Abstract
Nitric oxide produced by inducible nitric oxide synthase (iNOS) regulates sepsis-induced hypotension. During septic shock, interleukin (IL)-1β is synthesized in endothelial cells and smooth muscle cells by endotoxin. Ethanol (EtOH) suppresses endotoxin-induced hypotension. The present study aimed to elucidate the effect of EtOH on gradual relaxation and iNOS expression induced by IL-1β in isolated rat superior mesenteric arteries (SMAs). Exposure to IL-1β–induced contraction in SMA rings, followed by a gradual relaxation of phenylephrine precontracted tone. Contraction was abolished by indomethacin (IM), cycloheximide (Chx), and endothelium denudation. In contrast, the gradual relaxation was abolished by NOS inhibitors, Chx, endothelium denudation, and inhibited by EtOH (50 and 100 mM). However, IM had no effect on relaxation. Western blot analysis demonstrated that iNOS expression was induced by IL-1β and was inhibited by EtOH and endothelium denudation. Furthermore, messenger RNA expression of iNOS, but not endothelial NOS, was inhibited by EtOH. These data suggest that IL-1β–induced contraction is mediated by thromboxane A2, whereas IL-1β–induced relaxation occurs via NO derived from iNOS. The endothelium plays an important role in vasorelaxation. Taken together, EtOH inhibits IL-1β–mediated vasorelaxation by suppressing endothelium iNOS expression. This study provides the first evidence of EtOH -induced inhibition of IL-1β–mediated vasorelaxation.
Introduction
Bacterial sepsis is a systemic inflammatory state characterized by vascular smooth muscle dysfunction, leading to hypotension, inadequate tissue perfusion, and multiple organ failure. 1 Inducible nitric oxide synthase (iNOS)-derived nitric oxide (NO) plays a key role in sepsis. 2 Patients with sepsis have elevated levels of interleukin (IL)-1β, IL-6, and tumor necrosis factor α (TNF-α). 2 Endotoxemia-associated vascular iNOS expression is induced by a variety of cytokines, including IL-1β and TNF-α, and lipopolysaccharide (LPS), resulting in a high NO production. 3
LPS and IL-1β activate the expression of iNOS 4,5 in the rat superior mesenteric artery (SMA) and abdominal aorta. Increased iNOS-mediated NO production suppresses phenylephrine (Ph)-induced vascular contractile responses, resulting in decreased arterial pressure. 3,6 Thus, in endotoxemia, the impaired contractile function of the smooth muscle and the resulting decreased blood pressure have been attributed to increased NO-mediated dilatation secondary to iNOS activity.
IL-1β is a cytokine produced by monocytes and macrophages that contributes to disease pathogenesis. It is an important mediator of host responses to infection and injury. 3,7 Prolonged exposure of blood vessels to IL-1β decreases vascular tone and causes systemic vasodilation. IL-1β can produce a shock-like syndrome, characterized by hypotension and decreased vascular resistance when administered to animals. 8,9 Interestingly, ethanol (EtOH) is a recognized immunosuppressant. For example, chronic abuse of EtOH increases susceptibility to bacterial infections and pneumonia. 10,11 Previous studies have shown that acute in vitro administration of EtOH inhibits LPS-mediated induction of iNOS gene expression and release of NO from Kupffer cell, alveolar macrophages, and glial cells in experimental animals. 12 –19 Therefore, the increased sensitivity to bacterial infection associated with EtOH seems to be due to EtOH-induced suppression of NO production. On the other hand, Ajisaka et al. 20 reported that acute low-dose EtOH administration also inhibits endotoxin-induced hypotension and inflammatory cytokine responses in intravenously treated rats. They suggested that acute EtOH administration may play a protective role in endotoxin-induced shock. Furthermore, Greenberg et al. 21 reported that EtOH suppresses endotoxin-induced hypotension in rats administered with LPS intravenously. Greenberg et al. suggested that EtOH may increase morbidity and mortality during endotoxemia by masking the hypotension and humoral changes characteristic to early endotoxemia. Thus, the explanation for the inhibitory effect of EtOH on endotoxin-induced hypotension is a matter of debate. To obtain a better understanding of the inhibitory effect of EtOH, we aimed to elucidate the mechanism underlying the EtOH-induced suppression of endotoxin-mediated hypotension. We evaluated the effect of EtOH on vascular reactivity induced by IL-1β in vitro using isolated rat SMAs.
Methods
Tissue preparation
All procedures were approved by the Committee on Ethics in Animal Experiments of Nara Medical University and were conducted in conformity with institutional guidelines. Male Wistar rats (10–12 weeks old, 330–350 g) were anesthetized by intraperitoneal pentobarbital injection (0.05 g kg−1) and killed by exsanguination. SMAs were removed, cut into 1 mm segments, and mounted horizontally on tension hooks with a 50 μm diameter in 4 mL tissue baths containing Krebs–Ringer solution (pH 7.4). Baths were maintained at 37°C and aerated with a mixture of 95% oxygen and 5% carbon dioxide.
Drug application
IL-1β, Ph, and EtOH were obtained from Wako Pure Chemical (Osaka, Japan). Cycloheximide (Chx), 1400W, indomethacin (IM), nitro-
Measurement of tension
Isometric tension was monitored using a force–displacement transducer (Nihon Kohden Kohgyo Co., Tokyo, Japan). One side of the tension hook was connected and recorded with a pen recorder. The rings were equilibrated for approximately 1 h, replacing the medium every 15 min, and were adjusted for a resting tension of 0.2 g. Arteries were then precontracted with 10–6 M Ph. At peak precontraction, 20 ng mL−1 IL-1β was added to the organ baths. Thereafter, tissue tension was monitored for 3 h. To investigate the role of iNOS in IL-1β–induced relaxation, the rings were pretreated with Chx, IM, the iNOS inhibitors AMT and 1400W,
To examine the role of the endothelium in IL-1β–induced relaxation, endothelium was denuded by gently rubbing the intimal surface with stainless wire. Removal of the endothelium was confirmed by the loss of the relaxation in response to 10–6 M acetylcholine. Relaxation was expressed as the percent contraction in response to 10–6 M Ph 3 h after the addition of IL-1β.
Preparation of isolated SMA for Western blotting and RT-PCR analysis
Rat SMAs were isolated and washed with Krebs–Ringer solution. The SMA specimens were divided into the following three groups: control, IL-1β, and IL-1β and EtOH. In the control group, SMAs were incubated with Krebs–Ringer solution for 3 h. In the IL-1β group, SMAs were incubated with IL-1β for 3 h. In the IL-1β and EtOH group, specimens were incubated with EtOH 30 min before incubation with IL-1β and, thereafter, EtOH was co-incubated with IL-1β for 3 h.
Western blotting
Several specimens from each group were homogenized and centrifuged, and the supernatant was resolved by sodium dodecyl sulfate polyacrylamide gel electrophoresis. Samples were transferred onto a polyvinylidene fluoride membrane, which was blocked and incubated with a primary antibody against iNOS (ab15323, Abcam, Cambridge, UK). Membranes were washed with Tris-saline buffer containing 0.05% Tween-20 and incubated with a secondary antibody Rabbit immunoglobulin G horse radish peroxidase (170-6515, Bio-Rad Laboratories Inc., Hercules, California, USA). We used an antibody against actin (A2103, Sigma-Aldrich) as a loading control. After an additional wash, the signal was visualized by enhanced chemiluminescence using an LAS-1000 image analyzer (Fuji Photo Film Co. Ltd., Tokyo, Japan). The band intensity was calculated using Image J software (National Institutes of Health, Bethesda, Maryland, USA).
RNA isolation and quality control
Total RNA was extracted from cell samples using the RNeasy Mini kit (Qiagen, Valencia, California, USA) according to the manufacturer’s instructions and treated with DNase I to eliminate contamination from genomic DNA. RNA samples were diluted in Tris-ethylene diamine tetraacetic acid buffer, and the concentration was measured at a wavelength of 260 nm with a UV-1600 spectrophotometer (Shimadzu, Kyoto, Japan). Real-time reverse transcriptase polymerase chain reaction (RT-PCR) analyses were performed using samples with a 260/280 nm absorbance ratio that was between 1.9 and 2.1.
Gene expression analysis
Total RNA (50 ng) was reverse transcribed using the Superscript First Stand system (Life Technologies, Carlsbad, California, USA) with random hexamers, as described in the manufacturer’s protocol. To assess endothelial NOS (eNOS) and iNOS expression, quantitative RT-PCR analysis was performed using the StepOne real-time PCR system (Applied Biosystems, Foster City, California, USA), with a universal PCR master mix according to the manufacturer’s specifications. The expression of the following genes was examined using TaqMan probes and primers: eNOS (NOS3, assay ID; Rn02132634_s1) and iNOS (NOS2, assay ID; Rn00561646_m1). The expression of hypoxanthine phosphoribosyl transferase (Hprt1, assay ID; Rn01527840_m1) was used as an endogenous control. For quantification of iNOS gene expression, complementary DNAs derived from pooled IL-1β–treated lungs were used to generate standard reference curves. For quantification of eNOS expression, we employed the comparative cycle threshold (ΔΔCt) method, because adequate reference samples for the standard curve could not be found. Data were expressed relative to the value of untreated SMA.
Statistical analysis
Data are presented as the mean ± SEM, and were analyzed using a one-way analysis of variance and Dunnett’s post hoc test. The values of p < 0.05 were considered statistically significant.
Results
IL-1β–induced relaxation in SMAs
Ph produced sustained contraction in endothelium-intact or -denuded SMA (data not shown) over a 3 h period (Figure 1). In contrast, IL-1β induced a peak contraction approximately 30 min after IL-1β. After 1.5 h, it recovered to levels similar to those at the time of addition (Figure 1). Gradual relaxation followed the observed contraction. IM, an inhibitor of cyclooxygenase, abolished the contractile response seen after the addition of IL-1β, but did not inhibit the gradual relaxation (Figures 1 and 2). Chx, an inhibitor of protein synthesis, almost completely abolished both the contractile and relaxation response (Figures 1 and 2). Similarly, SMA denudation inhibited both contractile and relaxation responses to IL-1β (Figure 1). Therefore, the following experiments were conducted in the presence of IM to exclude the effect of the contractile response on relaxation.
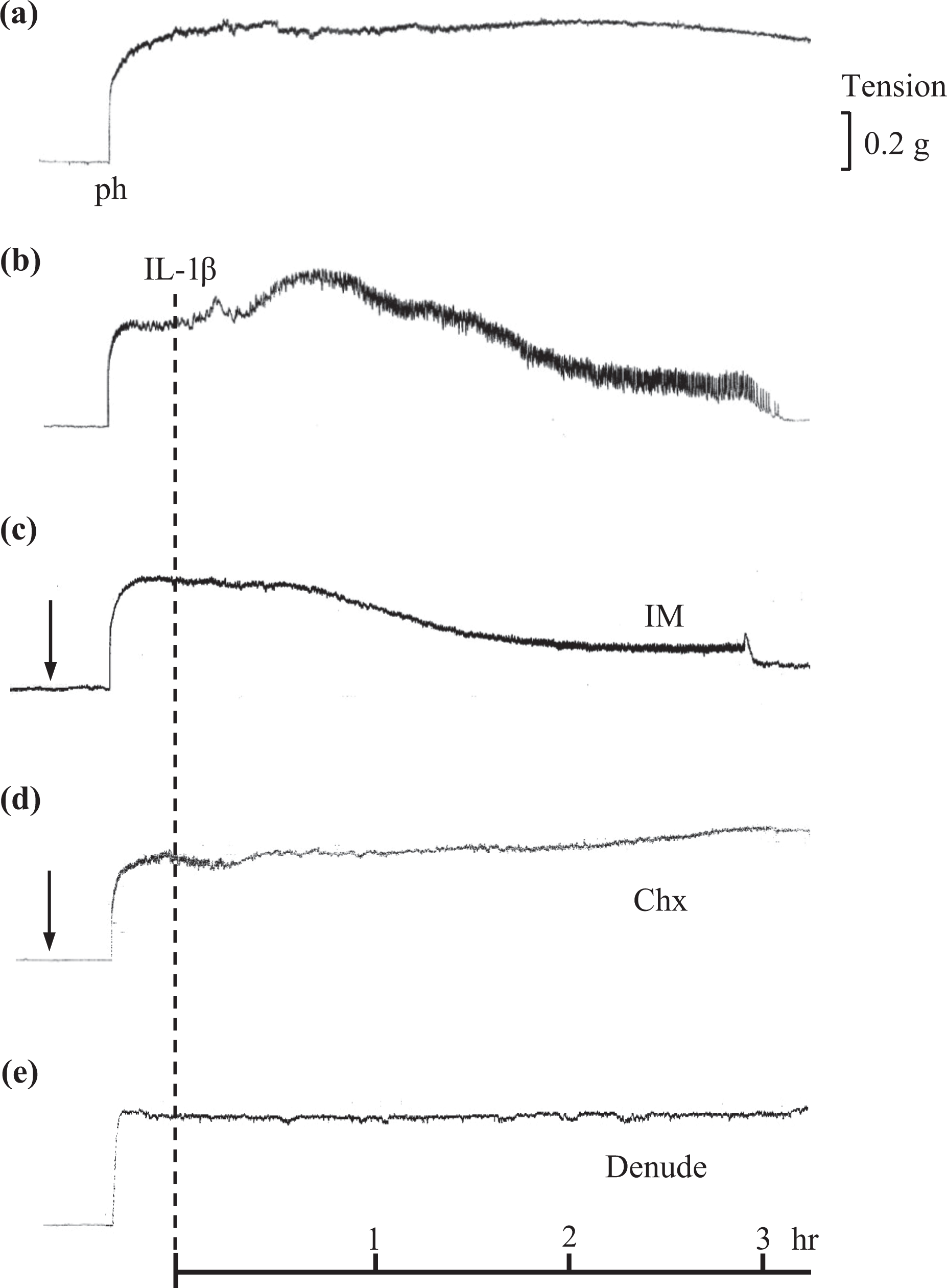
Representative traces showing the effect of inhibitors and endothelium denudation on IL-1β–induced relaxation of Ph precontracted rat SMAs. (a) Time control trace showing a sustained contractile response to Ph over a 3 h period. (b) A contractile response followed by a gradual relaxation response induced by IL-1β. (c) Absence of IL-1β–induced contractile response in the presence of IM. (d) Absence of IL-1β–induced relaxation in the presence of Chx and (e) Absence of IL-1β–induced contractile and relaxation responses in endothelium-denuded SMAs. The arrow and dashed line indicate when IM and Chx, and IL-1β were added, respectively. SMA: superior mesenteric arteries; Ph: phenylephrine; IL: interleukin; IM: indomethacin; Chx: cycloheximide.

Summary data showing the effects of various inhibitors and endothelium denudation on IL-1β–induced relaxation. Results represent the mean ± SEM and are expressed as a percent of the contraction elicited by Ph. †p < 0.01 versus control. SEM: standard error of mean; IL: interleukin.
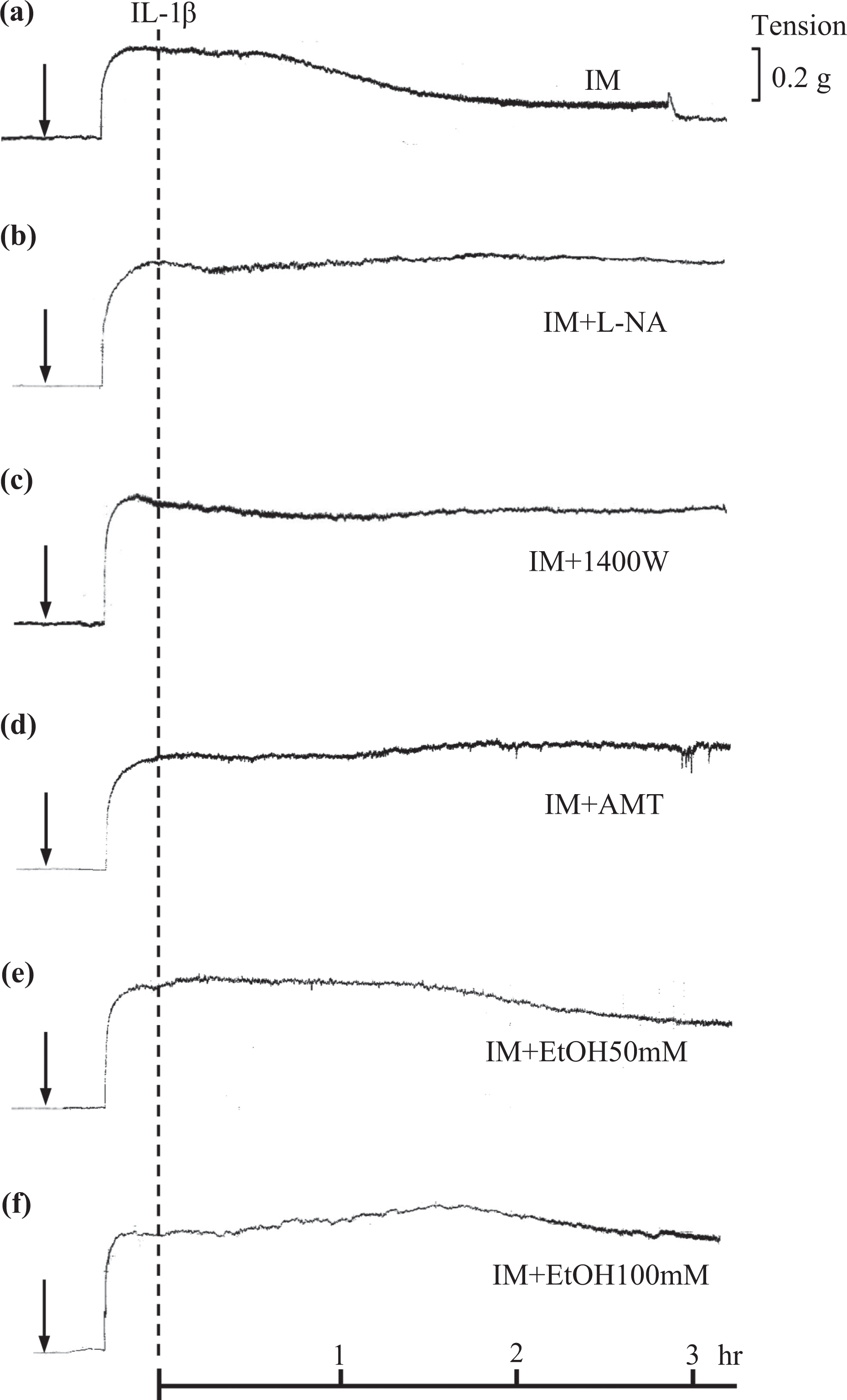
Representative traces showing the effect of inhibitors and EtOH on IL-1β–induced relaxation responses to Ph precontracted isolated rat SMAs in the presence of IM. (a) Gradual relaxation response induced by IL-1β. (b) Absence of IL-1β–induced relaxation in the presence of

Summary data showing the effects of various NO synthase inhibitors and EtOH (50 mM and 100 mM) on IL-1β–induced relaxation. Results represent the mean ± SEM and are expressed as a percent of the contraction elicited by Ph. *p < 0.01 versus IM and IL-1β. NO: nitric oxide; IL: interleukin; SEM: standard error of mean; Ph: phenylephrine; EtOH: ethanol.
Expression of iNOS protein in IL-1β–treated SMAs
When SMAs were unstimulated, iNOS was not expressed in the presence or absence of endothelium (Figures 5 and 6). However, IL-1β exposure for 3 h enhanced iNOS expression in SMAs, both in the presence and in the absence of endothelium. In SMAs without endothelium, the degree of IL-1β–induced iNOS expression was 30% compared to SMAs with endothelium (Figure 5). IL-1β–induced iNOS expression in SMAs with endothelium was inhibited by co-incubation with 50 mM or 100 mM EtOH for 3 h, respectively (Figure 6).
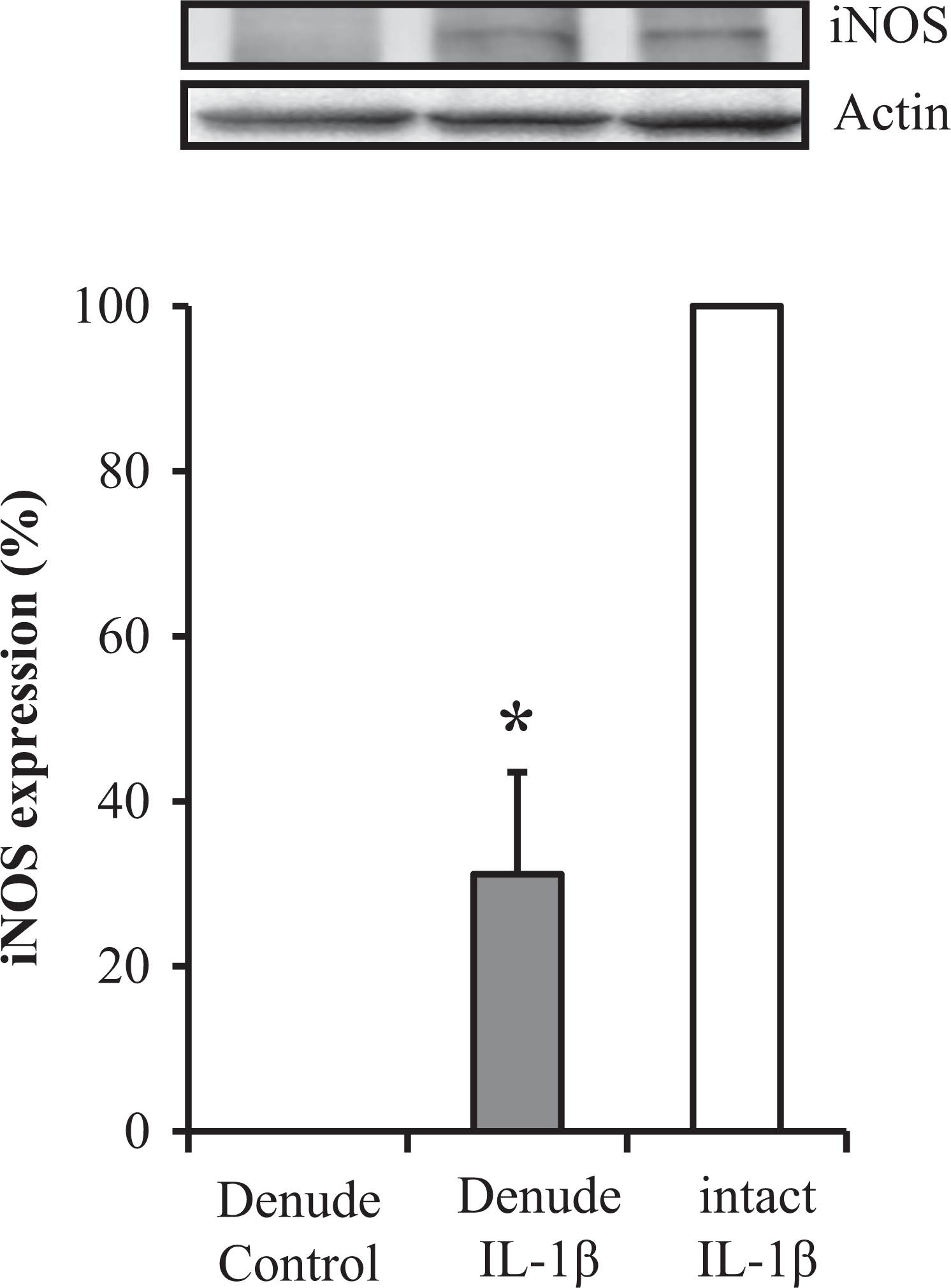
Effect of endothelium denudation on IL-1β–induced iNOS expression effect of endothelium denudation on iNOS protein expression induced by IL-1β. iNOS and actin were evaluated by Western blot analysis in IL-1β–treated SMAs with or without endothelium (upper). The relative iNOS protein expression is expressed as a percent of the maximal iNOS band (lower). *p < 0.01 versus intact IL-1β. iNOS: inducible nitric oxide synthase; IL: interleukin.
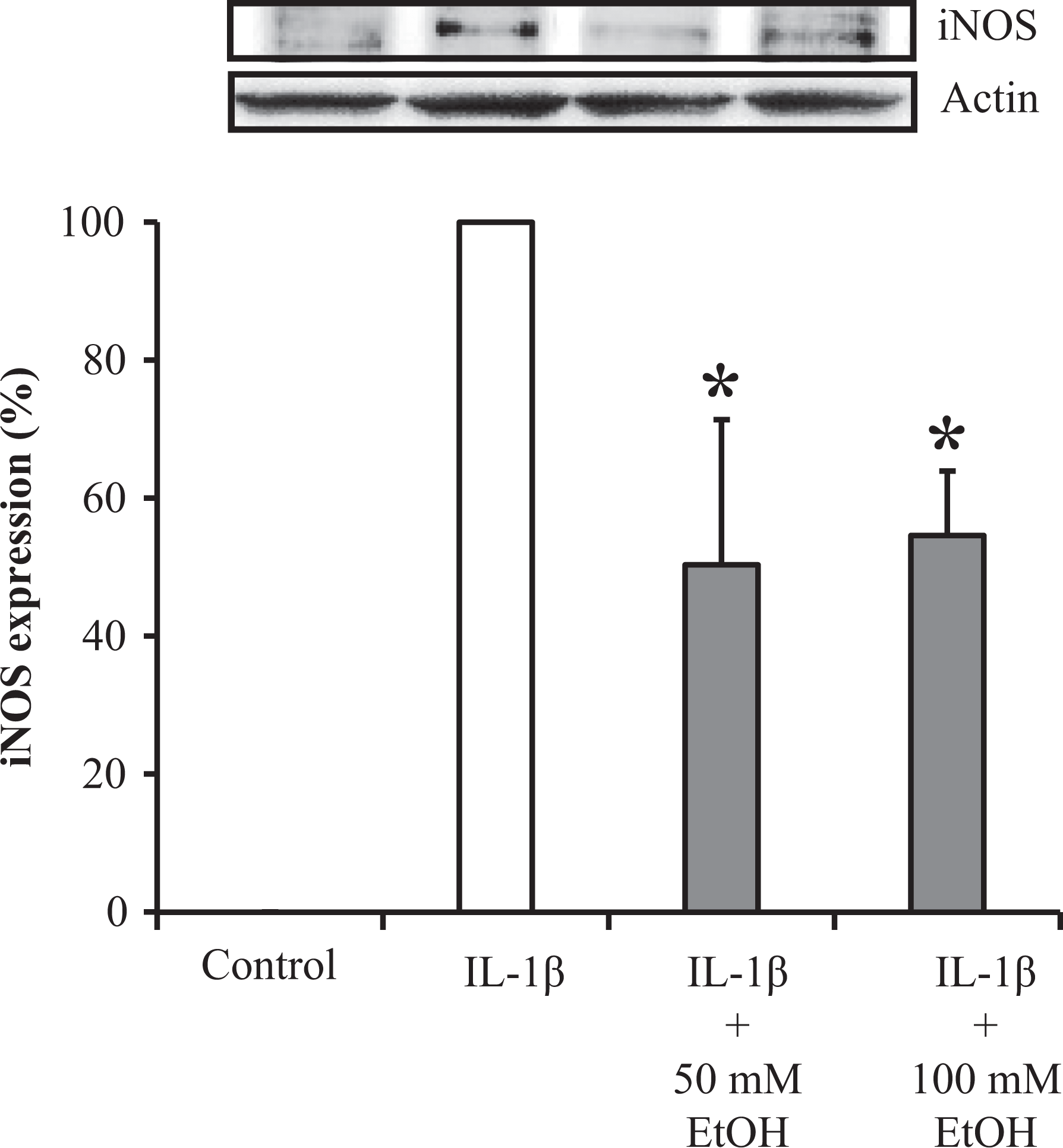
Effect of EtOH on IL-1β–induced upregulation of iNOS expression. Effect of EtOH (50 and 100 mM) on iNOS expression induced by IL-1β. iNOS and actin were analyzed by Western blot in IL-1β–treated SMAs with endothelium (upper). The relative iNOS protein expression was expressed as a percent of the maximal iNOS band (lower). *p < 0.05 versus IL-1β. EtOH: ethanol; IL: interleukin; iNOS: inducible nitric oxide synthase; SMAs: superior mesenteric arteries.
Effect of IL-1β on iNOS and eNOS messenger RNA (mRNA) expression in SMAs
Real-time PCR analysis revealed that eNOS mRNA expression was slightly, although not significantly, decreased in IL-1β–treated SMAs, as compared to untreated SMAs. However, iNOS mRNA expression was enhanced in a time-dependent manner when SMAs were expressed to IL-1β for 1 or 3 h. Moreover, pretreatment with 50 or 100 mM EtOH for 3 h suppressed IL-1β–induced iNOS mRNA expression (Figure 7).

Relative mRNA expression profile of iNOS and eNOS in SMAs. Expression levels were determined by real-time PCR and then normalized to the expression of Hprt1. (a) With regard to iNOS, data are expressed as a comparative ratio to IL-1β–treated rat lung. (b) For quantification of eNOS expression, we employed the ΔΔCt method because adequate reference samples for the standard curve could not be found. Data were expressed relative to the value of untreated SMA. *p < 0.05 versus IL-1β 3h. mRNA: messenger RNA; eNOS: endothelial nitric oxide synthase; iNOS: inducible nitric oxide synthase; SMAs: superior mesenteric arteries; PCR: polymerase chain reaction; HPRT: hypoxanthine phosphoribosyl transferase; IL: interleukin; ΔΔCt: comparative cycle threshold; N.D.: not detected.
Discussion
In the present study, we showed that IL-1β induces early contraction of SMAs, followed by a gradual relaxation. SMA contraction was abolished by IM, an inhibitor of cyclooxygenase, and Chx, an inhibitor of protein synthesis. These data suggest that the contractile response was caused by cyclooxygenase-induced thromboxane A2, which produces a contractile response. Thromboxane A2 is reported to regulate vascular tone via its effect on the expression of iNOS. 22 Therefore, our experiments were conducted in the presence of IM to exclude the effect of thromboxane A2 on relaxation.
IL-1β–induced relaxation was not inhibited by IM; therefore, relaxation was likely not caused by cyclooxygenase products, including prostacyclin, which is known to induce relaxation. In contrast, relaxation was abolished by Chx, the iNOS inhibitors, AMT and 1400W, and the nonselective NOS inhibitor,
IL-1β–induced relaxation responses were inhibited by endothelium denudation. These data suggest that the relaxation response is endothelium dependent. Lu et al. 25 reported that IL-1β can decrease Ph-induced contraction in rat aortic rings with endothelium and that the removal of the endothelium eliminated this effect. Additionally, Vo et al. 4 found eNOS inhibition in the endothelium by specific inhibitors suppresses LPS-induced iNOS expression, thereby delaying the associated vascular effects during endotoxemia. These data suggest that eNOS activation is an essential step in the induction of iNOS expression. Therefore, in the present study, the loss of IL-1β–induced relaxation in the absence of endothelium is likely due to the loss of NO derived from eNOS activation by endothelium denudation. Our data indicated that iNOS was expressed in denuded SMA, suggesting that iNOS expression in smooth muscle cells was not involved in relaxation. Nevertheless, this possibility is unlikely because eNOS mRNA expression was not increased by IL-1β exposure. Alternatively, immunofluorescence analyses in rat SMAs revealed that iNOS was expressed in both endothelial and smooth muscle cells after IL-1β treatment. 26 Thus, a more plausible explanation is that iNOS expressed in endothelial cells may be largely involved in relaxation.
In the present study, EtOH inhibited IL-1β–induced relaxation. Several mechanisms may regulate EtOH-induced suppression of relaxation. First, IL-1β–induced relaxation is regulated by iNOS-mediated NO release. NO induces relaxation via cyclic guanosine monophosphate (cGMP), through the activation of soluble guanylate cyclase (sGC) in smooth muscle cells. EtOH may impair the ability of the vasculature to vasodilate, by preventing NO-mediated sGC activation in smooth muscle cells. However, this is unlikely, because EtOH does not inhibit sodium nitroprusside-induced relaxation, which induces sGC activation. 27 Alternatively, EtOH could inhibit eNOS activity, thereby decreasing eNOS-derived NO and downregulating IL-1β–induced iNOS induction. Indeed, EtOH inhibits endothelium-dependent relaxation in response to acetylcholine, which is mediated by eNOS-derived NO. 27 However, this is also unlikely because eNOS mRNA was not inhibited by EtOH in the present study. EtOH may inhibit the upregulation of iNOS expression induced by IL-1β. The present study indicated that EtOH suppresses IL-1β–induced iNOS mRNA expression 1 and 3 h after treatment and iNOS protein expression 3 h after treatment. It is reasonable that IL-1β–induced iNOS mRNA expression 1 h after addition because relaxation was observed at 1.5 h, at that point iNOS protein was expressed. Furthermore, in porcine smooth muscle cells and lung alveolar macrophages, EtOH negatively regulates iNOS transcription and protein production. 28,29 These studies also support the possibility that EtOH inhibits iNOS protein expression. Although iNOS is expressed in both endothelial and smooth muscle cells after IL-1β treatment, 26 EtOH may have inhibitory effects on endothelial cell iNOS expression, and not smooth muscle cells, because the relaxation response was not observed in denuded SMAs. Thus, the present study shows that the endothelium plays a critical role in the in vitro inhibitory effect of EtOH on IL-1β–induced vasorelaxation. Husain et al. 30,31 reported that chronic EtOH ingestion induces hypertension in rats. Chronic EtOH ingestion is related to aortic inflammation, oxidative endothelial injury, and downregulation of the aortic endothelial NO-generating system leading to a loss of the endothelium-dependent vascular relaxation response and hypertension in rats. 30 Thus, the endothelium also plays a major role in chronic EtOH-induced hypertension. In experiments similar to those conducted in the present study, when the endothelium was intact or was not injured, EtOH may have played a protective role in endotoxin-induced hypotension. However, in experiments involving chronic EtOH ingestion, when the endothelium is injured, EtOH seems to be related to an increase in blood pressure. Further investigation is required to determine the validity of this explanation.
In conclusion, in rat SMA, EtOH was considered to inhibit IL-1β–mediated vasorelaxation probably by suppressing endothelium iNOS expression. This study provides the first evidence of EtOH-induced inhibition of IL-1β–mediated vasorelaxation.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
