Abstract
Objective:
The aim of this work was to investigate the cytotoxic, antioxidative, and enzyme inhibition effects of alizarin, quinizarin, and purpurin, which are anthraquinones (AQ).
Methods:
Cytotoxic effects were evaluated with cell inhibition rate by 3-(4,5-dimethylthiazol- 2-yl)-2,5-diphenyltetrazolium bromide assay. Different chemical assays, including free radical scavenging activity (1,1-diphenyl-2-picrylhydrazyl and 2,2-azino-bis(3-ethylbenzothiazloine-6-sulfonic acid)), phosphomolybdenum and reducing power (ferric reducing antioxidant power and cupric ion reducing activity), were used to evaluate the antioxidant properties. Moreover, enzyme inhibitory activities were analyzed against acetylcholinesterase, butrylcholinesterase, tyrosinase, α-amylase, and α-glucosidase.
Results:
These components have antioxidant and enzyme inhibition activity. Especially, purpurin showed the strongest antioxidant and good enzyme inhibitory effects. According to our cytotoxicity results, alizarin, purpurin, and quinizarin induced dose- and time-dependent cell proliferation. Furthermore, when we applied AQs with mitomycin C (MC) on L929 cell line, we demonstrated that cell proliferation in MC-AQ groups compared with MC group was increased. The most effective component was alizarin at 100 µM concentration. These AQs showed positive effects on L929 cell lines with high half-maximal inhibitory concentration values.
Conclusion:
Our results demonstrate that AQs may be used as antioxidative compounds in food and medicinal applications.
Introduction
Antioxidant compounds can inhibit or delay the oxidative damage/stress by interfering with the production of free radicals and/or inactivate them once they are formed. 1 For this purpose, some antioxidants (butylated hydroxyanisole, butylated hydroxytoluene, propyl gallate, and tertiary butylhydroquinone) are synthetically developed and used for industrial processing, but they may possess some side effects and toxic properties to human health. 2 Enzyme inhibitors are commonly used as treatment options for global health problems such as Alzheimer’s disease (AD) and diabetes mellitus (DM). However, these inhibitors are reported to have side effects including cytotoxicity, hepatotoxicity, gastrointestinal disturbances, and diarrhea. 3,4 In this direction, the development and utilization of alternative and potentially more effective and less toxic substances are indicated to be very necessary. Identification of cytotoxic effects of synthetic and natural products is not only important in diseases but also in the usage of drugs, cosmetics, and food industries. When they have been used or consumed they can cause toxic effects in our cells. Therefore, natural and chemical substances have to be tested before they use. 5
Anthraquinones (AQ) are the most common group of naturally occurring quinones. Both natural and synthetic AQs have been used as colorants in food, drugs, and cosmetic industries. 6 Although the importance of the AQ or derivatives is apparent from their widespread applications in these industries, there is actually very little information about biological properties of these compounds. 7 From this point, new research studies on biological effects of AQ have particular interest for applications in the areas mentioned above.
AQs are shown to possess important biological activities, including antibacterial, antioxidant, antifungal, and anticancer.
8
–11
On the other hand, there is no information in the literature on their proliferative or antiproliferative effects of three AQs (alizarin, purpurin, and quinizarin) on eukaryotic cells. (Figure 1). Takahashi et al.
12
showed that purpurin and alizarin have anti-genotoxic effects in the
Structure of the AQs. AQ: anthroquinone.
Materials and methods
Materials
Alizarin, quinizarin, and purpurin were provided by Sigma (St Louis, Missouri, USA). They were dissolved in Dulbecco’s modified Eagle’s medium (DMEM). Stock solutions were diluted to the desired final concentrations with growth medium just before use. MTT was purchased from Roche (Switzerland). DMEM was purchased from Gibco (Istanbul, Turkey). Penicillin/streptomycin was purchased from Biochrom (Germany). Fetal bovine serum (FBS) was purchased from Biochrom.
Antioxidant activity assays
Phosphomolybdenum method
The total antioxidant activity of the samples was evaluated by phosphomolybdenum method according to Berk et al. 13 with slight modifications. Sample solution (0.3 mL) was combined with 3 mL of reagent solution (0.6 M sulfuric acid, 28 mM sodium phosphate, and 4 mM ammonium molybdate). The sample absorbance was read at 695 nm after a 90-min incubation at 95°C. The total antioxidant capacity was expressed as equivalents of trolox as determined by the equation obtained from the standard trolox graph.
DPPH radical scavenging activity
The effect of the samples on 1,1-diphenyl-2-picrylhydrazyl (DPPH) radical was estimated according to the method followed by Sarikurkcu. 14 Sample solution (1 mL) was added to a 4 ml of a 0.004% methanol solution of DPPH. The sample absorbance was read at 517 nm after a 30-min incubation at room temperature in dark. The DPPH scavenging activity was expressed as equivalents of trolox according to the equation obtained from the standard trolox graph.
ABTS cation radical scavenging activity
The scavenging activity aganist 2,2 azino-bis (3-ethylbenzothiazloine-6-sulfonic acid) (ABTS) cation radical was measured according to the method described by Re et al. 15 with slight modifications. Briefly, ABTS-positive cation radical was produced directly by reacting 7 mM ABTS solution with 2.45 mM potassium persulfate and allowing the mixture to stand for 12–16 h in dark at room temperature. Prior to beginning the assay, ABTS solution was diluted with methanol to an absorbance of 0.700 ± 0.02 at 734 nm. Sample solution (1 mL) was added to ABTS solution (2 mL) and mixed. The sample absorbance was read at 734 nm after a 30-min incubation at room temperature. The ABTS cation radical scavenging activity was expressed as equivalents of trolox according to the equation obtained from the standard trolox graph.
CUPRAC method
The cupric ion reducing activity (CUPRAC) was determined according to the method of Apak et al. 16 Sample solution (0.5 mL) was added to premixed reaction mixture containing cupric chloride (CuCl2; 1 mL, 10 mM), neocuproine (1 mL, 7.5 mM), and ammonium acetate buffer (1 mL, 1 M, pH 7.0). Similarly, a blank was prepared by adding sample solution (0.5 mL) to premixed reaction mixture (3 mL) without CuCl2. Then, the sample and blank absorbances were read at 450 nm after a 30-min incubation at room temperature. The absorbance of the blank was subtracted from that of the sample. CUPRAC activity was expressed as equivalents of trolox according to the equation obtained from the standard trolox graph.
FRAP method
The ferric reducing antioxidant power (FRAP) assay was carried out as described by Aktumsek et al.
17
with slight modifications. Sample solution (0.1 mL) was added to premixed FRAP reagent (2 mL) containing acetate buffer (0.3 M, pH 3.6), 2,4,6-tris(2-pyridyl)-
Enzyme inhibitory activity
ChE inhibition
The ChE inhibitory activity was measured using Ellman’s method as previously reported. 18 Sample solution (50 µL) was mixed with 5,5-dithio-bis (2-nitrobenzoic) acid (125 µL) and acetylcholinesterase (AChE or butrylcholinesterase (BChE)) solution (25 µL) in tris(hydroxymethyl)aminomethane-HCl buffer (pH 8.0) in a 96-well microplate and incubated for 15 min at 25°C. The reaction was then initiated with the addition of acetylthiocholine iodide or butyrylthiocholine chloride (25 µL). Similarly, a blank was prepared by adding sample solution to all reaction reagents without enzyme (AChE or BChE) solution. The sample and blank absorbances were read at 405 nm after a 10-min incubation at 25°C. The absorbance of the blank was subtracted from that of the sample and the ChE inhibitory activity was expressed as equivalents of galanthamine.
α-Amylase inhibition
α-Amylase inhibitory activity was performed using Caraway-Somogyi iodine/potassium iodide (IKI) method. 18 Sample solution (25 µL) was mixed with α-amylase solution (50 µL) in phosphate buffer (pH 6.9 with 6 mM sodium chloride) in a 96-well microplate and incubated for 10 min at 37°C. After preincubation, the reaction was initiated with the addition of starch solution (50 µL, 0.05%). Similarly, a blank was prepared by adding sample solution to all reaction reagents without enzyme (α-amylase) solution. The reaction mixture was incubated 10 min at 37°C. The reaction was then stopped with the addition of HCl (25 µL, 1 M). This was followed by addition of the IKI solution (100 µL). The sample and blank absorbances were read at 630 nm. The absorbance of the blank was subtracted from that of the sample, and the α-amylase inhibitory activity was expressed as equivalents of acarbose.
α-Glucosidase inhibition
α-Glucosidase inhibitory activity was performed by the previous method.
18
Sample solution (50 µL) was mixed with glutathione (50 µL), α-glucosidase solution (50 µL) in phosphate buffer (pH 6.8), and 4-
Tyrosinase inhibition
Tyrosinase inhibitory activity was measured using the modified dopachrome method with
Cell lines and cell culture
L929 cells are one of the widely used cells in cytotoxicity tests. 20 L929 cell line was provided by American Type Culture Collection. Fibroblast cells were cultured in DMEM including 10% FBS and 1% penicillin/streptomycin. Cells were incubated in a humidified atmosphere (37°C, 5% carbon dioxide (CO2)), and the screw cap of the flask was kept flexible to allow circulation of CO2 into flask. The day after medium was refreshed to allow elimination of the dead cells. Subculture of cells was repeated at about 3–4 days intervals according to the cell types and doubling times.
MTT assay
L929 cell line was seeded in triplicate at 1 × 104 cells/mL to the 96 well plates in final volume of 100 μL/well culture medium and incubated overnight at 37°C under a 5% CO2 atmosphere for 24 h before the treatment. The medium in the wells was then replaced with fresh medium containing alizarin, quinizarin, and purpurin (1, 5, 10, 50, 100 μM) and incubation continued for 48 h. In another set of experiment, evaluating the protective effects of AQ against MC at 5 µL/mL concentration was performed for 48 h. Briefly, 10 μL of MTT solution was added to each well, and the plates were incubated for 4 h. The reaction was terminated by adding 100 μL of 10% sodium dodecyl sulfate in 0.01 M HCl. The plates were incubated overnight (37°C, 5% CO2). The absorbance of samples was measured at 550–600 nm with an enzyme-linked immunosorbent assay reader.

Half-maximal inhibitory concentration (IC50) values were calculated by using Graphpad 6 prism program.
Statistical analysis
For all the experiments, all the assays were carried out in triplicate. The antioxidant and enzyme inhibitory results are expressed as mean values and standard deviation. The differences between the different compounds were analyzed using one-way analysis of variance (ANOVA), followed by Tukey’s honestly significant difference post hoc test with α = 0.05.
For statistical analysis of MTT values, Mann–Whitney
Results
Antioxidant capacity
Free radical scavenging activities of tested sample were evaluated by DPPH and ABTS assays. DPPH and ABTS free radicals are frequently used to determine the radical scavenging activity of natural or chemical compounds that act as hydrogen donors. The investigated AQs demonstrated notable antiradical activities. in both DPPH and ABTS assays, the greatest scavenging activity was caused by purpurin, closely followed by alizarin. Quinizarin was also inactive on DPPH free radical (Table 1). Similar to the results, AQs are known as very good electron and/or hydrogen donors.
Radical scavenging activity and total antioxidant activity (by phosphomolybdenum method) of the samples (mean ± SD).a

TEs: trolox equivalents; na: not active; DPPH: 1,1-diphenyl-2-picrylhydrazyl; ABTS: 2,2-azino-bis(3-ethylbenzothiazloine-6-sulfonic acid); SD: standard deviation.
a
The reducing power of a compound may serve as a significant indicator of its potential antioxidant activity. FRAP and CUPRAC assays were used to evaluate the reducing powers of tested AQs. The results are listed in Table 2. In these assays, antioxidants react by donating an electron, thus converting Fe+3 to Fe+2 in FRAP assay or Cu+1 to Cu in CUPRAC assay. Similar to DPPH and ABTS results, alizarin and purpurin were the most active compounds in FRAP and CUPRAC assays. Phosphomolybenum assay is based on the reduction of Mo(VI) to Mo(V) by the antioxidants and subsequent formation of green phosphate/Mo(V) complex, which is measured spectrophotometrically at 695 nm. Apparently, alizarin had the highest activity, followed by purpurin and poorest activity was observed in quinizarin (Table 1).
Reducing power of the samples (mean ± SD).a

TEs: trolox equivalents; FRAP: ferric reducing antioxidant power; CUPRAC: cupric ion reducing activity1,1-diphenyl-2-picrylhydrazyl; SD: standard deviation.
a
Inhibitory activities on AChE, BChE, tyrosinase, α-amylase, and α-glucosidase
The enzyme inhibitory activities were measured with spectrophotometric methods, and the results were illustrated in Table 3. Similar to antioxidant assay results, purpurin displayed the strongest enzyme inhibitory activities, except for α-amylase. Alizarin and quanizarin showed no ability to inhibit AChE and BChE enzymes. All the compounds studied have remarkable inhibitory effect on tyrosinase and α-glucosidase. Interestingly, quinizarin possessed the highest anti-amylase effect, whereas purpurin was not active on α-amylase. Clearly, there is strong relationship between antioxidant properties and enzyme inhibitory effects of the investigated AQs.
Enzyme inhibitory activity of the samples (mean ± SD).a

GALAEs: galanthamine equivalents; KAEs: kojic acid equivalents; ACEs: acarbose equivalents; FRAP: ferric reducing antioxidant power; na: not active; SD: standard deviation.
a
Cytotoxic activity
To evaluate the direct effects of alizarin, quinizarin, and purpurin on L929 cell line, MTT assay was used. We treated cells with increasing concentrations (1–100 µM) of alizarin, quinizarin, and purpurin at 48 h. As shown in Figures 2(a), 3(a), and 4(a), these AQs significantly increased proliferation of the L929 cells in a dose-dependent manner. When cells were cultured for 48 h with increasing concentrations of AQs (1–100 µM) with MC (5 µM), the cell inhibition rate decreased. Especially Figures 1(b), 2(b), and 3(b) show decreasing cell inhibition rate, which was lower at the highest concentration of AQs. Decreasing cell inhibition rate means that AQs have protective effects on negative impacts of MC. Alizarin and quanizarin were more effective than purpurin. These results express that quinizarin and alizarin have promising and positive effect on cell growth, as shown in Figures 2(c) and 3(c). However as shown in Figure 4(b) when purpurin with MC are applied coherently, only the 50 and 100 µM concentrations have a higher effect on inhibition in cell growth when compared to other groups (

(a) Results of the MTT assay treated for 48 h with different concentrations of alizarin on L929 cells. (a
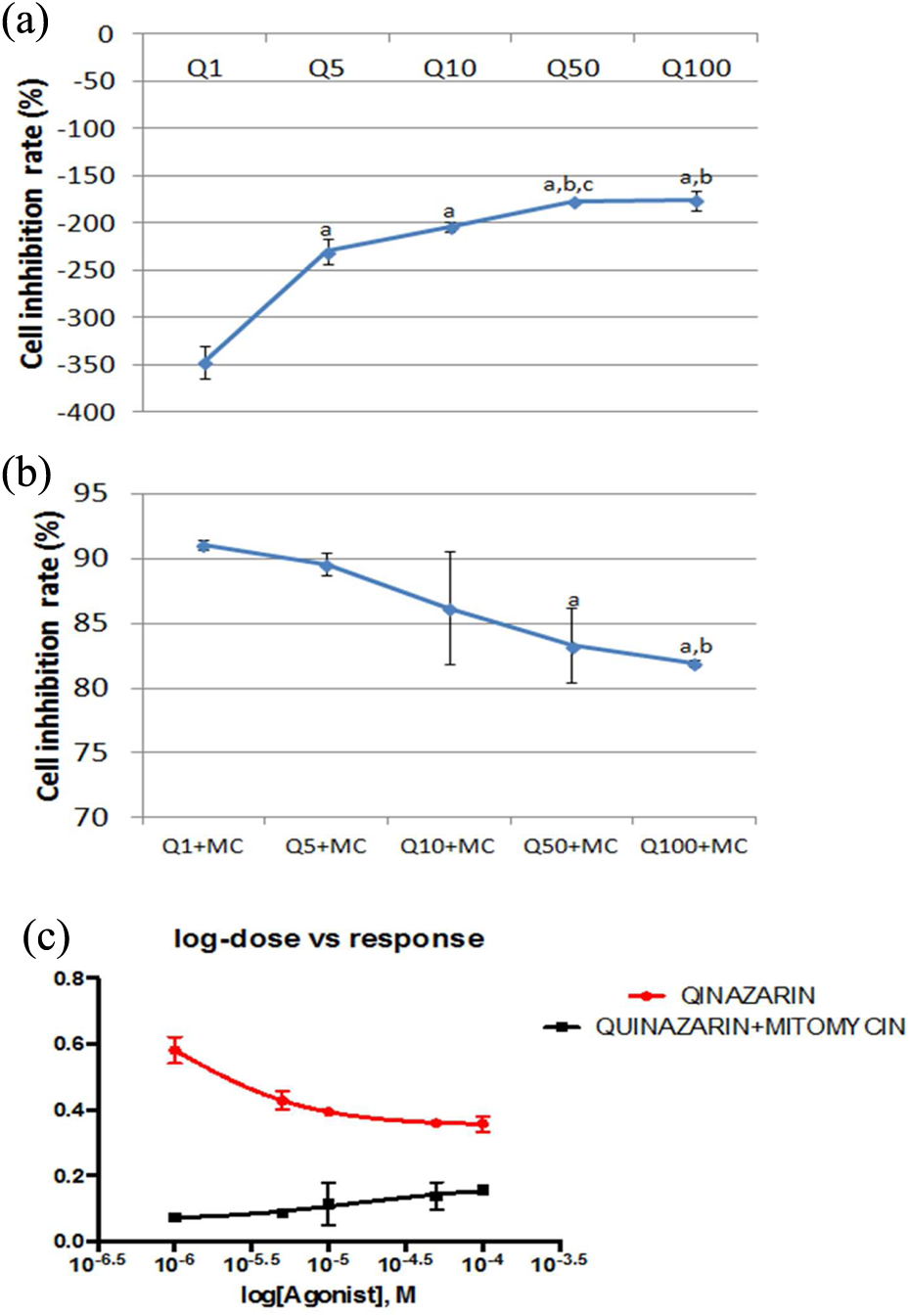
(a) Results of the MTT assay treated for 48 h with different concentrations of quinizarin on L929 cells. (a
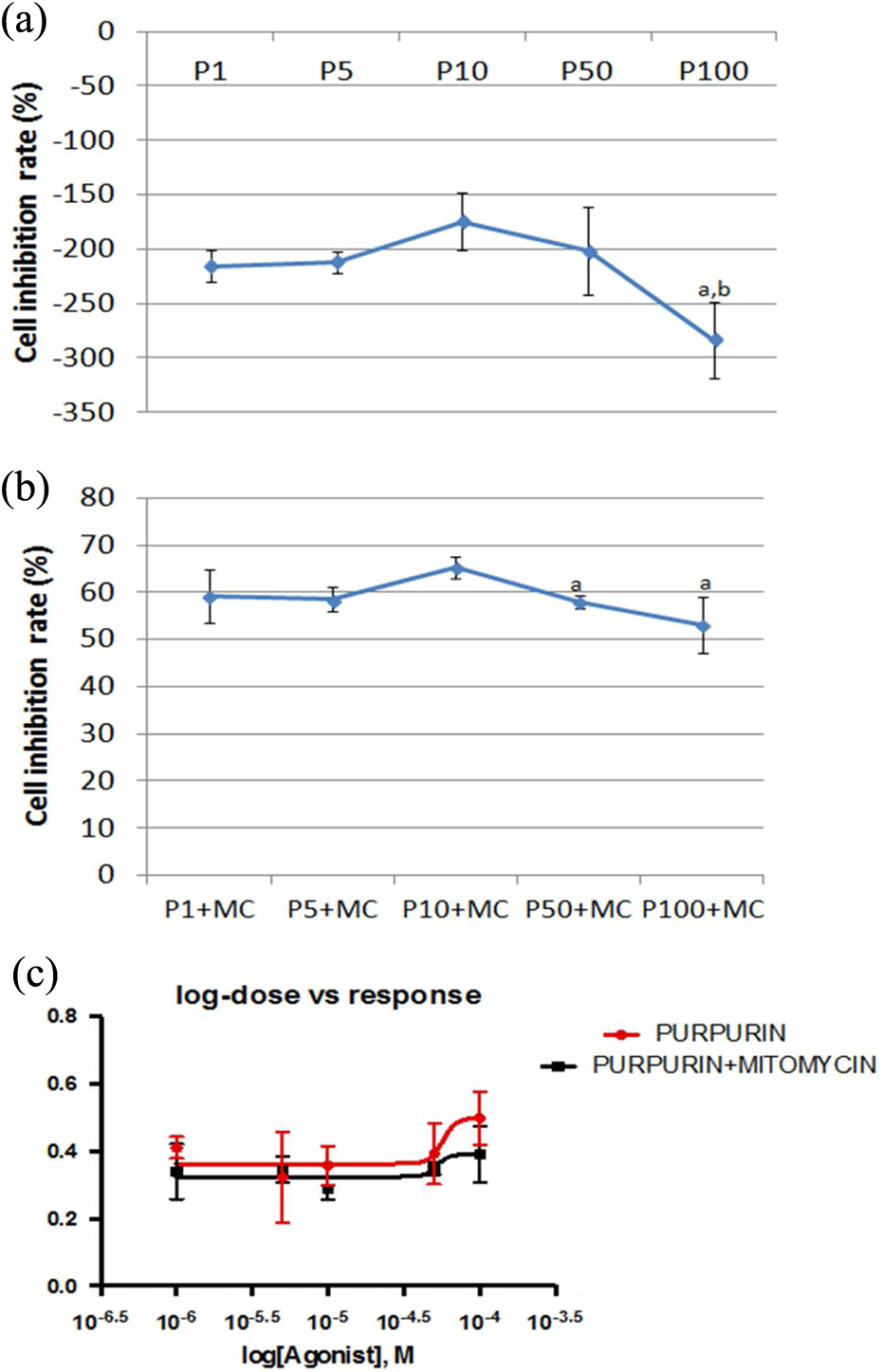
(a) Results of the MTT assay treated for 48 h with different concentrations of purpurin on L929 cells (a
IC50 values for AQs and AQs + MC in MTT assay.

AQ: anthroquinone; MTT: 3-(4,5-dimethylthiazol- 2-yl)-2,5-diphenyltetrazolium bromide; MC: mitomycin C; IC50: half-maximal inhibitory concentration.
Discussion
AQs are more common quinones that have been used in food, drugs, and cosmetic industries. 6 They have antibacterial, antioxidant, antifungal, and anticancer activities. 8 –11 The biological activities of AQs usually explained related to their antioxidative properties, which determined by using single test method. The antioxidant capacity cannot be fully described using a single method because antioxidant capacity is influenced by many factors. Therefore, it is pertinent to use several assays instead of a single one to evaluate and compare the antioxidant activity of chemicals. 21 Also antioxidant activities of AQs are difficult to interpret because of different views on the molecular mechanism of radical scavenging and the structural dependency of the antioxidant action. 7 For this purpose, the antioxidant activity of the investigated AQs was evaluated using different methods: free radical scavenging activity (ABTS and DPPH), reducing power assays (CUPRAC and FRAP), and total antioxidant capacity (phosphomolybdenum)
According to the results of DPPH and ABTS assays, purpurin and alizarin have the greatest scavenging activity. Quinizarin was inactive on DPPH free radical. Similarly, in FRAP and CUPRAC assays, the most active substances are alizarin and purpurin. All in all, alizarin had the highest activity, followed by purpurin and poorest activity was observed in quinizarin (Table 1). For potential utilization of natural and synthetic antioxidants, it is essential to explore and clarify the contribution of key structural elements of antioxidant, especially the OH groups. 22 In this direction, the hydroxyl substitution groups may be explained the highest antioxidant activities of alizarin and purpurin. These results are in good agreement with Yen et al. 9 who reported that the antioxidant properties of AQs depend on hydroxyl substitution in the structure.
AD and DM are two major health issues nowadays. The inhibition of key enzymes is one of the most accepted treatment approaches for these disturbances. For example, neuropathological occurrence of AD symptoms and cognitive deficits is consistent with the presence of plaques and cholinergic deficiency due to the degeneration or atrophy of cholinergic neurons in the basal forebrain. 23 Thus, AChE inhibition is very important for AD. 24 Again, α-amylase and α-glucosidase, which catalyze hydrolysis of starch, increase the blood glucose level. Therefore, an important strategy for DM is to inhibit the activities of α-amylase and α-glucosidase. 25 Moreover, tyrosinase is known as the key enzyme for melanin biosynthesis and tyrosinase inhibitiors play a crucial role against skin photocarcinogenesis. 26
Currently, many compounds are developed as enzyme inhibitors for the above-mentioned diseases. For example, serine, tacrine, and galantamine are used to treat AD. 27 Likewise, kojic acid and arbutin are used as tyrosinase inhibitor in the treatment of hyperpigmentation. Acarbose and viglibose are conventionally used in the management of DM. However, the inhibitors may possess various side effects such as flatulence, diarrhea, abdominal discomfort, and cell toxicities. 28 –30 Therefore, there is considerable interest in the development of new and safer enzyme inhibitory agents. In this direction, many compounds have been reported as potential and safe enzyme inhibitors. 31 –34
Similar to antioxidant assay results, the strongest enzyme inhibitory activities was in purpurin after α-amylase. Alizarin and quanizarin haven’t inhibited AChE and BChE enzymes. Apparently, there are strong relationship antioxidant properties and enzyme inhibitory effects of the investigated anthraquinones. The earlier published data about the relationship between antioxidant capacity and enzyme inhibitory effects of chemicals were controversial. Some authors have found positive correlation between antioxidative properties and enzyme inhibitory effects. 35 –38 On the other hand, several studies showed lower correlation between antioxidant and enzyme inhibitory effects. 39,40 At this point, it seems to the contradictory results are most probably due to differences in chemical structure, functional groups, and in the methodology used. Up to date, there have been a few reports on enzyme inhibitory effects of AQs. Orhan et al. 31 previously described that two AQs (rhein and aloe emodin) displayed moderate inhibitory effects on AChE and BChE. Our study is therefore the first report of in vitro enzyme inhibitory activities of the investigated AQs.
Previously, AQs used in this study have not been studied on healthy cells. In the literature, alizarin, purpurin, and quanizarin have antitumor activity because of their antioxidant properties. 41,42 Fotia et al. 43 reported that alizarin inhibited the proliferation of bone cancer cells although it didn’t have inhibition on human mesenchymal stromal cells at 5 to 50 µg/mL concentration. According to researchers, alizarin suppressed cell proliferation in cancer cells by Erk and Akt signaling pathways. 43 In addition, alizarin, purpurin, and quanizarin inhibited cell proliferation in different cancer cells. Another study showed that alizarin and purpurin have inhibition of CYP activities for eliminating mutagens. B16-F10 melanoma murine cells and breast carcinoma cell line MCF-7 have been inhibited by using quanizarin and purpurin respectively. 41,44
According to the results of cytotoxicity assay, AQs have protective effects on MC treatment. Alizarin and quanizarin were more effective than purpurin. Also the IC50 values of alizarin, quinizarin, and purpurin showed that they have positive effects on L929 cell lines. By possessing antioxidant activity and inhibiting CYP 450 enzyme systems, these substances protect cells against mutagenic and toxic agents. 12 Thus, the high level of antioxidant capacity reduces the toxic effect of MC when these substances are given with MC.
Conclusion
In conclusion, the present study showed that AQ tested have antioxidative and enzyme inhibitory effects in vitro. Besides, these compounds had no cytotoxic effects on L929 cell line. The compounds also stimulated the growth of the cells. Moreover, these compounds decrease the inhibitory activity of MC on normal cells. The stimulator activity can be caused from the antioxidative effects of these compounds. Hence, these compounds could be recommended for the formulation of antioxidant-rich therapeutic diets and supplementary food with no cytotoxic properties for the food and drug industry. However, further studies are needed to evaluate the safety and efficacy of these compounds with different test systems.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
