotoxic effects of pesticides at various time intervals following exposure as well as the specific impact of their administration on growing hearts are needed. 45
Previous studies have reported that OPs can produce typical signs of toxicity, such as fasciculation of skeletal muscles associated with muscle fiber damage in mammals. 46,47 Cardiotoxicity of diazinon has previously been reported using a rat model, where it was shown that the myocardiotoxic activity of diazinon caused a degenerative and inflammatory reaction on the heart. 48 Furthermore, Abdou and ElMazoud reported widespread disorders in the rat myocardial structure accompanied by subendocardial necrosis with capillary dilation in the heart muscles of diazinon-treated rats, which is similar to our findings in the rabbit model. In the present study, fibrosis was detected in the cardiac muscle tissue by both diazinon and propoxur exposure. Furthermore, the histopathologic analysis detected hemorrhage after the high-dose treatment, which is an indication of an ongoing tissue damage response. In order to determine whether the involvement of these two pesticides could be related to this ongoing damage, cardiac tissues were analyzed for pesticide residues. Traces, mainly of diazinon but also of propoxur, were observed in heart tissues after the completion of the exposure scheme. Diazinon persisted longer in the cardiac muscle tissues when compared to propoxur, thereby potentially accounting for its hemorrhage-inducing properties. 49
It is well established that the heart is susceptible to free radical damage, due to its intrinsic elevated oxidative metabolic activity and its fragile antioxidant resistance, in comparison to other parts of the body. The induction of oxidative stress by diazinon is well documented. 50 Indeed, diazinon and propoxur increased lipid peroxidation in plasma, as shown by the elevated TBARS levels, and decreased the activity of catalase, one of the most important antioxidant enzymes, while protein carbonyls were not influenced. Interestingly enough, TAC levels increased at low doses, while they remained unchanged at high doses. The explanation for this apparent contradiction is that sometimes under tolerable oxidative stress conditions the organism increases the production of antioxidant molecules as a compensatory mechanism against free radicals. 51,52 The pro-oxidant activity of CPF was higher compared to the other two pesticides. CPF increased lipid peroxidation and protein oxidation as shown by the increased TBARS and protein carbonyl levels, while it decreased the catalase activity. Moreover, unlike the other two pesticides, CPF was the only one that decreased TAC levels, indicating that the induced oxidative stress was too high to be outbalanced by antioxidant mechanisms.
A recent study 53 has reported that the diazinon cardiotoxicity can be caused by the oxidation of myocardial cell membranes and internal lipid structures. Another report has documented that the enhanced production of ROS was correlated with the OP-induced toxicities. 54 It is widely known that ROS promote the peroxidation of lipids, thus causing significant damage to membrane structures. Consecutively, it is proposed that the heart tissue may be susceptible to oxidative damage due to the presence of polyunsaturated fatty acids (PUFAs) and oxygen, which may produce oxidative changes in myocytes. 55 Our study has identified another possible model of cardiac cell destruction, namely the introduction and accumulation of oxidative DNA modifications in the genomic DNA content. These modifications are caused by the oxidative environment and are routinely repaired by a cellular prophylactic mechanism. 56 However, under extreme conditions the repair mechanism can be overrun and the modifications accumulated, possibly causing cell death. 57
Telomeres are indicators of oxidative stress. Under increased oxidative stress, telomere shortening is accelerated. 58 The increased telomerase activity in PBMCs observed in the present study depicts systemic inflammation, 59,60 in agreement with the histopathological findings recorded. The nonsignificant increase in telomerase activity found in the heart, following exposure to propoxur, diazinon, and CPF, which corresponds to an extension of the life span of the cells, could possibly represent a counteracting survival mechanism. 61,62 Such a protective function has already been shown for telomerase, which is excluded from the nucleus under oxidative stress and is localized in the mitochondria in order to protect them from stress. 63
Summarizing, in the present study a mild cardiotoxic activity was observed by echocardiography for all pesticides tested. Severe histological alterations were identified especially to the diazinon-treated animals in agreement with its increased persistence in the cardiac tissues. Furthermore, our data provided possible evidence of the cardiotoxicity mechanism, according to which due to the oxidative environment, cardiac muscle cells accumulate oxidative DNA modifications resulting in cardiac tissue damage and potentially cell death.
Footnotes
Acknowledgments
No external funding was received for this study.
Authors’ note
Alexandros Zafiropoulos, Konstantinos Tsarouhas, Christina Tsitsimpikou and Ioannis Germanakis have contributed equally.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
