Abstract
Hydroxyapatite nanoparticles (HAP NPs) are widely used for preparations of biomedical and biotechnological fields such as drug delivery, gene therapy, and molecular imaging. However, the current toxicological knowledge about HAP NPs is relatively limited. The present study was designed to investigate the toxicity potentials of various concentrations (0–1000 µg cm−2) of HAP NPs in cultured primary rat hepatocytes. Cell viability was detected by 3-(4,5-dimethyl-thiazol-2-yl) 2,5-diphenyltetrazolium bromide (MTT) assay and lactate dehydrogenase (LDH) release, while total antioxidant capacity (TAC) and total oxidative stress (TOS) levels were determined to evaluate the oxidative injury. The DNA damage was also analyzed via scoring liver micronuclei rates and determining 8-oxo-2-deoxyguanosine (8-OH-dG) levels. The results of MTT and LDH assays showed that the higher concentrations of dispersed HAP NPs (300, 500, and 1000 µg cm−2) decreased cell viability. Also, HAP NPs increased TOS (500 and 1000 µg cm−2) levels and decreased TAC (300, 500, and 1000 µg cm−2) levels in cultured hepatocytes. On the basis of increasing doses, the NPs as depending on dose caused significant increases of the number of micronucleated hepatocytes and 8-OH-dG levels as compared to control culture. Furthermore, the highest concentration of HAP NPs (1000 µg cm−2) exhibited cytotoxic activity. Based on these results, HAP NPs have a dose-dependent toxic effect in rat hepatocytes. Further extensive research in this field is promising and reasonable.
Introduction
With the rapid development of nanotechnology, more and more nanomaterials had been fabricated and manipulated to perform particular functions, such as adhesive, cosmetics, drug delivery, and artificial organ and tissue. 1 However, in a recent report, it was suggested that before these novel materials could be safely applied in a clinical setting and/or any other areas, their toxicity should be carefully assessed. 2 The discussion about safety concerns associated with small particles is ongoing for many decades and is to a large extent related to potential risks following oral, parenteral, or dermal exposure. 3 Especially, ultrafine or nanoparticles are in the focus of the debate, for example, 4 usually meant to have one or more size dimensions between 0.1 nm and 100 nm.
In the last 20 years, mechanisms of micro-sized natural and man-made mutagens’ toxicity and protective effects of therapeutic substances were studied extensively. 5 –7 But the toxic effects of nano-sized particles were not fully detailed except for some titanium, tungsten, cadmium, and zinc oxide nanoparticles. 8,9 However, the so-recent report indicated that there was a lack of systematic assessment of the DNA damaging and carcinogenic potential of NPs in spite of their extensive use in nanotechnological applications. 10
Hydroxyapatite nanoparticles (HAP NPs) are one of the most important members of ultrafine materials, which have been found extensive applications in biomedical and biotechnological fields, such as drug carrier, gene therapy, and molecular imaging. 11 In general, engineered HAP materials and their composites are intended for a variety of biomedical applications, often serving as bone substitute in intraosseous implantation or as implant coating materials, including small particle sizes down to the nano range. 12,13 Thus, possible health impact of HAP NPs upon introduction into the body is of great interest. With the increased applications of HAP NPs, the concerns about their potential human toxicity and their environmental impact were also increased. Cytotoxicity, inflammation, and increased oxidative stress through reactive oxygen species (ROS) formation are prominently discussed to be relevant factors regarding the safety of small particles down to the nano-range. 14,15 Cellular oxidative stress is a fundamental mechanism whereby toxic particles like quartz exert their pathogenicity following inhalation. 16,17 It has been reported that different sizes and morphologies of particles, including HAP NPs, have the potential to influence the interaction with many kind of biomolecules including cytochrome, hemoglobin, and DNA. 18 –21 Moreover, liver is a target site for NP toxicity, because of NP accumulation after ingestion, inhalation, or absorbsion. 22 However, our knowledge about hepatotoxicity of HAP NP in humans is not profound.
In the present study, specific measurements were aimed to perform in cultured rat primary hepatocytes as in vitro model system for assessing the impact of HAP NPs on human health. Therefore, cytotoxic effects were measured by using two colorimetric functional assays, mitochondrial dehydrogenase (MTT) activity, and lactate dehydrogenase (LDH) activity assay. In fact, assessing mitochondrial activity gives a fair indication of cell health. And, LDH activity is commonly used as an indicator of cell membrane integrity. In addition to these cytotoxicity assays, total antioxidant capacity (TAC) and total oxidative stress (TOS) levels were determined to evaluate the oxidative injury. At the same time, genotoxicity was assessed via scoring liver micronucleus (LMN) formations and determining 8-hydroxy-2′-deoxyguanosine (8-OH-dG) levels.
Materials and methods
Synthesis and characterization of HAP NPs
In the synthesis, analytical grade chemicals were selected. Anhydrous calcium nitrate (Ca(NO3)2·4H2O; assay: 99–102%, impurity: ≤ 0.0005% heavy metals (as lead), ≤ 0.005%; Merck KGaA, Germany) and ammonium dihydrogen phosphate (NH4H2PO4(; assay: ≥98% trace metals basis, impurities: 0.0005% heavy metals (as lead) ≤ 0.005% water-insoluble matter; Carlo Erba, Italy) were used as starting calcium and phosphorus precursors, respectively. Ammonia (extra pure; Merck KGaA) was used for adjusting the pH of the solution. Ca(NO3)2·4H2O (1.00 M) aqueous solution (pH≈10) was added dropwise to 0.60 M NH4H2PO4 solution (Ca/P = 1.67) under vigorous stirring at 60°C. A white precipitate solution was obtained and this was followed by aging the solution for 12 h at 60°C. The gel obtained after the aging process was filtered and washed repeatedly using double-distilled water and then acetone (CH3COCH3; assay: ≥99.8%; Merck KGaA) to avoid agglomeration. The filter cake obtained by this process was then dried at 80°C overnight in a dry oven and the dried powder was calcined at 750°C for 4 h, using an electrical furnace, employing a heating rate of 10°C min−1 in air atmosphere.
The phase composition and crystallinity of the calcined HAP NPs were analyzed by X-ray diffraction (XRD) using a Bruker AXS, D8 Advance diffractometer (Billerica, Massachusetts, United States) with copper K α radiation λ = 0.15406 nm at 40 kV and 30 mA. Data were collected over the 2θ range 20–60° at a scan rate of 0.05° min−1. Phase analysis was done using Powder diffraction file (PDF) card no. 009–0432 for HAP. The Raman analysis was performed with a Renisaw InVia Raman System using a 532 nm laser beam and a Leika microscope (Turkey) (objective of 50 times). The morphology studies were performed with a transmission electron microscope (TEM; JEM 2100 HRTEM, Jeol, Japan) and scanning electron microscope (SEM; JSM-6335F FEGSEM, Jeol).
Animals
Male rats of Sprague–Dawley strain (Medical Experimental Research Center, Atatürk University, Erzurum, Turkey) of 200–300 g body weight were used throughout the present study. They were provided water and standard laboratory chow ad libitum and were maintained under standard light, temperature, and relative humidity conditions. The local ethical committee approved the study protocol. All experiments were performed in accordance with the Guide for the Care and Use of Laboratory Animals
Hepatocyte isolation and cell cultures
Rats were killed by carbon dioxide (CO2) overdose and the livers were removed immediately. Isolated hepatocytes from rats were prepared by the collagenase perfusion technique. 23 The liver was perfused through the hepatic portal vein with calcium-free Hanks balanced salt solution to remove blood for about 10 min at a flow rate of 2.5 mL min−1. As soon as the liver became grayish brown in color, a second buffer solution containing collagenase (Hank’s balanced salt supplemented with 4 mM calcium chloride and 0.5 mg collagenase/mL) was perfused at the same rate until the liver appeared to have broken up. After treatment, the liver was minced into 3–4 mm pieces with a sterile scalpel. Following mechanical dissociation, the cells were filtered through gauze and centrifuged at 1350 r min−1 for 5 min. Then, the hepatocytes were collected in medium containing bovine serum albumin and bovine insulin. The cell suspension was filtered through gauze again and allowed to sediment for 20 min to eliminate cell debris, blood, and sinusoidal cells. The cells were then washed three times by centrifugation at 50 g, tested by Trypan blue dye exclusion for viability (always in the range of 82–93%). The hepatocytes were then suspended in a mixture of 75% Eagle’s minimum essential medium and 25% medium 199, supplemented with 10% fetal calf serum containing streptomycin, penicillin, bovine insulin, bovine serum albumin, and sodium bicarbonate (2.2 mg). For the experimental procedure, hepatocytes were plated in multi-well tissue culture plates (3 × 105 cells in a well area of 3.8 cm2; 8 × 105 cells in a well area of 9.6 cm2). The medium was changed 4 h later. The effect of HAP NPs was studied after 72 h of exposure in cultures maintained with a medium deprived of fetal calf serum but supplemented with hydrocortisone hemisuccinate (7 × 10−7 M). Hepatocytes were cultured for an additional 8 h before treatment.
Treatments
After 8 h of plating, when primary hepatocytes got adhered and attained their epithelial morphology, culture media was aspirated and replaced with an equal volume of the media supplemented with different concentrations of dispersed aqueous HAP NPs (5, 10, 20, 50, 75, 100, 150, 300, 500, and 1000 µg cm−2) followed by incubation in CO2 incubator for 72 h (n = 6). Mitomycin C (MMC; at 10−7 M; Sigma®, St Louis, Missouri, USA) alone added group was considered as a positive control (control+).
MTT assay
Viability of cells was assessed by measuring the formation of a formazan from MTT spectrophotometric test. At the end of the experiment, the hepatocytes were incubated with 0.7 mg mL−1 MTT for 4 h at 37°C. After washing with phosphate-buffered saline the blue formazan was extracted from cells with isopropanol/formic acid (95:5) and was photometrically determined at 560 nm. 24,25
LDH assay
LDH activity was measured in the culture medium as an index of cytotoxicity, employing an LDH kit (Cayman Chemical Company, Ann Arbor, Michigan, USA). Enzyme activity was expressed as the extracellular LDH activity percentage of the total activity on the plates. 26,27
TAC and TOS assays
The automated Trolox equivalent TAC and total oxidative stress (TOS) assays were carried out in the culture medium by commercially available kits (Rel Assay Diagnostics®, Gaziantep, Turkey).
LMN assay
LMN assay was performed using the method of Suzuki et al. 28 Immediately prior to evaluation, 10–20 μL of hepatocyte suspension was mixed with an equal volume of acridine orange (AO)–4′,6-diamidino-2-phenylindole dihydrochloride (DAPI) stain solution (AO, 0.5 mg mL−1; DAPI, 10 μg mL−1) for fluorescent staining. Approximately 10–20 μL of the mixture was dropped onto a glass slide and covered with a cover glass. Samples of well-isolated hepatocytes were evaluated with the aid of a fluorescence microscope counting the number of micronucleated hepatocytes (MNHEPs) in 2000 hepatocytes for each animal. MNHEPs were defined as hepatocytes with round or distinct micronuclei that stained like the nucleus, with a diameter 1/4 or less than that of the nucleus and confirmed by focusing up and down, taking into account hepatocyte thickness by one observer.
Nucleic acid oxidation
DNA oxidation was determined by measuring the amount of 8-OH-dG adducts. DNA was digested by incubation with DNAase I, endonuclease, and alkaline phosphatase. 29 The amount of 8-OH-dG was measured by high-performance liquid chromatography with electrochemical detection as described previously. 30
Statistical analysis
The experimental data were analyzed using one-way analysis of variance and Fischer’s least significant difference tests to determine whether any treatment significantly differed from the controls or each other’s. Results presented as mean ± SE values and the level of 0.05 were regarded as statistically significant.
Results
Synthesis and characterization of HAP NPs
The XRD pattern of the HAP nanocrystals calcined at 750°C is shown in Figure 1. It is revealed that all major peaks of standard HAP were present in the synthesized powder.

The XRD pattern of synthesized hydroxyapatite sintered at 750°C. XRD: X-ray diffraction.
Raman spectrum of sintered HAP particles is shown in Figure 2. The strong peak observed at 962 cm−1 is assigned to υ1 phosphate [PO4]3− symmetric stretching mode of the phosphate group and it is the characteristic Raman peak of HAP. Anti-symmetric stretching mode (υ3) of [PO4]3− was observed 1045 cm−1. The weak peaks observed at 427 cm−1 (υ2) and 585 cm−1 (υ4) are assigned to split, doubly degenerate and split, triply degenerate bending vibrations of [PO4]3−, respectively.

Raman spectrum of hydroxyapatite nanoparticles.
The morphologies of the as-dried and calcined HAP particles are shown in Figure 3 and Figure 4, respectively. The as-dried particles showed nano-sized elongated morphology with 30 nm average particle width and 100 nm average particle lengths. Calcination caused not only agglomeration of particles but also their swelling and twisting, as can be seen in SEM and TEM micrographs.

Secondary electron SEM images of HAP NPs before and after sintering. SEM: scanning electron microscopic; HAP NP: hydroxyapatite nanoparticle.
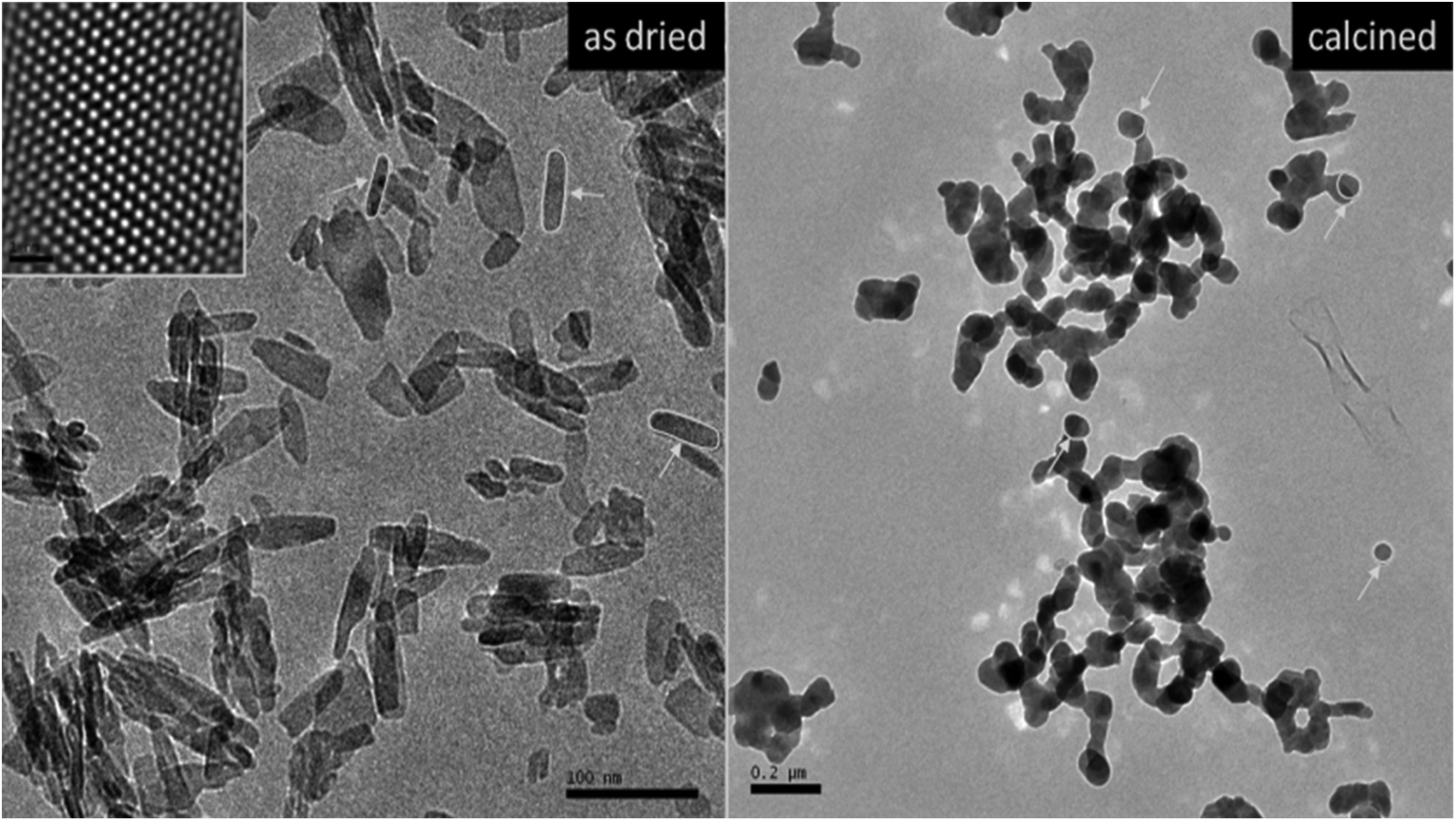
TEM micrographs of as dried and calcined HAP NPs. TEM: transmission electron microscopy; HAP NPs: hydroxyapatite nanoparticles.
Cytotoxicity evaluation
The results of cell viability measured by MTT assay are shown in Figure 5. When assayed in vitro on the hepatocyte cells using the MTT assay, the value for the MMC (10−7 M)-treated cells (as control+) was approximately 2.4-fold lower than that for the control−. Likewise the higher concentrations of HAP NPs (300, 500, and 1000 µg cm−2) caused significant (p < 0.05) decreases of the cell viability. However, the hepatocyte cells exposed to lower doses than 300 µg cm−2 of HAP NPs did not show any significant change in cell viability during 72 h as determined by MTT assay. And no cytotoxicity was observed for control− cells. MMC-induced hepatic damage was clearly evidenced by six-fold increases in the activity of LDH compared with the observations of the negative controls (Figure 6). Although LDH was not affected by low doses of HAP NPs alone but the increases of the levels of enzyme reached statistical significances at 300, 500, and 1000 µg cm−2.
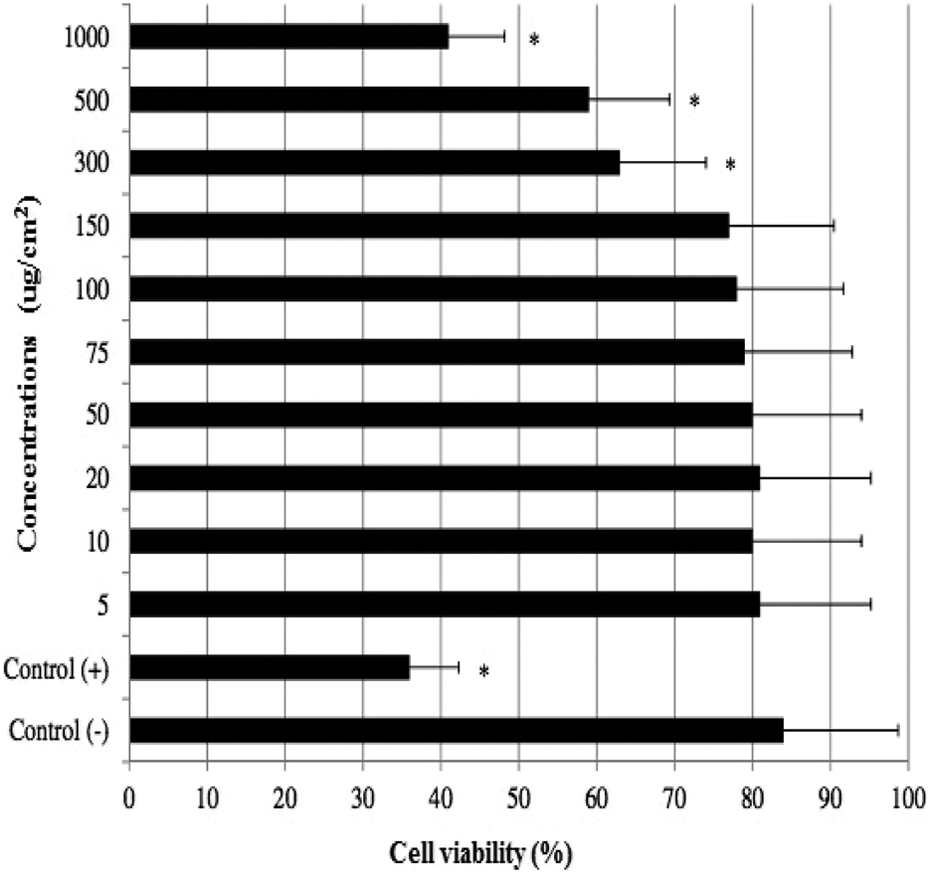
MTT reduction in cultured rat hepatocytes maintained for 72 h in the presence of different HAP NP concentrations. Each individual hepatocyte culture without HAP NPs was studied as a negative control group (control−). MMC alone-added group was considered as a positive control (control+). Values are means ± standard error (n = 6). *p < 0.05: significant differences from the control− group; MTT: 3-(4,5-dimethyl-thiazol-2-yl) 2,5-diphenyltetrazolium bromide; HAP NP: hydroxyapatite nanoparticle; MMC: mitomycin C.
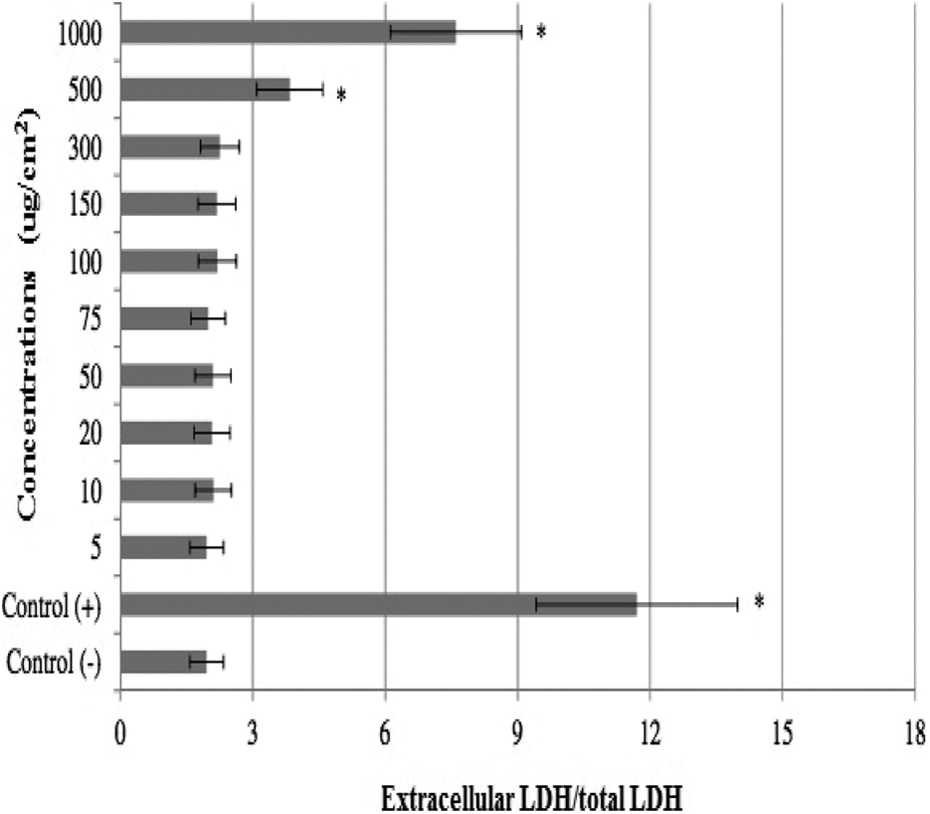
Extracellular level of LDH in cultured rat hepatocytes maintained for 72 h in the presence of different HAP NP concentrations. LDH: lactate dehydrogenase; HAP NP: hydroxyapatite nanoparticle.
DNA damage analyses
The levels of 8-OH-dG in cultured rat hepatocytes of controls and experimental groups are shown in Figure 7. Firstly, the levels of 8-OH-dG, a sensitive marker of oxidative DNA damage, were quantified with regard to MMC treatment. It was observed that MMC significantly increased 8-OH-dG concentrations in the hepatocyte cultures after 72 h. Likewise 8-OH-dG levels also increased in the hepatocyte cells that were treated with 300, 500, and 1000 µg cm−2 of HAP NPs.

8-OH-dG adducts in cultured rat hepatocytes maintained for 72 h in the presence of different HAP NP concentrations. 8-OH-dG: 8-oxo-2-deoxyguanosine; HAP NP: hydroxyapatite nanoparticle.
The results of the observed LMN rates in primary rat hepatocyte cells after 72 h HAP NP treatment are presented in Figure 8. LMN (at 150, 300, and 500 µg cm−2) analyses showed statistically significant differences (p < 0.05) between control− and HAP NP applied cultures. But the highest concentration of HAP NPs (1000 µg cm−2) caused sterility of the cultures due to its cytotoxic activity.

Results of liver MN assay in cultured rat hepatocytes maintained for 72 h in the presence of HAP NPs. MN: micronucleus; HAP NPs: hydroxyapatite nanoparticles; HEP: hepatocyte; MNHEPs: number of micronucleated hepatocytes.
Oxidative effects
Table 1 shows the effects of HAP NPs on oxidant status in cultured rat hepatocytes that were determined by TAC and TOS analysis. As shown in Table 1, the TAC value decreased with the addition of MMC while TOS value increased. In contrast, the treatments with 5, 10, 20, 50, 75, and 100 µg cm−2 of HAP NPs did not alter both TAC and TOS levels. However, nanomaterials’ applications at higher doses changed the TAC (150, 300, 500, and 1000 µg cm−2) and TOS (300, 500, and 1000 µg cm−2) levels. Thus, HAP NPs had dose-dependent effects on oxidative damage in hepatocytes in vitro.
Extracellular TAC and TOS levels in cultured rat hepatocytes maintained with HAP NPs for 72 h.

TAC: total antioxidant capacity; TOS: total oxidative stress; HAP NPs: hydroxyapatite nanoparticles; H2O2: hydrogen peroxide.
aSignificant differences at the p < 0.05 level from the control− group.
Discussion
We aimed to assess the cytotoxicity, genotoxicity, and oxidative damage in cultured primary rat hepatocyte cells in response to different concentrations of HAP NPs. Trends in cytotoxicity as observed for the different concentrations of HAP NPs were overall highly similar using the two independent assays (MTT and LDH) in cultured rat hepatocytes. LDH and MTT assays were performed to measure the cytotoxicity at different points in the death pathway. Because MTT assay quantifies mitochondrial activity by measuring the formation of a dark blue formazan product formed by the reduction of the tetrazolium ring of MTT. The reduction in MTT is considered to mainly occur in the mitochondria through the action of succinate dehydrogenase, therefore providing a measure of mitochondrial function. LDH is a soluble cytosolic enzyme that is released into the culture medium following loss of membrane integrity resulting from either apoptosis or necrosis. LDH activity, therefore, is used as an indicator of cell membrane integrity and serves as a general means to assess cell viability by measuring plasma membrane permeability. A particular contrast was observed for the highest concentrations (300, 500, and 1000 µg cm−2) of HAP NPs that induced pronounced LDH release and decrease of MTT. Potential effects on cytotoxicity as observed with HAP NPs have been shown. 31 Rothen-Rutishauser et al. 32 reported that the size of nanoparticles or their aggregates can be considered a potential determinant for uptake and subsequent macrophage responses, which could explain for the observed differences on LDH, release at the highest concentration of HAP NPs. 33 Since particles in the fine size range may induce more pronounced responses than in the nano-range, 32 the ‘ultrafine hypothesis’ has been challenged previously by experimental studies. 34 Albrecht et al. 33 also found that cytotoxicity as observed at high concentrations did not necessarily represent a compound-related effect and may at least partly be due to physical coverage of the cells by the test with HAP NPs. On the contrary, the recent obtained results of the MTT assay indicated that HAP NPs did not induce cytotoxicity up to 800 μg mL−1 in mouse bone marrow mesenchymal stem cells. 35
In primary rat hepatocyte cultures, no cytotoxic effects were observed with any of the lowest concentrations (< 300 µg cm−2) of HAP NPs in the MTT and LDH assays. Therefore, the survival of hepatocytes is one of the key prerequisites for the effective elimination of nanoparticles (> 1500 µg cm−2) of HAP NPs, the observed low cytotoxicity provides evidence for a good biocompatibility of the tested material. Also, the observed low toxicity toward rat hepatocyte cells exposed to low concentrations (< 150 µg cm−2) of HAP NPs in our investigations correlates with other investigations using nano-HAP and composites thereof, which made use of a wide spectrum of different specifications, test systems, and testing conditions, with some of the studies focusing on efficient biodegradation. 36,37
A complete understanding of how HAP NPs interact with hepatocytes, especially at the molecular level, is still unclear. Wang et al. 38 found cell growth inhibition and apoptosis in MC3T3-E1 cells cocultured with HAP NPs. Our findings demonstrated that direct exposure of hepatocytes to HAP NPs induced the intracellular ROS generation, DNA damage, 8-OH-dG adducts, reduced the TAC, and subsequently caused dysfunction of these cells. In accordance with present findings, Motskin et al. 39 reported that HAP NPs eluded the phagocytic pathway and entered the nuclei through nuclear pores. Mechanisms involved in the reduction of cell viability through oxidative stress by HAP NPs are yet to be determined in detail. Recently, it was recorded that in vitro results showed that HAP NPs had dose-dependent effects on inducing oxidative damage, genotoxicity, and cytotoxicity in human blood cells. 21 Oxidative stress is associated with protein and lipid oxidation, ultimately leading to a profound alteration in mitochondrial function. 40 Any change in the mitochondrial membrane permeability is known to be an early event in apoptosis. 41 At this point, Xu et al. 42 suggested that a mitochondrial-dependent pathway mediated the nano-HAP-induced apoptotic pathway. It is known that ROS activates multiple signaling pathways, including mitogen-activated protein kinase (MAPK) family and p53 expression signal transduction cascades. 43 P53, Bax, and Bcl-2 are also generally thought to be involved in mitochondria-dependent apoptosis. 41 p-JNK, p-c-Jun, p-p53, Bax, and cleaved caspase-3 were significantly increased in human umbilical vein endothelial cells (HUVECs) after exposure to silica nanoparticles, while Bcl-2 was dramatically suppressed. ROS scavenger could markedly inhibit c-Jun-N-terminal kinase (JNK), c-Jun, and p53 activity, indicating that ROS may be upstream effectors of JNK and p53. 41 Also, Hsin et al. 44 found that nano silver-induced apoptosis was mediated by ROS via JNK and p53 activation. ROS could also directly activate p53, most probably by the induction of DNA damage. 6,43,45 Based on the results of Liu and Sun, 41 the following silica nanoparticle-mediated signaling pathways for apoptosis-related events were proposed as ROS production (JNK/c-Jun phosphorylation), P53 activation, Bax upregulation and Bcl-2 downregulation.
Studies of Oesterling et al. 46 , Schanen et al. 47 and Liu and Sun 41 showed that higher dose exposure to nanomaterials including silica nanoparticles, titanium dioxide, zinc oxide, and alumina nanoparticles led to pro-inflammatory and procoagulant responses in endothelial cells. It is an accepted viewpoint that ROS are involved in many of the processes underlying endothelial activation (e.g., the upregulation of adhesion molecules and chemokines and increased expression of tissue factor). Many of the key signal transduction molecules involved in endothelial activation, such as various MAPKs and the transcription factors nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB), are known to be redox sensitive. 48 To assess the biological effect of different concentrations of HAP NPs on rat hepatocyte cells, cell viability was determined. In the present study, MTT and LDH assays showed that the higher concentrations of HAP (300, 500, and 1000 µg cm−2) decreased cell viability.
Liu and Sun 41 found that exposure to high concentrations (100–200 µg cm−2) of silica nanoparticles caused an increase in cytotoxicity in HUVECs. The LDH release was also increased by silica nanoparticles only at the highest concentration, indicating that exposure to high concentrations of silica nanoparticles can affect cell membrane integrity and lead to cell death. They also reported induction of apoptosis or necrosis by silica nanoparticles and oxidative stress as silica nanoparticles induced toxicity mechanism. Liu and Sun 41 showed that intracellular ROS generation of HUVECs by silica nanoparticles was gradually increased in a time- and dose-dependent manner, suggesting that oxidative stress occurred not at once, but continuously during cell culture. ROS played a central role in silica nanoparticle-mediated apoptosis. Disturbance of membrane integrity has been recently suggested as possibly one of the mechanisms for cytotoxic action in nanoparticles. 49 However, there are some contradictory findings on cytotoxicity by HAP NPs. The results of the MTT assay indicated that HAP NPs do not induce cytotoxicity up to 800 μg mL−1 in bone marrow mesenchymal stem cells. 35 Again, Geetha et al. 50 found that there was no cytotoxicity (up to 600 μg mL−1) and oxidative damage (up to 100 μg mL−1), when exposed to HAP NPs in Guinea pigs. These controversial results could be explained via differences in tested cell or tissue types. In fact, the particle effects with the concentration of HAP NPs on cytotoxicity, oxygen species generation, production of inflammatory cytokines (tumor necrosis factor α and interleukin (IL) 6), particle–cell association, and cellular uptake were evaluated on BEAS-2B and RAW264.7 cells and was revealed that these NPs induced cell-specific cytotoxicity and IL-6 expression. 51
Data from the current study showed that exposure to HAP NPs at high concentrations caused ROS generation and decreased TAC in cultured primary rat hepatocytes. Similar to our finding, the results of the study by Chen et al. 52 showed that exposure to HAP NPs at a concentration above 200 µg mL−1 induced hepatic oxidative stress with elevated levels of hydrogen peroxide and malondialdehyde. Likewise, it was determined that HAP NP exposure caused oxidative stress, leading to expression of antioxidant enzymes, such as catalase, superoxide dismutase (SOD), and heme oxygenase-1, via a Src family kinase (SFK)-JNK and p38-NF-κB signaling pathway in human lung epithelial A549 cells. 53 The measurement of SOD generation also showed that HAP NPs decreased the total SOD of cellular levels in C6 glioma cells. 54 Again, in an in vivo study, a dose-dependent decrease in the activity of glutathione peroxidase was observed in dermally HAP NP exposed rats. 55
In summary, HAP NPs caused decreased cell viability, which was detected by increased MTT and LDH release. Exposure to HAP NPs at high concentrations also caused genotoxicity and cytotoxicity due to oxidative stress in hepatocytes. HAP NPs, at the highest concentration (1000 µg cm−2), caused sterility of the cultures due to its cytotoxic activity. It also increased DNA damage by significant increases in micronuclei (LMNs) and 8-OH-dG. Moreover, our overall findings suggested that exposure to HAP NPs possibly has cytotoxic and genotoxic effects and oxidative damage potentials on rat hepatocyte cells in a clear dose-dependent manner. So, these excessive concentrations and their toxicological potentials must be seriously taken into consideration when HAP NPs are used for biomedical applications.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
