Abstract
Probiotics are live microorganisms ingested for the purpose of conferring a health benefit on the host. Development of new probiotics includes the need for safety evaluations that should consider factors such as pathogenicity, infectivity, virulence factors, toxicity, and metabolic activity. Clostridium butyricum MIYAIRI 588® (CBM 588®), an anaerobic spore-forming bacterium, has been developed as a probiotic for use by humans and food animals. Safety studies of this probiotic strain have been conducted and include assessment of antimicrobial sensitivity, documentation of the lack of Clostridium toxin genes, and evaluation of CBM 588® on reproductive and developmental toxicity in a rodent model. With the exception of aminoglycosides, to which anaerobes are intrinsically resistant, CBM 588® showed sensitivity to all antibiotic classes important in human and animal therapeutics. In addition, analysis of the CBM 588® genome established the absence of genes for encoding for α, β, or ε toxins and botulin neurotoxins types A, B, E, or F. There were no deleterious reproductive and developmental effects observed in mice associated with the administration of CBM 588®. These data provide further support for the safety of CBM 588® for use as a probiotic in animals and humans.
Introduction
Probiotics are defined internationally as live microorganisms that, when administered in adequate amounts, confer a health benefit on the host. 1 The safety in use of many probiotics has been extensively studied and documented. 2 –4 The most common and well-studied probiotics include Lactobacillus spp., Bifidobacterium spp., Pediococcus spp., Bacillus spp., and Escherichia coli, along with Enterococcus spp., Streptococcus spp., and Leuconostoc spp. Probiotics have been studied in healthy individuals as well as in “at-risk” subjects, such as preterm infants, 5,6 the elderly, 7 hospitalized children, 8 and adults. 9
However, the safety of new probiotics must be established. In 2002, Food and Agriculture Organization/World Health Organization (WHO) published guidelines for the evaluation of microbes for their use as probiotics. Among the safety assessments recommended were the determination of antibiotic resistance patterns and testing for toxin production if the strain under evaluation belongs to a species known to produce toxins. 10 Antibiotic resistance, particularly in light of the growing number of resistant pathogens, has become a public health concern. Acquired antibiotic resistance (via plasmids or bacteriophages) is considered to have a high potential for the horizontal transfer of resistance genes, 11 while intrinsic resistance is presumed to possess a minimal potential for horizontal spread.
The genus Clostridium consists of a heterogeneous group of bacteria, classified by a few shared characteristics: rod-shaped vegetative forms, Gram positive, the formation of heat-stable endospores, anaerobic growth, fermentative growth with no dissimilatory sulfate reduction, and genomic DNA with a low percent of guanine and cytosine. 12 A recent study indicates that non-toxigenic Clostridium colonization may increase the percent and function of regulatory T cells in the colon and draining lymph nodes of mice. 13 However, several Clostridium species (i.e. C. botulinum, C. difficile, C. tetani, and C. perfringens) are known human and animal pathogens due to their toxin production. 14 One of the virulent pathogenic Clostridium spp. is C. perfringens, which has been isolated from soil and fecal samples of animals and humans. The major virulence factors of C. perfringens include α, β, and ε toxins. 15 In addition, the highly potent botulinum neurotoxins (BoNT), the causative agent in all cases of botulism 16 are known to be produced by many Clostridium species.
C. butyricum MIYAIRI588® (CBM 588®) was isolated from soil in Nagano, Japan, in the 1960s. CBM 588® has been widely used as a probiotic in humans and animals 17,18 in Japan. It is also currently authorized in the European Union as a feed additive for turkeys, chickens and related minor avian species, and weaned piglets and minor porcine species 19 –21 and as a novel ingredient for use in food supplement. 22 In previous studies, concentrated CBM 588® powders did not produce toxicity in rats or dogs during short-term administration 23,24 or when fed to rats, up to 5000 mg/kg in diet (241 mg/kg body weight (BW) or 288 mg/kg BW male and female, respectively), for 12 months. 25
The purpose of the studies reported in this article was to assess the safety of CBM 588® with regard to antimicrobial resistance, the presence of Clostridium toxin genes, and the effect on reproduction and development.
Methods
Bacteria and other reagents
CBM 588® was obtained from Miyarisan Pharmaceutical Co, Ltd (Tokyo, Japan). CBM 588 was originally isolated from soil, and it has been deposited in the National Institute of Technology and Evaluation, Patent Microorganism Depository (NPMD), Japan, under the Budapest Treaty as an internationally recognized depository for the microorganisms with the deposited no. FERM BP-2789. The biochemical and genetic characteristics were well identified and reported previously. 26,27 Standard culturing conditions for CBM 588 are anaerobic conditions (nitrogen 80%, carbon dioxide 10%, and hydrogen 10%) at 37°C for 24 h in brain heart infusion (BHI) broth (Oxoid, Hampshire, UK). Evaluation of minimum inhibitory concentration included incubation with various antibiotics.
ATCC strains (Bacteroides fragilis ATCC 25285, C. butyricum ATCC 19398T) were obtained directly from the American Type Culture Collection (ATCC. JCM strains (C. perfringens JCM 1290T; C. perfringens JCM 3816; C. perfringens JCM 3817; C. perfringens JCM 3818; C. perfringens JCM 3819 were obtained directly from Japan Collection of Microorganisms (JCM). KZ strains (C. perfringens KZ 210; Type A BoNT producing C. botulinum KZ 2; Type B BoNT producing C. botulinum KZ 1710; Type F BoNT producing C. botulinum KZ 47; Type E BoNT producing C. botulinum KZ 1896) were obtained through a partnership with Kanazawa University.
Antimicrobial powders were obtained from the following sources: benzyl penicillin, lincomycin, colistin, bacitracin, chlortetracycline, oxytetracycline, doxycycline, amikacin and fosfomycin from Wako Pure Chemical Industries Ltd (Osaka, Japan); ampicillin, chloramphenicol, clindamycin hydrochloride, erythromycin, gentamicin sulfate, kanamycin sulfate, metronidazole, streptomycin sulfate, tetracycline hydrochloride, vancomycin hydrochloride, carbenicillin, cefmetazole, cinoxacin, norfloxacin, ofloxacin, ciprofloxacin, lomefloxacin, cephalothin, cefazolin, and cefoperazone from Sigma-Aldrich Co. (St Louis, Missouri, USA); nalidixic acid, enoxacin, tosufloxacin, ceftizoxime, ceftazidime, minocycline, neomycin, and spiramycin from Tokyo Chemical Industry Co. Ltd (Tokyo, Japan); and imipenem and paromomycin from LKT Laboratories, Inc. (St Paul, Minnesota, USA). Antimicrobial powders were diluted as recommended and prepared fresh for each experiment.
Mice
ICR mice (CLEA Japan, Inc., Tokyo, Japan) were used in the reproduction/teratology studies. Ether was used for anesthesia and euthanasia for all mouse experiments.
Evaluation of MIC
The antimicrobial susceptibility tests were performed in a consistent manner using an internationally recognized and standardized method for anaerobic bacteria, the broth microdilution procedure following the Clinical and Laboratory Standard Institute (CLSI) document M11-A8. 28 Antibiotic resistance minimum inhibitory concentration (MIC) results of C. butyricum were compared with those break points for several important antimicrobials, ampicillin, chloramphenicol, clindamycin, erythromycin, gentamycin, kanamycin, metronidazole, streptomycin, tetracycline, and vancomycin. B. fragilis ATCC 25285 was used as a control strain for chloramphenicol, clindamycin, metronidazole, and imipenem. All MICs obtained were considered to be valid since control MIC results were within control ranges described by CLSI. 29
Isolation of total DNA
C. botulinum toxin genes: All bacterial strains were cultured in BHI medium (BBL™, Becton Dickinson and Company, Baltimore, Maryland, USA) prior to total DNA extraction. Briefly, all strains were cultured overnight in an anaerobic chamber. Bacterial cells were suspended in 400 µl of Tris-ethylenediaminetetraacetic acid (EDTA)-sucrose buffer (50 mM Tris-hydrochloric acid (HCl; pH 8.0), 1 mM EDTA (pH 8.0), and 6.7% sucrose) and lysed by treating with lysozyme and then with sodium dodecyl sulfate. The DNA was extracted with phenol–chloroform (1:1) following RNase treatment and precipitated with isopropanol. The extracted DNA was suspended in 20 μl of distilled water and used for polymerase chain reaction (PCR) template. The DNA solution was kept at −20°C until use.
C. perfringens α, β, and ε toxin genes: All bacterial strains were cultured in the same manner with C. botulinum strains. Harvested bacteria were resuspended in TE buffer (10 mM Tris-HCl, 1 mM EDTA (pH 8.0)) with 20 µL of 10% sodium dodecyl sulfate and 2 µL of proteinase K and incubated at 37°C for 1 h. After incubation of the reaction mixture with 66.7 µL of 5 M sodium chloride (NaCl) and 53.3 µL of cetyltrimethylammonium bromide (CTAB)-NaCl solution (10% CTAB in 0.7 M NaCl) at 65°C for 10 min, the solution was extracted with phenol–chloroform–isoamyl alcohol (25:24:1) solution, and DNA was precipitated with absolute ethanol by incubation at room temperature for 20 min. The DNA was harvested by centrifugation, washed with 70% ethanol, dried, and resuspended in 100 µL of TE buffer. RNA was removed by incubation of the solution with RNase (1 µg/µL) at 37°C for 1 h. The concentration of DNA was measured at 260 nm with a spectrophotometer (BioPhotometer; Eppendorf, Hamburg, Germany). The DNA solution was kept at −20°C until use.
Polymerase chain reaction
All PCRs were carried out using a DNA PJ480 cycler (Perkin-Elmer Cetus, Berkeley, California, USA). 16S Ribosomal RNA (rRNA) 30 gene was used as a positive control for all reactions.
C. perfringens α, β, and ε toxin genes: Detection of the C. perfringens toxin genes was performed by Multiplex PCR. Primers used were previously described by Yoo et al. 31 The reaction volumes of 50 µL contained 100 µg of template DNA, 0.25 µL of Ex Taq polymerase (5 units/µL, TaKaRa Bio Inc., Shiga, Japan), 5 µL of 10X PCR buffer (20 mM Mg2+, TaKaRa Bio Inc.), 4 µL of deoxynucleotide phosphate (dNTP) mixture (2.5 mM each) and 100 pmol of each primer. Pretreatment at 94°C for 5 min was followed by 30 amplification cycles (94°C for 1 min, 55°C for 1 min, and 72°C for 1 min). The amplified DNA was analyzed following electrophoresis on a 1.5% agarose gel.
C. botulinum toxin (BoNT) genes: Multiple PCR to detect BoNT was carried out with the methods described by Lindström et al. 32 Ex Taq Hot Start Version (TaKaRa Bio Inc.) was used in amplification. The reaction was performed using 10 ng template DNA, 0.3 µM each primer, 0.2 µM each dNTP, 1X Ex Taq buffer, and 1.25 units Ex Taq DNA polymerase. After pretreatment at 95°C, DNA underwent 27 amplification cycles (95°C for 30 s, 60°C for 30 s, and 72°C for 1 min 30 s). The amplified DNA was analyzed following electrophoresis on a 2% agarose gel.
Teratogenicity and reproduction
The teratogenicity and reproductive toxicity study was carried out in accordance with internationally accepted guidelines 33 and in accordance with good laboratory practices. The studies were conducted by the research laboratory of Miyarisan Pharmaceutical Co., Ltd with technical support from Japan’s Central Institute for Experimental Animals. All mice were acclimated to the rearing environment for 14–30 days and evaluated during the acclimation period. The housing conditions were 23 ± 2°C, 50 ± 10% humidity, 10–13 times/h air circulation and 12 h light/dark cycle (8:00 a.m. to 8:00 p.m.). Feed (CA-1, purchased from CLEA Japan Inc., Tokyo, Japan) and sterilized tap water were provided ad libitum.
A concentrated powder preparation of CBM 588® (Miyarisan Pharmaceutical Co., Ltd; lot no. 011, 2.4 × 109 colony-forming unit (CFU)/g) was used as the test substance. CBM® powder also contains pharmaceutical-grade lactose and corn starch as excipients and is manufactured under WHO good manufacturing practice (GMP) conditions. The powder was freshly suspended in water with 0.5% (w/v) carboxymethyl cellulose (CMC) salt prior to each administration.
Teratology
The teratology study was conducted with healthy 10-week-old male and 8-week-old nulliparous female ICR mice. Males (F0) and females (F0) were pair mated, and females were examined daily for evidence of a vaginal plug. Detection of the vaginal plug was determined as day 0 of pregnancy. Pregnant mice were separated, weighed, and randomly distributed into one of four feeding groups (37–38 females/feeding group).
Females in each feeding group were administered 0, 24, 240, or 4800 mg/kg BW of the CBM powder by oral gavage, daily, from days 6–15 of gestation. Total volume administered was adjusted for BW (0.15 mL/10 g BW). The mice were weighed daily, from gestational day 0, days 6–18, and then on postpartum days 0, 7, 14, and 21. Two-thirds of the pregnant F0 mice (26 females/feeding group) were anesthetized and killed on day 18 of gestation. The abdominal wall was cut along the medioventral line to expose the uterine horn. The numbers of implantations, live fetuses, dead fetuses, and absorbed fetuses were recorded. The uterine horn was removed, and the viable fetuses were examined for external abnormalities, weighed, and sexed. A gross visceral examination of the major thoracic/abdominal organs (lung, liver, spleen, kidneys, adrenal gland, testis, and ovaries) was conducted in the viable F1 fetuses, followed by fixation with 95% ethanol and alizarin red staining 34 to examine the skeletal structures.
The remaining one-third of the pregnant F0 mice (11–12 females/feeding group) were continuously reared for spontaneous delivery in order to collect data on postnatal development in live fetuses, delivery conditions, nursing behavior, implantation site counts, and reproductive performance. All F1 live mice were nursed through postpartum day 4. On postpartum day 4, any F1 pups with external abnormalities were euthanized and subjected to a gross observation of thoracic/abdominal organs. The remaining F1 pups were weighed daily and notes on general condition were taken. Examinations of pinna detachment (day 4), dorsal hair growth (day 11), tooth germ eruption (day 12), external acoustic pore (day 14), and eyelid opening (day 15) were conducted. Additionally, one male and one female were randomly selected from each litter to undergo observation for motor development. Mice not part of the reproductive study were euthanized on postpartum day 22. Gross observations were made of the same major thoracic/abdominal organs as in the F1 fetuses.
Reproduction studies
A single male and female F1 mouse from each litter were randomly selected and continuously reared for the F1 reproductive examination. Non-siblings were pair mated, and females were monitored for the presence of a vaginal plug. F1 dams were weighed, and feed consumption was recorded on days 0, 6, 15, 18, and 19. All established pregnancies were spontaneously delivered. Delivery conditions, nursing behavior, number of live births, and number of dead fetuses were examined in the F2 generation. All live F2 offspring were allowed to nurse until postpartum day 7 at which point all F2 pups and remaining F1 mice were euthanized, and gross observations were made of the major thoracic/abdominal organs.
Statistical analysis
Means and standard deviations were calculated for all data. Data were analyzed using a Student’s t-test between each groups. In addition, χ2 test was used for the parameters that were recorded as frequency of occurrence.
Results
CBM 588® lacks acquired antibacterial resistance
First, the MIC of several antimicrobials were determined for CBM 588® (Table 1). CBM 588® was susceptible to most antibiotics in all of the antimicrobial classes except the aminoglycosides. Additionally, there were a few antibiotics that also had high MIC values, such as cinoxacin (64 mg/L) and colistin (>256 mg/L). However, cinoxacin has been discontinued in the United States, and colistin is used primarily against respiratory infections in cystic fibrosis patients. 35
CBM588® antibiotic susceptibility.

CBM 588®: Clostridium butyricum MIYAIRI 588®; MIC: minimum inhibitory concentration.
Next, the susceptibility of CBM 588® to antibiotics in clinical use was determined (Table 2). CBM 588® was susceptible to all of the clinically relevant antimicrobials used, with the exception of the aminoglycoside antibiotics (i.e. gentamicin, kanamycin, and streptomycin). European Food Safety Authority break points 36 for gentamicin, kanamycin, and streptomycin for Gram-positive bacteria are 4.0, 16.0, and 8.0 mg/L, respectively. The MIC values for CBM 588® against these antibiotics were 64.0, 128, and >256 mg/L, respectively. To determine whether these high MIC values were unique to the CBM 588® strain, the cryopreserved, deposited sample (FERM BP-2789) of CBM 588® as well as C. butyricum ATCC 19398T were tested. Both the cryopreserved CBM 588® and the ATCC strain of C. butyricum were resistant to aminoglycosides, indicating that aminoglycoside resistance is not a specific trait of CBM 588®.
Sensitivity of CBM 588® to clinically relevant antibiotics.
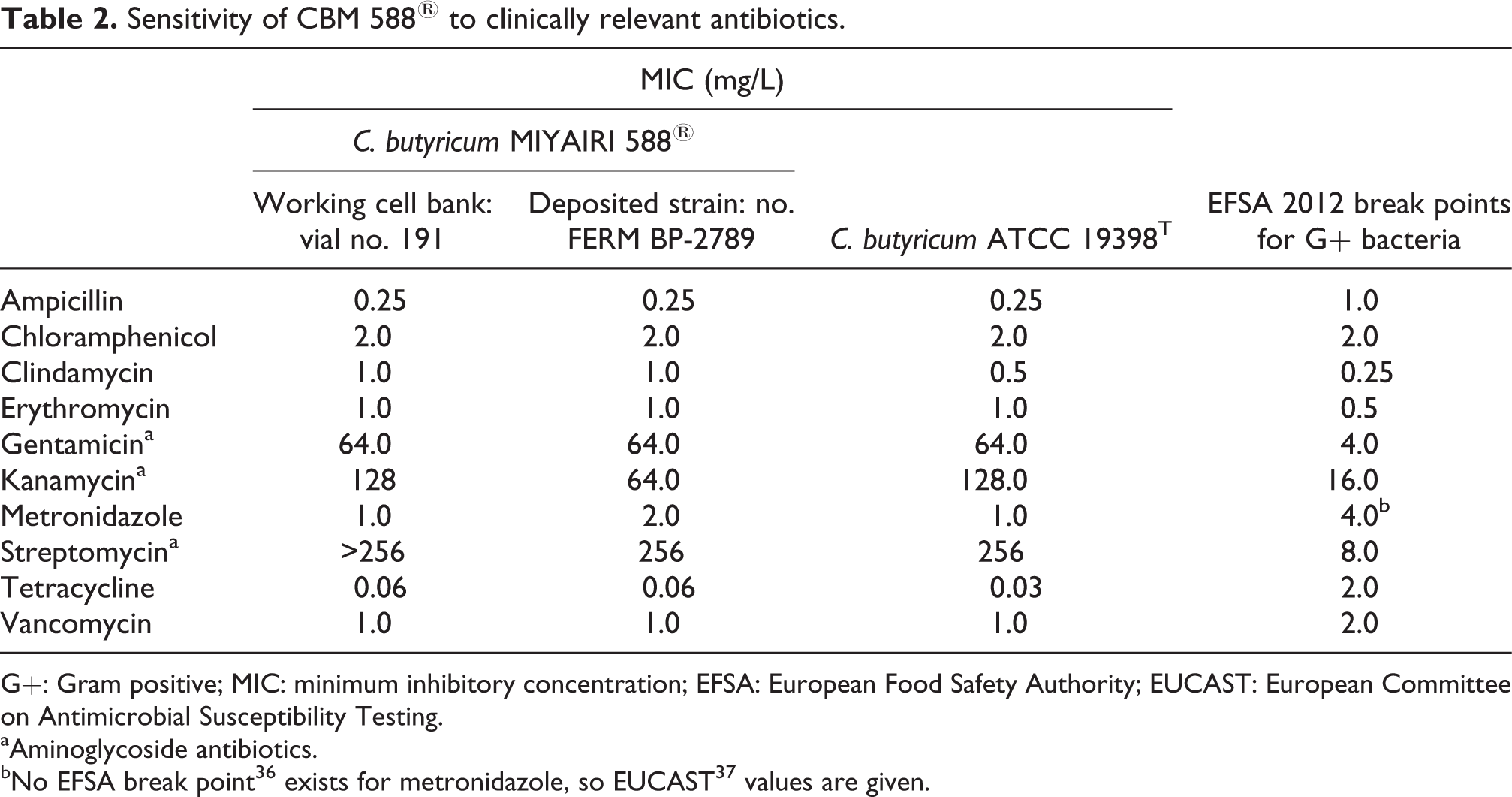
G+: Gram positive; MIC: minimum inhibitory concentration; EFSA: European Food Safety Authority; EUCAST: European Committee on Antimicrobial Susceptibility Testing.
aAminoglycoside antibiotics.
CBM 588® lacks Clostridium toxins
CBM 588® has thus far shown no evidence of pathogenicity, although the presence of toxins produced by other Clostridium species has not been fully assessed. Therefore, the genome of CBM 588® was analyzed for the presence of Clostridium toxins (Figure 1(b) and (d)). 16S rRNA 30 gene was amplified as an internal control for all PCR reactions (Figure 1(a) and (c)). The primers for all reactions are listed in Table 3.

Demonstration of absence of genes encoding Clostridium toxins in CBM 588®. (a) Determination of 16S rRNA gene for C. perfringens with universal primers. (b) Detection of C. perfringens α, β, and ε toxins. All JCM strains show the presence of toxin genes, but CBM 588® does not. (c) Determination of 16S rRNA gene for C. botulinum with universal primers. (d) Detection of BoNTs: A, B, E, and F. All KZ strains show the presence of toxin genes, but CBM 588® does not. CBM 588®: Clostridium butyricum MIYAIRI 588®; rRNA: ribosomal RNA; JCM: Japan Collection of Microorganisms; KZ: Kanazawa University; BoNT: botulinum neurotoxin.
Nucleotide sequences of primers.

rRNA: ribosomal RNA.
Many strains of Clostridium perfringens produce α, β, and ε toxins and are known virulence factors. The C. perfringens JCM strains 1290T, 3816, 3817, 3818, and 3819 were used as positive controls for α, β and ε toxin genes (Figure 1(b)). Sequence heterogeneity has been shown for α toxin gene that may affect the binding of the PCR primers. We confirmed the absence of α toxin gene using additional primers described by Siqueira et al. 38 (data not shown). The presence of α, β, or ε toxin genes was not detected in the genome of CBM 588®.
BoNT is a group of seven, serologically distinct toxins produced by many members of the Clostridium genus. Of these, A, B, E, and occasionally F are the main causes of botulinum poisoning in humans. The genomes of the positive control C. botulinum strains (C. botulinum KZ 2, KZ 1710, KZ 47, and KZ 1896) showed the genes encoding their respective toxins (toxin A, B, F, and E; Figure 1(d)). CBM 588®, however, did not contain any genes encoding for BoNT A, B, E, or F toxins. Additionally, nontoxic non-hemagglutinin (NTNH) protein is also present in all BoNT-producing bacteria. 39 The highly conserved NTNH gene is located directly upstream from the BoNT genes. The four positive control C. botulinum strains and CBM 588® were examined for the NTNH gene. The C. botulinum strains producing BoNTs also encoded for the NTNH gene, while CBM 588® lacked genes for BoNT A, B, E, or F as well as NTNH (data not shown). These results indicate that CBM 588® lacks Clostridium toxin virulence factors α, β, and ε toxins; types A, B, E, or F neurotoxins; and NTNH.
CBM 588® does not adversely affect reproduction or development
Dams were administered the CBM 588® via gavage at 0, 24, 240, or 4800 mg/kg BW, daily through days 6–15 of gestation. Evaluation of fetuses for gross visceral and skeletal malformations at day 18 of gestation along with monitoring live pups for survival, growth, and development were carried out on the resulting F1 litters. Additionally, 10–11 mating pairs from the F1 generation were followed to determine whether CBM 588® had any adverse effects on second generation development.
A total of three F0 dams died during the experiment (Table 4). Two were in the 4800 mg/kg BW group and died due to technical error on days 10 and 15 of gestation. Tracheal damage and thoracic retention of the test substance were observed and the cause of death was determined to be intrathoracic pressure due to incorrect administration of the test substance. A control animal died on day 16 postpartum due to unknown causes. None of these deaths were attributed to CBM 588®. One dam was killed for necropsy since all live offspring were deceased on postpartum day 9. Body weights and feed intake were not affected by CBM 588® consumption.
Effects of CBM 588® powder on maternal growth and survival.a

CBM 588®: Clostridium butyricum MIYAIRI 588®; BW: body weight; CFU: colony-forming unit; SD: standard deviation.
aApproximately 24 mg/kg BW: 5.8 × 107 CFU/kg BW; 240 mg/kg BW: 5.8 × 108 CFU/kg BW; 4800 mg/kg BW: 1.2 × 1010 CFU/kg BW.
aNumber of live females/number of females allowed to carry litters to term.
bp < 0.05.
cp < 0.01.
CBM 588® did not produce any dose-related adverse effects on F1 fetal survival. At 18 days gestation (approximately 1 day before expected delivery), no differences in placental remnants, reabsorption sites, sex ratio, dead embryos, or gross external or organ malformations were seen in CBM 588®-treated animals compared with the controls (Table 5). The dams receiving CBM588® at 240 mg/kg BW had significantly fewer live pups/litter (p < 0.05) as well as fewer corpora lutea and implantations/dam compared with the control (p < 0.01). A small but significant (p < 0.01) increase in the weight of the live fetuses was seen in the litters from the dams receiving CBM 588® at 24 and 240 mg/kg BW. However, there were no significant adverse effects in any of these parameters in fetuses from dams receiving the highest dose, 4800 mg/kg BW CBM 588®.
Effects of CBM 588® powder on survival, growth, and development of F1 fetuses.a

CBM 588®: Clostridium butyricum MIYAIRI 588®; BW: body weight; CFU: colony-forming unit; SD: standard deviation.
aApproximately 24 mg/kg BW: 5.8 × 107 CFU/kg BW; 240 mg/kg BW: 5.8 × 108 CFU/kg BW; 4800 mg/kg BW: 1.2 × 1010 CFU/kg BW.
bp < 0.01: compared with the control group.
cp < 0.05: compared with the control group.
dAverage and SD values of female and male F1 mice in each group were calculated with the average BW of two to six mice randomly selected per litter.
eFusion of occipital bone with atlas or 7th cervical vertebra with 1st thoracic vertebra.
CBM 588® did not negatively affect the development of the F1 fetuses examined at day 18 of gestation (Table 5). The weight of viable fetuses, both male and female, was slightly higher in the litters from dams given 24 or 240 mg/kg BW CBM 588® (p < 0.05–0.01) compared with the controls. The skeletal malformation rate did not differ significantly in treated groups compared with the control group, with the exception of a statistically significant increase in the incidence of extra cervical ribs noted in F1 fetuses from the group receiving CBM 588® at 4800 mg/kg BW compared with the control. Additionally, the number of ossified caudal vertebrae was higher in the fetuses in the 240 mg/kg group compared with the control group.
CBM 588® had no dose-related adverse effect on the survival or physical and behavioral development of the F1 mice evaluated up to 21 days postnatal age (Table 6). No effect was seen on gestational length, live birth index, stillborn count, sex ratio, external abnormalities, day 4 survival rate, weaning rate, and physical or behavioral development. There was a significant reduction in BW of both male and female pups at day 21 from dams receiving CBM 588® at 240 and 4800 mg/kg BW compared with the control group (Table 6).
Effects of CBM 588® powder on survival, growth development of F1 mice.a

CBM 588®: Clostridium butyricum MIYAIRI 588®; BW: body weight; CFU: colony-forming unit; SD: standard deviation.
aApproximately 24 mg/kg BW: 5.8 × 107 CFU/kg BW; 240 mg/kg BW: 5.8 × 108 CFU/kg BW; 4800 mg/kg BW: 1.2 × 1010 CFU/kg BW.
bp < 0.05: compared with the control group.
cLive F1/live newborn nursed.
dLive pups at postnatal day 22/live pups at postnatal day 4.
eAverage and SD values of female and male F1 mice in each group were calculated with the average BW of two to six mice randomly selected per litter.
fp < 0.01: compared with the control group.
CBM 588® did not produce any dose-related adverse effects on the reproductive capabilities of the F1 generation or the survival and growth of the F2 generation (Table 7). There was no significant difference in gestation length, implantations, and number of live pups/litter in all test groups compared to control. F1 dams fed with CBM 588® gained more weight between 3 and 11 weeks than the F1 dams in the control group. All mating pairs copulated, but the fertility rate for animals receiving CBM 588® at 24 and 4800 mg/kg BW was only 90% compared to 100% for the control and 240 mg/kg BW CBM 588®. Additionally, all confirmed pregnancies were delivered, with the exception of one litter in the 240 mg/kg BW group.
Effects of CBM 588® powder on F1 reproduction and survival and growth of F2 mice.a

CBM 588®: Clostridium butyricum MIYAIRI 588®; BW: body weight; CFU: colony-forming unit; SD: standard deviation.
aApproximately 24 mg/kg BW: 5.8 × 107 CFU/kg BW; 240 mg/kg BW: 5.8 × 108 CFU/kg BW; 4800 mg/kg BW: 1.2 × 1010 CFU/kg BW.
bp < 0.05: compared with the control group.
cp < 0.01: compared with the control group.
dAverage and SD values of female and male each group were calculated with the average BW of all live pups per litter.
eData were calculated based on eight litters since all pups from one litter died by postnatal day 5 due to maternal neglect.
CBM 588® did not alter the survival of the F2 pups (Table 7) through day 7. One F2 litter in the 4800mg/kg BW group was lost on postpartum day 5 due to neglect by the F1 dam. No statistically significant dose-related malformations were noted in the F2 pups. BWs of male and female F2 pups were also unaffected by CBM 588® administration.
Discussion
CBM 588® is used as a probiotic in Japan for both humans and animals and is approved as a probiotic for animal feed 40 –42 and as a novel food 22 in Europe. CBM 588® is the first Clostridium species and strain to be used effectively as a probiotic. 17 However, because there are many pathogenic Clostridium species, it is essential that the safety and toxicity of CBM 588® is thoroughly assessed.
The increasing prevalence of antibiotic resistant strains of bacteria is a global public health concern. Probiotics should have minimal antibiotic resistance for clinically relevant antibiotics. The MICs of several antibiotics were determined (Table 1), and CBM 588® is susceptible to most classes of antibiotics with the exception of aminoglycosides. However, the resistance to aminoglycosides, in anaerobic bacteria, is a physical, intrinsic feature rather than genetic or metabolic property of anaerobic bacteria due to the inability of anaerobes to transport these antibiotics across the microbial cell membrane. 43 In addition to the aminoglycosides, there were a few specific antibiotics with elevated MICs. Cinoxacin (MIC = 64 mg/L) has been discontinued in the United States. Colistin, also known as polymixin E, (MIC = >256 mg/L) is used intravenously, primarily as a drug of last resort in multidrug-resistant, Gram negative bacterial infections in the respiratory tract of cystic fibrosis patients. 35 The susceptibility of CBM 588® to clinically relevant antibiotics was also assessed (Table 2). CBM 588® was resistant to gentamicin, kanamycin, and streptomycin, all aminoglycosides. The nonpathogenic C. butyricum ATCC 19398T strain was also resistant to these antibiotics, indicating that the resistance is not specific to CBM 588® but similar across multiple C. butyricum strains. Aside from the aminoglycosides, CBM 588® does not show resistance to any class of antibiotics.
CBM 588® does not produce α, β, or ε toxins commonly associated with C. perfringens, or BoNTs A, B, E, or F (Figure 1). While it is known that some toxin genes are located on plasmids, it has been reported that CBM 588 has only one plasmid and critical genes, such as toxin genes or antibiotic resistance genes, relevant to safety were not found. 44 The only toxin C. butyricum has been reported to produce is BoNT type E. 45,46 BoNT type E producing C. butyricum have been isolated from soil 47 and suspect food in a botulism outbreak in China 48 as well as from the feces of two infants suffering from botulism in Italy. 49 –51 The strains producing the type E toxin were confirmed as C. butyricum by 16S rRNA gene sequencing. 52 Due to the sequence homology (97%) of the type E toxin produced by C. butyricum and C. botulinum, 53 it is likely that C. butyricum acquired the toxin gene (bont/E) through horizontal gene transfer. However, genetic analysis of the toxins from the C. butyricum isolated from Italy and China represent two distinctive subtypes, indicating that two distinct horizontal gene transfer events occurred. 54 –56
Although possible, horizontal genetic transfer of type E toxin has proven to be very difficult to induce in a laboratory setting since it is well known that Bont/E is chromosomally located. However, one study has shown type E toxin can be stably transferred from a toxigenic C. butyricum BL 5839 strain to a nontoxigenic C. botulinum type E-like S-5 in the presence of a bacteriophage transducing agent and the “helper” strain, nontoxigenic C. butyricum ATCC 19398T. The transfer rate was 1 in 10. 4,57 The transfer appears to be facilitated by a temperate prophage. Mature bacteriophages have been implicated in toxins C and D transfer between C. botulinum strains and have resulted in much higher transfer rates. 58 The low in vitro transfer rates, combined with the low frequency of type E producing C. butyricum isolated from food and soil, suggests that the chances of CBM 588® becoming toxigenic through horizontal gene transfer is very low.
The developmental toxicity of CBM 588® was assessed through two generations. F0 dams were administered concentrated CBM 588® powder (2.4 × 109 CFU/g) at 0, 24, 240, or 4800 mg/kg BW, daily during days 6–15 of gestation. No dose-related adverse effects related to CBM 588® administration were noted. The resulting F1 fetuses and live pups from dams given CBM 588® showed no adverse effects on physical or behavioral development. CBM 588® administration in the F0 generation did not affect the survival and growth of the F2 generation. The skeletal malformation rate did not differ significantly in treated groups compared with the control group, with the exception of a statistically significant increase in the incidence of extra cervical ribs noted in F1 fetuses from the group receiving CBM 588® at 4800 mg/kg BW compared with the control. Supernumerary or accessory ribs (SNR), either lumbar or cervical (CR), are a common finding in standard developmental toxicology bioassays. The biological significance of these anomalies has been the subject of some debate in the literature. In rodents, the spontaneous incidence of SNR is species and strain related and ranges from <1% to >30%. 59 In this study, the incidence of CR was within reported historical range, and there were no associated changes in any other skeletal parameters supporting the conclusion that this finding is not related to test article administration.
Conclusion
These data support the safe use of CBM 588® as a probiotic for use in food supplements and animal feeds. CBM 588® is susceptible to most antibiotics, with the exception of amnioglycosides, which are known to have limited effect on anaerobes. The genome of CBM 588® lacks genes encoding known Clostridium toxins. Additionally, CBM 588® does not produce developmental or reproductive toxicity in mice.
Footnotes
Acknowledgments
Teratogenicity and reproduction studies were conducted with the superb technical support of the Central Institute for Experimental Animals, Japan. The authors would like to thank the institute for their efforts. All study experiments were sponsored by Miyarisan Pharmaceutical Co. Ltd, Tokyo, Japan.
Authors’ Note
Authors KI and KO contributed equally to this work.
Declaration of Conflicting Interests
KI, KO, MS, KW, SN, MT and MT are employees of Miyarisan Pharmaceutical Co. Ltd., Tokyo, Japan.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
