Abstract
This study aims to investigate the mechanisms involved in the action of lutein (LU) alleviating arsenic-induced reproductive toxicity using mice model. Forty male Kunming mice were received following treatments by gavage: normal saline solution (control), arsenic trioxide (ATO; 5 mg/kg/day), LU (40 mg/kg/day), and ATO + LU (5 mg/kg/day + 40 mg/kg/day). At the end, the mice were killed by cervical dislocation and weighed. Pathological examination was done on the testis. The biomedical parameters including superoxide dismutase (SOD), glutathione (GSH), total antioxidative capability, malondialdehyde (MDA), 8-hydroxydeoxyguanosine (8-OHdG), and reproductive indexes were analyzed. The messenger RNA (mRNA) and protein expression of Nrf2, heme oxygenase 1 (HO-1), glutathione S-transferase (GST), nicotinamide adenine dinucleotide phosphate dehydrogenase, quinone 1 (NQO1) in testis were detected by real-time polymerase chain reaction and Western blot. We found that there was a decrease in sperm count; testis somatic index; the activities of SOD, GSH, total antioxidative capacity (p < 0.01, respectively) in ATO-treated mice, while there was an increase in the levels of sperm abnormalities, MDA, and 8-OHdG than control (p < 0.01, respectively). The groups treated with ATO + LU showed recovery of the measured parameters between those of ATO or saline-treated group. The antagonized interaction between ATO and LU was statistically significant (p < 0.01). Mice treated with ATO + LU also showed greater mRNA expression of Nrf2, HO-1, NQO1, and GST than ATO or saline-treated groups. These findings suggest that LU alleviates reproductive toxicity induced by arsenic in male mice via Nrf2 signaling, which implicates a possible mechanism of LU in preventing the reproductive injury, and elucidates that consuming the rich plant sources of LU will alleviate the reproductive toxicity induced by chemicals.
Introduction
Arsenic poisoning is a medical condition caused by elevated levels of arsenic in the body. The dominant basis of arsenic poisoning is from ground water that naturally contains high concentrations of arsenic. Arsenic is related to heart disease (hypertension-related cardiovascular), cancer, stroke (cerebrovascular diseases), chronic lower respiratory diseases, and reproductive effects. 1 –4 Arsenic produces steroidogenic dysfunction in male rats and mice, which leads to infertility. 5 –8 Chronic arsenic exposure may have contributed to male infertility, 9 impacted erectile function, 10 and reduced semen quality. 11
The free radical-mediated theory becomes more acceptable to explain the damage induced by arsenic. Oxidative stress is acceptable as one of the mechanisms of arsenicosis, which can be mediated by excess production of reactive oxygen species (ROS). 12 –16 Furthermore, inorganic arsenic could bind protein thiol groups and nonprotein thiols such as glutathione (GSH). 17 Arsenic is also reported to regulate the activity of antioxidant enzymes, including superoxide dismutase (SOD), glutathione peroxidase (GSH-Px), and catalase. 18 Therefore, synthetic scavengers of ROS and antioxidants are to reduce arsenic-induced toxic effects.
Lutein (LU) is a xanthophyll, which is synthesized only by plants, and like other xanthophylls is found in high quantities in green leafy vegetables such as spinach, kale and yellow carrots, lettuce, broccoli, peas, lima beans, orange juice, celery, string beans, and squash. 19 The biological mechanisms of the protective role of LU in the eye has been reviewed. 20 LU was found to be concentrated in the macula, a small area of the retina responsible for central vision. The hypothesis for the natural concentration is that LU helps keep the eyes safe from oxidative stress and the high-energy photons of blue light. Various research studies have shown that a direct relationship exists between LU intake and pigmentation in the eye. Recent studies showed the various biological properties of LU in different disease models including diabetic retinopathy, 21 uveitis, 22 light-induced retinopathy, 23,24 and ischemia/reperfusion injury. 25
The transcription factor nuclear factor (erythroid-derived 2)-like 2 (Nrf2) is a basic leucine zipper protein that regulates the expression of antioxidant proteins which protect against oxidative damage triggered by injury and inflammation. The main role of Nrf2 signaling is to protect against insults such as xenobiotics and oxidative stress by activating the cellular antioxidant response. 26 –29 However, few studies have addressed the mechanism of LU’s effects and the involvement of Nrf2 pathway in these effects in the testis.
This study was designed to investigate the combined effect of LU by gavage and arsenic on male mice to elucidate the function of Nrf2 pathway. The findings can provide better understanding of the mechanism of LU, which also guides for the prevention and therapy of arsenicosis.
Materials and methods
Chemicals
Arsenic trioxide (ATO; As2O3; arsenite) was purchased from Beijing Chemical Reagent Corp. (Beijing, China). ATO was dissolved in 1 N sodium hydroxide at 0.1 M as a stock solution. LU (>95% purity) was obtained from Day-Natural Tech Co., Ltd. (Xi’an, China) dispersed in coin oil from Arawana (Tianjin, China). SOD, GSH-Px, total antioxidative capacity (T-AOC), 8-hydroxydeoxyguanosine (8-OHDG), and malondialdehyde (MDA) assay kits were purchased from Nanjing Jiancheng Bioengineering Institute (Nanjing, China). All other chemicals were of analytical grade and obtained commercially.
Animals and treatment
Forty healthy Kunming male mice weighing 18–22 g with the approval of the Ethics Committee of Shihezi University were chosen as the subjects. The mice were obtained from the laboratory center of XinJiang Medical University (license numbers: SYXK 2011-0004; Urumqi, China). Animals were kept in well-ventilated cages in a temperature-controlled room at 22–25°C under specific pathogen-free conditions on a 12-h light/12-h dark cycle. Mice received a standard laboratory diet composed of 60% ground corn meal, 15% ground beans, 10% bran, 10% corn oil, 3% casein, 1% mineral mixture, and 1% vitamin mixture and supplied with water ad libitum throughout the experimental period. After 2 weeks of acclimatization, the animals were divided into 4 groups of 10 mice each. All mice were administered their respective treatments daily by gavage for 5 weeks.
The mice were divided into 4 groups as follows: (1) control group received normal saline (NS) solution and corn oil; (2) ATO-treated group received ATO solution (5 mg/kg/day). This dose was reported to produce clinical effects without affecting survival 30 ; (3) LU-treated group received LU (40 mg/kg/day). This dose was reported to counteract semicarbazide-induced toxicity 31 ; and (4) ATO + LU-treated group received ATO (5 mg/kg/day) in the morning and LU (40 mg/kg/day) in the evening.
Preparation of plasma and collection of testis tissues and testis somatic index
At the end of the treatment period, mice were killed by cervical dislocation. The blood samples were collected into evacuated tubes containing heparin solution. The body weight and testes were weighed to the nearest milligram on a Shimadzu electronic balance (model BL-220H, Tokyo, Japan). Testis somatic index was calculated using the following formula: index weight = organ weight / body weight (10−3g/g).
Measurement of sperm quality
The cauda epididymal sperm suspension was prepared in normal saline at 37°C. The indexes including sperm count (× 106/mL) and sperm abnormalities (%) were used to evaluate the sperm quality according to the reported methods. 32,33
Histopathological examination of testis tissue
Sections were taken from testis tissues from mice in each group immediately after killing. The tissues were washed with normal saline solution to remove blood, fixed in 10% neutral formalin for a period of at least 24h, dehydrated in different grades of alcohol, and processed for paraffin embedding. Sections of 5-μm thickness were cut using a rotary microtome. The sections were processed and passed through a graded alcohol series, stained with hematoxylin and eosin, cleared in xylene, and examined microscopically according to the previously described methods. 34
Preparation of testis homogenates
Tissue testis homogenates were prepared by soaking, in a lysis buffer containing 20 mM tris(hydroxymethyl)aminomethane (Tris; pH 7.5), 150 mM sodium chloride, 1% Triton X-100, and the protein inhibitors sodium pyrophosphate, β-glycerophosphate, ethylenediaminetetraacetic acid, sodium orthovanadate, and leupeptin (Beyotime Biotechnology, Shanghai, China), and then homogenized with a TissueLyser (Qiagen, Valencia, California, USA). The protein concentration in the homogenate was measured with a Bradford protein assay kit (Beyotime Biotechnology, Shanghai, China).
Measurement of lipid peroxidation
MDA in the tissue homogenates was measured with an MDA analysis kit (Nanjing Jiancheng Bioengineering Institute, Nanjing, China) following the manufacturer’s instructions (absorption maximum at 532 nm).
Measurement of antioxidant enzyme assay
GSH, SOD, and T-AOC were measured using the analysis kits (Nanjing Jiancheng Bioengineering Institute, Nanjing, China), which could react with some certain chemicals and form the colored products and can be assayed by colorimetry at 532, 560, and 550 nm, respectively, according to the instructions. 8-OHdG was assayed using a commercial enzyme-linked immunosorbent assay kit (Nanjing Jiancheng Bioengineering Institute, Nanjing, China) according to the manufacturer’s instructions. Results were normalized to the total amount of protein as measured by bicinchoninic acid.
Detection of mRNA of Nrf2-related genes by real-time PCR
Testis tissue was snap frozen in liquid nitrogen and total RNA was extracted by the TRIzol extraction method (Invitrogen, Grand Island, New York, USA) according to the manufacturer’s instructions. Equal amounts of RNA (2 µg) were reverse transcribed into complementary DNA (cDNA) using the Transcriptor First Strand cDNA Synthesis Kit (Roche, Indianapolis, Indiana, USA). Primers were synthesized by Sigma-Aldrich (St Louis, Missouri, USA) for the following Mus musculus genes: heme oxygenase 1 (HO-1; 184 bp amplicon; F: 5′-CAGGTGATGCTGACAGAGGA-3′; R: 5′-ACAGGAAGCTGAGAGTGAGG-3′); glutathione S-transferase (GST; 199 bp amplicon; F: 5′-ATCGTTCCCTTTCTCGGCAT-3′; R: 5′-GCAGCCTGTAAGCCATTGAC-3′); nicotinamide adenine dinucleotide phosphate dehydrogenase, quinone 1 (NQO1; 112 bp amplicon; F: 5′-TGGCCGAACACAAGAAGCTG-3′; R: 5′-GCTACGAGCACTCTCTCAAACC-3′); nuclear factor (erythroid-derived 2)-like 2 (Nrf2,173 bp amplicon; F: 5′-TTCCATTTACGGAGACCCAC-3′; R: 5′-ATTCACGCATAGGAGCACTG-3′); and β-actin (240 bp amplicon; F: 5′-CACGATGGAGGGGCCGGACTCATC-3′; R: 5′-TAAAGACCTCTATGCCAACACAGT-3′).
The relative gene expression of targets was detected using a comparative cycle threshold method. 35 All samples were tested in triplicate. Real-time polymerase chain reaction (PCR) was performed on a mixture containing 10 μL of PCR Supermix (Bio-Rad Laboratories, Hercules, California, USA), 1 μL of forward and reverse primers (Sangon, Beijing, China), 1 μL of template DNA, and 8 μL of distilled water.
The quantitative PCR conditions were as follows: 1 cycle of initial denaturation (94°C for 3 min), 30 cycles of amplification (94°C for 30 s, 57°C for 30 s, and 72°C for 25 s), 1 cycle of melting curve measurement (95°C for 5 s, 65°C for 60 s, and a gradual increase in temperature to 97°C), and a cooling period (40°C for 30 s). The data presented were relative messenger RNA (mRNA) levels normalized to β-actin.
Detection of protein
Expression of Nrf2-related protein was performed using Western blot. Testis tissues were homogenized in one volume of sample buffer (50 mM Tris–chloride, 100 mM dithiothreitol, 10% glycerol, and 2% sodium dodecyl sulfate (SDS)), and centrifuged at 14,800g at 4°C for 15 min to move debris. The samples were subjected to SDS-polyacrylamide gel electrophoresis and transferred to polyvinylidene difluoride membranes. After blocking with skim milk (5%), the blots were probed with the primary antibodies (Abcam, Cambridge, UK) for Nrf2 (1:1000), Hmox-1 (1:1000), NQO1 (1:1000), GST (1:1000), and β-actin (1:1000) at 4°C overnight. Incubation with the primary antibodies was followed by incubation with secondary antibodies (conjugated to horseradish peroxidase) after washing in Tris-buffered saline and Tween 20. Blots were processed using an enhanced chemiluminescence kit (Santa Cruz Biotechnology, Inc., Dallas, Texas, USA) and exposed to film. All experiments were repeated three times.
Statistical analysis
The results were expressed as the mean ± standard deviation. A general linear model was used to analyze the interactions of the effects of the combination of ATO and LU in a two-factor, two-level factorial design experiment. The model was as follows, in which a significant p value for b3 indicated an interaction between the effects of ATO and LU.
where “y” is a measured parameter such as body weight, testis weight, testis/body weight ratio, sperm count, and so on. Bonferroni’s multiple comparison method is used to determine whether the differences were significant. The data were analyzed using SPSS software for Windows version 15.0 (SPSS Inc., Chicago, Illinios, USA), and values of p < 0.05 were considered to be statistically significant.
Results
The effect of ATO and/or LU on the indexes related with reproductivity of mice
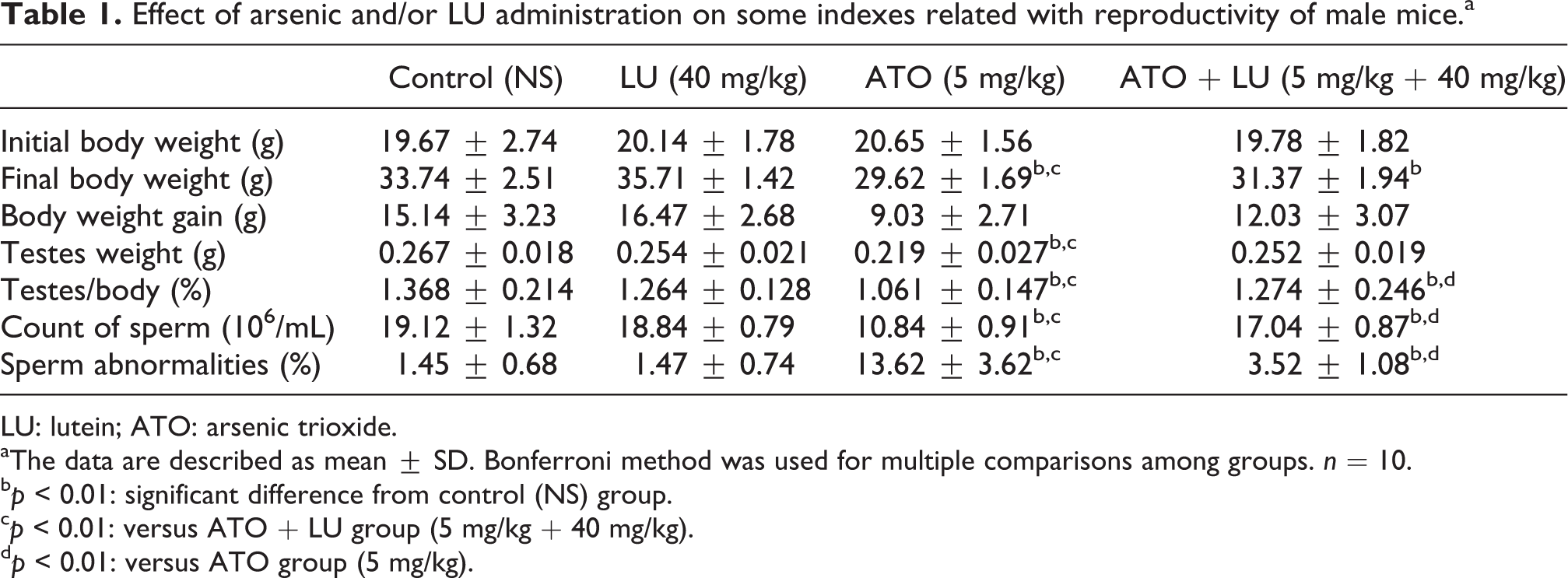
The body weight, testes weight, and testes somatic index of mice treated with ATO were decreased as compared to the control group (p < 0.01). The body weight, testes weight, or testes somatic index of LU group were not significantly different from the control group. In addition, there was a statistically significant interaction between the effects of ATO and LU on body weight gain (F = 12.878, p < 0.001) and testes somatic index (F = 4.633, p = 0.007; see Table 1).
Effect of arsenic and/or LU administration on some indexes related with reproductivity of male mice.a
LU: lutein; ATO: arsenic trioxide.
aThe data are described as mean ± SD. Bonferroni method was used for multiple comparisons among groups. n = 10.
bp < 0.01: significant difference from control (NS) group.
cp < 0.01: versus ATO + LU group (5 mg/kg + 40 mg/kg).
dp < 0.01: versus ATO group (5 mg/kg).
LU had no effect on sperm count or sperm abnormalities, whereas the ATO group had a lower sperm count (p < 0.01) and higher rate of sperm abnormalities (p < 0.01) than the control group. The significant interactions of sperm count (F = 150.697, p < 0.001) and sperm abnormalities (F = 88.614, p < 0.001) were found between ATO and LU. There was an increase in sperm count and decrease in sperm abnormalities in ATO + LU-treated group compared with the ATO group (p < 0.01).
The effect of ATO and/or LU on the testicular oxidative damage and antioxidant enzymes
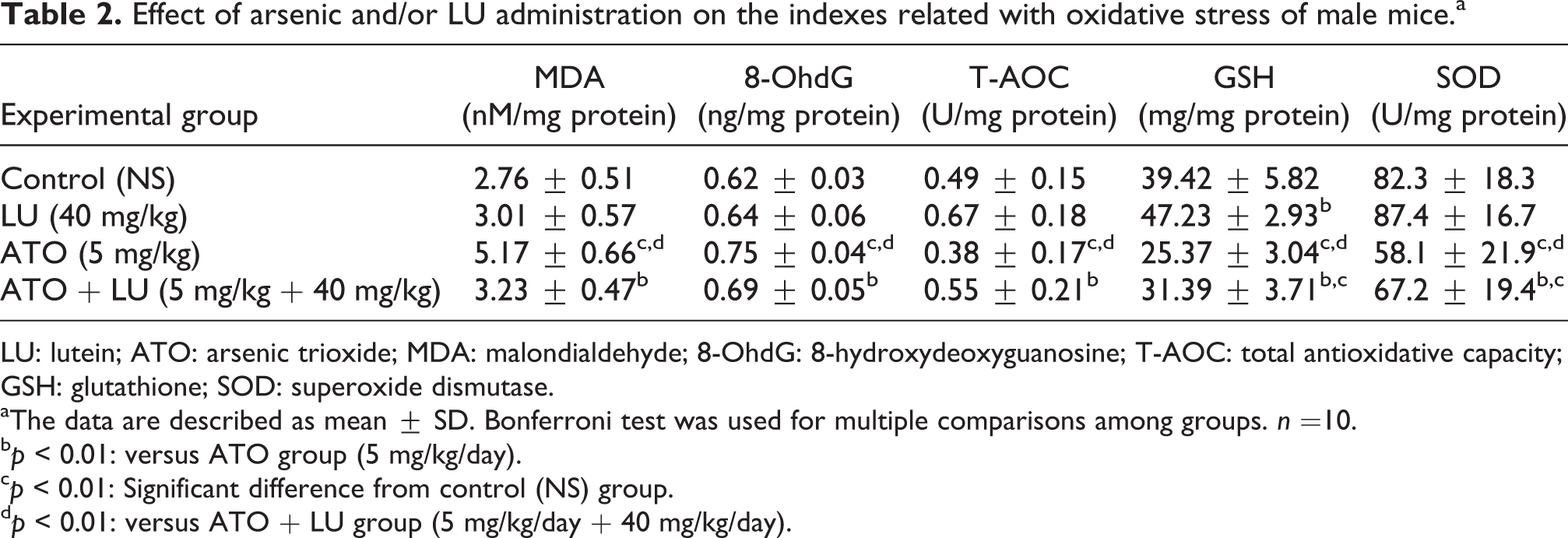
ATO treatment increased oxidative damage level (MDA and 8-OHdG) and decreased the activity of antioxidant enzymes (GSH and SOD) compared with the control group (p < 0.01). ATO + LU (40 mg/kg/day) group showed decreased MDA and 8-OHdG levels and increased T-AOC, as well as the activities of GSH and SOD (p < 0.01), than the ATO group. The statistically significant interactions were found between the effects of ATO and LU on MDA (FMDA = 39.117, PMDA < 0.001), 8-OHdG (F8-OHdG = 15.659, P8-OHdG < 0.001), as well as on GSH activity (FGSH = 55.394, PGSH < 0.001; see Table 2).
Effect of arsenic and/or LU administration on the indexes related with oxidative stress of male mice.a
LU: lutein; ATO: arsenic trioxide; MDA: malondialdehyde; 8-OhdG: 8-hydroxydeoxyguanosine; T-AOC: total antioxidative capacity; GSH: glutathione; SOD: superoxide dismutase.
aThe data are described as mean ± SD. Bonferroni test was used for multiple comparisons among groups. n =10.
bp < 0.01: versus ATO group (5 mg/kg/day).
cp < 0.01: Significant difference from control (NS) group.
dp < 0.01: versus ATO + LU group (5 mg/kg/day + 40 mg/kg/day).
Morphological changes in the testis in the treatment groups are shown in Figure 1. ATO administration caused a reduction in germ cell number, necrosis of sperm, congestion of interstitial blood vessels, degeneration, and necrosis of germ cells lining seminiferous tubules (Figure 1(b)). The groups treated with ATO + LU showed some normal and some ectopic seminiferous tubules (Figure 1(d)). While control and LU groups showed the normal structure of germ cell and sperm.

Histopathological changes of testis treated with ATO and/or LU (×200). (a) Testis of normal negative control group showed the normal histopathological structure of seminiferous tubules and spermatogenous cells. (b) Testis of mice group administered ATO. It reveled reduction of germ cells, necrosis of sperm cell, congestion of interstitial blood vessel, degeneration, and necrosis of germ cells lining seminiferous tubules. (c) Testis of mice group received LU showed normal structure of germ cell and sperm. (d) Testis of mice group administered ATO and treated with LU. It revealed partly normal and ectopic seminiferous tubule. ATO: arsenic trioxide; LU: lutein.
The effect of ATO and/or LU on Nrf2 signaling-related gene mRNA expression
The mRNA expression levels of Nrf2 and its downstream genes (HO-1, GST, and NQO1) of ATO group were higher than those of the control group (p < 0.01). LU increased the mRNA expression levels of these genes compared with the control group (p < 0.01). The mRNA expression level of the genes of ATO + LU group were the highest among the groups (p < 0.01; see Figure 2).

(a) Real-time PCR analysis of treatment of ATO and/or LU. Nrf2 (molecular weight 173 bp), NQO1 (molecular weight 112 bp), HO-1 (molecular weight 184 bp) and GST (molecular weight 199 bp) mRNA expression levels were measured by real-time PCR. (b) Quantitative mRNA analysis of treatment of ATO and/or LU. Y-axis indicates mRNA expression of test marker versus β-actin. Each bar represents the mean ± SD. The gene expression of Nrf2, NQO1, HO-1, and GST of LU-treated group were higher than those of control group. The group treated with ATO + LU showed the most prominent mRNA expression of Nrf2-related genes. Significant differences relative to the control and ATO groups are indicated as follows: *p < 0.01: versus control group; #p < 0.01: versus ATO group. PCR: polymerase chain reaction; ATO: arsenic trioxide; LU: lutein; Nrf2: nuclear factor erythroid 2-related factor 2; NQO1: NAD(P)H dehydrogenase, quinone 1; HO-1: heme oxygenase-1; GST: glutathione transferase; mRNA: messenger RNA.
The effect of ATO and/or LU on Nrf2 signaling-related protein expression
The effects of LU on protein expression related to the Nrf2 signaling pathway are shown in Figure 3. Interestingly, protein expression levels of HO-1, GST, NQO1, and Nrf2 were greater in both ATO group and LU group than in control group (p < 0.01). The group treated with ATO + LU (40 mg/kg/day) showed elevated expression of genes related to the Nrf2 signaling pathway compared to the other groups (p < 0.01).

(a) Western blot analysis of protein levels of treatment of ATO and/or LU. Nrf2 (molecular weight 57 kDa), NQO1 (molecular weight 31 kDa), HO-1 (molecular weight 32 kDa), and GST (molecular weight 26 kDa) protein expression were measured in the livers of mice treated with ATO and/or LU by Western blot. (b) Quantitative protein analysis of treatment of ATO and/or LU. Nrf2, NQO1, HO-1, and GST protein expression levels were measured. Y-axis represents protein expression of test protein relative to β-actin. Each bar represents the mean ± SD. The protein expression of Nrf2, NQO1, HO-1, and GST of LU-treated group were higher than those of control group. The group treated with ATO + LU showed the most prominent protein expression of Nrf2-related genes. Significant differences relative to the control and ATO groups are indicated as follows: *p < 0.01: versus control group; #p < 0.01: versus ATO group. ATO: arsenic trioxide; LU: lutein; Nrf2: nuclear factor erythroid 2-related factor 2; NQO1: nicotinamide adenine dinucleotide phosphate dehydrogenase, quinone 1; HO-1: heme oxygenase 1; GST: glutathione transferase.
Discussion
Recently, more and more studies focused on the arsenic-induced reproductive toxicity. Arsenic is reported to play a role in the endocrine system. 36 –38 Based on the mice model, arsenic could decrease the reproductive ability, including sperm count, sperm motility, and testicular weight, 39,40 and disrupt meiosis of spermatogenesis, even rapid and extensive disruption with acute arsenic exposure to mice. 8 Our study also showed the similar reproductive toxicity of mice treated with ATO as the references. ATO could induce the significant decrease of sperm quality in mice. The obvious abnormal pathological changes in testis of ATO group could provide strong evidence for the arsenic-induced reproductive toxicity. In addition, the present study shows that arsenic could induce the oxidative stress. The oxidative injury (MDA and 8-OHdG) level of ATO group was higher than the control group, while the concentration of antioxidative stress (GSH) was reduced. These results elucidate that arsenic induced significant reproductive toxicity linked with oxidative damage in the testis, which hints that better antioxidant therapies may be beneficial for relevant cases of hypo spermatogenesis. 41,42
LU is synthesized only by plants and like other xanthophylls is found in high quantities in green leafy vegetables such as spinach, kale, and yellow carrots, with protective effects against oxidative stress 43,44 LU may have ameliorated the oxidative status by promoting the antioxidant network. 45 The present study shows that LU blocks arsenic-induced pathological changes and oxidative damage. Our study found statistically significant interaction between LU and ATO on index of sperm quality, levels of MDA and 8-OHdG and GSH concentration. In comparison among the groups, we found that these parameters of ATO + LU group ranged between the levels observed in the ATO and control groups. These results indicate that LU has an antagonistic effect on ATO-induced reproductive toxicity and oxidative stress.
Previous studies have shown that LU inhibits oxidative damage and protects hepatic function, 31 but few reports have focused on the relationship between LU and the Nrf2 signaling pathway. Our study also demonstrated that LU activated Nrf2 to alleviate oxidative damage induced by arsenic. Nrf2 has been found to induce a cellular defense system that detoxifies drugs and environmental pollutants. 46 Our study showed that the mRNA expression levels of Nrf2 and its downstream genes GST, HO-1, and NQO1 of LU group increased compared with the control group. The similar effect of protein expression level of Nrf2 signaling of LU group was found. These results implicated that LU possibly activates Nrf2 signaling to decrease MDA and 8-OHdG levels and increased GSH concentration. Therefore, this is a possible mechanism of LU alleviating the arsenic-induced reproductive toxicity and oxidative stress.
Conclusions
Based on these measured parameters and coordinated mRNA and protein expression of Nrf2 signaling pathway components, our findings suggest that LU possibly counteracts the arsenic-induced reproductive toxicity and oxidative stress by activating Nrf2 signaling. This study also provided more evidence that consuming the rich plant sources of LU will alleviate chemicals-induced reproductive toxicity.
Footnotes
Acknowledgments
The constructive critics and helpful suggestions from reviewers are deeply appreciated. The authors are grateful to the Key Laboratory of Xinjiang Endemic and Ethnic Diseases (Ministry of Education), Shihezi University School of Medicine, Xinjiang, China, for assistance with this work.
Authors’ Note
Authors SGL and SZX contributed equally to this work. SGL and SZX prepared the design of this study, collected the data, and wrote the manuscript. FL, QN, YSD, and LJP participated in the experiment. RLM, MXJ, KW, XMM, and GLF contributed to acquiring reagents/materials. HLX and XFZ analyzed data. FL edited and revised the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from Xinjiang Production and Construction Corps (2014BA039, 2015AG014) and Shihezi University (RCZX201112).

